Quantum Numbers
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Electron configuration
When writing electron configuration, follow the order in which the arrows go (see image)
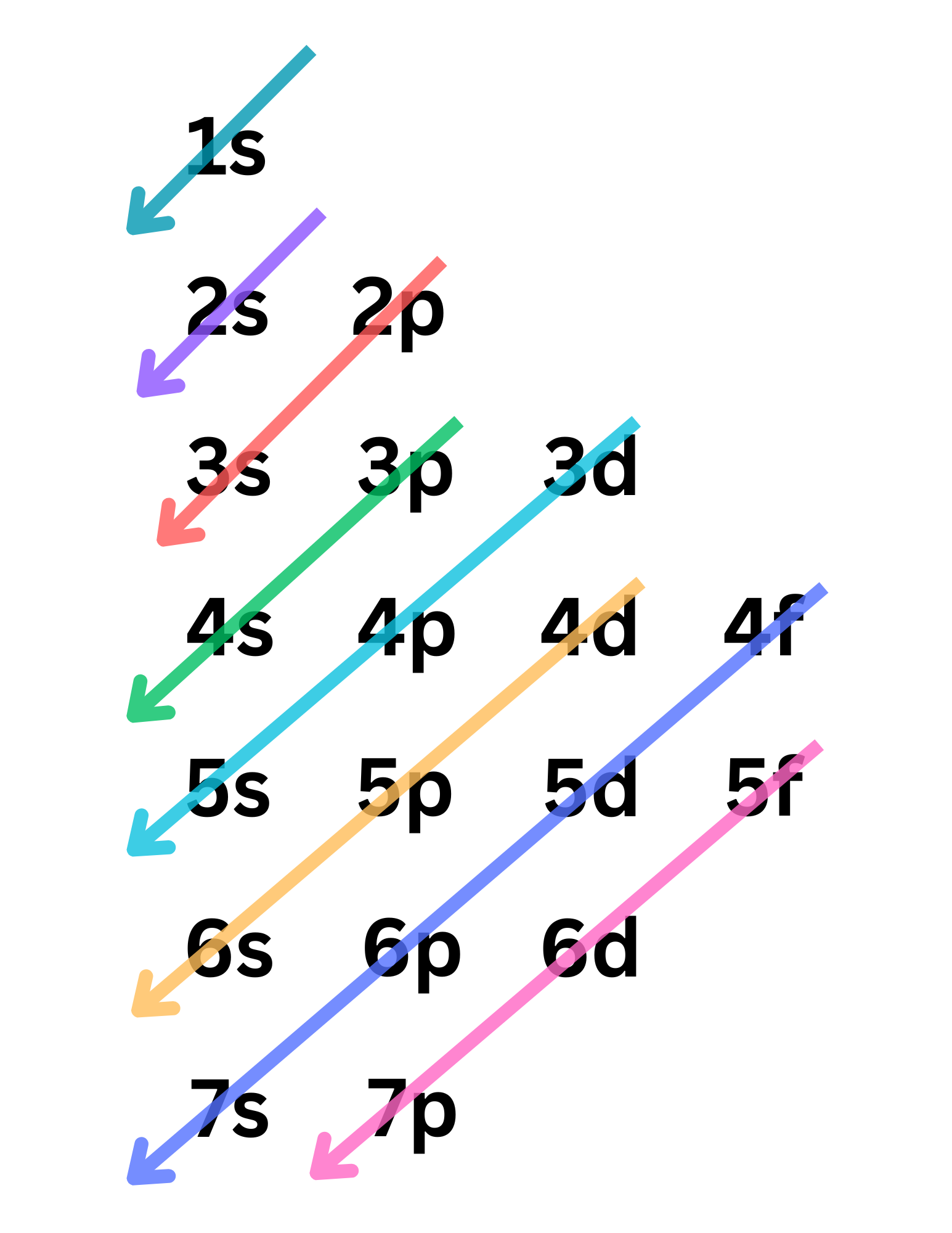
Quantum Numbers
There are 4 quantum numbers. Quantum numbers are like a "GPS coordinate" or a "home address" for an electron in an atom. They describe the location, energy, and behavior of electrons according to the laws of quantum mechanics
Principle Quantum Number
n. n = , 1, 2, 3, 4, 5, 6, 7 (any positive integer, nonzero value). This describes the size and energy level (shell) of the electron. As increases, the electron is farther from the nucleus and has higher energy. An n of 1 is the ground state (the shell closest to the nucleus)
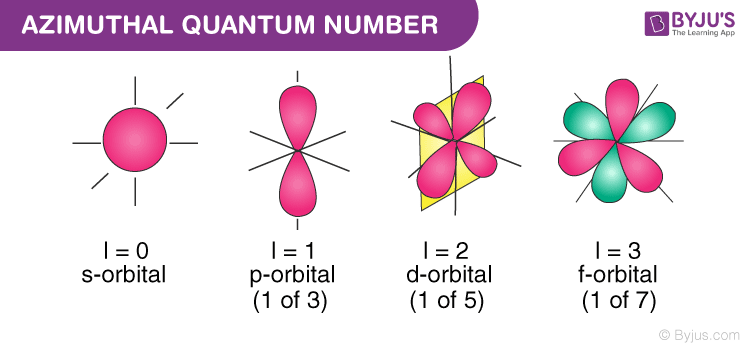
Angular Momentum Quantum Number
𝑙. (4):
s (sphere) (𝑙 = 0) (ground floor)
p (loop) (𝑙 = 1)
d (cross) (𝑙 = 2)
f (flower) (𝑙 = 3)
This defines the shape of the orbital. It tells us whether the electron is moving in a simple sphere or a more complex "dumbbell" shape. This number describes the shape of the sublevel within an energy level. 𝑙 can be zero or any positive integer, but not larger than (n-1).
𝑙 must be…
𝑙 must be less than or equal to n-1
EX 1: n = 1, 𝑙 must be 0.
EX 2: n = 2, 𝑙 = 0,1
EX 3: n = 3, 𝑙 = 0,1,2
Relationship between 𝑙 and n
when n is 1, we are in s sublevel (𝑙 = 0).
when n is 2, we can have s & p sublevel (𝑙 = 0,1).
when n is 3, we can have s, p and d sublevel (𝑙 = 0, 1, 2).
when n is 4, we have have the s, p,d or f sublevels ( 𝑙 = 0, 1, 2, 3).
Sublevel s, 𝑙 =
Sublevel s , 𝑙 = 0
EX: 1s, n = 1, 𝑙 = 0
Sublevel p, 𝑙 =
Sublevel p, 𝑙 = 1
EX: 2p, n =2, 𝑙 = 1
Sublevel d, 𝑙 =
Sublevel d, 𝑙 = 2
EX: 3d, n = 3, 𝑙 = 2
Sublevel f, 𝑙 =
Sublevel f, 𝑙 = 3
EX: 4f, n = 4, 𝑙 = 3
Magnetic Quantum Number
(m𝑙) This indicates the orientation of that orbital in space (for example, whether a -orbital is pointing along the x, y, or z axis). The The values for m𝑙 depend entirely on the 𝑙 value. It can be any integer from -𝑙 to +𝑙 (including zero).
If 𝑙 = 0, m𝑙 = 0 (1 orbital)
If 𝑙 = 1, m𝑙 = -1, 0, 1 (3 orbitals)
If 𝑙 = 2, m𝑙 = -2, -1, 0, 1, 2 (5 orbitals)
It’s called "magnetic" because of how these electrons behave in a magnetic field. If you place an atom in a strong magnetic field, the energy levels of these different orientations shift slightly. The number of possible values tells you how many orbitals of a certain shape exist in a subshell.
Relationship between 𝑙 and m𝑙
The number of possible values is calculated by the formula 2𝑙 + 1. This total tells you how many individual orbitals (orientations) exist within a specific subshell: (see image)
EX: 4f, n = 4, 𝑙 = 3, 𝑙 can vary between -3, -2, -1, 0, 1, 2, 3 (7 orbitals)

Sublevels and Orbitals
Every orbital can hold a MAXIMUM of 2 electrons (see image)
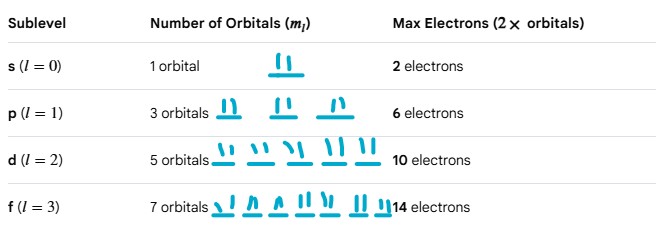
Sublevel s has ? orbitals
Sublevel s has 1 orbitals
Sublevel p has ? orbitals
Sublevel p has 3 orbitals
Sublevel d has ? orbitals
Sublevel d has 5 orbitals
Sublevel f has ? orbitals
Sublevel f has 7 orbitals.
EX: 4f, n = 4, 𝑙 = 3, 𝑙 can vary between -3, -2, -1, 0, 1, 2, 3 (7 orbitals)
Spin Quantum Number (Electron spin)
(ms) This describes the direction the electron is "spinning", one of two values, either "spin up" (+1/2) or "spin down" (-1/2).
If your electron goes up, your spin is + ½
- If your electron goes down, your spin is -1/2
Pauli Exclusion Principle
You can fit a MAXIMUM of 2 electrons in each orbital with an opposite spin.
EX: one up spin, one down spin max in each orbital.
Hand’s Rule
[Essentially: be fair first, then pair up] When you fill up your orbitals with spins, make sure to write at least one spin (going up) FIRST until all orbitals have one, THEN you can write a spin down and pair it with spin ups. (just make sure each orbital has one up BEFORE writing one spin down or vice versa).
3d8
n = 3, 𝑙 = 2. So -2, -1, 0, 1, 2. Write up arrows on all lines, once everyone has an up line, then start writing down lines, once you get to your exponent (8) whichever number it lands on is your m𝑙 (in this case it’s 0)(see image). Your ms would be -1/2, as that was your last line drawn.
What if you started with down spins instead of up spins? Well, your m𝑙 remains the same, but your ms changes, from -1/2 to now +1/2.
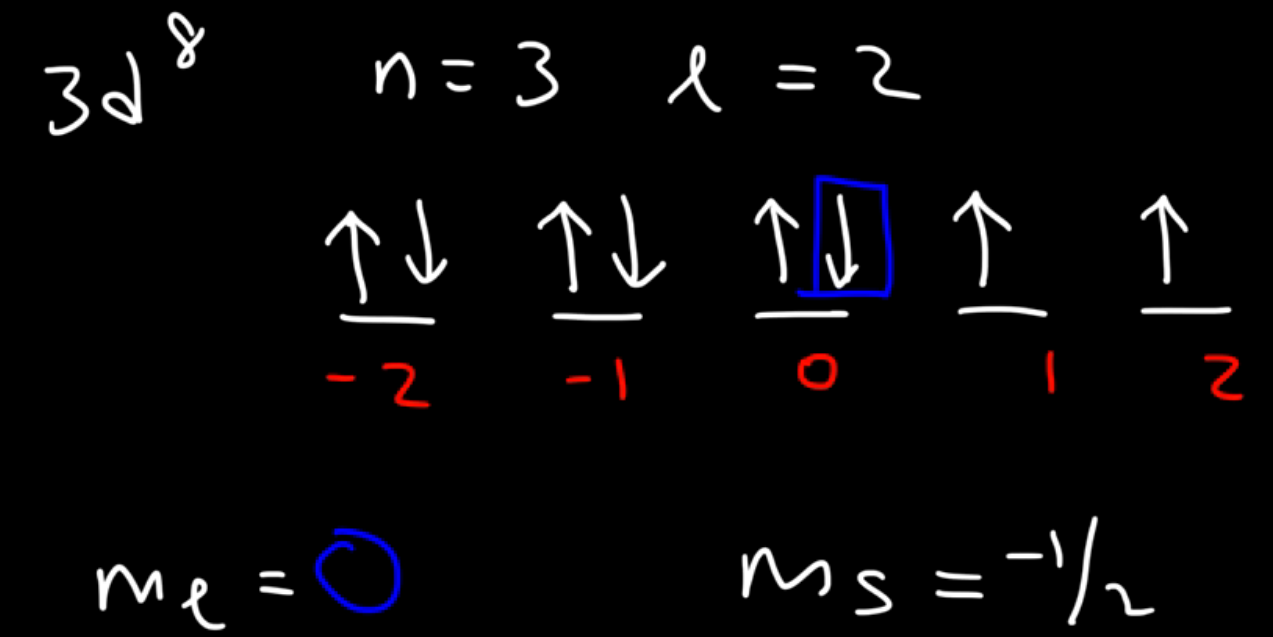
Paramagnetic
unpaired electrons
Diamagnetic
NO unpaired electrons
Valence Electrons
Outermost layer (only applies to main group elements).