Introduction to Hazardous Drugs 1 (Kirk)
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What is the primary purpose of USP <800>?
To provide standards for the safe handling of hazardous drugs to minimize the risk of exposure to healthcare personnel, patients, and the environment.
What are the two main criteria for a drug to be included on the NIOSH list?
The drug must be FDA-approved and either be accompanied by manufacturer special handling information (MSHI) or meet specific toxicity criteria such as carcinogenicity, teratogenicity, reproductive toxicity, organ toxicity, or genotoxicity.
What are the four primary routes of hazardous drug exposure in a clinical setting?
Inhalation, skin absorption, needle sticks, and accidental ingestion.
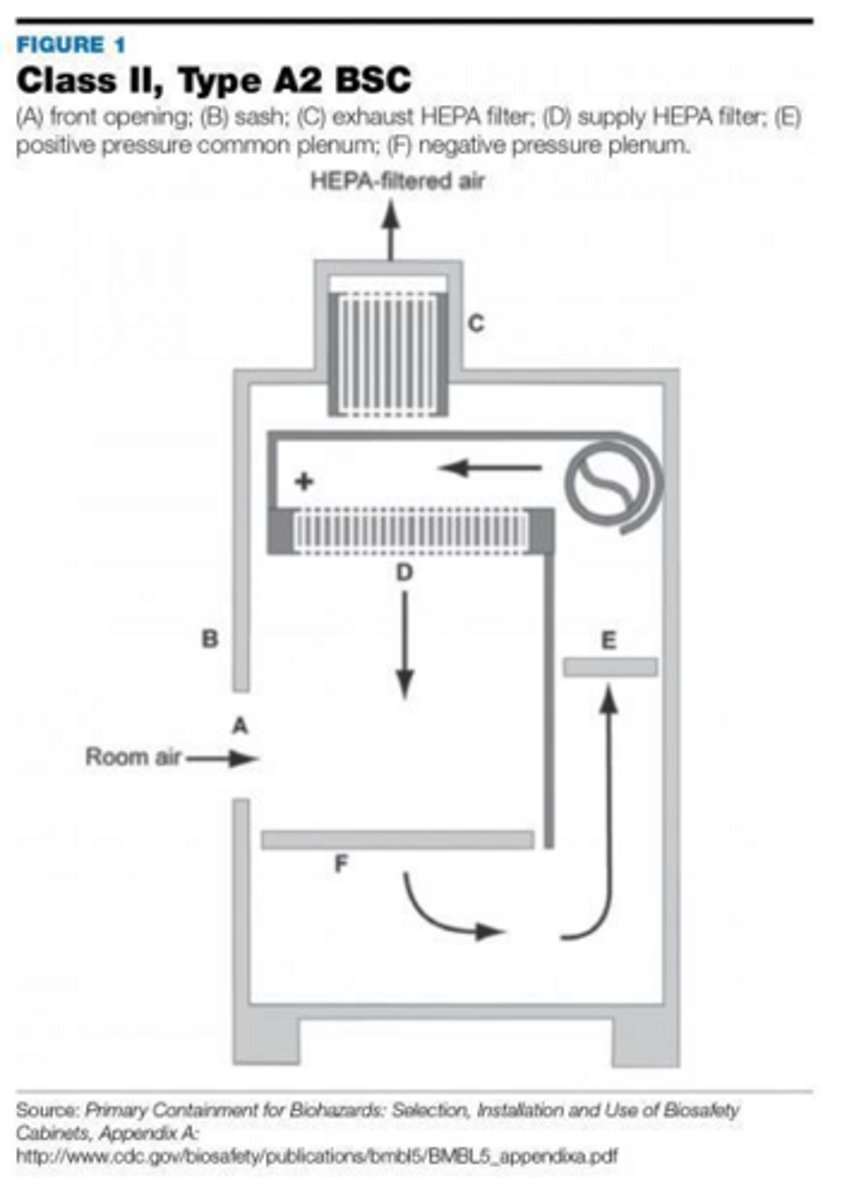
What does a 'Containment Primary Engineering Control' (C-PEC) provide for hazardous drug compounding?
It provides a sterile, negative-pressure environment with vertical airflow, typically vented to the outdoors, to contain hazardous drug vapors and particles.
List three examples of C-PECs used in sterile compounding.
Biological Safety Cabinets (BSCs), Compounding Aseptic Containment Isolators (CACIs), and Containment Ventilated Enclosures (CVEs).
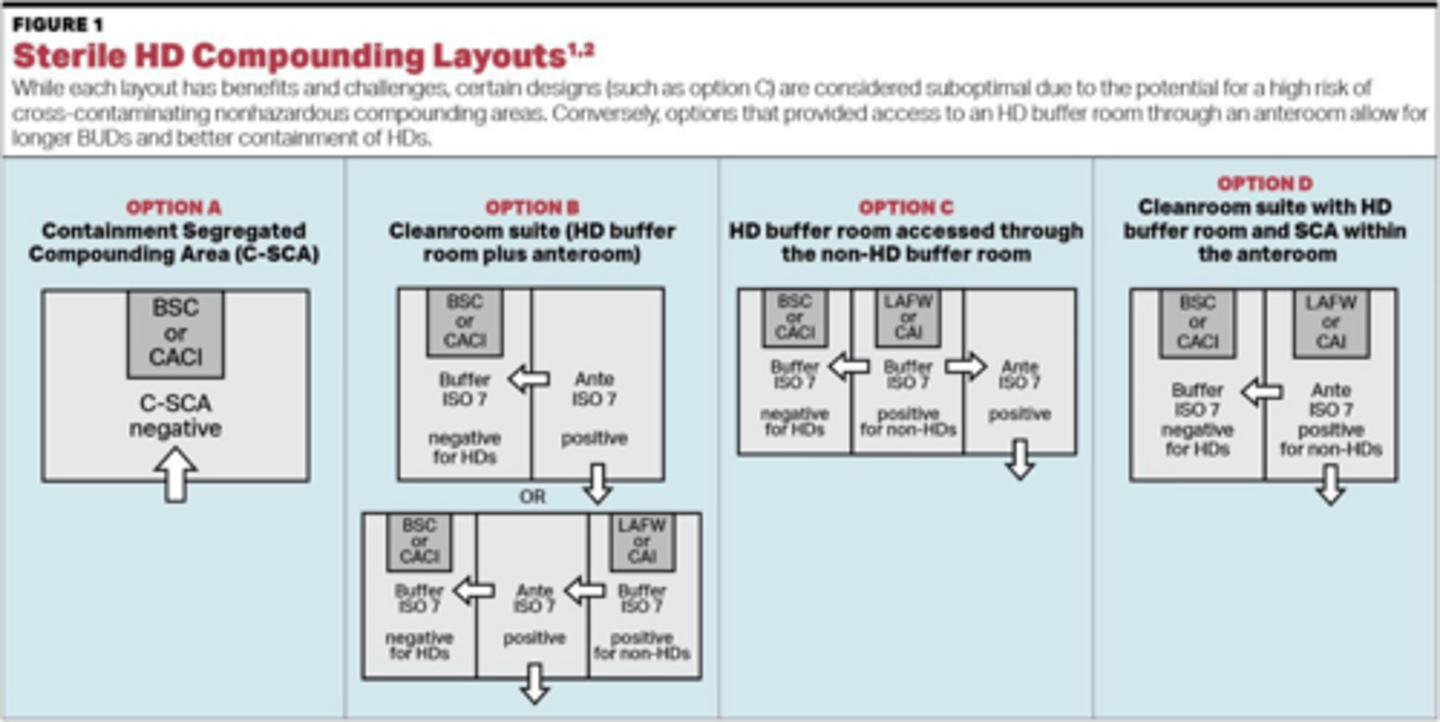
What are the required air pressure and ventilation conditions for a sterile C-SEC (Containment Secondary Engineering Control)?
The room must maintain negative pressure relative to surrounding areas and be externally vented.
What is the required ISO classification for an anteroom in a hazardous drug compounding facility?
ISO Class 7.
What is the function of a Closed System Transfer Device (CSTD)?
It is a supplemental engineering control designed to mechanically prohibit the transfer of environmental contaminants into the system and the escape of hazardous drug or vapor out of the system.
How should hazardous drugs be stored in a pharmacy setting?
They should be stored separately from non-hazardous drugs, often in a dedicated refrigerator or room that is externally ventilated and under negative pressure.
What is the purpose of an 'Assessment of Risk' regarding hazardous drug storage?
It allows for the determination of alternative containment strategies or work practices, and may permit certain lower-risk hazardous drugs to be stored with regular drugs.
What does the NIOSH Table 1 category represent?
Drugs that are carcinogenic to humans (NTP, IARC Group 1) or probably/possibly carcinogenic (IARC 2A/2B).

What does the NIOSH Table 2 category represent?
Drugs that pose adverse developmental effects, including risks to those who are pregnant, breastfeeding, or actively trying to conceive.
What is the required air change per hour (ACPH) for a sterile C-SEC?
The ACPH must be at least 12.
What is the required ACPH for an anteroom in a hazardous drug facility?
The ACPH must be at least 30.
How should hazardous drugs be packaged for transportation?
They should be placed in impervious plastic boxes or inner plastic bags, kept separate from other drugs, and clearly labeled as hazardous.
What is the significance of the 1990 ASHP Technical Assistance Bulletin?
It was an early professional guidance document regarding the handling of hazardous drugs.
What is the difference between a C-PEC and a C-SEC?
A C-PEC is the primary device (like a hood) where compounding occurs, while a C-SEC is the secondary engineering control (the room itself) that houses the C-PEC.
What is the role of NIOSH in hazardous drug regulation?
NIOSH identifies and publishes the list of hazardous drugs and provides guidance on how to handle them safely.
Are APIs (Active Pharmaceutical Ingredients) used in compounding included in the 'Assessment of Risk' process?
No, the Assessment of Risk process excludes any product considered an API used in compounding.
What is the primary air pressure requirement for an anteroom in a hazardous drug facility?
It must maintain positive air pressure.
What are the three factors considered when conducting an assessment of risk for hazardous drugs?
Risk of exposure, probability of exposure, and severity of effects.
What is a Containment Ventilated Enclosure (CVE) used for?
It is a C-PEC used specifically for non-sterile hazardous drug compounding.
What does the term 'genotoxicity' refer to in the context of hazardous drugs?
The ability of a substance to damage the genetic information within a cell, causing mutations.