Acids, bases & pH ✅
1/204
Earn XP
Description and Tags
200
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
205 Terms
3 types of bases
Metal oxides - NaO
Metal hydroxides - NaOH
Metal carbonates - Na₂CO₃
Neutralisation reaction
acid + base → salt + water
Can carry out titrations to find concentrations of acids and bases
Metal hydroxide + Acid
Metal oxide + Acid
Metal carbonate + Acid
Metal + Acid
Metal hydroxide + Acid → Salt + Water
Metal oxide + Acid → Salt + Water
Metal carbonate + Acid → Salt + Carbon dioxide + Water
Metal + Acid → Salt + Hydrogen gas
pH scale
1-6 Acidic
7 Neutral
8-12 Basic
Base vs Alkali
Alkali = A base that is soluble in water. All alkalis are bases
Bases = Insoluble in water
Are most hydroxides soluble or insoluble in water ?
Most hydroxides are SOLUBLE in water so are alkaline
E.g. the alkaline earth metals NaOH (aq)
Common acids, bases and alkalis
Acids: HCl, H₂SO₄, HNO₃
(Insoluble) Bases: Zn(OH)₂
(Soluble) Alkalis: NaOH, KOH, NH₃
What is a Bronsted-Lowry acid?
Proton donor
Releases H⁺ ions when mixed with H₂O
Which arrow sign should be put for weak acids and strong acids?
Weak acids partially dissociate so ⇌
Strong acids fully dissociate so →
How are protons in an aqueous solution represented ?
H₃O⁺
Hydronium ion
Concentration vs strength of acids
Concentration = The relative number of moles of acid in a given volume of water
Strength = How much the H⁺ dissociate
What is a Bronsted-Lowry base ?
Proton acceptor
Can H₂O act as a base?
Yes
What happens in a neutralisation reaction ?
● Acid donates a proton. HNO₃(aq) → H⁺(aq) + NO₃⁻(aq)
● Base accepts a proton, which then reacts with OH to form H₂O. KOH(aq) + H⁺(aq)→ K⁺(aq) + H₂O(l)
HNO₃ + KOH →
How are strong and weak acids/ bases different?
And so where does the equilibrium lie?
(check dis)
Strong acids/bases COMPLETELY dissociate/ ionise in aqueous solutions to release H⁺ ions
Weak acids/bases only SLIGHTLY dissociate/ ionise to release H⁺ ions
Name weak acids
Ethanoic acid CH₃COOH
Phosphoric acid H₃PO₄
and other carboxylic acids
Where does the equilibrium lie with weak acids?
Towards the LEFT so the backwards reaction is favoured so not many H⁺ is produced
Name strong acids
HCl hydrochloric acid
H₂SO₄ sulphuric acid
HNO₃ nitric acid
Where does the equilibrium lie with strong acids?
Towards the RIGHT so the forward reaction is favoured so lots of H⁺ is produced
Name Strong Bases
Group 1 metal hydroxides
NaOH, LiOH, KOH, Ca(OH)₂, Ba(OH)₂
Where does the equilibrium lie with strong bases ?
Towards the RIGHT so the forward reaction is favoured so lots of OH⁻ is produced
Name some weak bases
Ammonia (NH₃), Amines
Where does the equilibrium lie with weak bases?
Towards the LEFT so the backwards reaction is favoured so not many OH⁻ ions are produced
When acids and bases react with water, they form a ______________ reaction
Reversible
What ion is created when H+ ions react with water ?
H₃O⁺
Hydronium ion
Give the 2 equations for when water dissociates.
H₂O (l) ⇌ H⁺ (aq) + OH⁻ (aq)
Acid base equilibrium
H₂O (l) + H₂O (l) ⇌ H₃O⁺ (aq) + OH⁻ (aq)
The 1st water acts as a base as it accepts a proton. The 2nd water acts as an acid as it donates a proton.
Conjugate acid
Each acid is linked to a conjugate base on the other side of the equation
Base + H⁺ ⇌ Conjugate acid
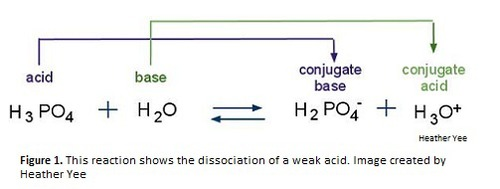
Conjugate base
Each base is linked to a conjugate acid on the other side of the equation
Acid ⇌ Conjugate base + H⁺
What is the conjugate acid of OH⁻ ?
OH⁻ + H⁺ ⇌ H₂O
OH⁻ and H₂O are called a conjugate acid-base pair
What is the conjugate base of HF ?
F⁻
HF ⇌ F⁻ + H⁺
HF and F⁻ are called a conjugate acid-base pair
Whenever a substance acts as a Brønsted-Lowry base, we call the product it forms its...
Conjugate acid
Whenever a substance acts as a Brønsted-Lowry acid, we call the product it forms its...
Conjugate base
Conjugate pairs according to Brønsted-Lowry model
Every acid-base reaction has 2 conjugate pairs
NH₃ (aq) + H₂O (l) ⇌ NH₄⁺ (aq) + OH⁻ (aq)
Conjugate pairs: (NH₃ and NH₄⁺) & (H₂O and OH⁻)
What happens when 2 acids are mixed together?
The stronger acid will act as a Brønsted-Lowry acid, donating a proton
The weaker acid will act as a Brønsted-Lowry base, accepting a proton
Complete the equation for HCl (aq) + CH₃COOH (aq)
HCl (aq) + CH₃COOH (aq) ⇌ Cl⁻ (aq) + CH₃C(OH)₂⁺ (aq)
The HCl is a stronger acid so acts as a proton donor
The ethanoic acid is a weaker acid so acts as a proton acceptor
H₂SO₄ (aq) + CH₃COOH (aq) → HSO₄⁻ (aq) + CH₃C(OH)₂⁺ (aq)
Identify the acid and base according to the Brønsted-Lowry model.
Brønsted-Lowry acid: H₂SO₄
Brønsted-Lowry base: CH₃COOH
Identify the acid and base according to the Brønsted-Lowry model.
NH₃ + NH₃ → NH₄⁺ + NH₂⁻
Give your answers as compound names, rather than chemical formulae.
Brønsted-Lowry acid: ammonia 1
Brønsted-Lowry base: ammonia 2
Ammonium ions can be used as a weak acid in organic reactions. Write an equation for the dissociation of this acid.
NH₄⁺ ⇌ H⁺ + NH₃
Consider the following acid-base reaction:
C₅H₅N + HCl → C₅H₅NH⁺ + Cl⁻
What are the conjugate pairs ?
Conjugate acid = HCl, Conjugate base = Cl⁻
Conjugate acid = C₅H₅NH⁺, Conjugate base = C₅H₅N
What is a monoprotic acid ? Give 3 examples
What is a monoprotic acid ? Give 3 examples
● An acid that donates ONE H⁺ ion for every acid molecule.
● So the concentration of the H⁺ ions is equal to the concentration of the acid HCl, HNO₃, HBr
What do square brackets represent? [ ]
Concentration
What is a diprotic acid ?
● An acid that donates 2 H⁺ ions for every acid molecule.
● So The concentration of H⁺ ions is 2x the concentration of the acid [H⁺]= 2[Acid]
E.g. H₂SO₄
How do diprotic acids dissociate? Use H₂SO₄ as an example.
First: H₂SO₄ → H⁺ + HSO₄⁻
Second: HSO₄⁻ ⇌ H⁺ + SO₄²⁻
Dissociates 1 proton at a time, and the 1st proton dissociates more fully than the 2nd
For 0.1 mol dm⁻³ HCl, what is the pH ?
-log₁₀[0.1] = 1.00
Acidity
The higher the concentration of H⁺ the higher the acidity
Define pH
-log₁₀[H⁺]
Where [H⁺] is the concentration of hydrogen ions in the solution.
How much should pH be rounded ?
ALWAYS to 2 d.p
2.00
11.67
How to find H⁺ ion concentration
[H⁺] = 10⁻ᵖʰ
What is the concentration of H+ ions in a solution with a pH of 3.5?
[H⁺] = 10⁻³.⁵
[H⁺] = 3.16 x 10⁻⁴
What do we -log the concentration with strong monoprotic acids to find the pH ?
Conc of the acid = the conc of H⁺ ions
-why? Because we assume it dissociates FULLY
How do you calculate the concentration of H⁺ ions when you know the pH?
Rearrange the pH equation
[H⁺]= 10⁻ᵖʰ
DONT FORGET THIS IS Inverse log (antilog), PRESS SHIFT THEN THE LOG BUTTON
Rearrange the pH equation
[H⁺]= 10⁻ᵖʰ
DONT FORGET THIS IS Inverse log (antilog), PRESS SHIFT THEN THE LOG BUTTON
When the pH increases by 1...
a tenfold difference in [H⁺]
E.g. pH 2 has 10x the H⁺ concentration of pH 3.
How much more acidic is pH 2 than pH 6 ?
10,000
Why is a logarithmic scale used for pH ?
The concentration of hydrogen ions in aqueous solution covers a very wide range.
What is the log of...
a) 100
b) 1,000,000
c) 10,000
d) 0.1
c) 0.001
a) 2
b) 6
c) 4
d) -1
e) -3
If y = 10ˣ
log(y) = x
Log(x) = 3
What is x equal to?
10³
= 1000
Log(a) = 1.2
What is a equal to?
10^1.2
= 15.8
Log(b) = -3.6
What is b equal to?
10^-36
= 2.51 x 10^-4
If the concentration of HNO₃ (aq) is 0.03 mol dm⁻³, then the concentration of H+ ions in the solution is...
0.03 mol dm⁻³
How to find pH of a solution that has been diluted with water?
Given Volume and Conc of acid.
● Find moles of acid
● Find moles of (strong) acid which is = moles of [H⁺]
● Find new volume (volume of acid + volume of water added)
● The number of [H⁺] stays the same, so find conc by doing moles of [H⁺] / new volume
● Find pH using -log₁₀[H⁺]
What happens to the pH and the volume of the solution if we add a solid base to a solution of acid?
pH increases
Volume stays the same (really small amount of water produced is negligible)
A student has 60mL of a perchloric acid (HClO₄) solution with concentration of 0.36mol dm⁻³. They add 50mL of a sodium hydroxide (NaOH) solution with a concentration of 0.25mol dm⁻³ to the acidic solution. The following reaction occurs:
HClO₄ (aq) + NaOH (aq) → NaClO₄ (aq) + H₂O (l)
What is the new pH of the solution after the reaction?
[H⁺] = [HBr]
Moles of H⁺ ions at start: 0.36 x 0.06 = 0.0216
Moles NaOH = 0.05 x 0.25 = 0.0125
Moles used = 0.0216 - 0.0125 = 0.0091
C = n / v
C = 0.0091 / 0.11 (the new volume)
C = 0.0827
pH = -log₁₀(0.0827)
pH = 1.08
A student has 50mL of a solution of hydrochloric acid (HCl), with a concentration of 5×10⁻³ mol dm⁻³.
How much water do they need to add to the HCl solution to raise its pH to 3?
Moles of H⁺ in initial solution:
0.05 x 0.005 = 0.00025
Calculate the concentration of H⁺ ions needed to get a pH of 3
[H⁺] = 10⁻ᵖʰ
[H⁺] = 10⁻³
[H⁺] = 0.001 mol dm⁻³
V = 0.00025 / 0.001
V = 0.25 dm³ = 250 mL
250 - 50 = 200 mL
Why is the concentration of water said to be a CONSTANT value ?
It only dissociates SLIGHTLY (equilibrium lies to the LEFT) so there are very little OH⁻ and H⁺ ions compared to the number of water molecules
What can be said about strong acids and the concentration of [H⁺] ions?
Strong acids fully dissociates so the concentration of the acid is equal to the concentration of [H⁺] ions
What is Kw? Expression and units
The ionic product of water, eqm lies to left as water hardly dissociates; so much water that it has a 'constant value'
Kw = [H⁺] [OH⁻]
Units mol²dm⁻⁶
How can you manipulate the Kw equation if using pure water?
Why does this work?
Kw = [H⁺]²
Because OH⁻ and H⁺ concentrations are equal
[H⁺] = [OH⁻]
How to find pH of water
[H+] = √Kw
pH = -log [H⁺]
Find the pH of pure water
Kw = [H⁺] [OH⁻]
Kw = [H⁺]²
[H⁺] = √Kw
[H⁺] 1 x 10⁻¹⁴ = 1 x 10⁻⁷ so pH = 7
What is the value of Kw at room temp ?
Always 1.00 X 10⁻¹⁴ mol²dm⁻⁶
at 25 degrees/ 298 K
At a given temp, Kw always has __________________ value in a solution
The SAME
Can the value of Kw change as the TEMP changes?
YES!
Why doesn't Kw include concentration of water?
[H₂O] is constant
[H₂O] is very high compared to [OH⁻] and [H⁺]
How can we predict the change in pH of pure water at different temperatures ?
● Use Le Chatelier's principle to predict the change in pH of pure water at different temperatures
● The dissociation of water is endothermic so increasing the temperature would push the equilibrium to the right
● Giving a bigger concentration of H⁺ ions and a lower pH
At 25°C, the Kw of pure water is 1.008×10⁻¹⁴ mol² dm⁻⁶.
Use the equation Kw = [H⁺]² to calculate the concentration of hydrogen ions in pure water at this temperature and the pH.
Kw = [H⁺]²
[H⁺] = √Kw
[H⁺] = √1.008×10⁻¹⁴
[H⁺] =1.004 x 10⁻⁷
pH = -log₁₀[1.004 x 10⁻⁷] = 7.00
Explain why the value of Kw increases as temperature increases
● Forward reaction is endothermic,
● Position of eqm shifts right to decrease temp so conc of H⁺ and OH⁻ increases
● So pH decreases, Kw increases ( Kw = [H⁺]² )
Give the effect of Kw if there is a temp increase/decrease for an endo/exo reaction
Endothermic reaction
● Temp increase = Kw increase
● Temp decrease = Kw decrease
Exothermic reaction
● Temp increase = Kw decrease
● Temp decrease = Kw increase
Why is the water still neutral ?
Even if pH decreases (due to temp ↑, forwards reaction endo, shifts right)
Equal conc of H⁺ and OH⁻ so is neutral
[H⁺] = [OH⁻]
What effect does changing concentration on a reactant or product have on Kw ?
No effect
● E.g. adding H⁺ ions to pure water will not affect Kw as position of eqm shifts to left decrease H⁺ ion conc, and Kw is the product pf H⁺ and OH⁻.
● H⁺ increases and OH⁻ decreases
If [H⁺] = 0.4 mol dm⁻³ and
Kw = 1.01×10⁻¹⁴ mol² dm⁻⁶,
what is [OH⁻] ?
b) Is this solution acidic, alkaline or neutral?
Kw = [H⁺] [OH⁻]
1.01×10⁻¹⁴ = [0.4] [OH⁻]
Rearrange to get OH⁻ = 2.53 x 10⁻¹⁴ mol dm⁻³
b) Solution is acidic as greater conc of H⁺ than OH⁻
What is the pH change if 20 cm³ of 0.1 mol dm⁻³ HCl has 30 cm³ water added ?
Strong acid fully ionises so [H⁺] = 0.1 mol dm⁻³
Starting pH: pH = -log₁₀[0.1] = 1.00
Adding water does not change moles HCl
n = c x v
n = 0.1 x 0.02 = 0.002
c = n / v
c = 0.002 / 0.05 = 0.04
Diluted pH = -log₁₀[0.03] = 1.40
pH change = 1.40 - 1.00 = 0.40
What do you have to do when calculating pH of a diprotic acid?
Multiply the concentration of the acid by 2
What happens when a small amount of sodium hydroxide is added to pure water?
● The concentration of hydroxide ions increases
● The concentration of hydrogen ions decreases
● The equilibrium constant of water dissociation stays constant
How can we work out the pH of a strong base ?
You use Kw!
Because Kw = 1.00 X 10⁻¹⁴ and Kw = [H⁺][OH⁻]
So, 1.00 X 10⁻¹⁴ = [H⁺][OH⁻]
You can use the pH given to work out the concentration of H⁺ then plug it into the equation,
[H⁺] = 10⁻ᵖʰ
Rearrange it to work out the concentration of OH⁻ ions
CONCENTRATION OF OH⁻ IONS = CONCENTRATION OF BASE (if monobasic - if dibasic multiply by 2)
Give the factors affecting the pH of a solution
● Amount of dissociation
● Solubility
● Conc of H⁺ ions
● Temperature
Group 2 hydroxides
Ba(OH)₂
Solubility of group 2 hydroxides
Solubility increases down the group, Ba(OH)₂ fully dissociates and is a strong base. Mg(OH)₂ is hardly soluble but the bit that does FULLY dissociates so it's still a strong base.
Be
Mg
Ca
Sr
Ba
Why does the conc of OH⁻ ions = the conc of the strong base ?
Because strong bases dissociate to produce 1 MOLE of OH⁻ ions for EVERY MOLE OF BASE
Which equilibrium constants always have the same units ?
Kw
Ka
What is Ka? What is it used for ? What does [HA] mean ?
● The acid dissociation constant for WEAK acids (same as Kc but for weak acids)
● is used to work out the pH of weak acids and bases
● Temperature dependent
● [HA]= conc of the weak acid
Kₐ = [H⁺] [A⁻] / [HA]
Write the expression for the acid dissociation constant (Ka) of methanoic acid.
Ka = [H⁺] [HCOO⁻] / [HCOOH]
What does a greater Ka indicate ?
● Higher Ka = Stronger the acid
● Because the acid dissociates into ions to a greater extent
● Eqm further to the right
How to calculate the pH of a weak acid
[H⁺] = √(Ka x [HA])
Find pH using -log₁₀[H⁺]
We assume [HA] is equal to initial [HA]
[HA] is the acid molecules