Chapter 7: Alkyl Halides
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
alkyl halides
single bonds only-molecules containing a halogen atom bonded to a sp3 carbon atom
how are alkyl halides classified
primary-1 R group bonded a carbon
secondary- 2 R groups bonded to a carbon
tertiary- 3 R groups bonded to a carbon
what are the 4 halogens
F, Cl, Br, I
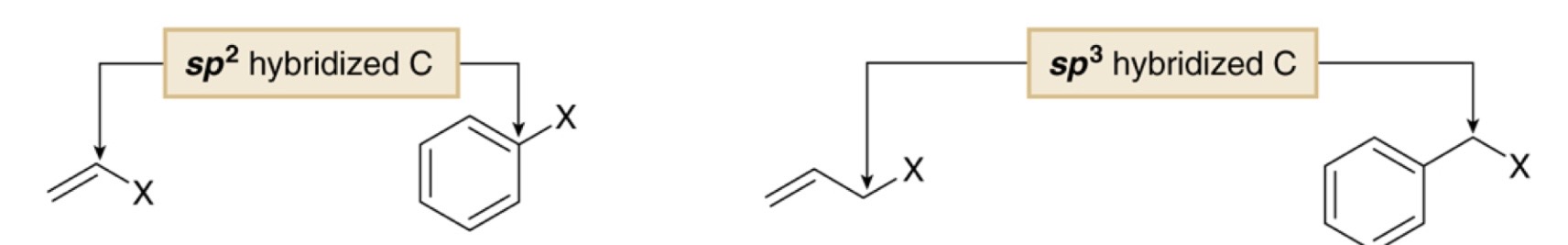
Match the types of halides: benzylic, aryl, allyic, vinyl
A= vinyl, B= aryl, C= allylic, D= benzylic
polarity increases what
boiling and melting point (alkyl halides are weakly polar/dipole-dipole interactions)
Chloromethane vs Dichloromethane vs Halomethane
chloromethane- produced by giant kelp and found in emissions from volcanoes
Dichloromethane- a solvent used to decaffeinate coffee
halomethane- general anesthetic
substitution vs elimination reactions
substitution- nucleophiles
elimination- bronsted-lowry bases
when a neutral nucleophile is used the substitution product bears what charge
positive (caused by a proton bonded to O or N)
what is a leaving group and what determines how strong
a leaving group is a halogen (F, Cl, Br, I)
the weaker the base the stronger the LG (I>Br>Cl>F)
what do nucleophiles and bases react with
nucleophile- reacts with C
bases- react with H and attack protons
basicity vs nucelophilicity
basicity- how readily an atom donates its electron pair to a proton(H)
nucleophilicity- how readily an atom donates its electron pair to other atoms
what makes a strong nucleophile
a strong base/a negative
(basicity increases right to left)
steric hindrance
decrease in reactivity from bulky groups
decreases nucleophilicity
how are Na+ cations and Br- anions solvated
Na+ cations- ion dipole interactions with H2O
Br- anions- strong Hydrogen bonds
Polar aprotic solvents
dipole-dipole interactions with no O-H or N-H bonding
first vs second order for nucleophilic substitution mechanisms
first order- depends on carboncation
second order-bond is broken as bond is formed
carbocation
three groups around C, trigonal planar, p orbital above and below
what happens to the rate of SN1 reaction as the number of R groups on the C with the leaving group. increases
SN1 reaction increases
how can you tell how stable a carbocation is
tertiary>secondary>primary>methyl