Practice Test: Acids, Bases, Redox, & Electro
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms

What type of reaction does Cu+ → Cu2+ represent?
Oxidation
In an electrochemical cell, what is the cathode?
Electrode at which matter gains electrons.
What is a species whose oxidation number increases in a reaction?
Oxidized

In the reaction X(aq) + H2O(l) → X(aq) + OH-(aq), what is X?
Conjugate acid
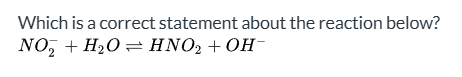
In the reaction NO2- + H2O ⇌ HNO2 + OH-, what is H2O classified as?
Acid
What is a property of bases?
They have a slippery feel.
Which statement about buffers is false?
A buffer is a solution that maintains a constant pH of 7.
What is an Arrhenius base?
One that increases the concentration of hydroxide ions in an aqueous solution.
What is the name for H2SO3?
Sulfurous Acid
When the name of an anion that is part of an acid ends in -ate, what is the suffix of the acid name?
-ic
Which represents a Bronsted-Lowry conjugate acid-base pair?
HNO2 and NO2-
What is the sum of oxidation numbers of the atoms in a polyatomic ion?
Is always the same as the ion's charge.
What is reduction?
Gain of electrons.
What is the pH of a solution with a pH of 4?
Acidic
Which is a diprotic acid?
H2SO4
What is the oxidation number for each atom in NaOH?
Na = +1, O = -2, H = +1
What is formed when a Bronsted-Lowry acid has given up its proton?
Conjugate base.
What is an oxidation number?
A value assigned to atoms and ions to keep track of electrons.
Where does oxidation take place in an electrochemical cell?
At the anode.
How does a weak acid dissociate in water, and give an example?
Partially; HNO2.
What is a solution with equal H+ ions and OH- ions considered?
Neutral.
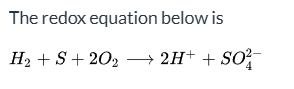
What is the redox equation H2 + S + 2O2 → 2H+ + SO4^2- classified as?
Correctly balanced.
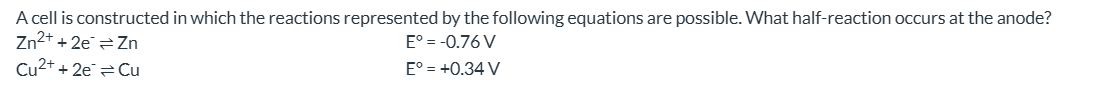
What half-reaction occurs at the anode in a cell with Zn2+/Zn and Cu2+/Cu?
Zn → Zn2+ + 2e-.
What is a titration?
The process of finding an unknown acid or base concentration by using a known acid or base concentration.
If an atom is oxidized in a redox reaction, what must happen to another atom?
It must be reduced.

In the reaction X(aq) + H2O(l) → X*(aq) + OH-(aq), what could X most likely be?
HCl.
Which statement is not true for oxidation numbers?
O will usually have an oxidation number of +2.
What is the oxidation number of chlorine in KCl?
-1.
What is the formula for phosphoric acid?
H3PO4.
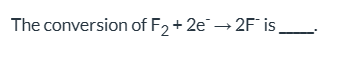
What is the conversion of F2 + 2e- → 2F- classified as?
Reduction.