Electrode potential and electrochemical cells
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is an oxidising agent
Electron acceptor
What is a reducing agent
Electron donor
What are electrode potentials
A value that indicates how readily a substance gains electrons
What is an electrochemical series
List of electrode potentials for half equations in numerical order
What is the role of the voltmeter in a cell
Measures potential difference (EMF)
I.e the difference in electrode potentials for the 2 half cells
Role of the salt bridge
Maintain electrical neutrality/complete the circuit
Does this because it has mobile ions
What can be used for the salt bridge and why
Potassium nitrate solution
Allows the movement of ions
And will not react with chemicals in the half cells
What conditions are standard electrode potentials measured under?
298k
1moldm-3 (conc for solutions)
100kPa (for gases)
When is a platinum electrode used and why?
When you have a cell that doesn’t contain solid metals (as electrodes)
Platinum provides a solid surface for electron transfer
And is inert (unreactive)
Equation to workout Ecell/EMF
Larger electrode potential - Smaller electrode potential
General formula for conventional representation for a cell
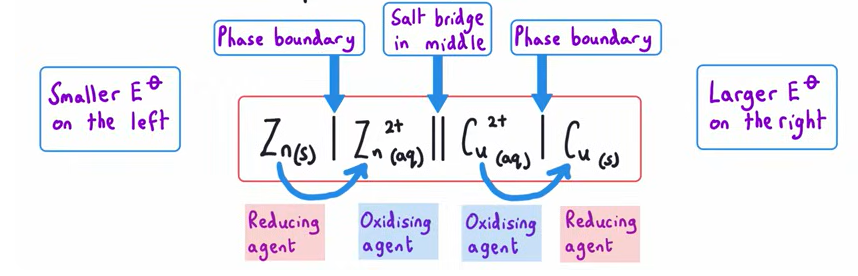
Why is a standard hydrogen electrode used ?
Used to measure unknown electrode potentials
What side of the cell representation is the standard hydrogen electrode always on?
LEFT
Equation for standard hydrogen electrode

What can be used for H+ source in SHE
1 moldm-3 solution of
HCL or HNO3
The potential of other half cells is compared to the potential of
SHE which has a potential of 0 V
What can cause the value of electrode potentials to change ?
Changing the conditions
Any change that causes the eqm to shift right (e.g increasing conc of reactants) will increase the electrode potential
Any change causing eqm to shift left will decrease electrode potential
Applying a change to what conditions changes electrode potential
concentration
Pressure (if gas is involved)
In an electrochemical cell when EMF is measured with a voltmeter…
no current is flowing
What happens when you replace a voltmeter with a lamp in a cell?
Electric current will flow through the circuit
EMF will decrease
3 types of cells
Non rechargeable
Rechargeable
Fuel
What happens in non rechargeable cells?
1 or both cell reactions cannot be reversed
Pros and cons of non-rechargeable cells
Pros
Cheaper to produce and buy
Cons
Single use - Thus use up finite resources
Can end up in landfill and pollute the environment by releasing toxic substances
What are rechargeable cells
Cells where electrode reactions can be reversed by applying a reverse potential
Pros and cons of rechargeable cells
Pros
Minimise waste going to landfills, therefore reduce pollution
Cons
More expensive
Applying a reverse potential requires electricity→ CO2 released from power stations → which causes global warming
Example of a rechargeable cell
Lithium ion cell
What reaction occur at the positive electrode of a lithium ion cell
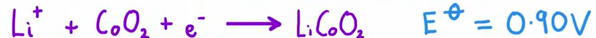
What reaction occurs at the negative electrode of a lithium ion cell

Overall reaction occurring in lithium ion cell?
Li + CoO2 → LiCoO2
How do fuel cells work?
Fuel cells are used to generate an electric current
Reactants for the fuel cell can be continuously supplied
Therefore fuel cells do not need to be electrically recharged
Meaning the concentration for the reactants remain constant
(EMF will not change with use)
Why are liquid fuels preferred to gas fuels in fuel cells?
Liquid fuels can be stored and transported more easily than gases
What happens in hydrogen-oxygen fuel cell
Converts hydrogen(fuel) and oxygen(air) into water
Which releases electrical energy in the process
What are the 2 main conditions for hydrogen oxygen fuel cells
Acid
Alkaline
Depends on the electrolyte used
What is the overall reaction in hydrogen-oxygen fuel cell
2H2 + O2→ 2H2O
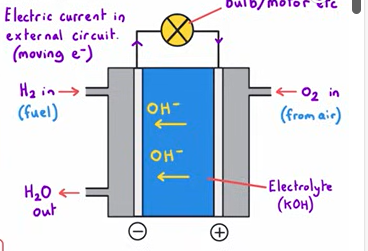
Explain how a hydrogen oxygen fuel cell works (alkaline)
2 electrodes separated by an electrolyte (KOH)
Each reactant is introduced from either side of the cell
Water is removed
Hydrogen is oxidised at the negative electrode - by reacting with OH from electrolyte, releasing electrons
H2+2OH- →2H2O + 2e-
These electrons travel through the external circuit and arrive at the positive electrode
Where oxygen gains the electrons at the positive electrode and reacts with water (from the electrolyte)
O2 +2H2O +4e- → 4OH-
Hydroxide ions produced move through the electrolyte to complete the circuit
Benefits and risks of a hydrogen-oxygen fuel cell?
(Essentially the benefits of producing electricity this way?)
Water is the only product /no greenhouse gases
Efficient compared to combustion
Risks
Generating hydrogen requires reacting methane with steam which produces CO2
Difficult to store and transport hydrogen gas
electrosde practical set up
How do u get a current to flow in a cell
Replace voltmeter with bulb