CELL DEATH ALL
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
Explain what homeostasis is
ongoing maintenance of physical and chemical conditions in a cell
What is hyperplasia
What is mannosidosis
What are the four most frequently affected cellular components by injury?
dna
ribosomes
phospholipid bilayer
mitochondria
name 7 things that can cause cell injury
infectious agents
genetic derangements
immunological reactions
oxygen deficiency
toxins, drugs, chemicals
physical agents (force)
nutritional imbalances
What is hypoxia, what causes this
deficiency of oxygen
reduced blood flow, severe blood loss, inadequate oxygenation of blood due to cardiorespiritory failure
How does hypoxia cause cell injury ?
low o2 → less atp made → Na/K channels close → Na accumulates in cell → messed up osmolarity → possible cell lysis due to osmosis of water into cell
Name two types of genetic derangements and mechanisms of action
Chromosomal abnormalities
Base pair substitution
alter proteins
enzymes, structural proteins, receptors
What is oxidative stress?
accumulation of free radicals
reactive oxygen species, unstable and bind to any molecule
important targets are lipids, proteins, dna.
important in the action of drugs, radiation, again and normal metabolism.
What are the consequences of loss of calcium homeostasis, what causes this?
injurious agents cause increased Ca+ into cells
increased cytosolic Ca+ activates enzymes
ATPase = decreased ATP
phospholipase = decreased phospholipids
endonuclease = nuclear chromatin damage
protease = disruption of membrane and cytoskeletal proteins
What are cellular adaptation, what can cause these?
reversible changes in size, number, phenotype metabolic activity or functions of cells.
can have physiological or pathological causes
eg stress, injury
What is atrophy, what can cause this
reduction in size / number of cells, occurring after organ development
disuse, denervation, reduced perfusion, poor nutrition, bad endocrine stimulation, senility, pressure.
What are the mechanism of atrophy
reduced protein synthesis, increased protein degradation, reduction in cellular organelles and cytoskeletal proteins, apoptosis.
What is involution
reduction in size and number of cells, after organ development. physiologic atrophy
what is hypoplasia
reduction in size and number of cells, during organ development
what is aplasia
absence of an organ during organ development, with primordium present.
what is agenesis
absence of an organ during organ development due to lack of primordium
what is hypertrophy
increase in size of cells
no cell division
no new cells
often accompanied by hyperplasia
when limit is reached, degeneration occurs.
what causes hypertrophy, what are the mechanisms of this?
increased functional demand
skeletal muscle
endocrine cells
mechanisms
increased cellular organelles
increased synthesis of growth factor
What is hyperplasia, what caused this, what is the mechanism?
increase in no of cells, only in cells that can divide. increased mitotic activity.
cells proliferate in response to hormones / growth factors
can be caues by inflammation, physical trauma
give examples of hormonal hyperplasia and compensatory hypoplasia
hormonal -
eg proliferation or mammary and uterine epithelium during pregnancy
compensatory -
with partial loss of parenchyma
in symmetrical organs with functional loss of one organ.
List three triggers for pathological hyperplasia
excessive hormone production or effect of growth factors, eg cystic endometrial hyperplasia
attempt of regeneration, eg nodular hyperplasia in spleen, liver, pancreas
with viral infections eg papilloma
What is metaplasia?
one differentiated cell type replaced by another.
What are potential causes of metaplasia?
reprogramming of local tissue stem cells
colonisation from other cell populations
usually a response to chronic irritation
Name three common intracellular accumulations
1) normal cellular components
eg water, lipids, proteins, carbs
2) abnormal substances eg:
a) exogenous minerals, products of infectious agents
b) endogenous, due to abnormal synthesis / metabolism
3) pigments
List three ways intracellular accumulations occur.
normal / increased production with inadequate removal of endogenous substance
genetic defects causing abnormal substances or storage
abnormal exogenous substance eg carbon/ sillica
How do accumulations of lipids occur?
steatosis / lipidosis (fatty change)
accumulation of triglycerides withing parenchymal cells of liver
caused by toxins, protein malnutrition, diabetes, obesity, anorexia.
What gross discolouration does lipid accumulation cause?
diffuse yellow
What does a protein accumulation look like, and what causes this?
round, eosinophilic droplets / vacuoles
caused by :
excesses of protein presented to cell or excessive synthesis
defects in protein folding
Explain in detail how protein accumulations occur in cells.
a) reabsorption of droplets in proximal renal tubules
proteinuria → tubular epithelium : pinocytosis → phagolysosomes → hyaline droplets
b) synthesis of excessive amount of normal secretory protein
Immunoglobulins → distension of ER → increased Russel bodies in plasma cells
Name some causes of defects in protein folding and what this causes intracellularly
genetic mutations
aging
environmental factors
TSEs
amyloidosis
causes defective intracellular transport and secretion of critical proteins, toxicity of aggregated abnormally folded proteins.
What causes intracellular glycogen accumulation
diabetes mellitus
glucocorticoid hepatopathy
glycogen storage diseases
What are some normal and abnormal pigment accumulations
normal
melanin
abnormal
exogenous - carbon, coal (anthracosis), tattoo ink
endogenous - lipofuscin
Describe two endogenous pigments that can accumulate
Lipofuscin
insoluble, polymers of lipids/phospholipids, not injurious, yellow-brown.
Melanin
brown-black, found in melanocytes, non-haemoglobin derived
Haemosiderin
Heamoglobin derrived, gold-yellow-brown
storage form of iron
When is haemosiderin present, and what are the different forms it can take?
from haemorrhage - degradation of haemoglobin.
Heme - red-blue → heamosidering (yellow, brown, gold)
Biliverdin (green)
Bilirubin (green / blue)
Where is bilirubin found, what does an excess cause?
bile, excess causes jaundice.
What are the three causes of jaundice (icterus)
pre-hepatic
excessive haemolysis
Hepatic
severe hepatic damage
Post-hepatic
bile obstruction
What is amyloid
pathological proteinaceous substance deposited between cells (i.e. extracellular)
Describe briefly the tissues amyloidosis effects and its components
affects various tissues and organs
uniform morphology but chemically variable
95% fibril proteins
5% P component and other glycoproteins
What are the four chemicals in amyloid?
give a brief description
AA (amyloid associated)
synthesised in liver, no Ig, derived from serum amyloid A (SAA)
AL (amyloid light chain
- derived from plasma cells, contains Ig light chains, associated with b cell proliferation
b-amyloid protein
deposited in spongiform encephalopathies
islet amyloid polypeptide
deposited in pancreatic islets
What is systemic amyloidosis
deposition in several organs
What is an example of localised amyloidosis
deposition in cutaneous plasmacytomas
What is primary amyloidosis
deposition with an immunologic disorder
eg multiple myeloma
What is hereditary / familial amyloidosis
genetic deposition of amyloid
What is an example of endocrine amyloidosis
deposition in the islet cells
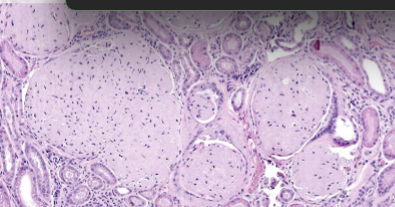
What is the stain for amyloid?
Congo red
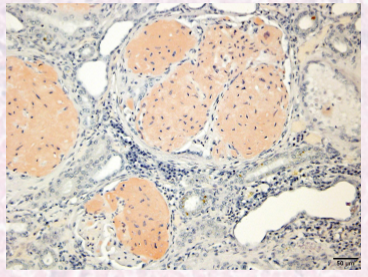
What does amyloidosis look like grossly?
yellow organ discolouration
What is pathologic calcification, name two types.
abnormal deposition of calcium salts
dystrophic calcification
metastatic calcification
Describe the pathogenesis of dystrophic calcification
initiation can occur via :
intracellular : Ca accumulates in mitochondria of dying cells
extracellular : phospholipids in membrane bound vesicles in dying cells
propagation :
membrane facilitated calcification
Ca binds to phospholipids in membrane
forms groups
leads to Ca deposition near membrane
microcrystal formation leads to membrane perforation.
Describe the pathogenesis of metastatic calcification
Occurs in normal tissues with hypercalcaemia
hypercalcaemia can occur due to :
increased PTH secretion
destruction of bone tissue
vitamin D intoxication
renal failure (due to secondary hypothyroidism)
Affects interstitial tissues in lungs, arteries, veins, kidneys, gastric mucosa
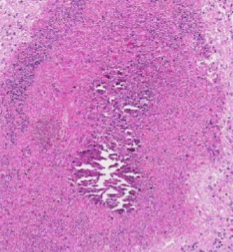
What stain do you use to visualise calcification on a microscope slide?
Von kossa - black.
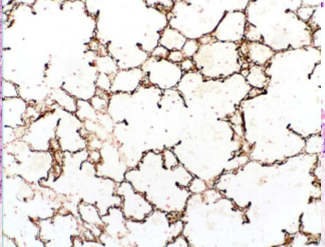
What two things can occur due to irreversible cell injury?
necrosis
apoptosis
What is necrosis?
Irreversible cell death leading to denaturation of proteins, leakage of cellular contents.
Why is necrosis irreversible?
inability to reverse mitochondrial dysfunction
disturbances in membrane function
What are the four mechanisms of necrosis
damaged cell membrane
protein denaturation
local inflammation (DAMP)
enzymatic digestion of necrotic cells.
What is meant by the terms autolysis and heterolysis?
autolysis : destruction of a cell through the action of its own enzymes
heterolysis : spontaneous death and disintegration of a cell from factors other than itself
Describe the cellular morphology of necrosis
increased cellular eosinophilia
nuclear shrinkage + increased basophilia (pyknosis)
fragmentation of nucleus (karyorrhexis)
loss of nuclear basophilia (karyolysis)
What is the gross morphology of necrosis
pale tan discolouration
Name 7 types of necrosis.
coagulative
lytic
caseous
liquefactive
steatonecrosis (adipose tissue)
gangrenous
fibrinoid
describe coagulative necrosis
protein denaturation
retention of cellular borders
ischemia
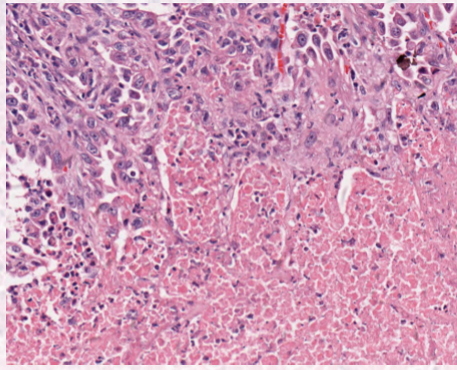
describe lytic necrosis
enzymatic digestion
loss of cell borders
neutrophil rich inflammation (bacterial)
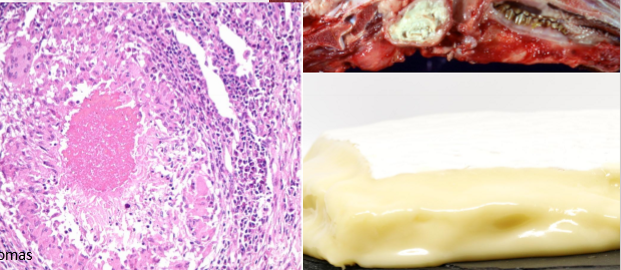
Describe caseous necrosis
subtype of lytic necrosis
mycobacterial granulomas
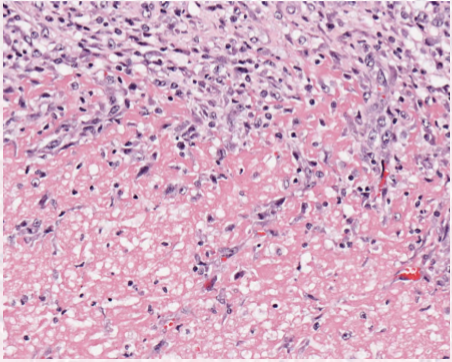
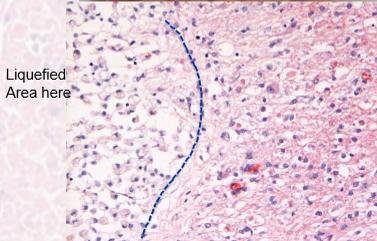
describe liquefactive necrosis
subtype of lytic necrosis
end stage = fluid filled lesion.
final stages of necrosis in brain
describe steatonecrosis
necrosis of adipose tissues, between lytic and coagulative.
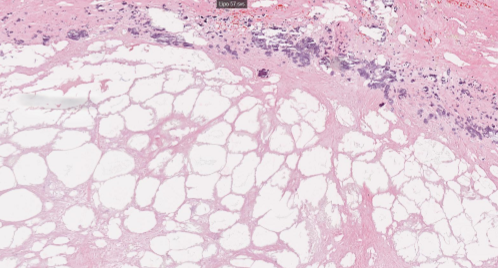
Describe gangrenous necrosis
subtype of lytic
occurs in distal limbs or dependant portions of organs
dry = vascular damage
wet = bacterial proliferation
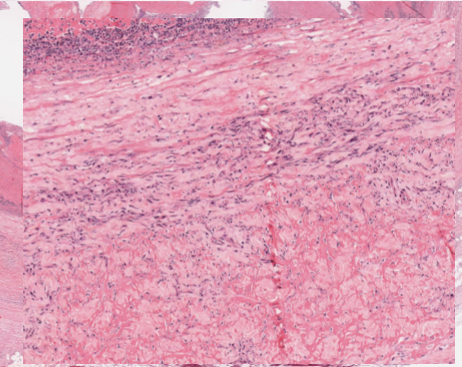
describe fibrinoid necrosis
specific form of lytic necrosis, occurs during vascular damage
intramural () accumulation of fibrin, other proteins and immunocomplexes in vessels ( associated with vasculitis)
What is apoptosis?
regulated cell death.
What enzyme mediates apoptosis
describe what happens when this is activated
Caspases
protein containing cystine in their active site, cleave proteins after aspartic residues
Endonuclease activation = DNA fragmentation
Breakdown of cytoskeleton
cytoplasmic buds form → apoptotic body → eaten by phagocyte.
List 5 causes of apoptosis
DNA damage (intrinsic)
accumulation of misfolded proteins (intrinsic)
pathogenic infection (via cytotoxic t cells)
tumour necrosis ligand factor TNF1 receptor - death domain. (extrinsic)
normal homeostasis
What are the intrinsic mechanisms of apoptosis (describe in detail)
intrinsic = mitochondrial pathway.
withdrawal from growth factors, hormones.
causes pro-apoptotic molecules eg cytochrome C to activate executioner caspases.
injury due to radiation, toxins, free radicals
causes DNA damage → P53 → mitochondria → cytochrome c → executioner caspases
Describe in detail the extrinsic mechanism of apoptosis
Via cytotoxic T lymphocytes
bind to surface and activate granzyme B → executioner caspases.
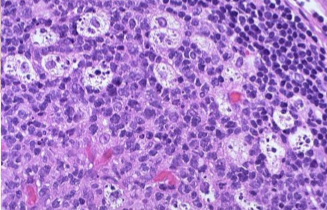
Describe morphology of apoptosis at a cellular level
Cell shrinkage and eosinophilia
chromatin condensation
formation of apoptotic bodies
phagocytosis of apoptotic bodies
What is the role of apoptosis inn homeostasis
programmed cell death during embryogenesis
cell depletion in proliferating tissues (turnover)
cell death in tumours
lymphocyte selection during immune system maturation