ch 18
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
frequency
waves (cycles) per second, Hz
v, nu
wavelength
λ
units depend on the part of the spectrum of interes
Ultraviolet-visible (UV-Vis)
usually in nanometers (nm) sometimes in angstroms (Å, 1 nm = 10 Å)
in a vacuum
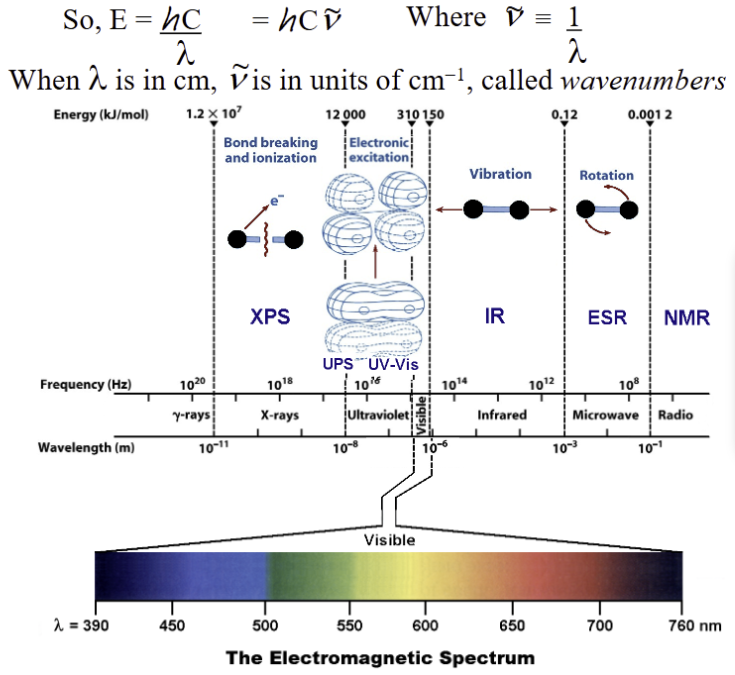
λ × ν = C
electromagnetic radiation variables in a vacuum
λ × ν = C
electromagnetic radiation variables in any medium
λ × ν = C/n where n is the refractive index of the medium
n > 1, so light slows down; λ is shorter but ν is constant in all media
Various wavelengths of light are used in analytical chem
The interaction of light with matter enables both qualitative and quantitative measurements
Spectroscopy can be classified based on types of energy and types of interactions
Energy of a photon
E = ℏν, where ℏ is Planck’s constant
Electronic spectroscopy
involves selection/detection of light
Basic parts of spectrometer
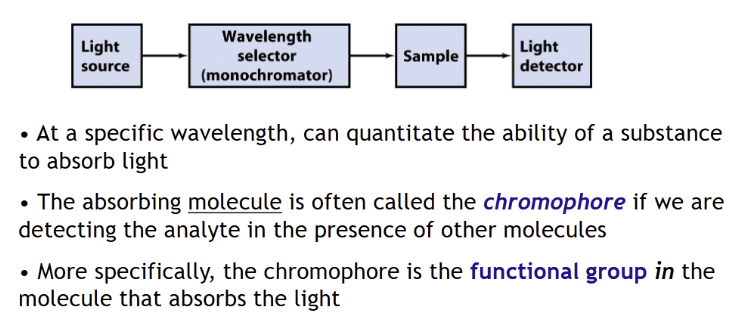
Analyze the light transmitted through a sample
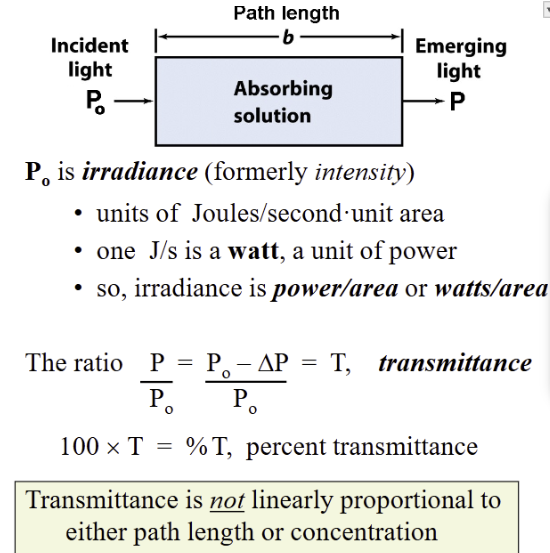
Consider a differential slice of a media with the variable
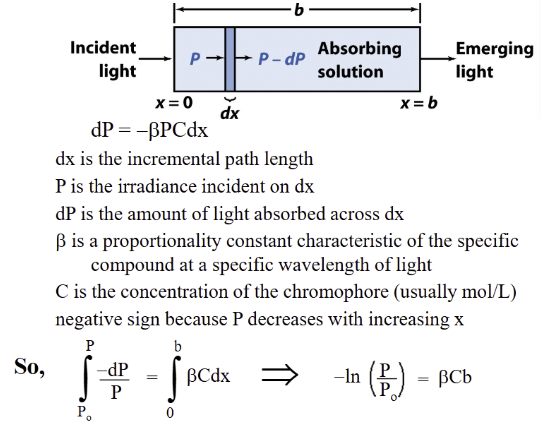
beer’s law

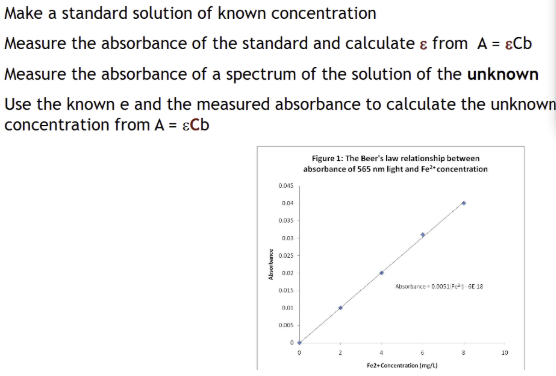
Calibration curves
give unknown concentrations
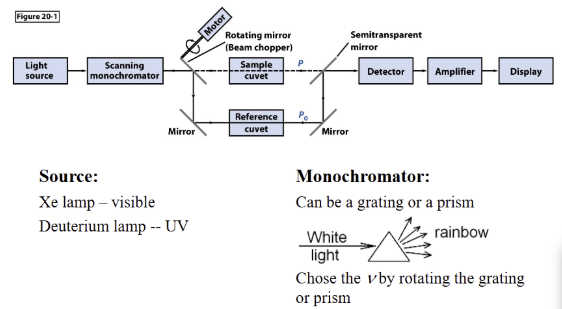
Dispersive Instruments
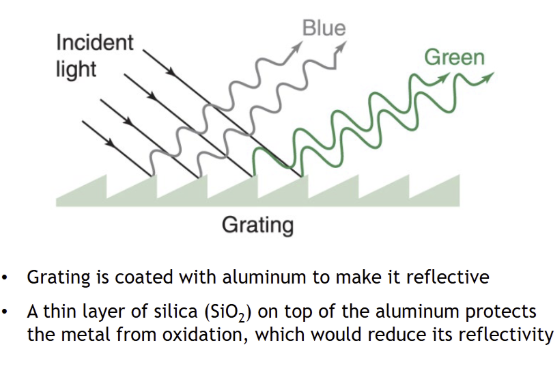
Diffraction gratings
help separate wavelengths of light
Important smaller components of spectrometer
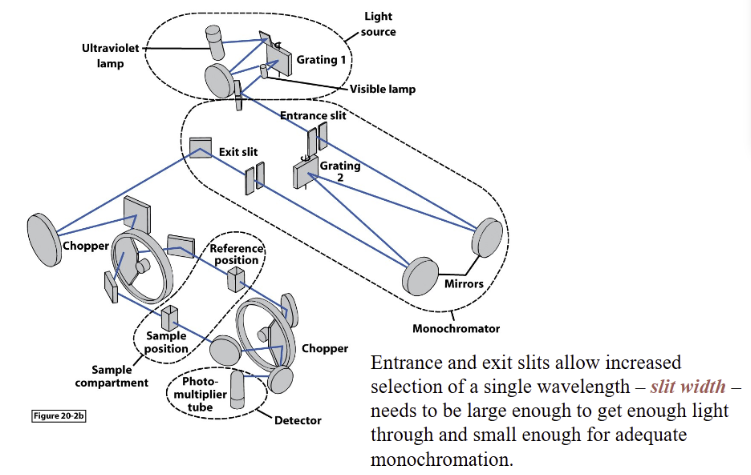
Single path instruments
require fewer parts, budget friendly
first run the solvent blank – then run the sample – the computer subtracts one from the other
photodiode array detector
photodiode array detector
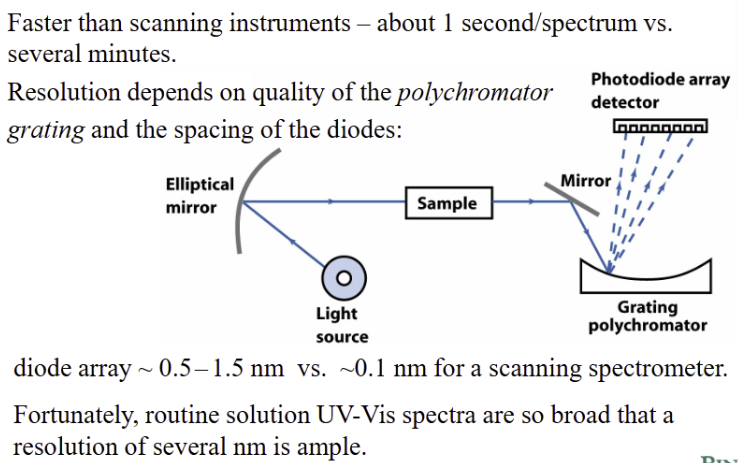
Spectrum can show multiple features depending on sample
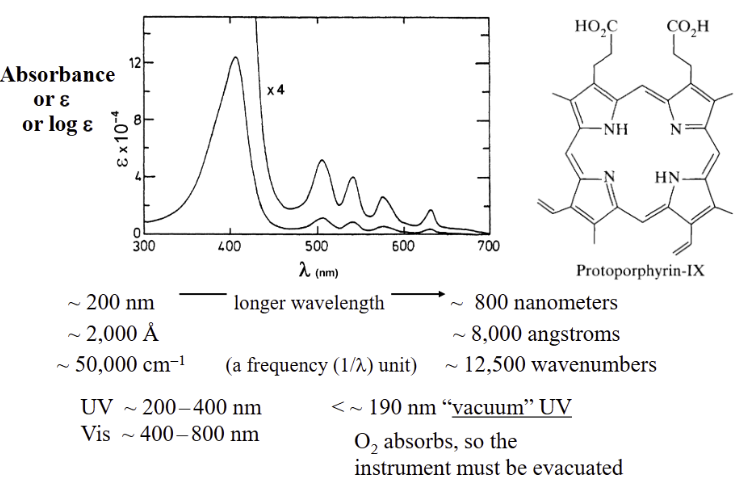
there are absorbed and observed colors
duh
Peak width related to how the compound reacts to light
as phase photon absorptions by single atoms cause transitions among quantized
electronic states with no vibrations and no rotations
∴ spectral lines are extremely sharp for atoms
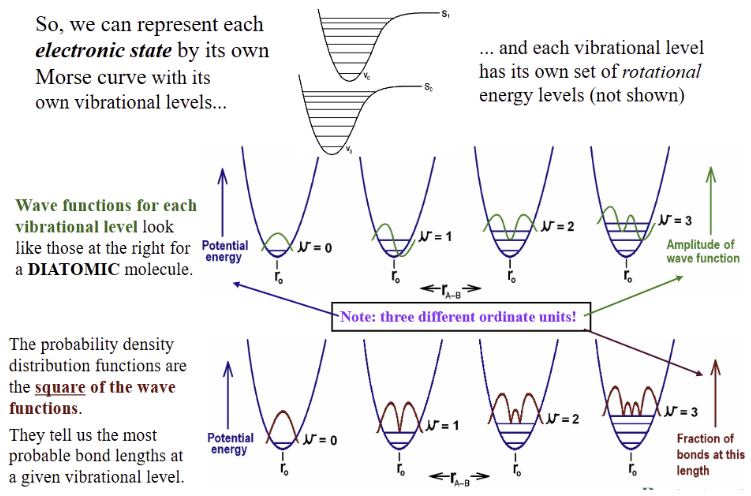
Molecules are more complicated. Each electronic state has a multitude of vibrational energy levels and rotational energy levels among which transitions can also occur
∴ spectral lines are generally very broad for molecules
Molecular orbitals across multiple atoms
Molecular orbitals delocalize across multiple atoms
molecular orbit is not confined to one atom, it’s a single wavelength spread over multiple atoms at once
on the molecule, there are omicron bonds (c-h, c-o), pi bonds (c=o) and nonbonding (n) electrons on oxygen
these combine into the molecular orbitals shown on right
pi orbital covers both c and o atoms, the electrons in this orbital are shared (delocalized) over both atoms, not sitting in just one
the omicron orbitals extend along bonds (c-h) (c-o), electron density is spread between atoms, not localized
the n orbital (non-bonding, on oxygen) is mostly centered on the oxygen atom (this is localized, not delocalized much)
when orbitals delocalize over multiple atoms, electrons can spread out, stabilizes bonding orbitals
When a molecule absorbs light it may change
geometry
S0 (ground state) - lowest energy state of the molecule, electrons occupy the lowest possible molecular orbitals, this is the molecules normal, stable state
S1 (first excited state) - a higher energy state reached after absorbing light, one electron has been promoted to a higher energy orbital, still a singlet - electron spins remain paired
slightly different shape because electron distribution changed
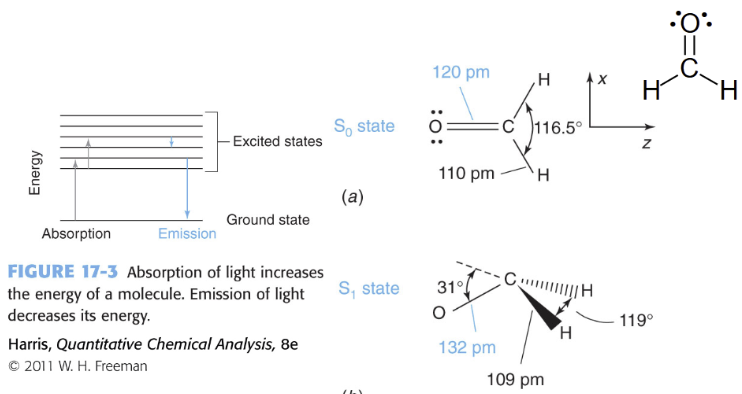
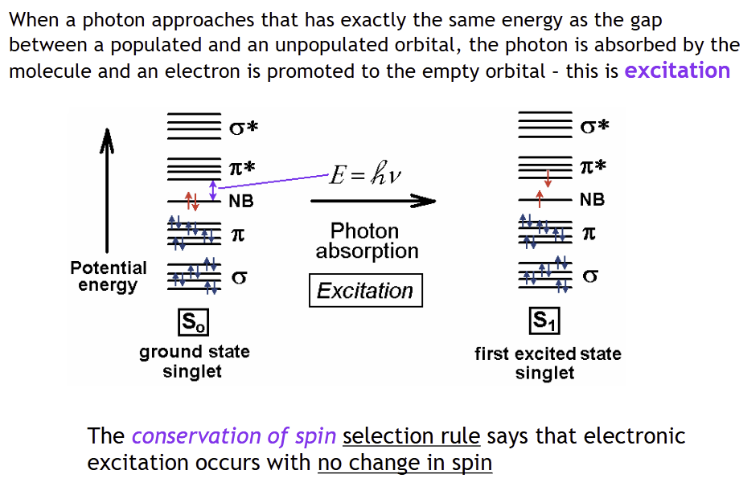
Photon is absorbed when
its energy matches the gap
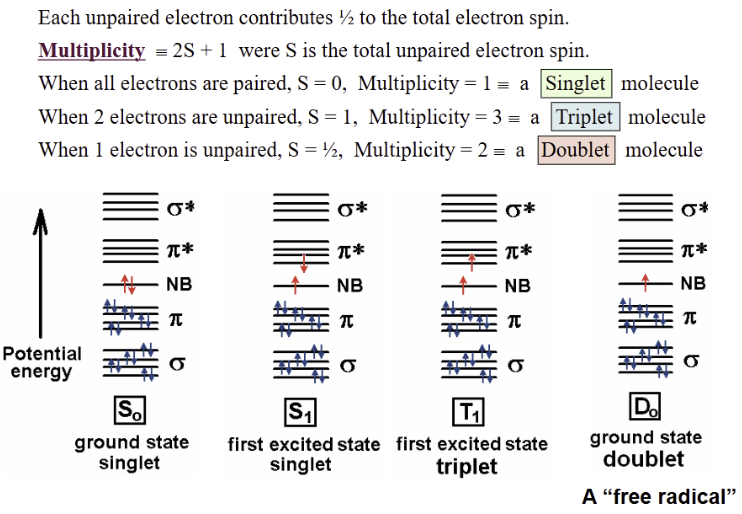
Single electrons in different orbitals can become unpaired
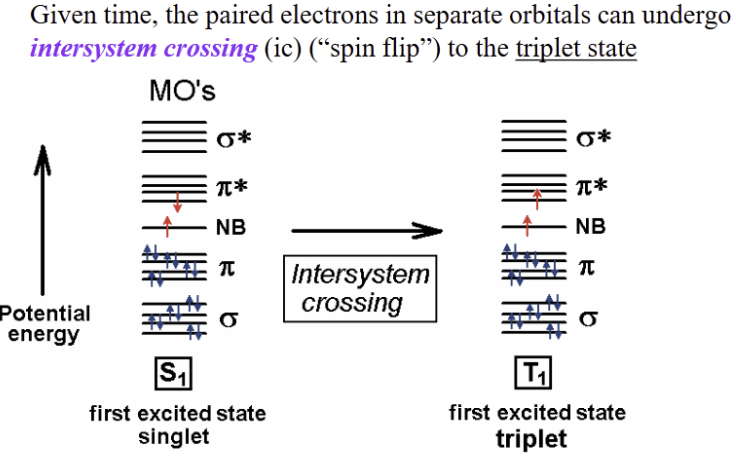
intersystem crossing
involves a change in the spin multiplicity
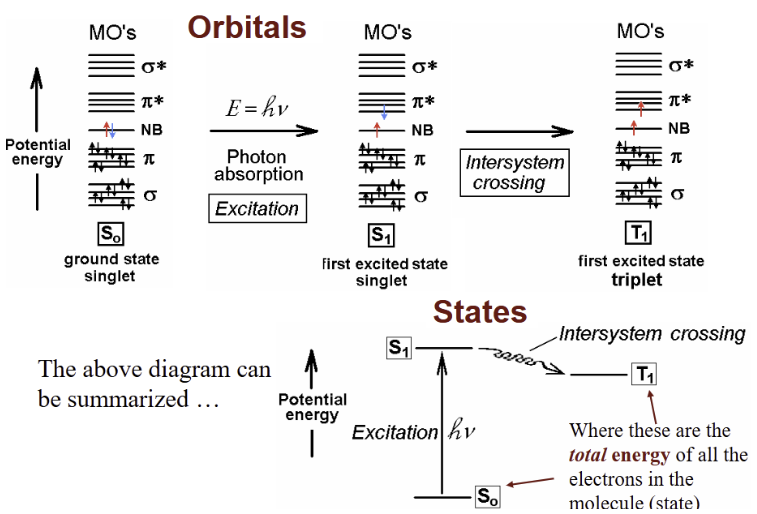
Summary of a typical absorption process
Some molecules have electron spin in the ground state
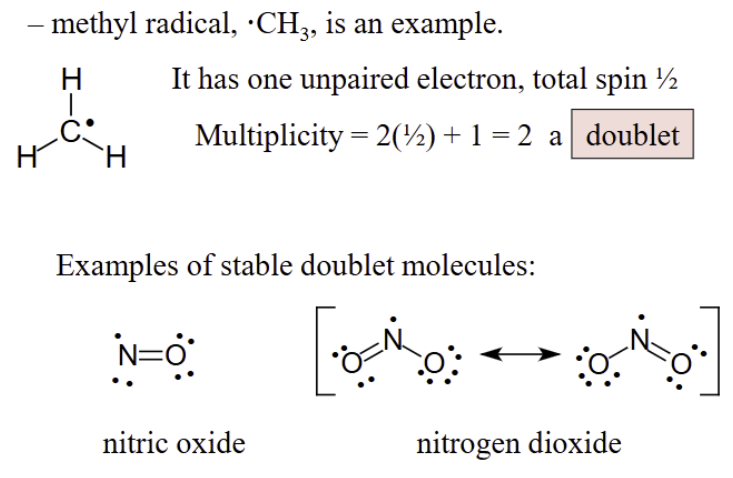
Most molecules have vibrations with varying symmetry

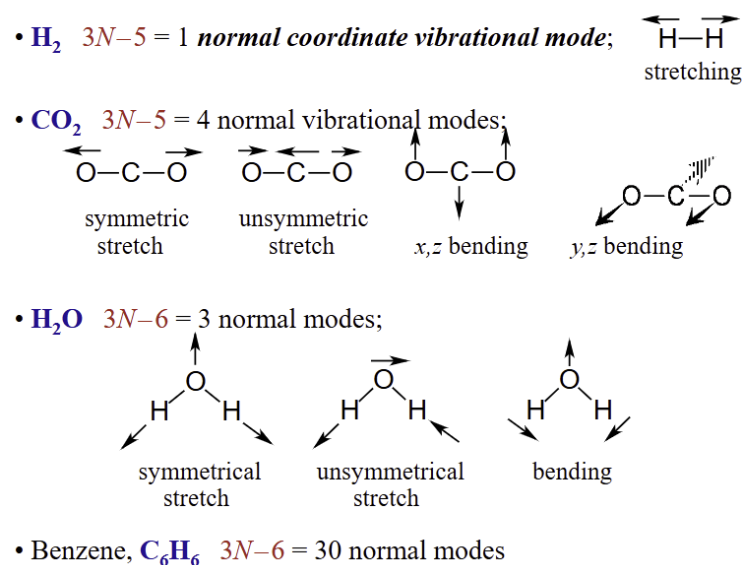
Counting potential vibrational modes

Vibrations in some simple molecules
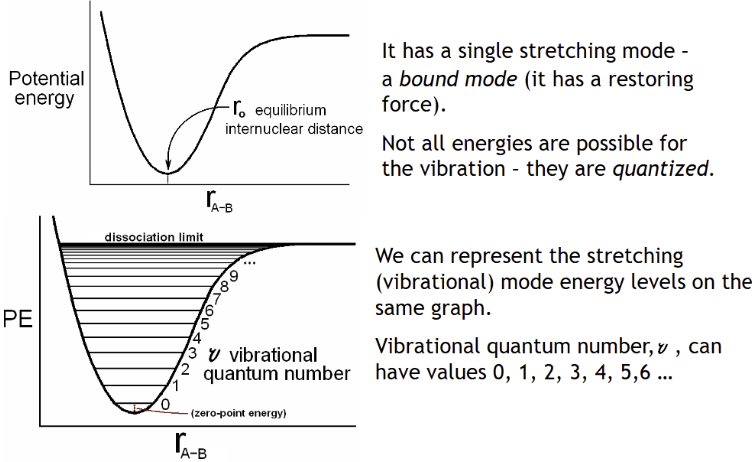
Morse Curve for a diatomic molecule, A–B
Morse Curve for benzene
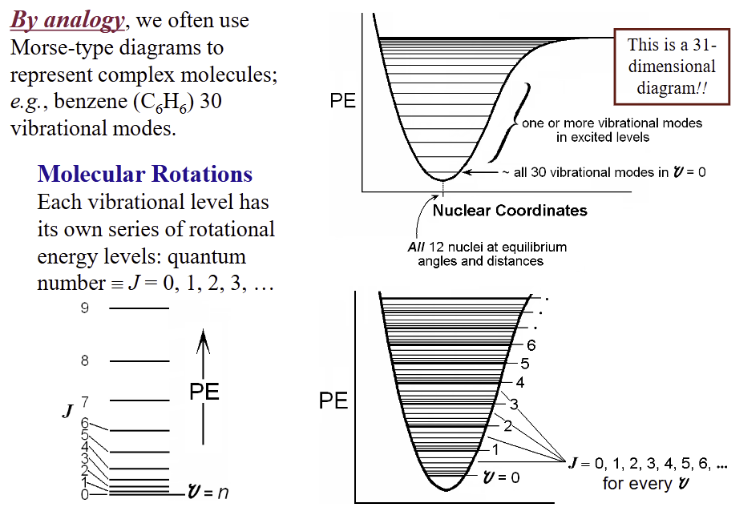
represent each electronic state by its own Morse curve
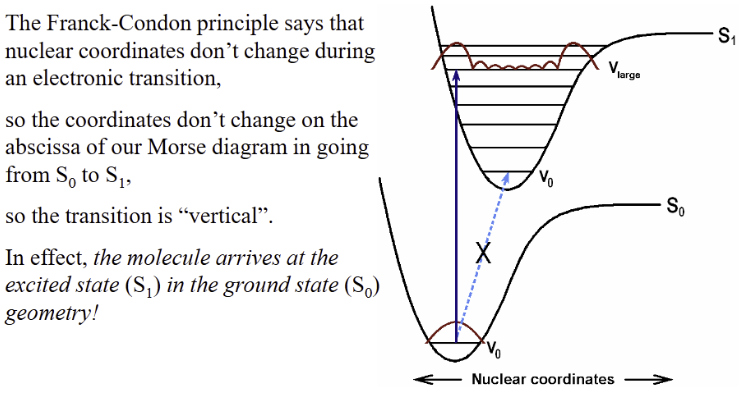
Franck-Codon Principle
Nuclei move much slower than electrons
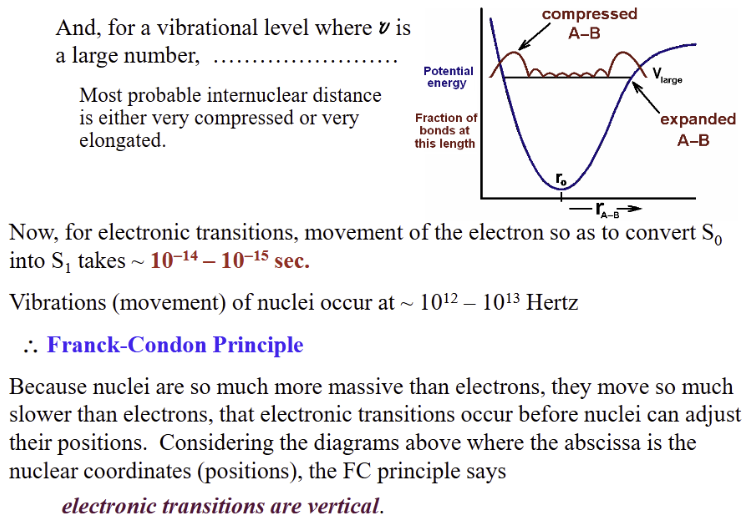
Nuclear positions don’t change during electronic transition