OCHEM 2211 Chapter 9 mechanisms
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
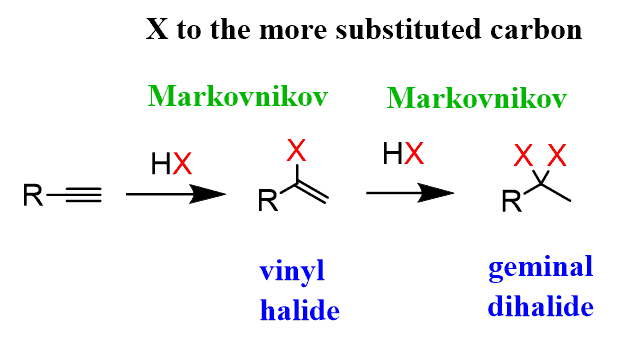
Hydrohalogenation
2 step process, an alkyne combining with excess HX, X being Cl, Br, or I to create a geminal halide- mark regio-selectivity and no stereo-specificity (1 HX creates a vinyl halide)
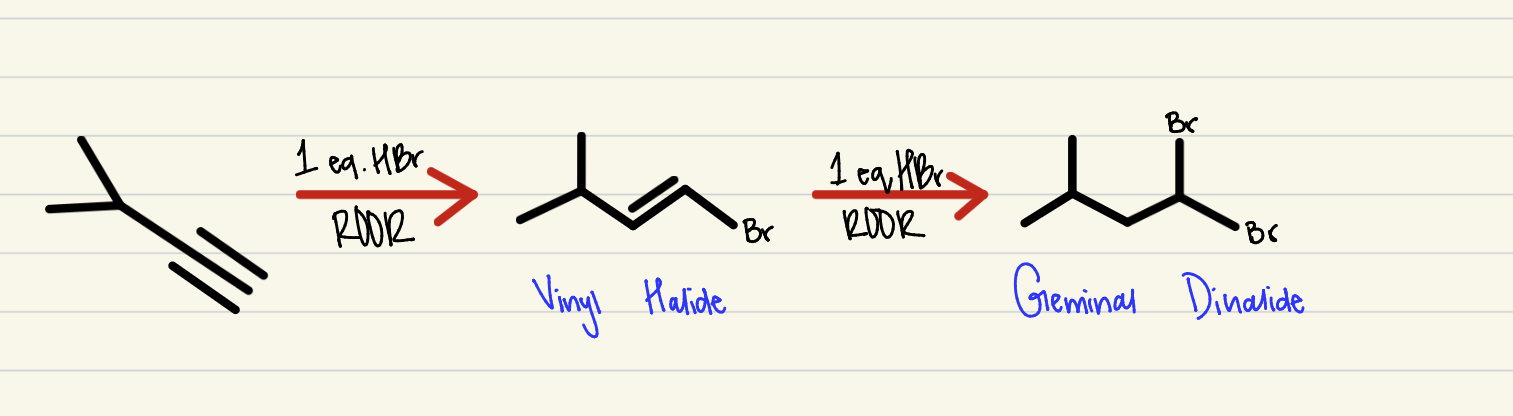
Hydrobromination
2 step process, an alkyne combining with excess HBr and peroxide to create a geminal dihalide- anti-mark regio-selectivity and anti stereo-specificity (1 HBR creates a vinyl halide)
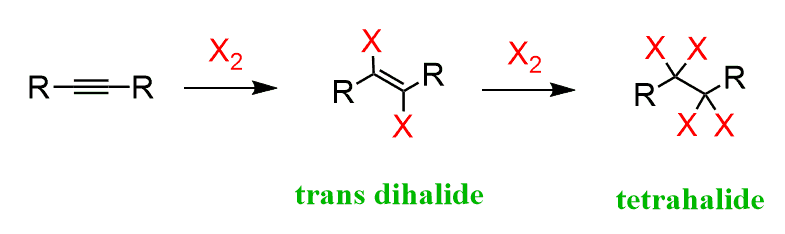
Halogenation
2 step process, an alkyne combining with excess X2, X being Cl or Br, and CCl4 to create a tetrahalide- mark regio- selectivity and anti stereo-specificity (1 X2 creates a Vicinal/trans Dihalide)
Tautomerization
2 step process, an alkyne-alcohol with H3O to create a ketone or aldehyde, depending on the structure
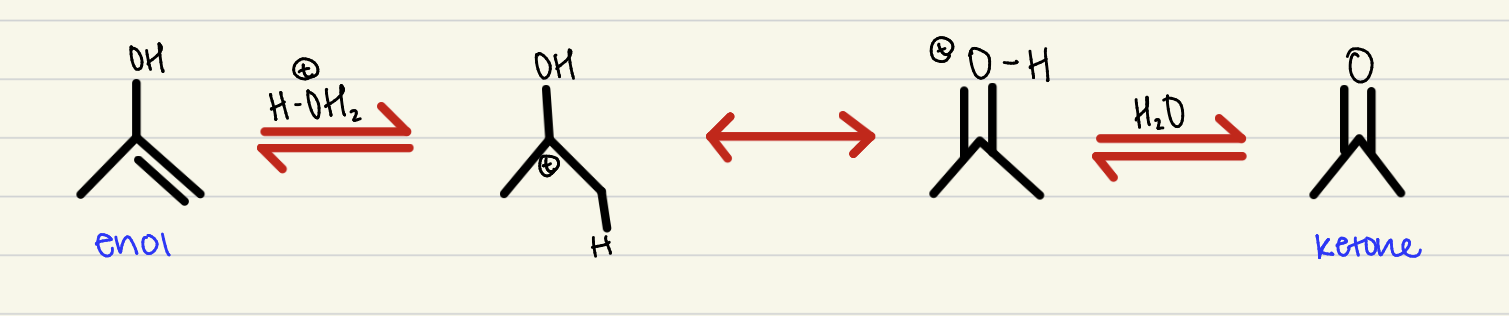
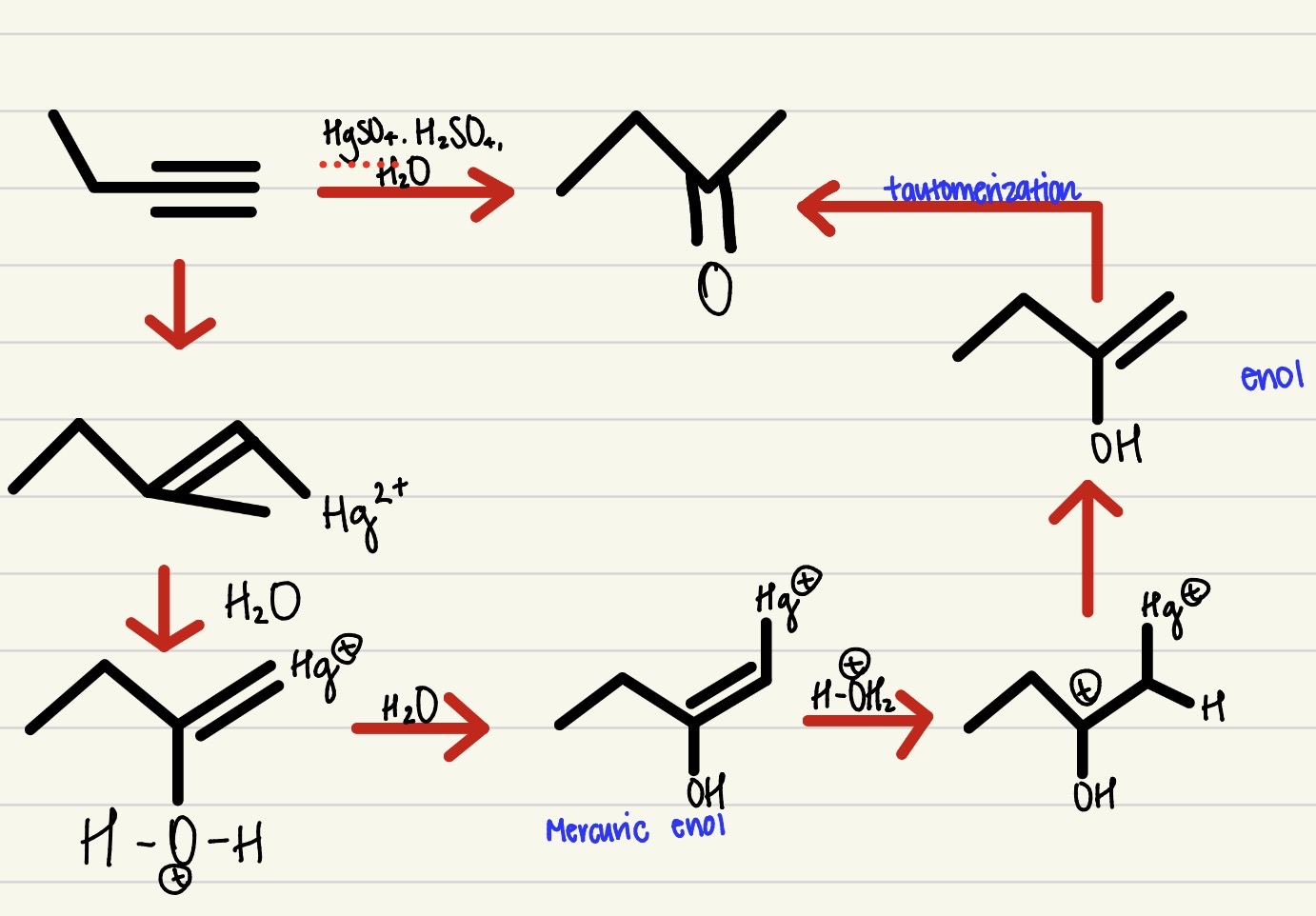
Mercury- Catalyzed Hydration
1 step process, an alkyne with H2O, H2SO4, and HgSO4 to create an alcohol, subject to go through tautomerization- mark regio-selectivity and no stereo-specificity
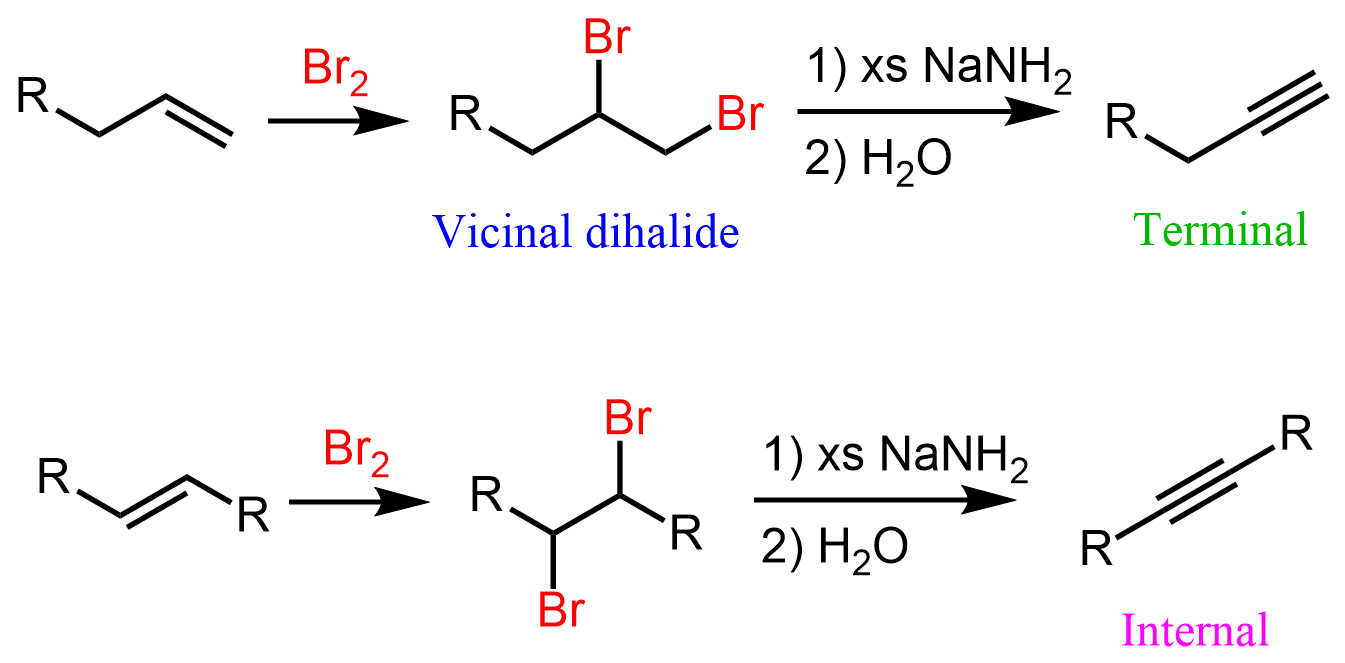
Alkyne formation
2 step process, an alkene with X2, X being Br or Cl, CCl4, and NaNH2 to create an alkyne
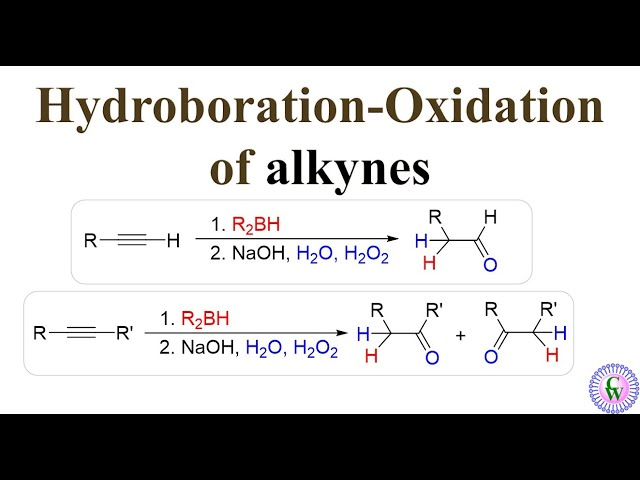
Hydroboration-Oxidation
2 step process, a terminal alkyne uses R2BH/9-BBN, THF then H2O2, NaOH then tautomerization to produce an alcohol- anti-mark regio-specificity and syn stereo-selectivity
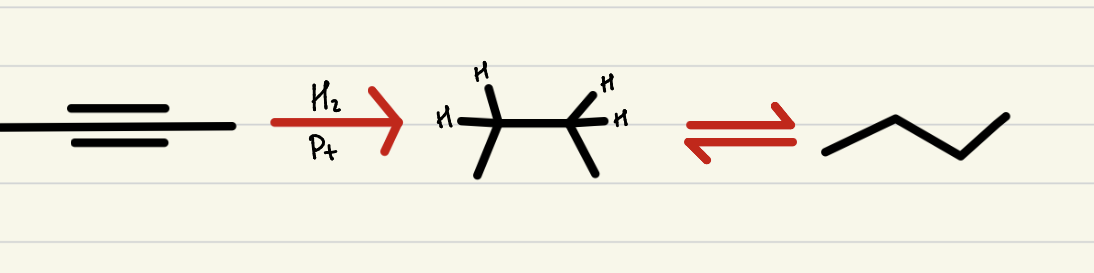
Catalytic Hydrogenation
2 step process, an alkyne with excess H2 and a metal to create an alkane- no regio-selectivity and syn stereo-specificity
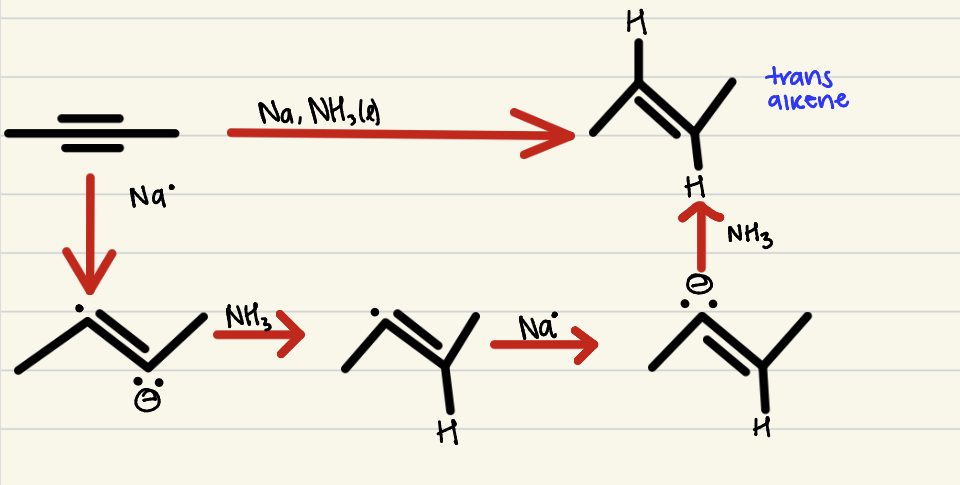
Dissolving Metal Reduction
1 step process, an internal alkyne with an alkali metal and liquid NH3 to produce a trans alkene- no regio-selectivity and anti stereo-specificity
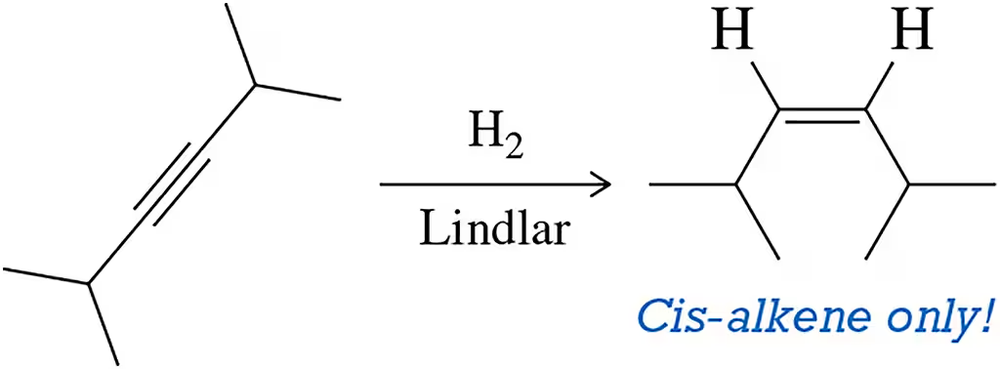
Lindlar’s Hydrogenation
1 step process, an alkyne with H2 and Lindlar’s catalyst produces a cis alkene- no regio-selectivity and syn stereo-specificity
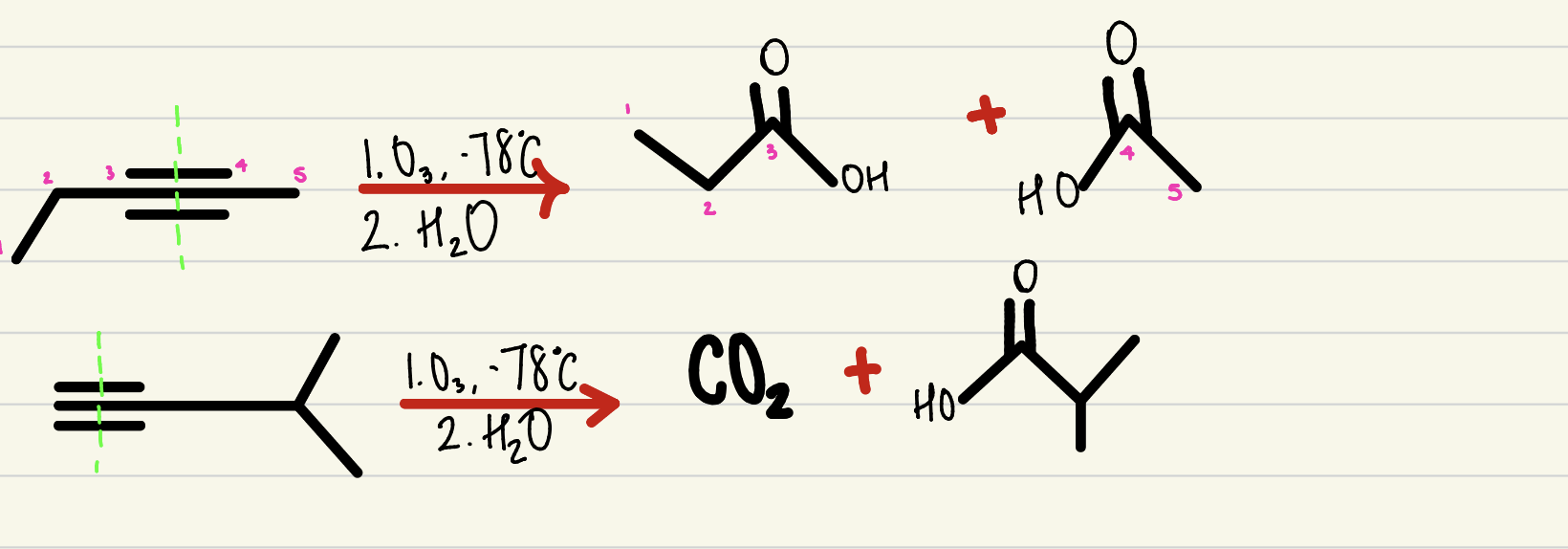
Oxidative Ozonolysis
2 step process, an alkyne with O3 @ -78C and H2O cleaves down through the triple bond to produce a carboxylic acid (no terminal H) or CO2 (1 terminal H)- no regio-selectivity nor stereo-specificity
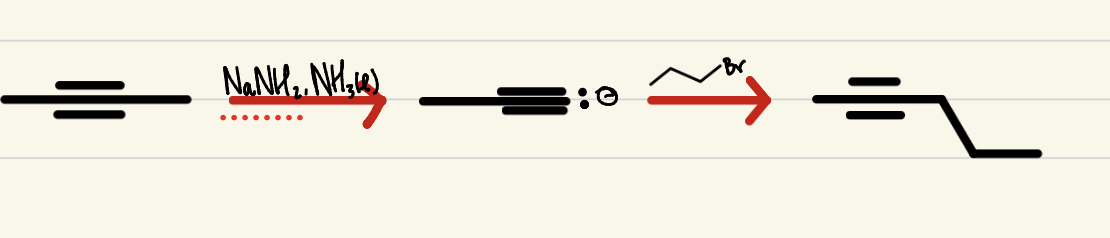
Chain Extension
2 step process, a terminal alkyne with NaNH2, liquid NH3 then an alkyl halide to add said halide to the original alkyne