chem 51c flashcards
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
bromination/chlorination of benzene
Br2/Cl2
—>
FeBr3
heat
add Br/Cl to benzene
Br2/Cl2
—>
FeBr3
heat
nitration of benzene
HNO3
—>
H2SO4
add NO2 to benzene
HNO3 + heat
—>
H2SO4
sulfonation of benzene with conc. H2SO4
oxygen of one H2SO4 deprotonates another H2SO4 with water leaving group
SO3H attaches to benzene
sulfonation of benzene with SO3 and H2SO4 (fuming sulfuric acid)
SO3 deprotonates H2SO4 with HSO4- byproduct
SO3H attaches to benzene
forward reaction of aromatic sulfonic acid
conc. fuming sulfuric acid
reverse reaction of aromatic sulfonic acid
heat in H2O or dilute sufuric acid
freidel crafts (F.C.) alkylation
benzene + R-Cl
AlCl3
—>
FC alkylation with 2 or 3 degree R group
form carbocation
benzene ring bonds with carbocation electrophile
AlCl4 equilibrium with Cl- + AlCl3
FC alkylation with primary R group
don’t form carbocation
Cl still bonded to Al
AlCl4 byproduct
carbocationic electrophile with alkene
H2SO4 —>
alkene deprotonates H2SO4
creates carbocation alkane and bonds with benzene
carbocationic electrophile with alcohol
H2SO4 —>
OH deprotonates H2SO4
H2O leaves, creating carbocation that bonds to benzene
FC alkylation carbocation rearrangement
primary electrophile - 35%
secondary/tertiary electrophile - 65%
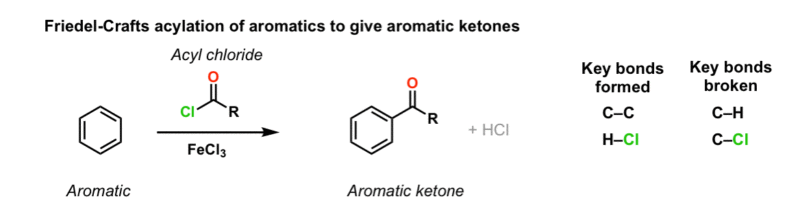
FC acylation with acid chloride
AlCl3
—>
H2O
acylium ion formed (H3C - C+ = O)
addition of acyl group to benzene
FC acylation with acid chloride
activators and ortho/para
if atom attached to ring has a lone pair
deactivators and meta
if arom attached to ring has a pi bond to an EN- atom (O or N) or positive charge
groups on benzene that are bad FC products
-COR, -N(CH3)3, —NO2, -CN, -CF3, -SO3H, -NH2
FC with NH2
aclylate the amine
CH3COCl, pyridine attach to N and then add the para sub. with AlCH3
1. H3O, H2O 2. NaOH to revert back to NH2
FC primary amine
NH2 group CH3COCl, pyridine —> HNROR group NHO3,H2SO4 —> add NO2 to para on benzene H3O,H2O,heat —> NH3+ group and NO2 group para NaOH—> NH2 and NO2 para
alkyl group on benzene to caboxylic acid
KMnO4, heat
NaOH, H2O
—>
H3O+
no reaction - b/c no H on the C attached to benzene
C(CH3)3 on benzene ring
KMnO4, heat
NaOH, H2O
—>
H3O+
NO2 group to NH2 group on benzene
H2
—>
Pd or Pt
NO2 to NH3+ to NH2 group
SN or Zn (Hg)
—>
HCl
then
NaOH
—>
wolff kishner reduction
basic conditions. reduces only ketone or aldehyde
Alcohols (can protonate/react)
Acetals (acid-labile)
Some alkenes
clemmenson reduction
acidic conditions. reduces only ketone or aldehyde
Esters (can hydrolyze in base)
Amides (can react under strong base)
Some fragile functional groups