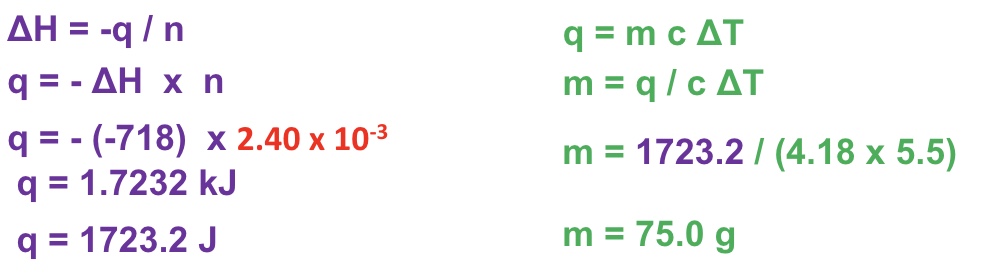3.1.4.2 calorimetry
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
what is calorimetry used to do?
to find the heat energy released in a reaction
explain how enthalpy of combustion can be found experimentally (ie explain how calorimetry works)
by burning a known mass of substance + using the heat energy released to increase the temperature of a known mass of water → the aim is to measure change in temp (△T)
after using calorimetry to find △T, how can you then find the heat energy transferred to the water?
by using the equation: q = m x c x △T
(q = mc△T)
give the units for the equation q = mc△T
q → the heat energy (Joules, J)
m → the mass of water (grams, g)
c → specific heat capacity (4.18 J K⁻¹ g⁻¹)
△T → change in temperature (℃ or K)
what is 1g of water in cm³?
1g of water = 1cm³ of water (the density of water is 1gcm⁻³)
the value of q can then be used to calculate what?
△H → enthalpy change
give the equation to calculate △H from q
△H = ∓q / n
(enthalpy change = ∓q / moles)
give the units of the equation △H = ∓q / n
△H has the units kJmol⁻¹
this means that q must be converted from J to kJ (by dividing by 1000)
what are the two ways to calculate moles?
by using either:
n = mass/Mr
or n = conc x vol
how do you know whether △H will have a positive or negative value?
if reaction is exothermic, then △H will have a negative value
if reaction is endothermic, then △H will have a positive value
required practical 2-
what are the two methods of carrying out calorimetry?
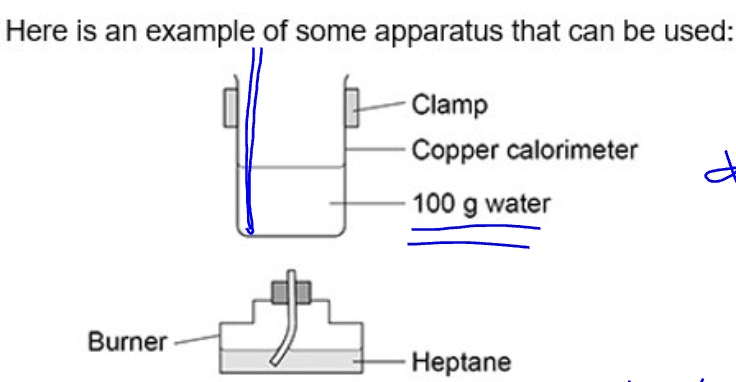
combustion reaction → a fuel is used to heat up a known amount of water
a reaction in solution is carried out in an insulated container (eg a polystyrene coffee cup) → eg hydration, displacement, neutralisation
describe the steps of the method of carrying out a combustion reaction (type 1) for calorimetry + so calculating △H

give 6 reasons why the value obtained from the combustion reaction for calorimetry will be less exothermic) than the data book value
heat loss to surroundings
incomplete combustion
non-standard conditions
evaporation of fuel/alcohol
heat loss to the can
poor stirring of the water in the can so that the temperature reading is incorrect
(first two are most important to learn)
describe the steps of the method of carrying out a reaction in solution in an insulated container (type 2) for calorimetry + so calculating △H
give two reasons why the value obtained from carrying out a reaction in solution in an insulated container will be less (or more) than the data book value
heat loss to the surroundings (for exothermic reaction / heat gain from the surroundings (for endothermic reaction)
incomplete reaction
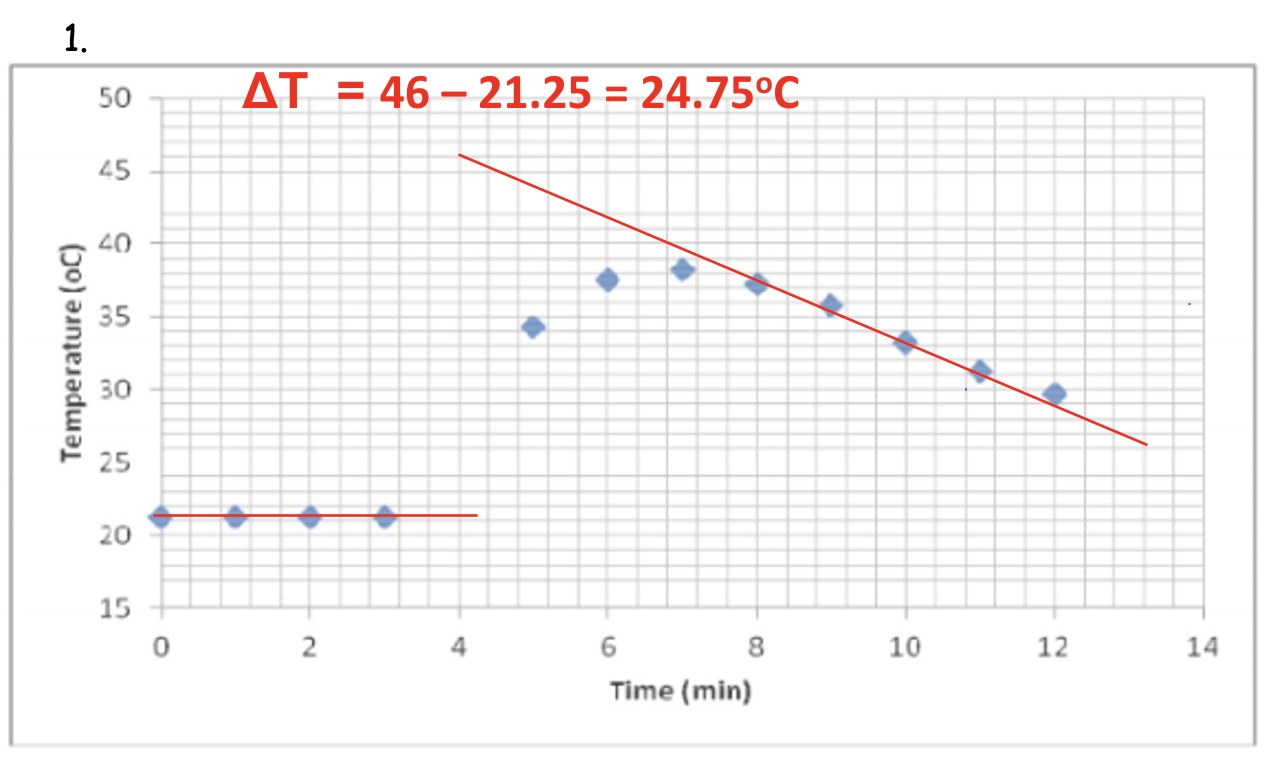
describe how you can calculate △T from a graph (plotted using results from the reaction in solution in an insulated container)
draw both lines of best fit + extrapolate both lines (past the minute of when the reactant was added) → remember not to include any data that doesn’t ‘fit’ the pattern
draw a vertical line where both lines ‘overlap’
calculate △T

using q = mc△T and △H = ∓q / n


using q = mc△T and △H = ∓q / n

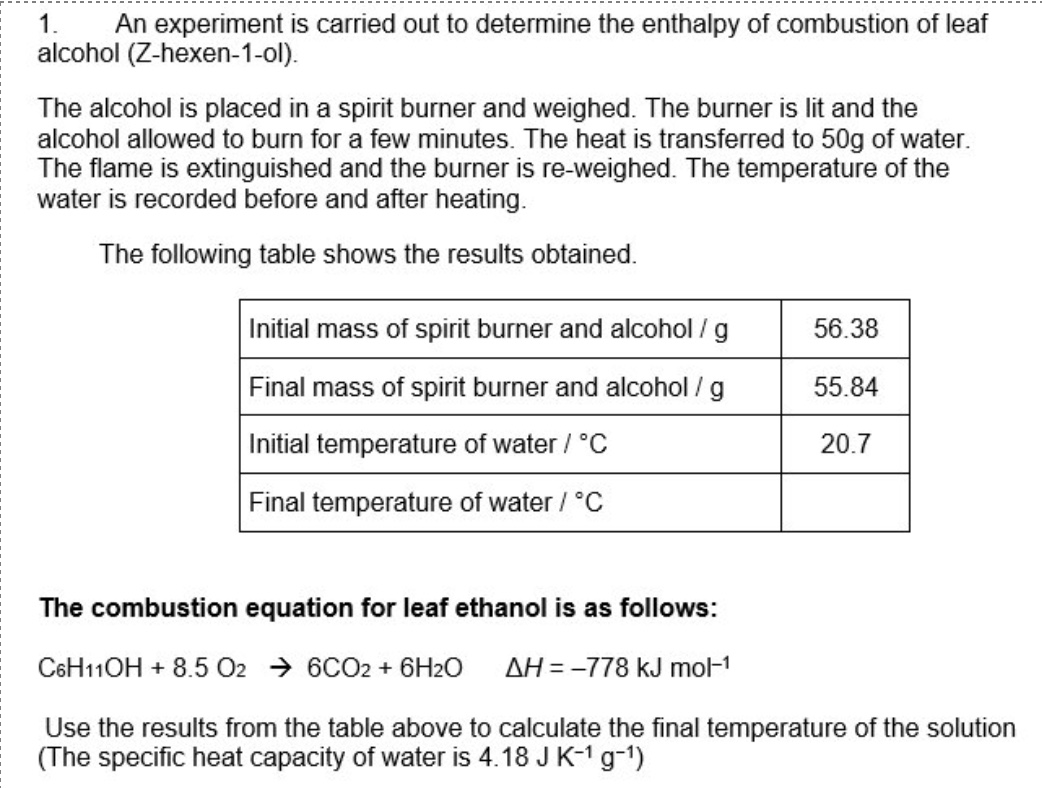
rearranging calorimetry calculations


rearranging calorimetry calculations