Materials T4 - Polymers
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
Natural Polymers
Wood, natural rubber, proteins, DNA
Saturated Hydrocarbon
Carbon is bonded to four hydrogens
Unsaturated Hydrocarbon
Contains double and/or triple bonds, reactive
Isomers
Two compounds with identical chemical composition but different structures
Addition (Radical) Polymerisation
Free radical attacks double bond, forming new radical out of monomer. Cycle repeats, forms a polymer chain until fully saturated.
Polymers from Addition Polymerisation
Polyethylene, polypropylene, poly(vinyl chloride), polystyrene
Polycondensation (Step) Polymerisation
Chemical reaction occurs between functional group of two monomers, forms new covalent bond to create long polymer chain.
Homopolymer
Composed of the same repeat unit
Co-polymer
Composed of two or more different repeat units
Functionality
Number of bonds a monomer can form
Number-Average Polymer Weight
Favours all molecules equally (lower g/mol)
Weight-Average Polymer Weight
Larger molecules weighted more heavily (higher g/mol)
Polydispersity Index (PDI)
Weight-Avg / Number-Avg
Degree of Polymerisation
Number of repeat units per chain (number-avg / molecular weight of repeat unit)
Number Average Molecular Weight Increases
Melting temperature increases (up to 100g/mol for liquid, 1000g/mol for wax, over 1000g/mol for solid)
Molecular Orientation (Conformation)
Can be changed by rotation around singular bonds without breaking needed
Retricts Molecular Orientation
Double bonds and bulky/side groups
Configuration
The direction monomers are linked together or the order (for copolymers), to change must break bonds
Stereoisomers
Compounds that are mirror images, cannot superimpose without breaking bonds
Isotactic
All R groups are on same side of the chain
Syndiotactic
R groups alternate sides of chain
Atactic
R groups randomly placed on chain
Cis Configuration
Bulky groups on same side of chain
Trans Configuration
Bulky groups on opposite sides of chain
Linear Polymers
Repeat units joined together end to end in single chains
Linear Polymer Properties
Flexible with Van der Waals forces acting between chains
Branched Polymers
Contains side branch chains connected to main ones
Branched Polymer Properties
Reduced chain packing efficiency, lower density
Cross Linked Polymers
Adjacent linear chains joined to one another at various positions via covalent bonds
Network Polymers
Multifunctional monomers forming 3 or more covalent bonds
Strength of Polymer
Greater as number of chains/links increase
Copolymer
Two or more monomers polymerised together
Block Copolymer
Large blocks of monomers alternate
Graft Copolymer
Chains of one monomer grafted onto another monomer backbone
Thermoplastics
Linear and branched polymers (normally additive) that are soft and can be recycled
Thermoplastics when Heated
Secondary bonds break and polymer becomes viscous, bonds reformed when cooled
Thermosets
Cross-linked or network polymers that are harder, stronger and cannot be recyled
Thermosets when Heated
Undergo cross-linking, which is irreversible
Thermoplastic Vs Thermoset Bonding
Thermoplastics are held together by secondary bonds, thermosets are held together by cross-linking bonds
Crystallinity
Molecular chains pack to produce an ordered atomic array, range from entirely amorphous to mostly crystalline
Factors of Crystallinity
Rate of cooling during solidification and simplicity of repeat unit
Crystalisation of Atactic Polymers
Difficult due to random R groups
Crystalisation of Isotactic and Syndiotactic Polymers
Easier
Crystallite
Small crystalline regions in a semicrystalline polymer
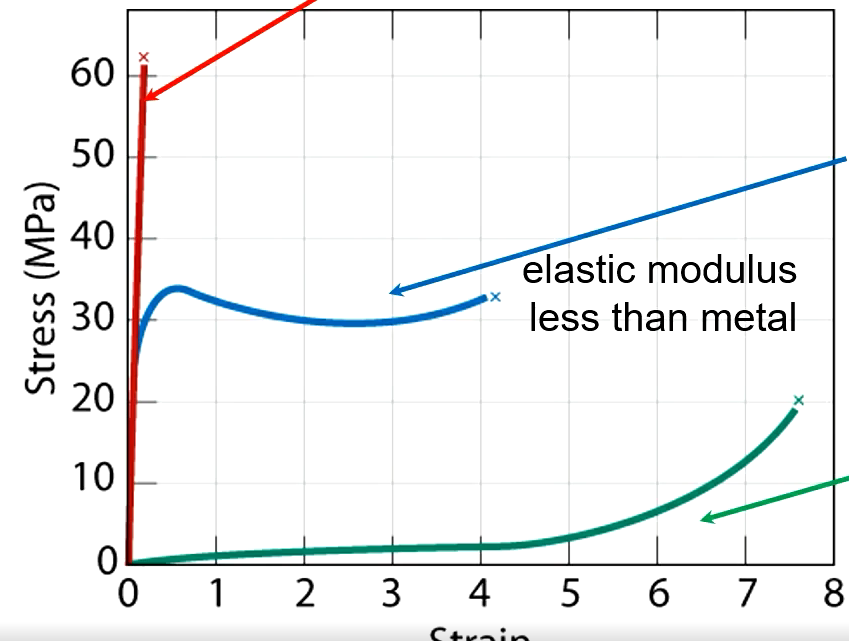
Brittle Polymer
Fractures during elastic deformation
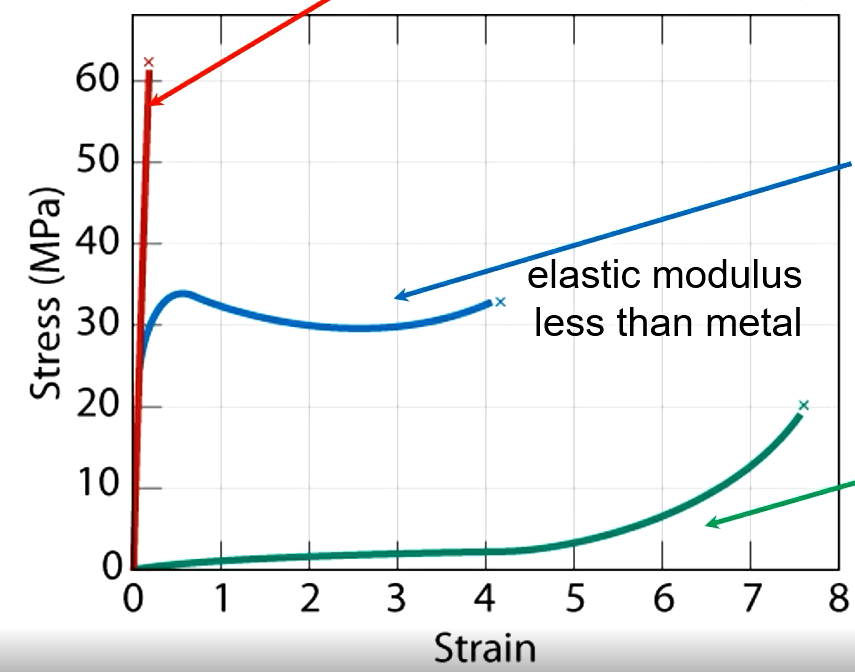
Plastic
Elastic then plastic deformation
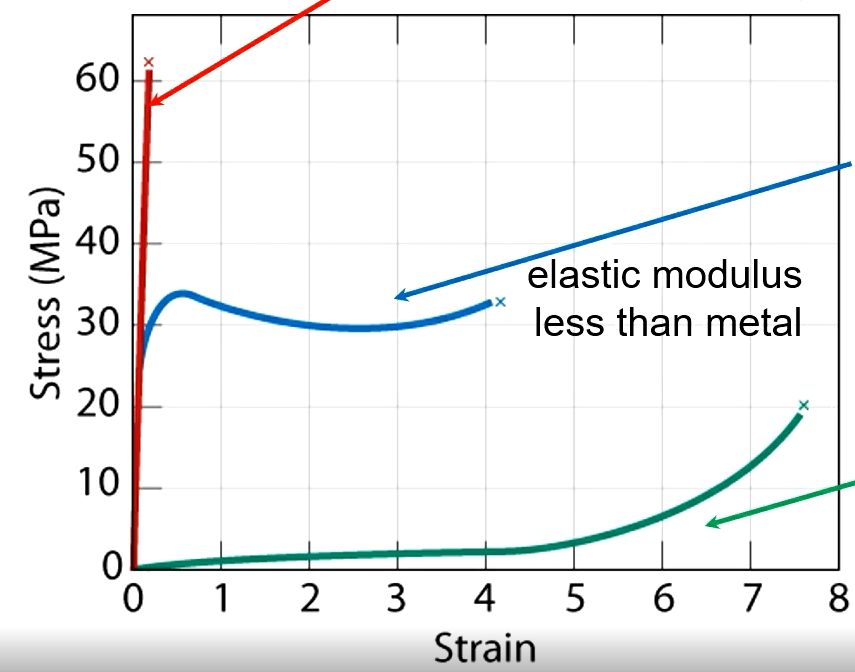
Elastomer
Experiences large recoverable strains
Effects of Increasing Temperature
Elastic modulus and tensile strength decrease, ductility increases
Viscoelasticity
Behaviour of polymers as rubbery solids at an intermediate temperature
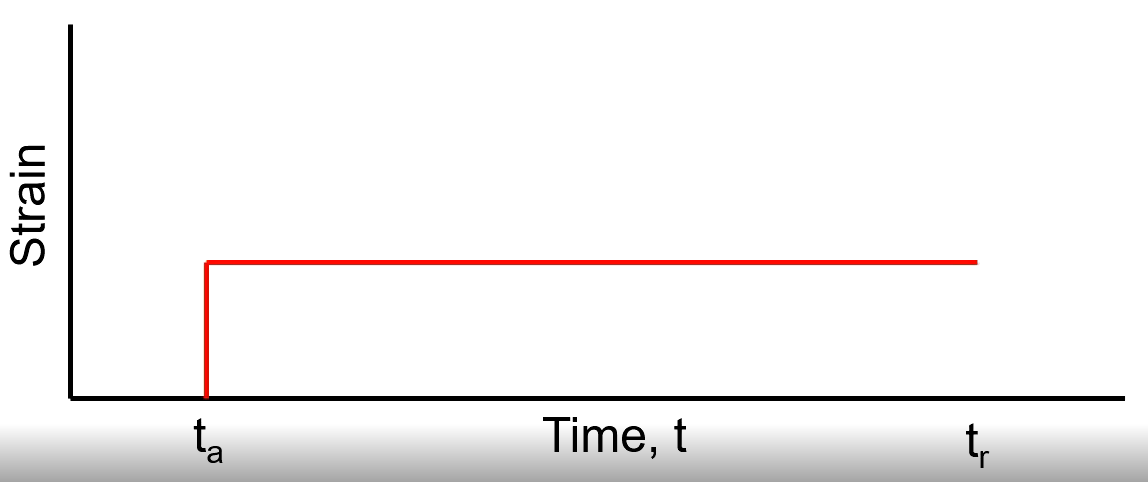
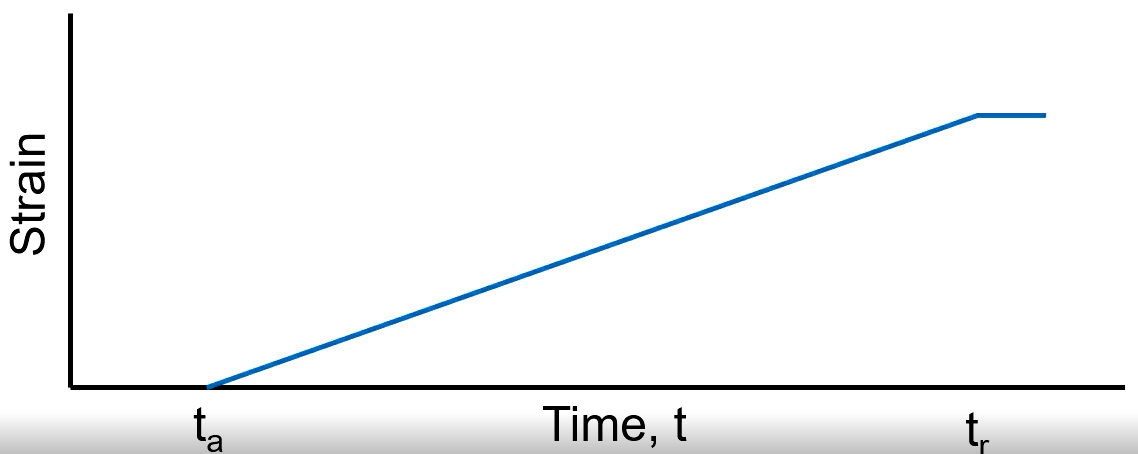
Elastic
Recovers fully
Viscous
Does not recover
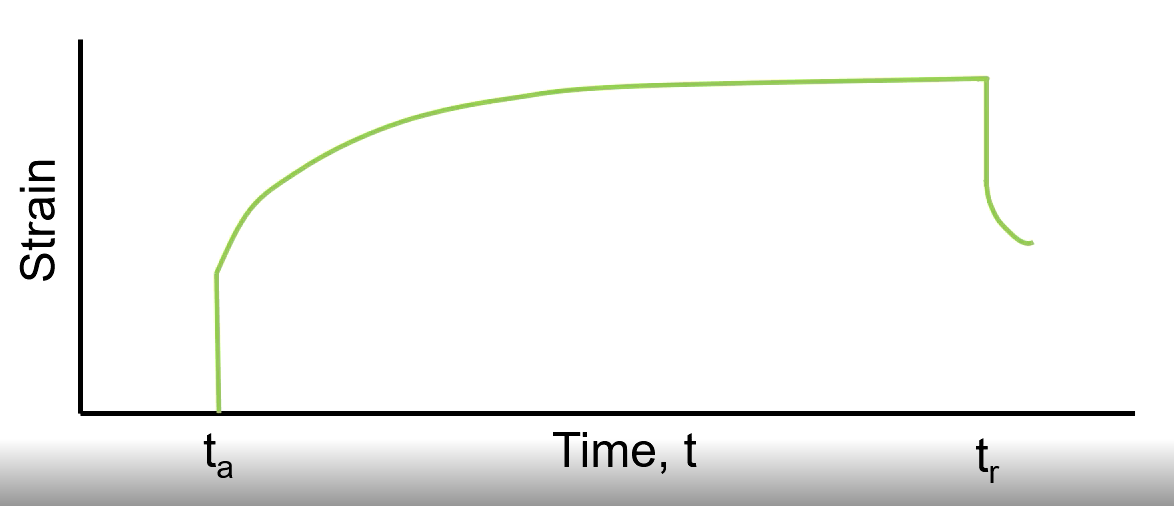
Viscoelastic
Recovers somewhat
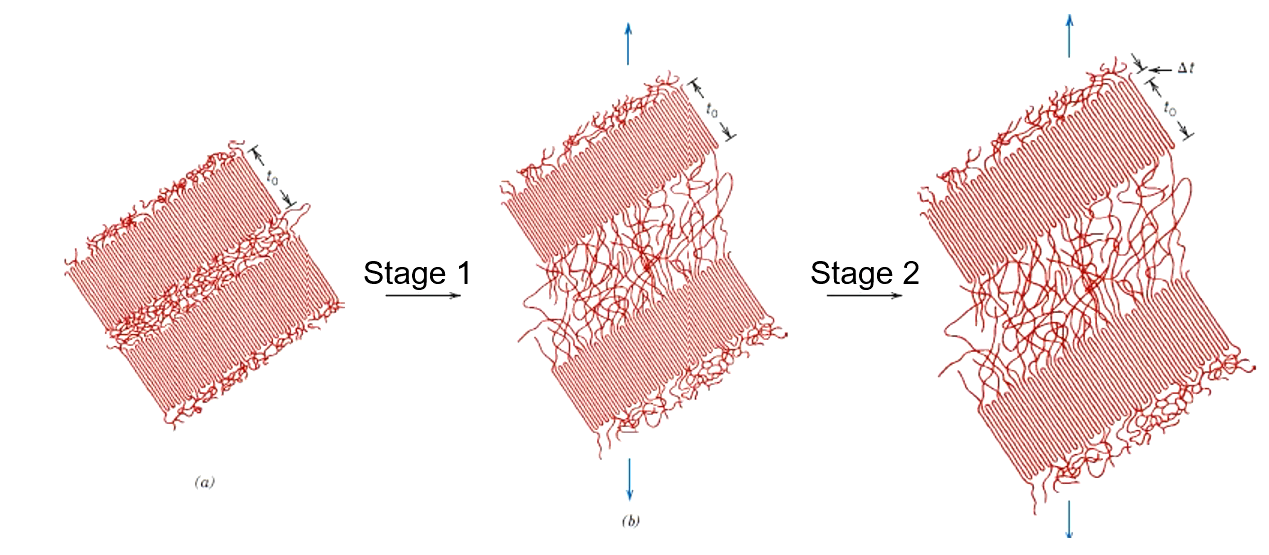
a
Chain molecules in amorphous regions elongate + align in direction of applied tensile stress
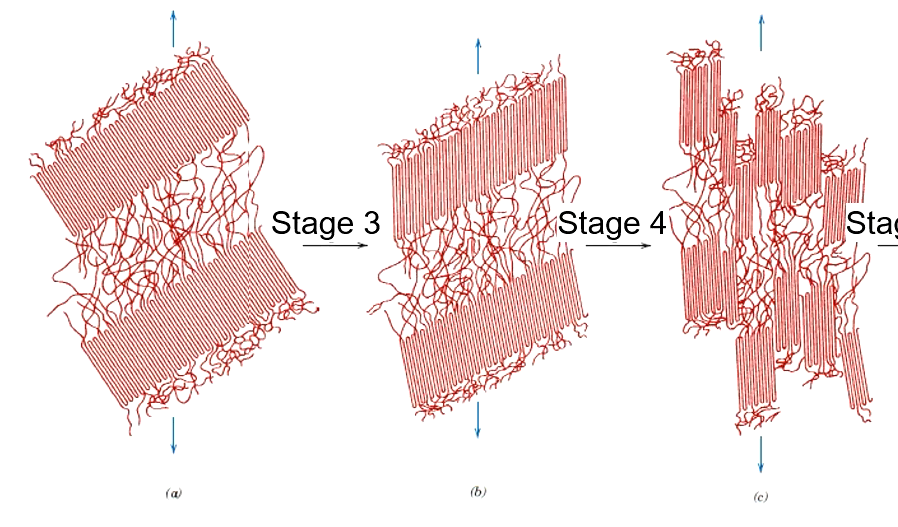
a
Block segments separate from lamellae + slide past one enother to orient w/ tensile axis
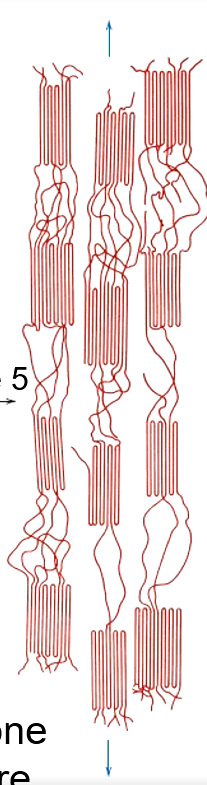
Blocks and tie chains become oriented in direction of tensile axis
Higher Degree of Crystallinity
Increased tensile strength and material becomes more brittle
Heat Treating
Increases % of crystallinity and size of crystallites
Carbon bonded to H, O, N, F
Backbone atoms of polymer molecules