Atomic and Electron structure
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Relative Atomic Mass
The weighted mean mass of an atom compared with 1/12th mass of an atom of carbon-12
Calculating R.A.M
Rearranging R.A.M Calculations
Isotopes
Atoms of the same element with the same number of protons but different number of neutrons
An atomic orbital
A region within an atom that can hold up to two electrons with opposite spins
S and P Orbitals
S Orbital - Spherical
P Orbital - Dumbbell
Electron configurations
1s22s22p63s23p64s23d104p65s2
Noble Gas configuration
A short hand configuration that can be written using the electronic configurations of the noble gases that come before the element
Configuration exceptions and reasons
Chromium - 1s22s22p63s23p64s13d5
Copper - 1s22s22p63s23p64s13d10
Reason: They represent more stable arrangements
Electron shells and the amount of electrons in them
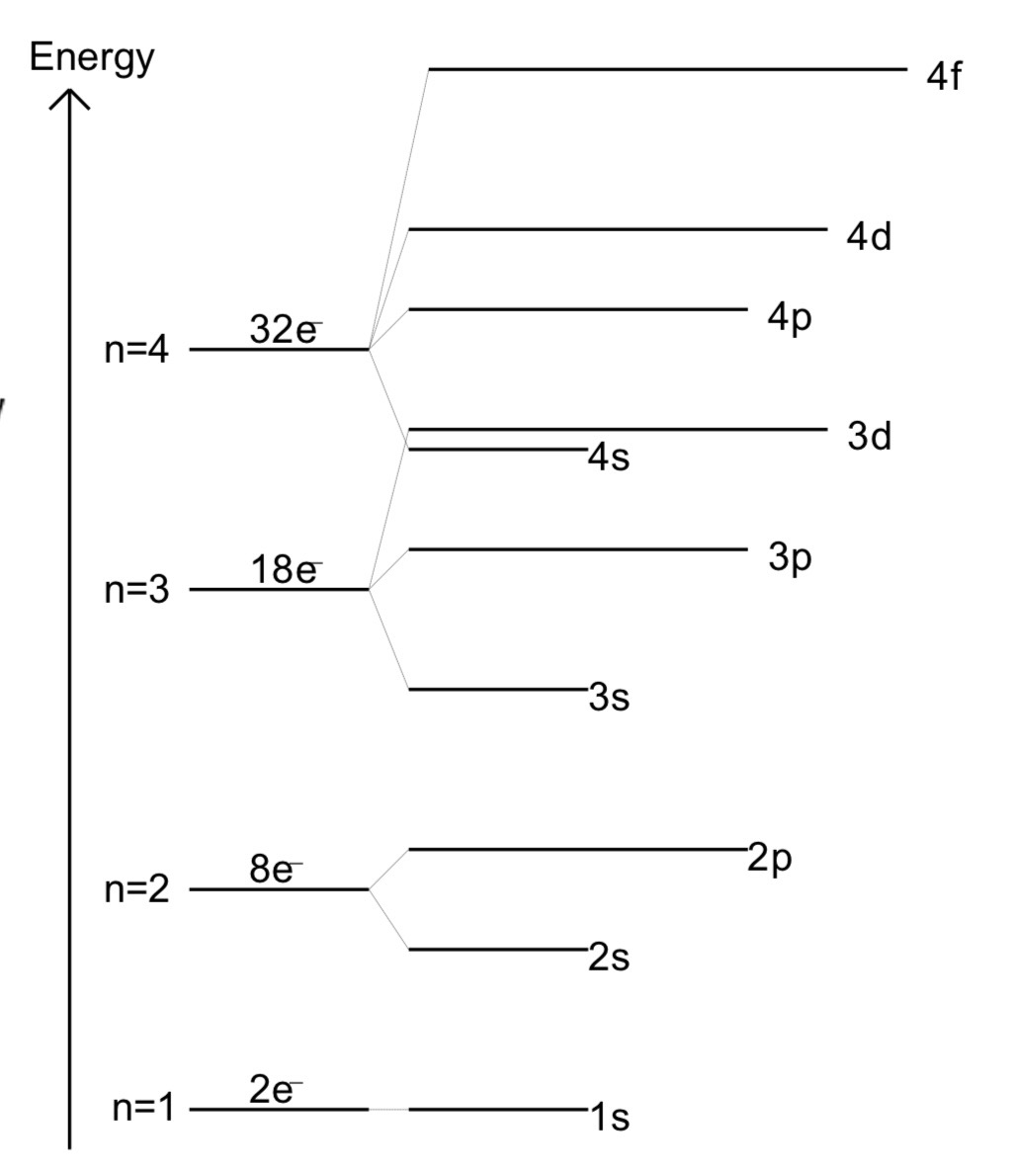
Describing energies between s and p orbitals
P - orbitals have greater energy than s - orbitals
P - orbitals have equal energy
Two differences between isotopes of the same element
Different physical properties
Different number of neutrons