Natural products
1/108
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
109 Terms
What are terpenoids?
Terpenoids are a large family of natural products linked by C5 units in a head to tail manner
they can be further classified by the number of C5 units used in construction of the basic carbon skeleton
what are monoterpenes?
C10 units (2X C5)
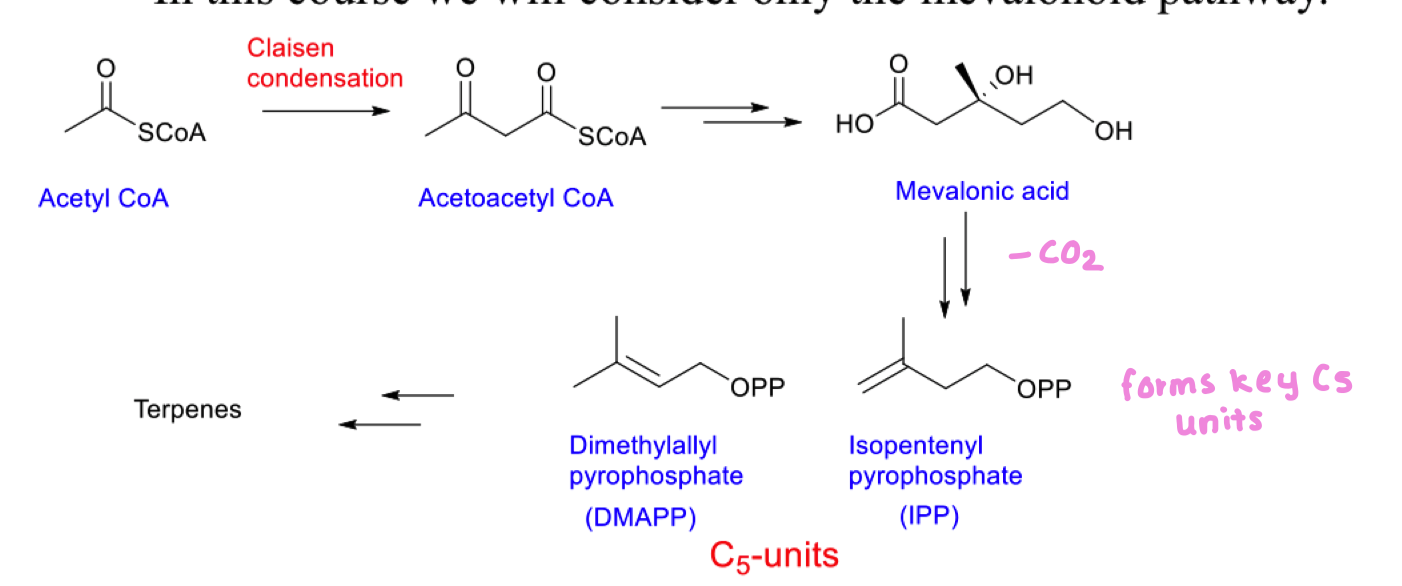
Describe the pathway of terpenoid biosynthesis?
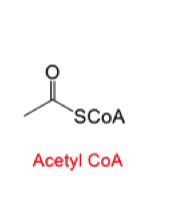
why does nature use coenzyme A for claisen condensation? what do synthetics use as the coenzyme A?
Used by enzymes for recognition
Acetyl CoA
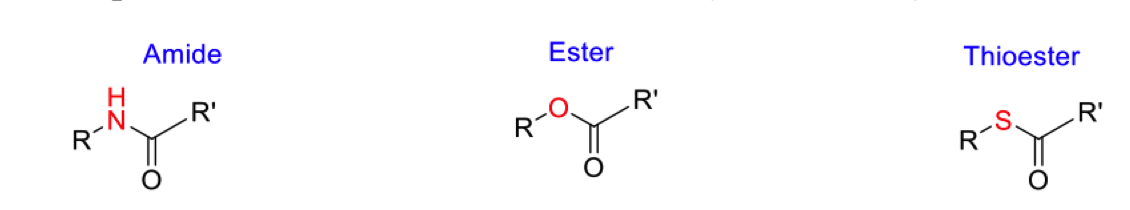
Order in the most to least reactive in ease of hydrolysis

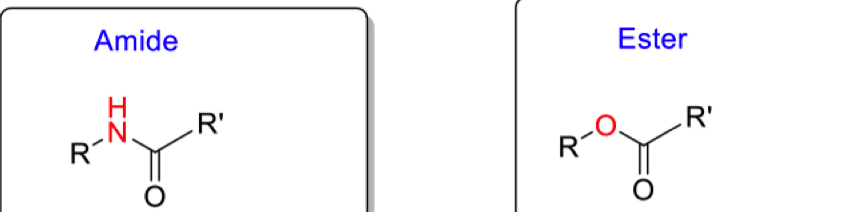
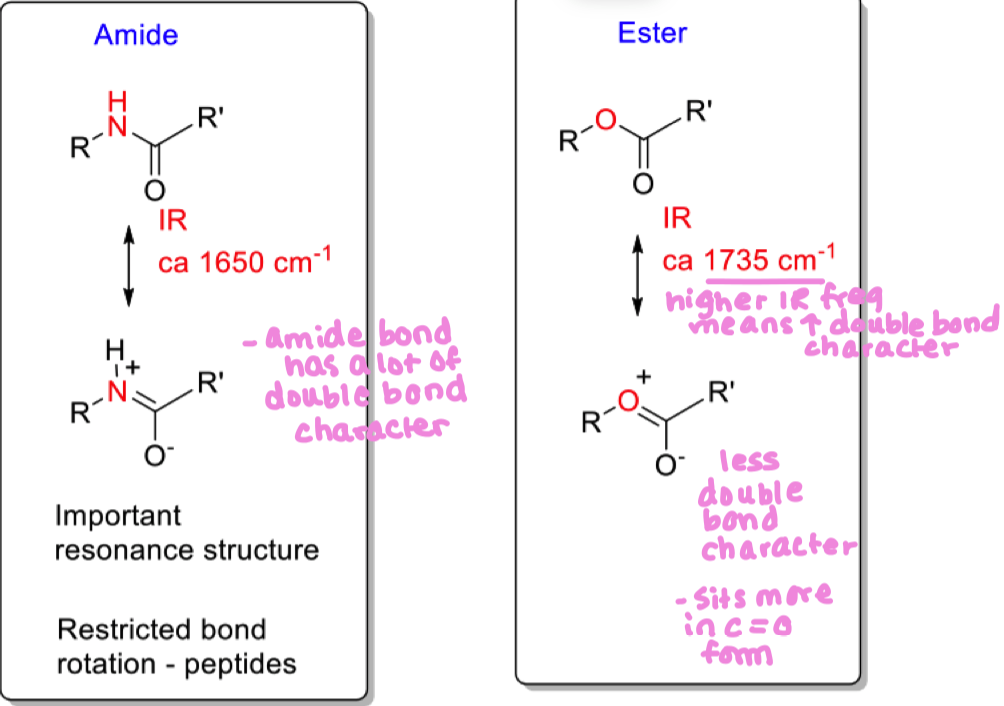
Compare reactivities of amide and ester (draw resonance structures)
amide has alot of double bond character and sits in that structure alot (less C=O character)
amide has restricted bond rotation
ester has more C=O character ( this is also reflected in its higher IR frequency
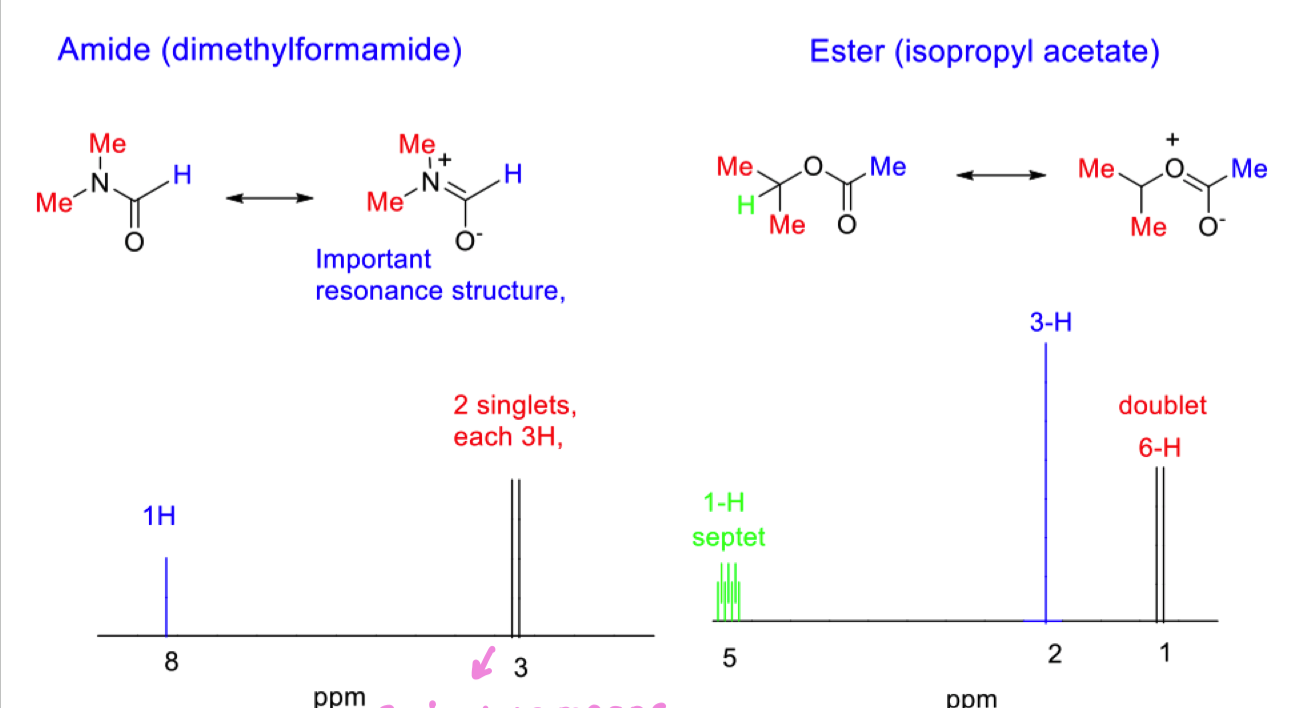
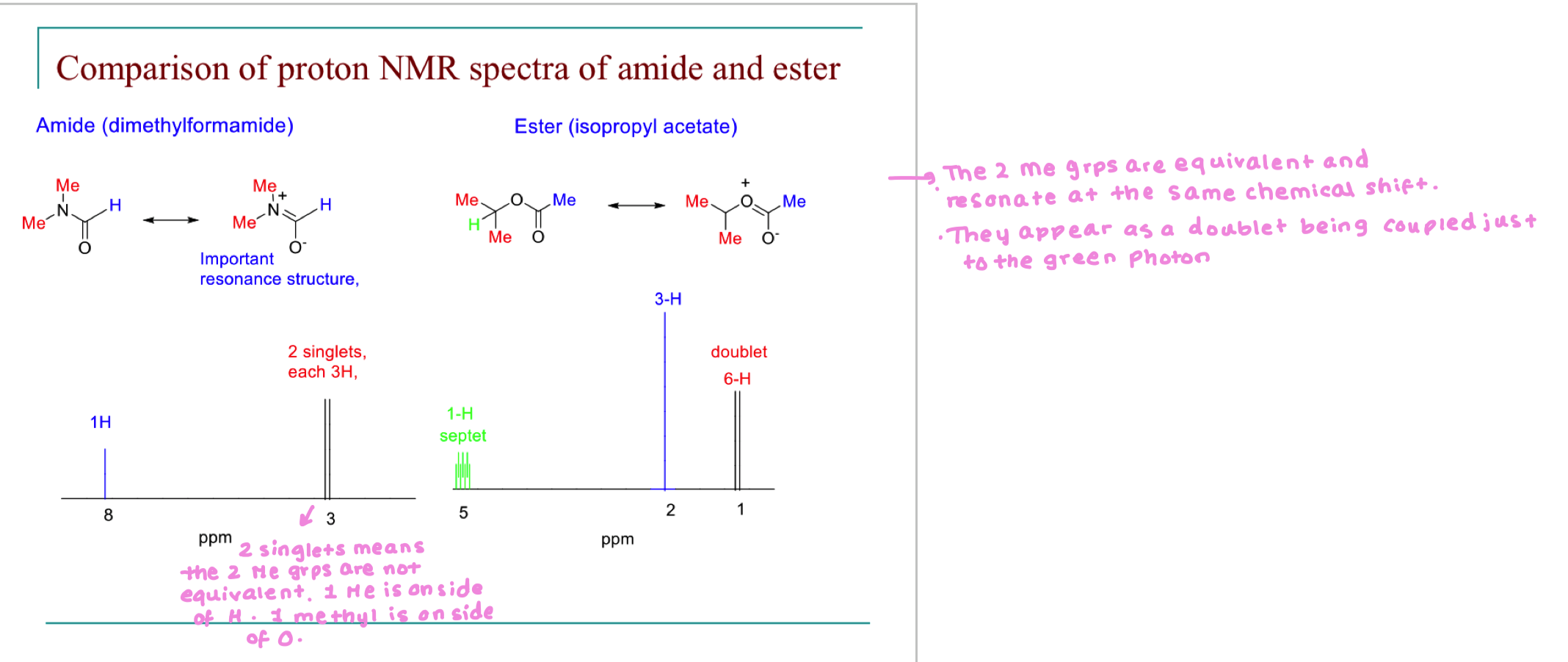
Compare the NMR spectra of amide and ester
the fact that methyl groups are not equivalent in amide means we have restricted bond rotation therefore methyls are in different chemical environments.
however, if temp was raised, energy barrier to rotation is overcome and this allows for rapid rotations making it seem like methyls are in the same environment.
Compare the ester with the thioester (13C)
thioesters are less stabilised by resonance
this leaves the carbonyl C to be more electrophilic so more reactive
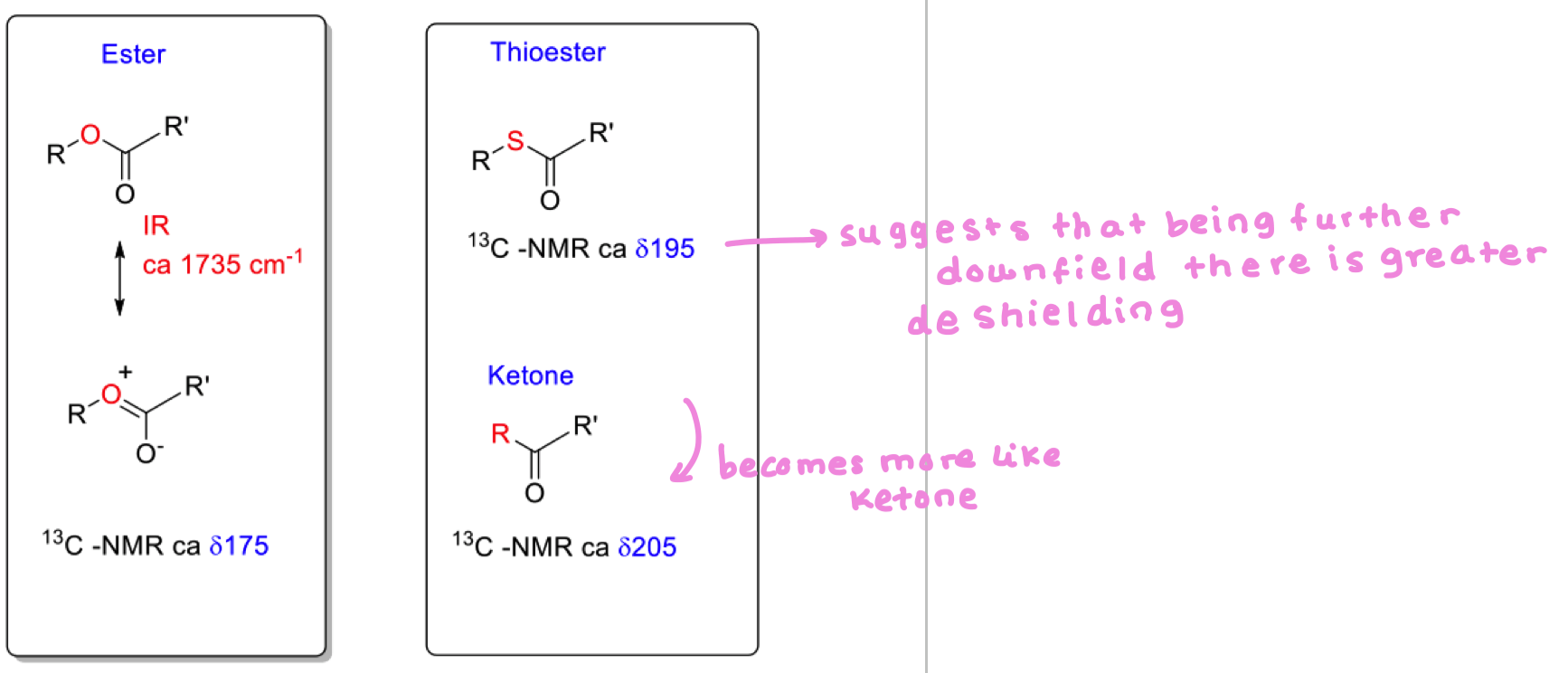
Why does nature use thioesters in biosynthesis?

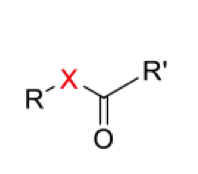
Draw the general mechanism for the hydrolysis of ester/thioester
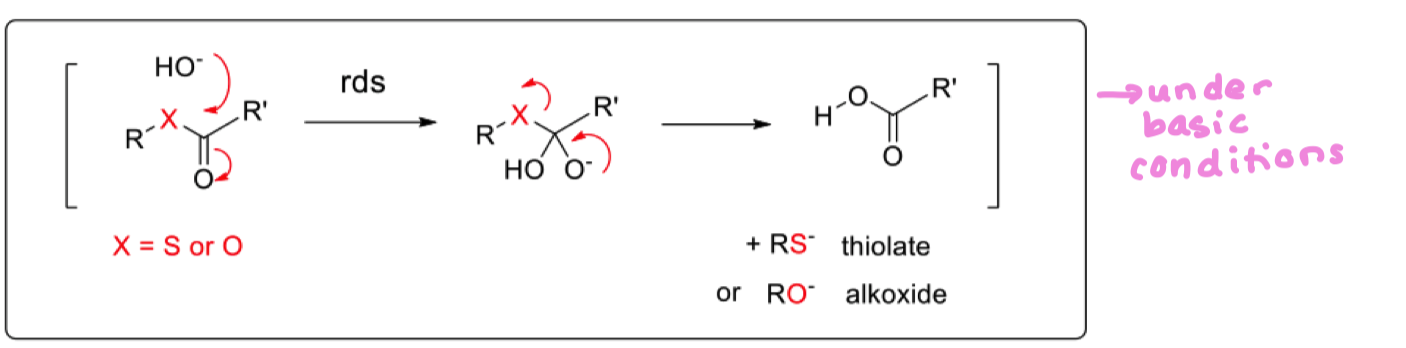
compare claisen condensation in organic synthesis and biosynthesis
Draw the mechanism of claisen condensation in organic synthesis
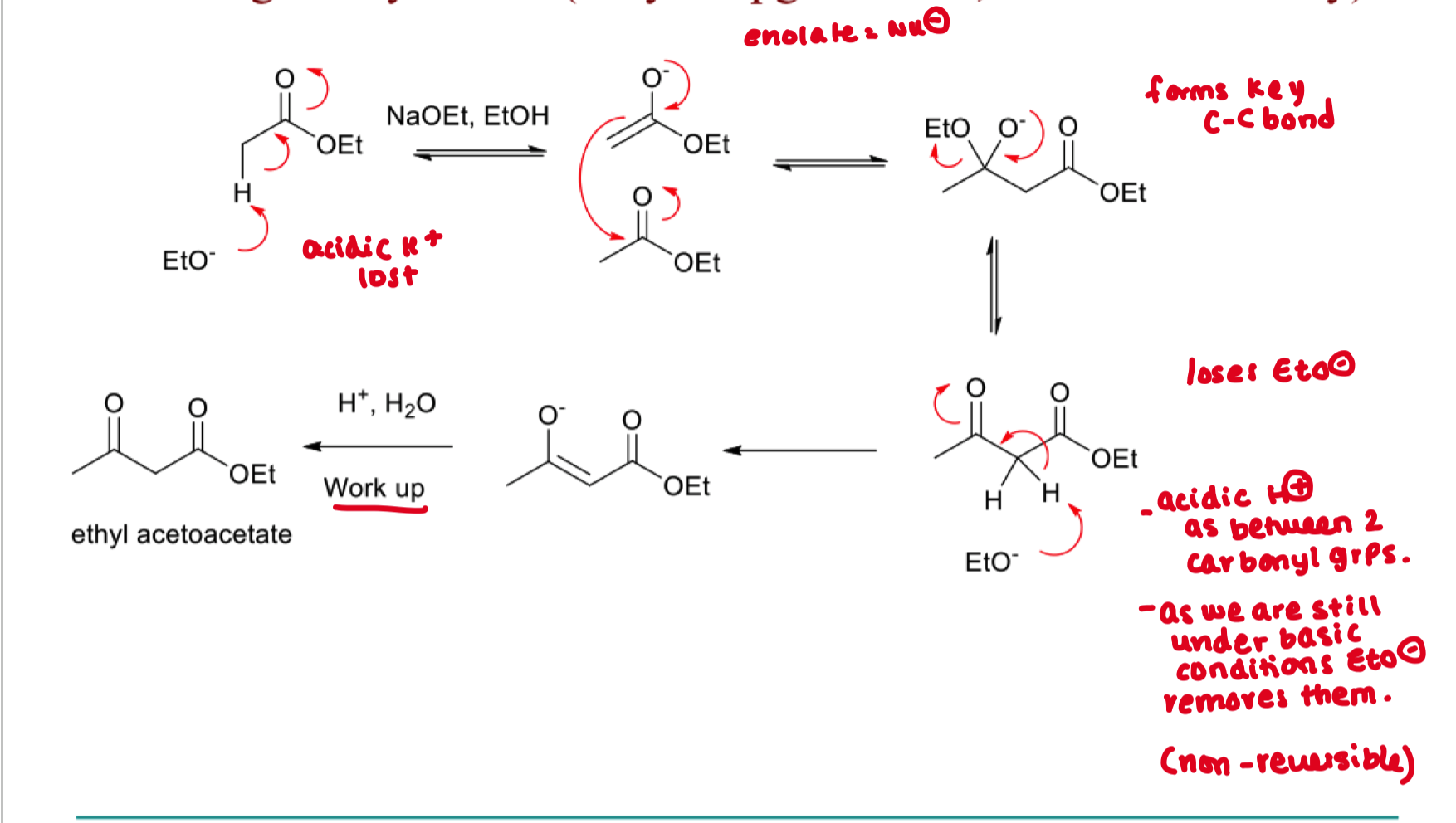
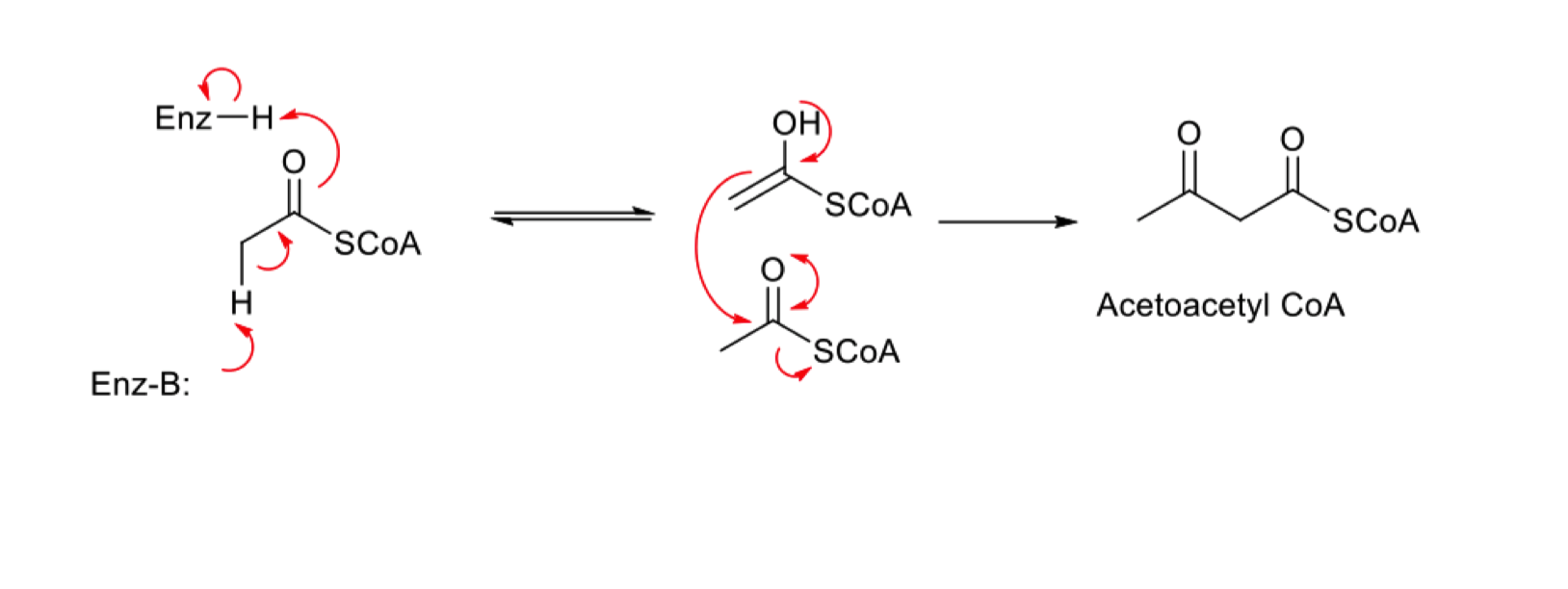
mechanism of claisen condensation in biosynthesis
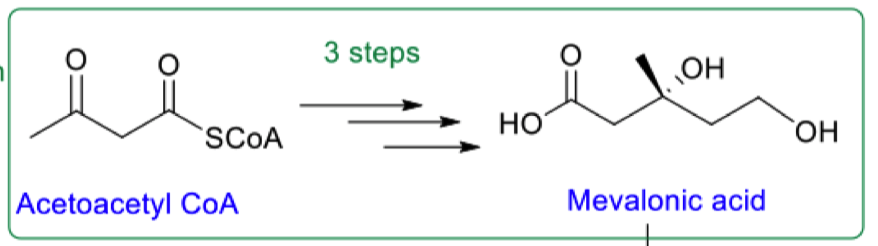
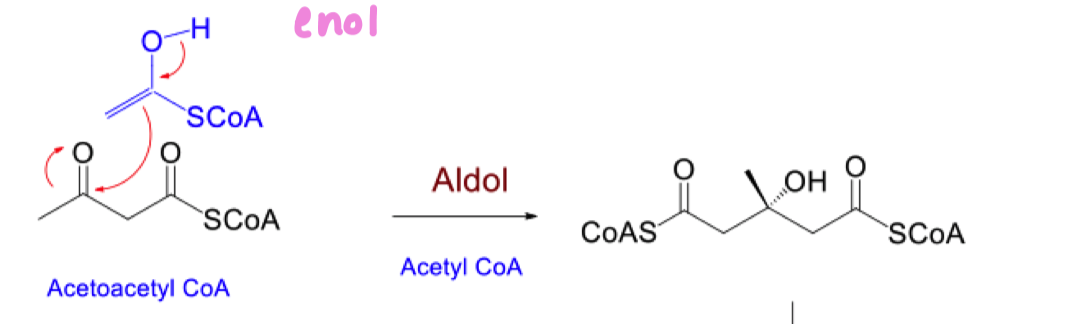
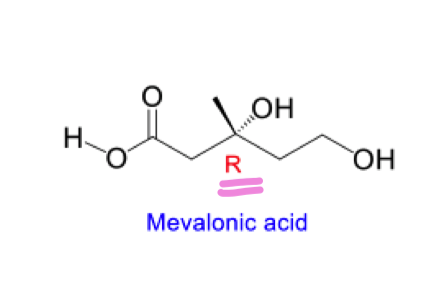
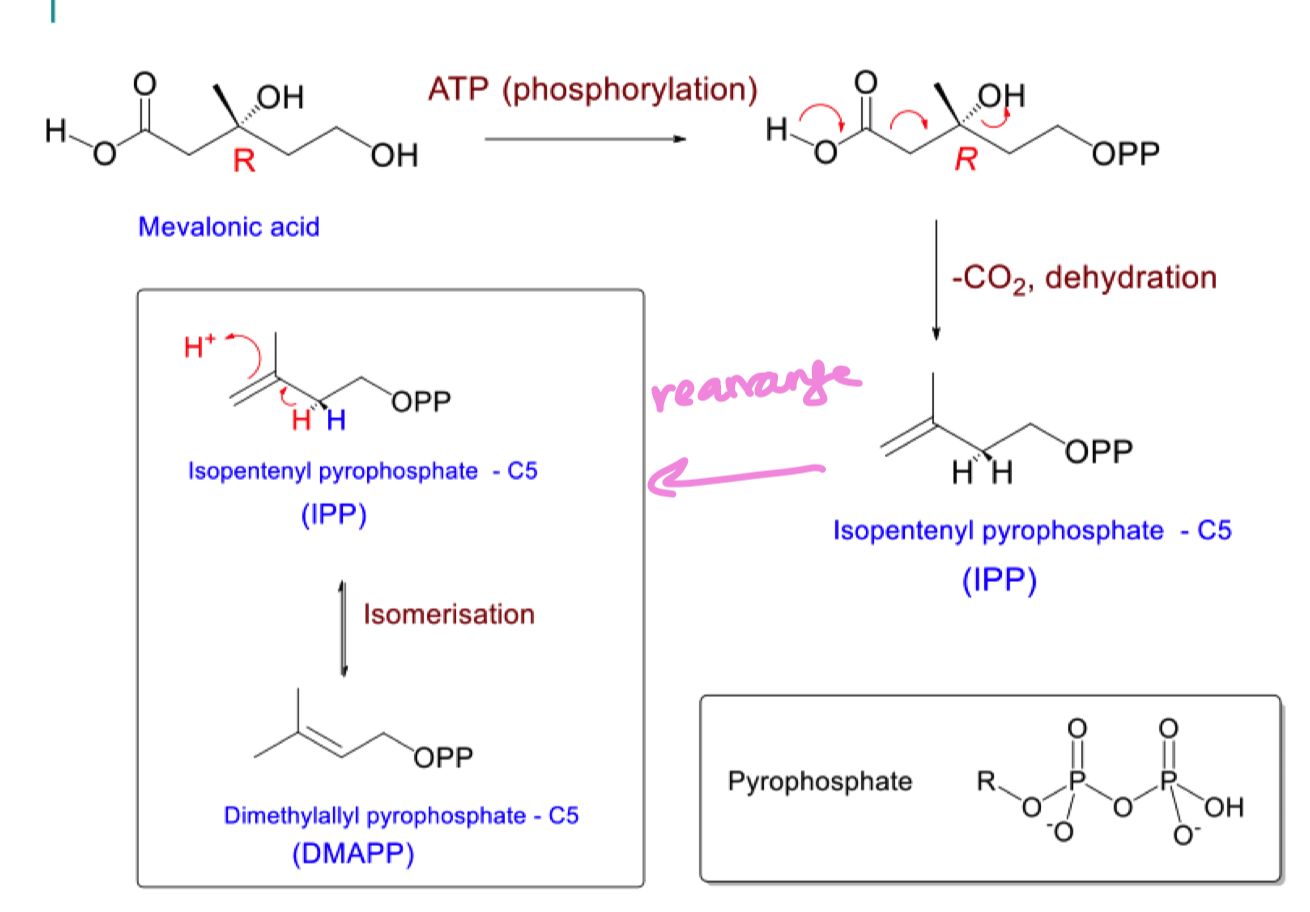
list the 3 steps to go from acetoacetyl coA to mevalonic acid
aldol reaction
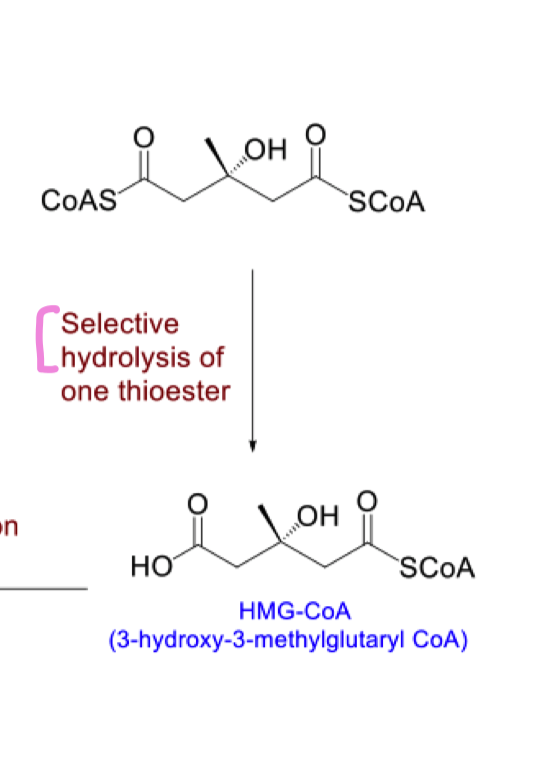
selective hydrolysis of one thioester
selective reduction of thioester
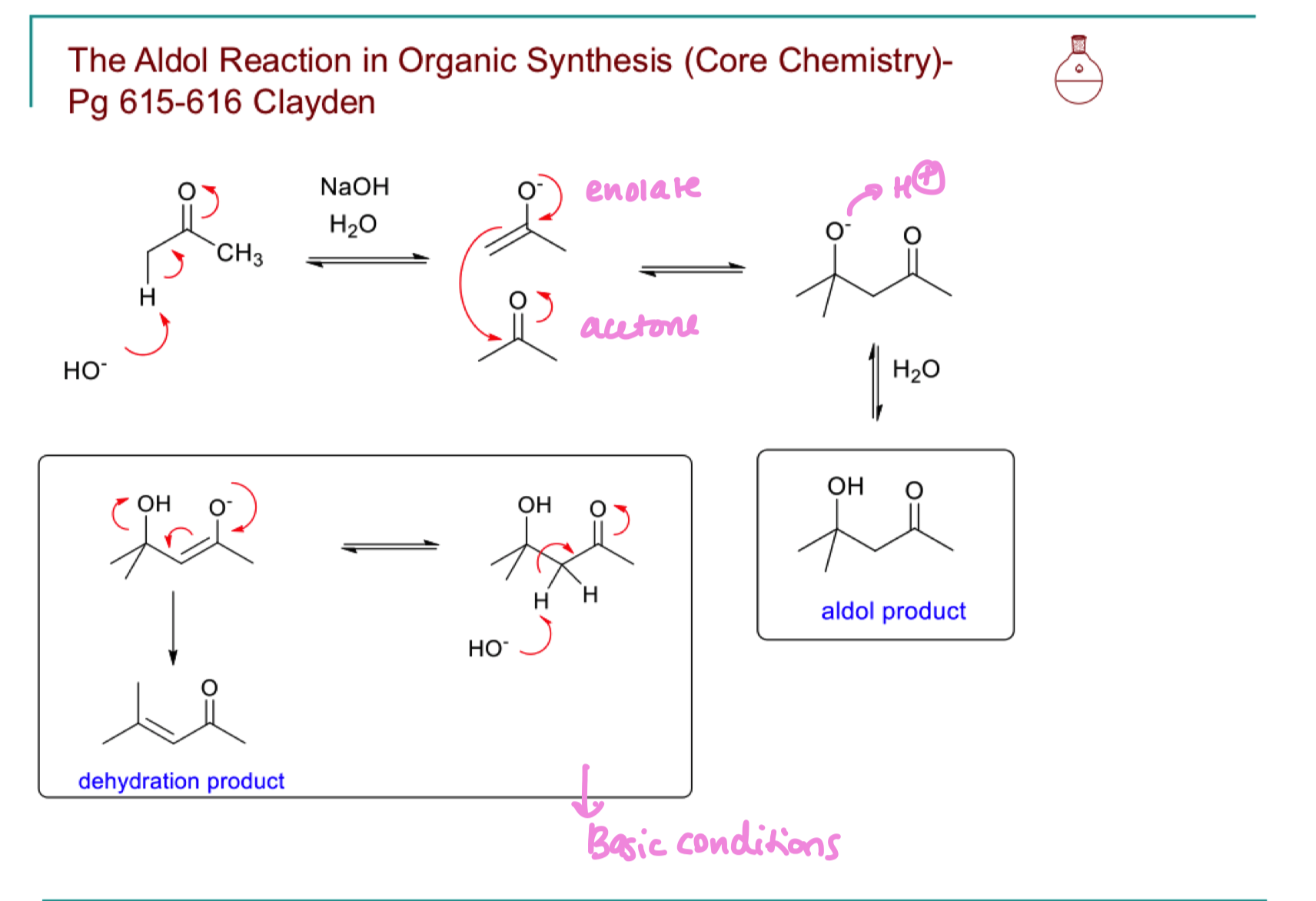
Draw the mechanism for aldol reaction in organic synthesis. under both acidic and basic conditions
draw the mechanism for aldol by biosynthesis
draw the product of selective hydrolysis of one thioester
thioesters are readily hydrolysed at room temp
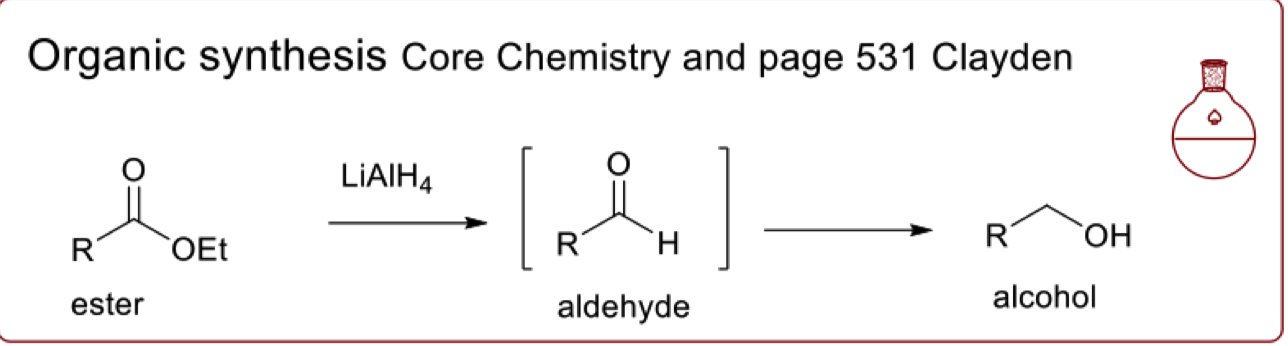
describe the selective reduction in organic synthesis
describe the selective reduction of thioester in biosynthesis
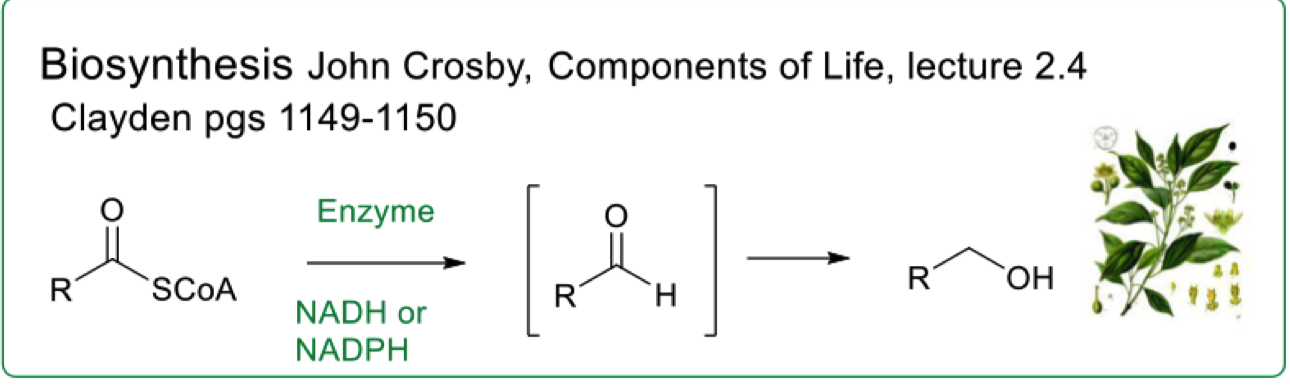
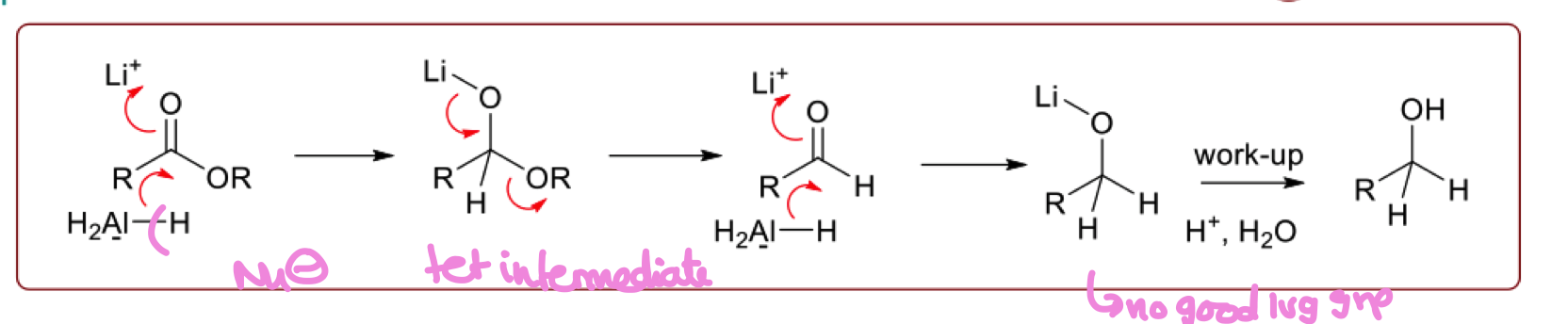
why is the aldehyde not isolated in the reaction ?

draw the mechanism for the selective reduction of ester in organic synthesis
draw the mechanism for the selective reduction of thioesters in biosynthesis
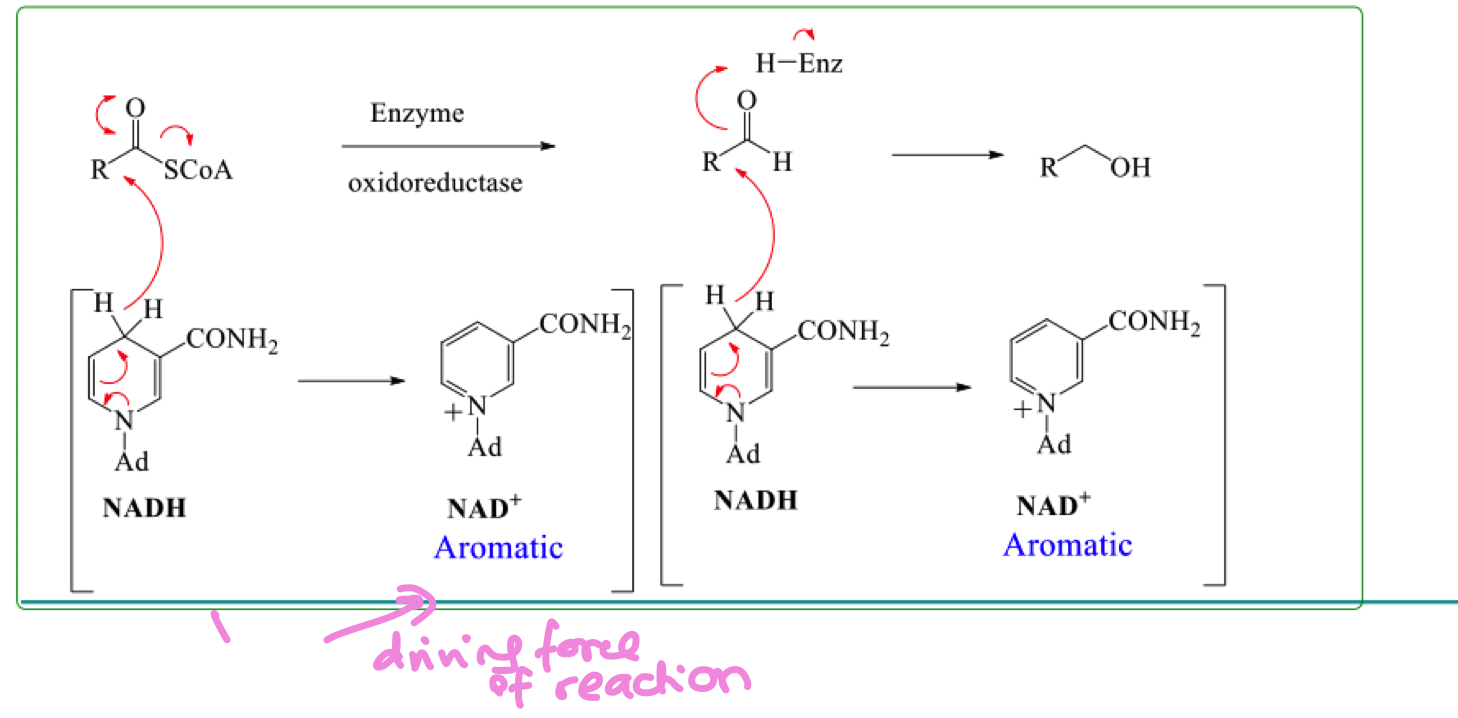
what is the stereochemistry of the product formed from selective reduction
draw the 2 C5 units formed from mevalonic acid : DMAPP and IPP
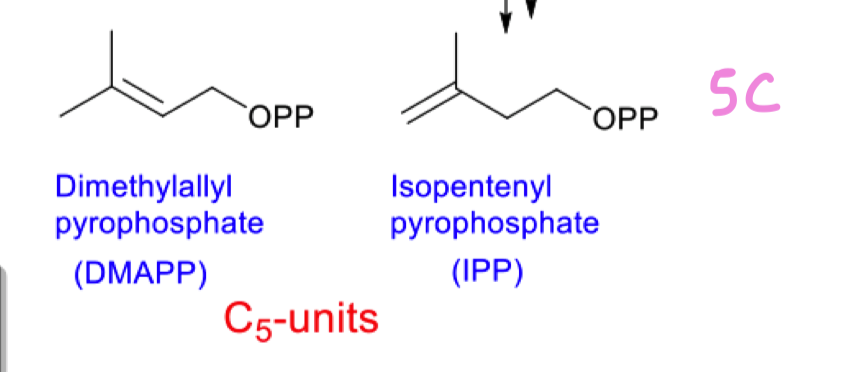
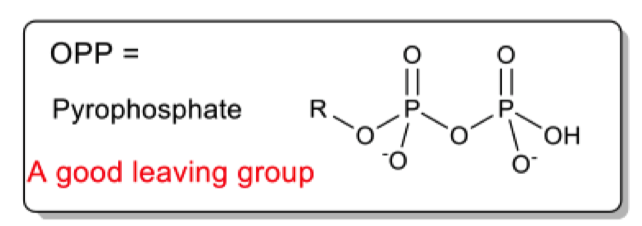
What is OPP?
draw the mechanism of the biosynthesis of IPP and DMAPP
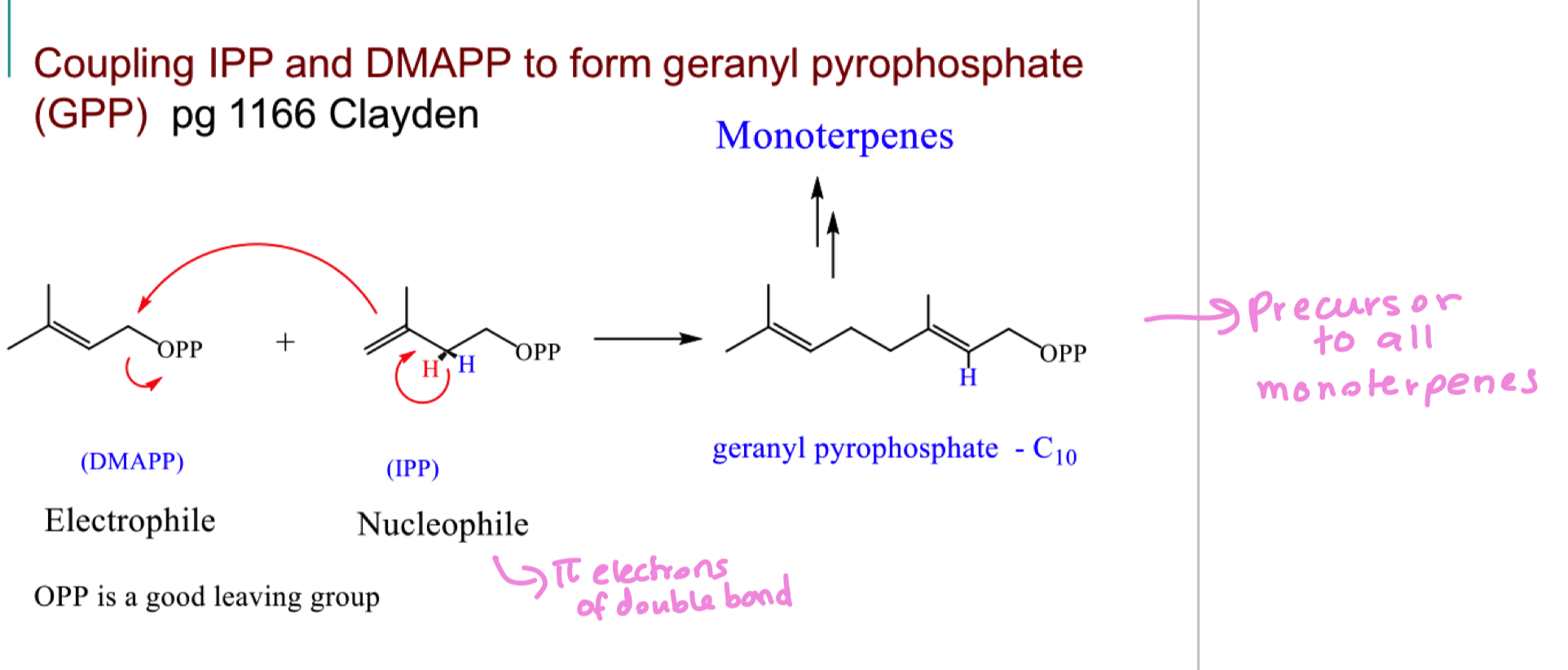
draw the mechanism of reaction IPP and DMAPP together to form a monoterpene
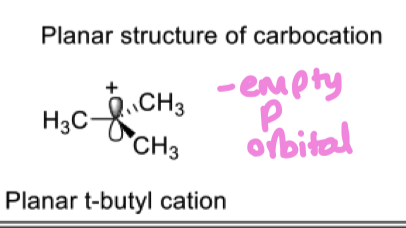
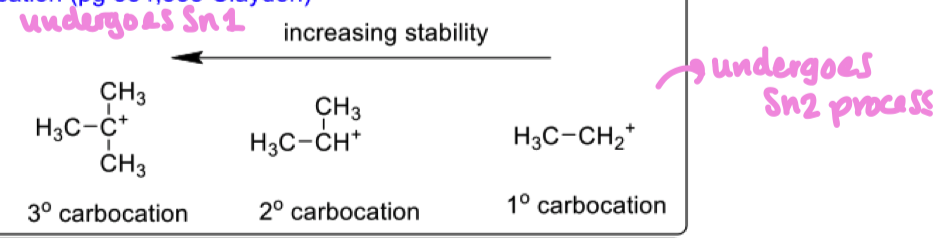
How do alkyl substituents stabilise a carbocation?
methyl groups overlap with the empty p orbitals of carbocation
show how adjacent C=C pi systems stabilise carbocations
they are able to delocalise the positive charge around the ring
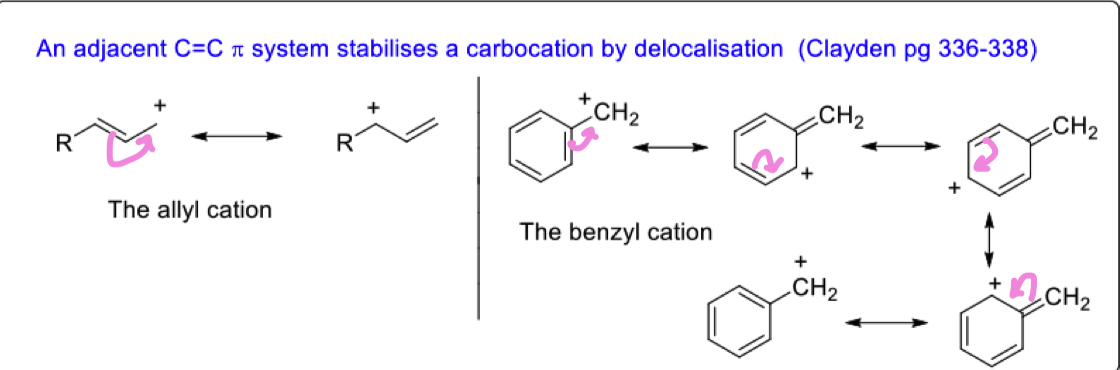
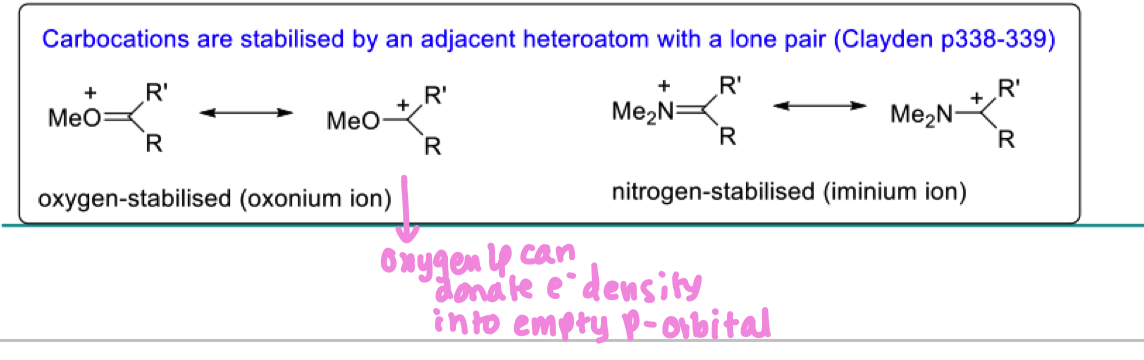
Describe how carbocations can be stabilised by an adjacent heteroatom like O and N
formation of a pi bond is a stabilising influence
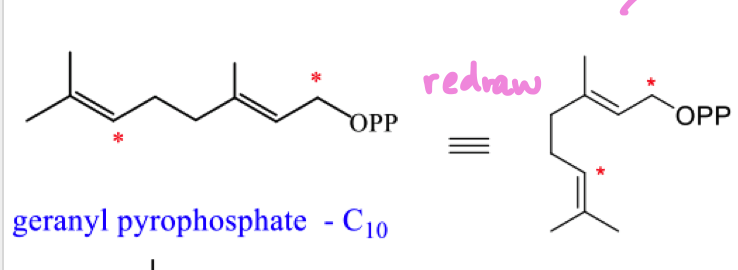
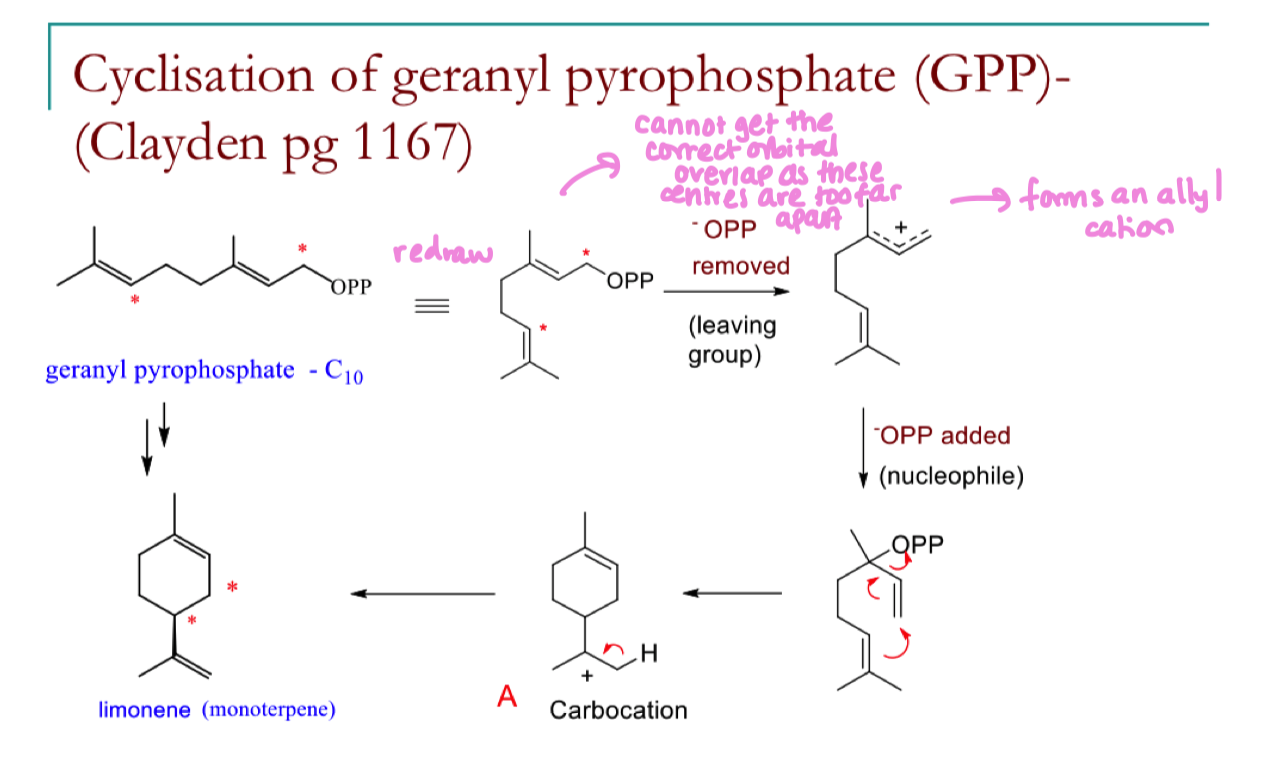
redraw geranyl pyrophosphate into a cyclic form and explain why we cant form a full cyclic structure from here
cannot get the correct orbital overlap as these centres are too far apart
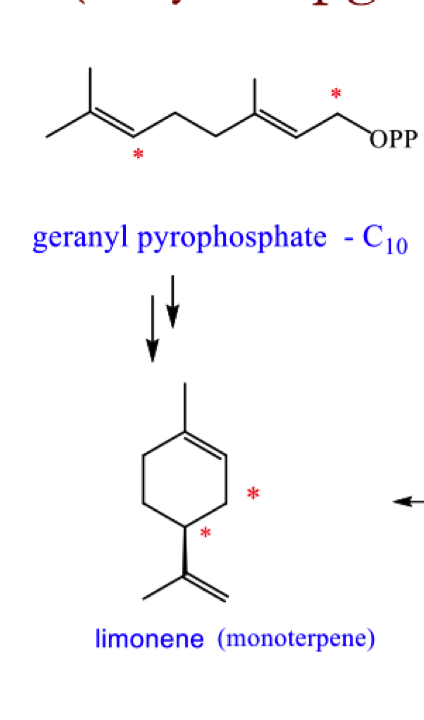
Draw the mechanism for the formation of limonene from geranyl pyrophosphate
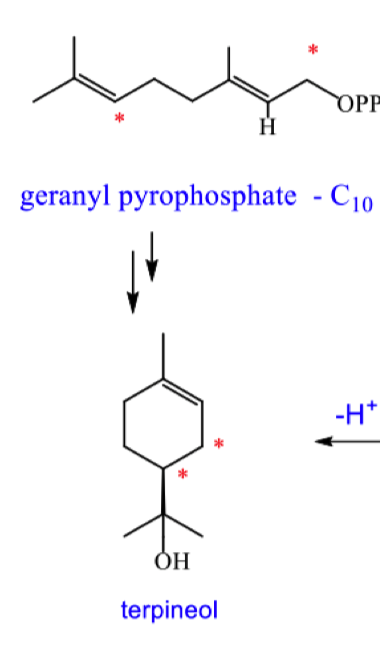
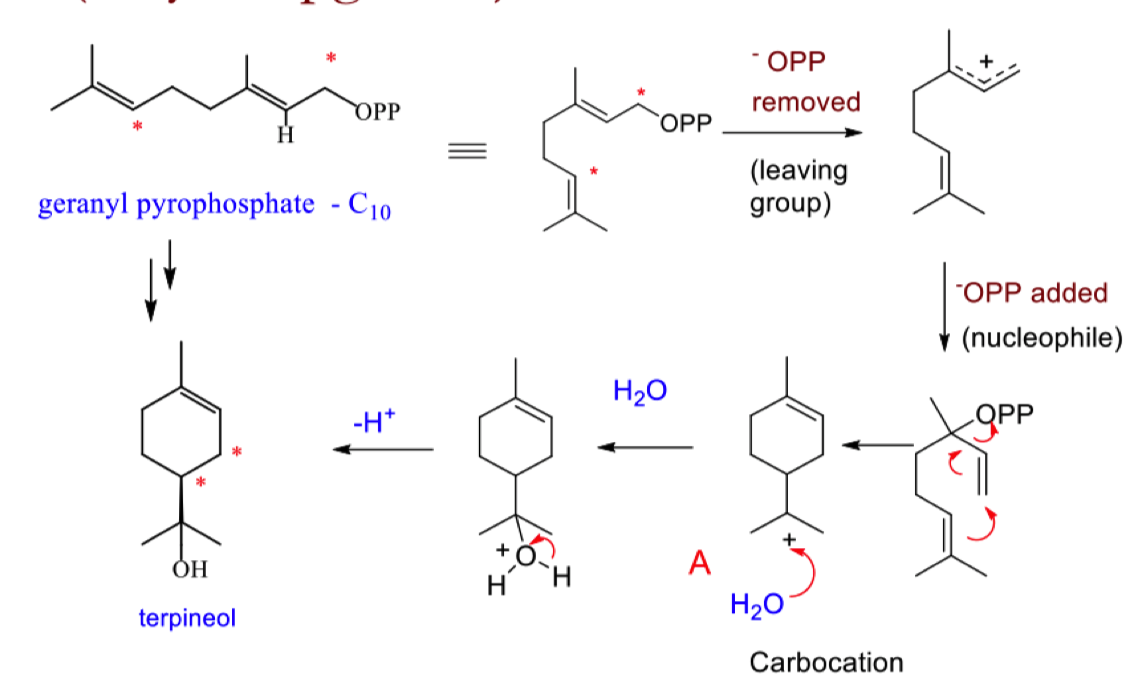
draw the mechanism for the formation of terpineol
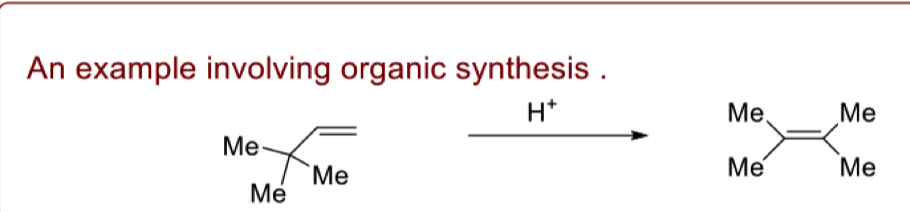
draw the mechanism for the migration of alkyl groups
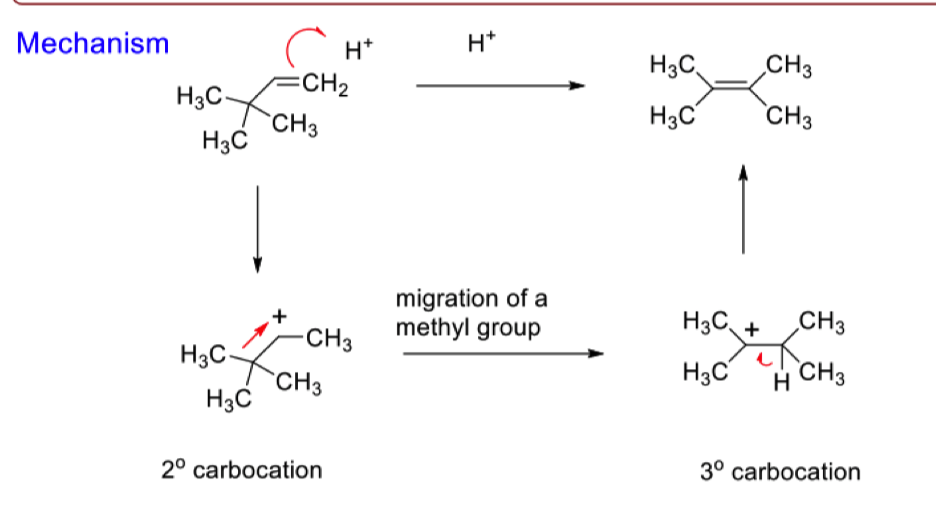
what is the driving force of carbocation rearrangements
reactions proceed from a less stable carbocation to form a more stable carbocation
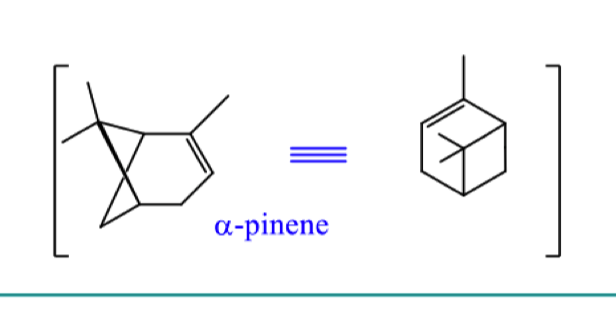
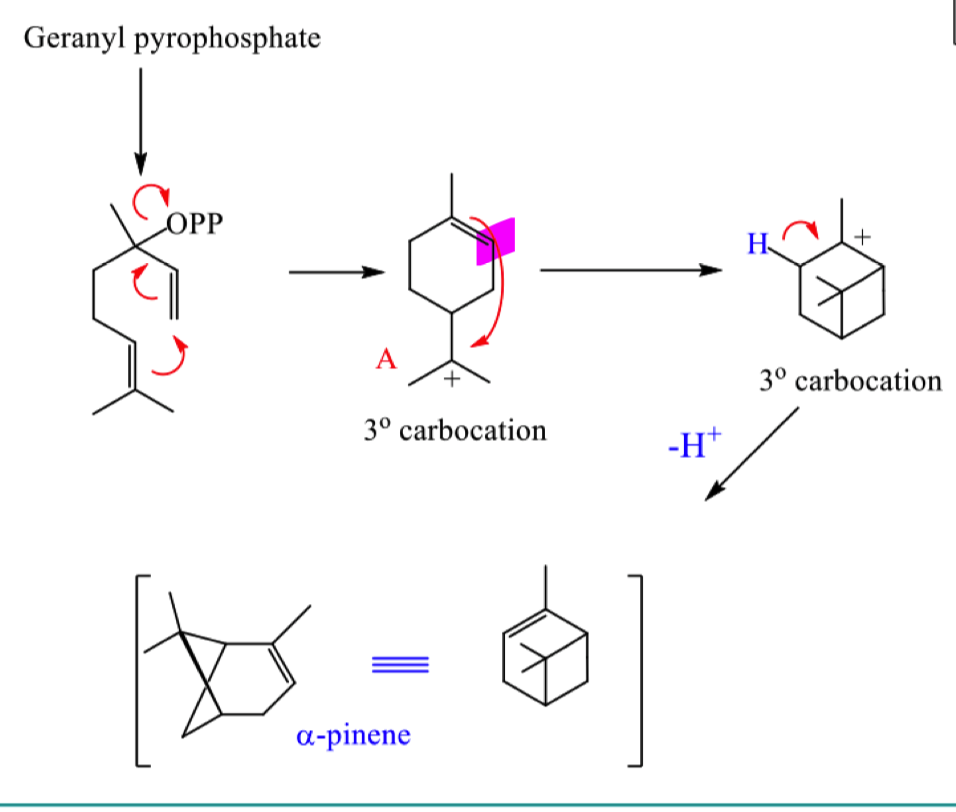
describe the mechanism for formation of alpha pinene starting from geranyl pyrophosphate
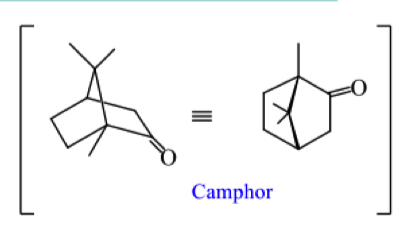
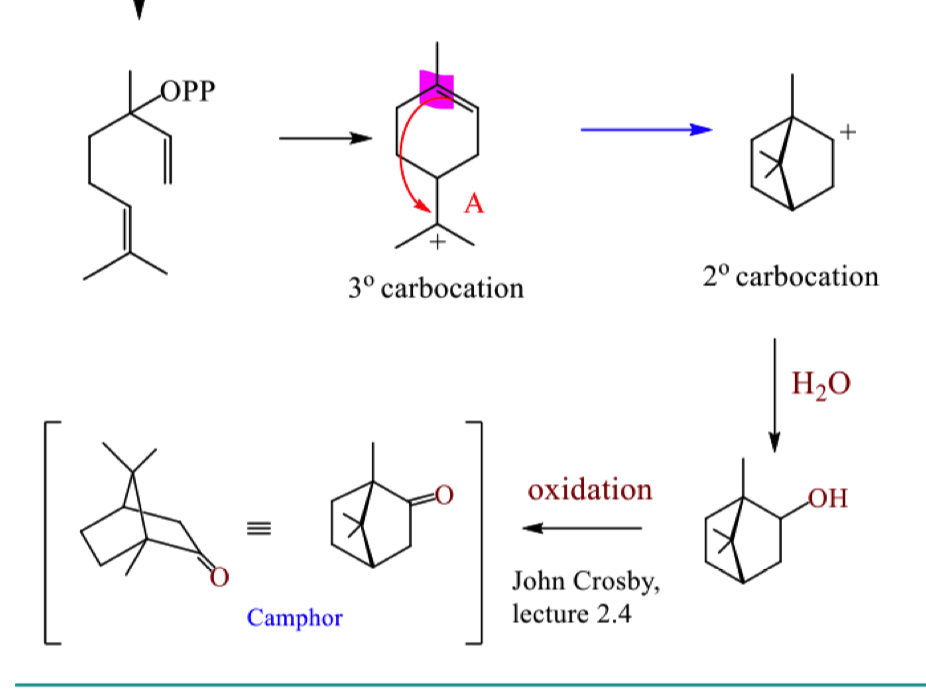
describe the mechanism for formation of camphor starting from geranyl pyrophosphate
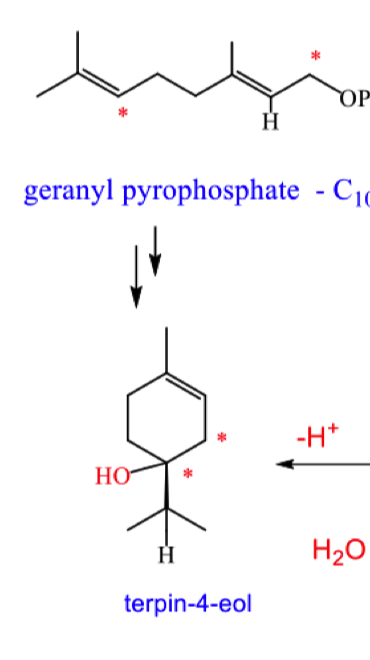
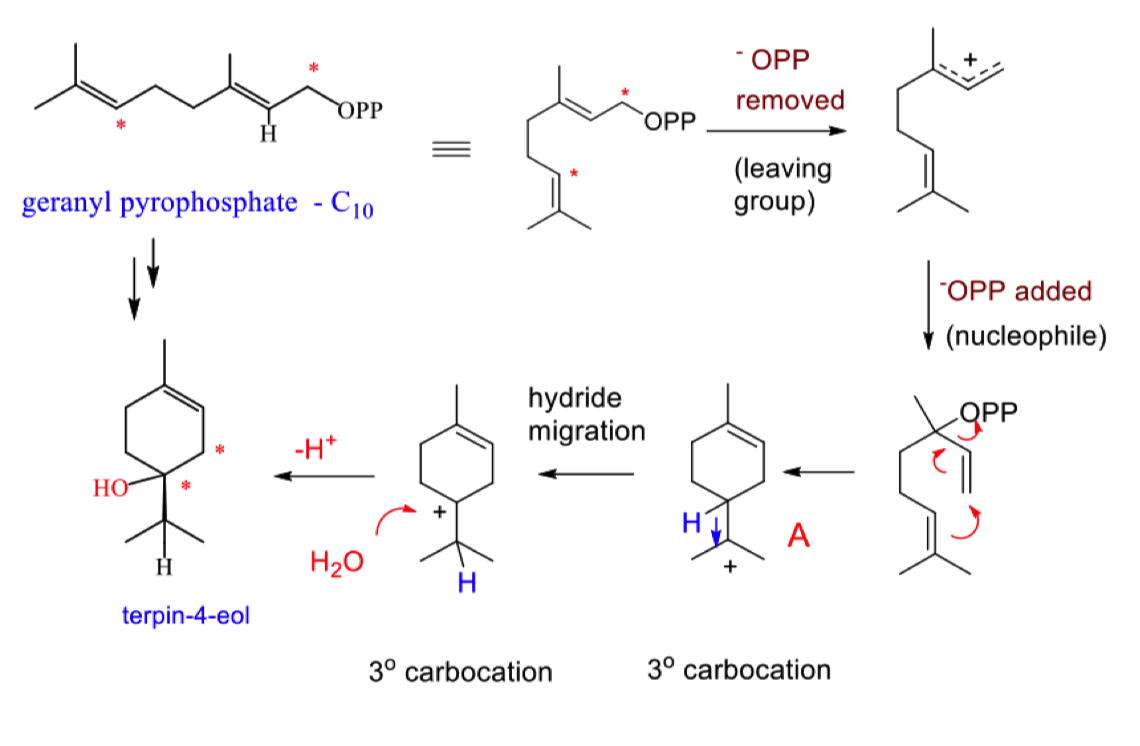
describe how hydride migration forms terpin-4-eol
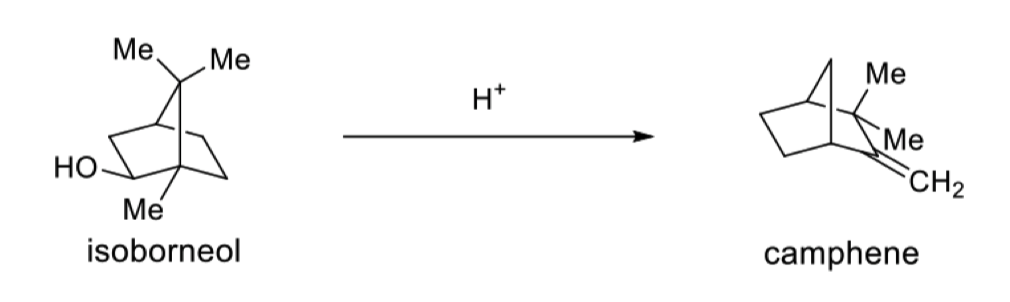
describe the interconversion between these two terpenes
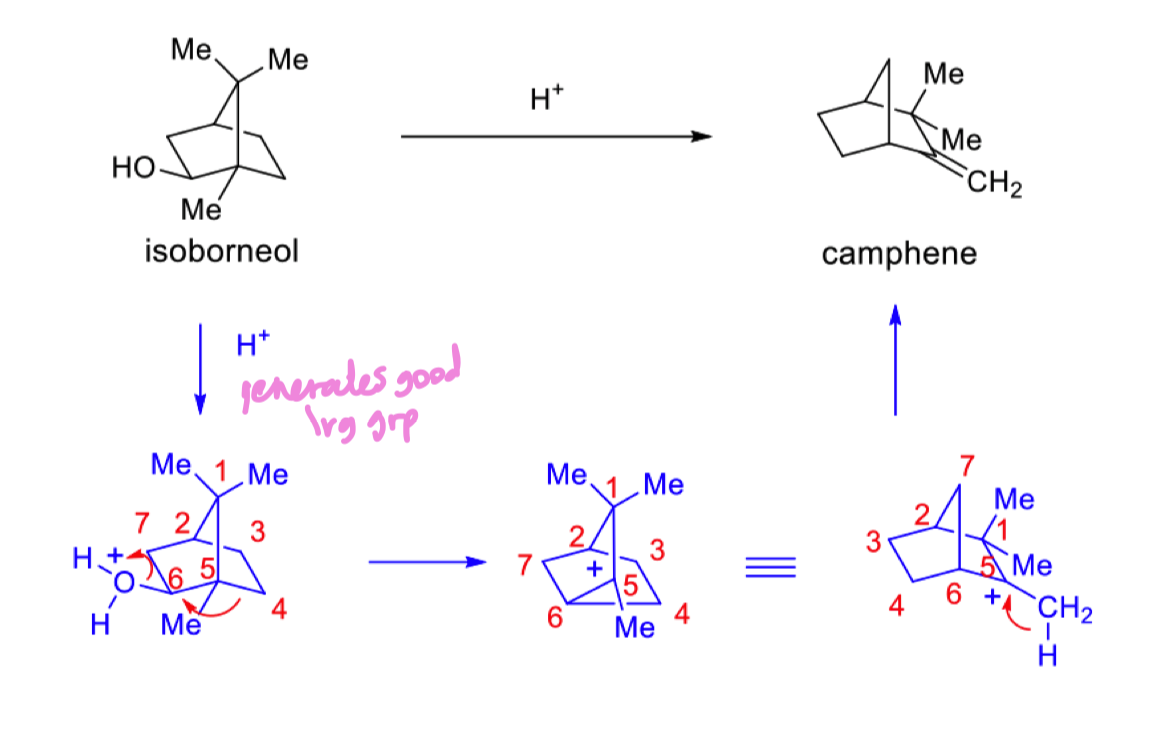
what are sesquiterpenes and how do we form them?
3 x C5 units
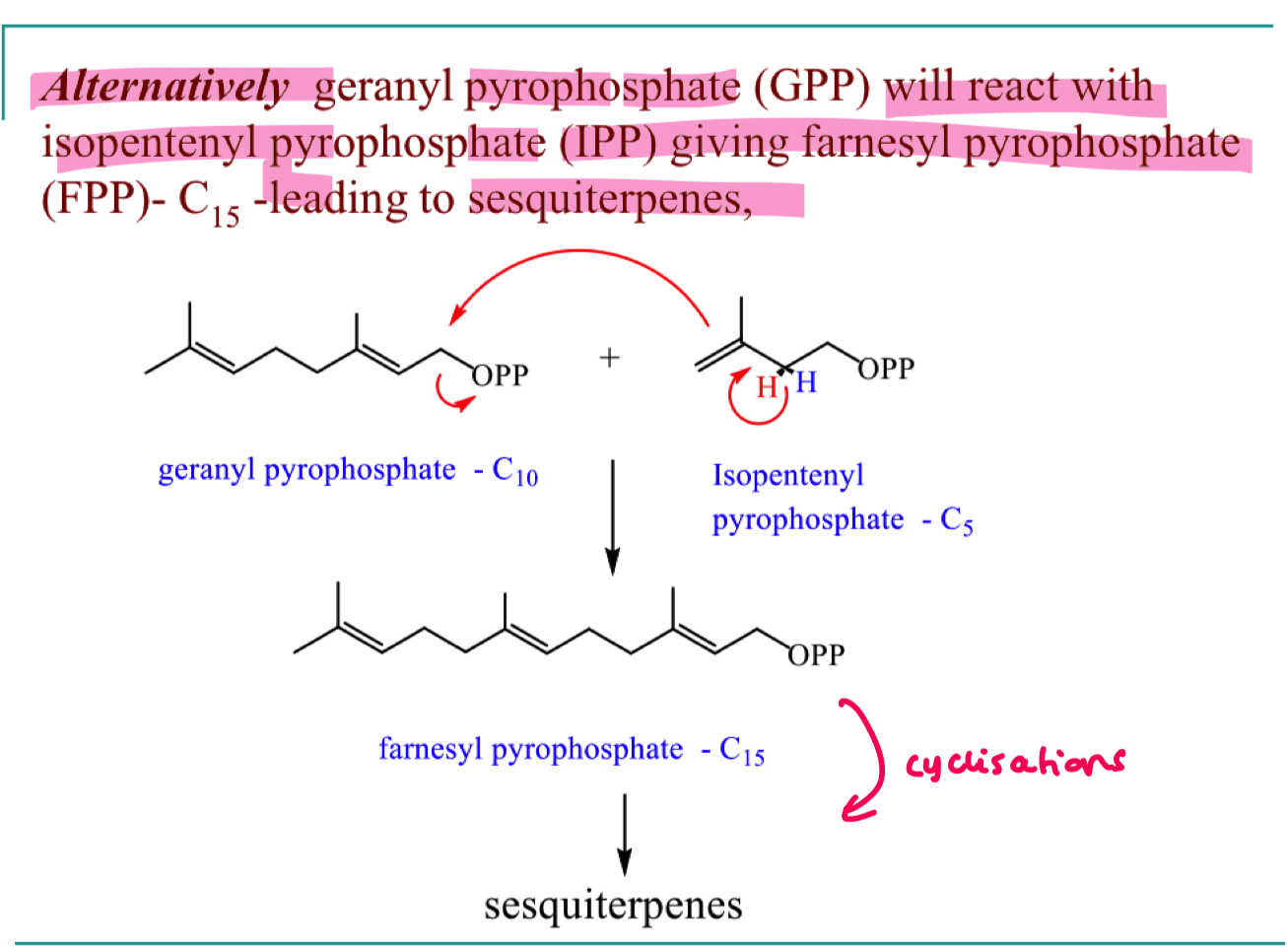
what are diterpenes and how do we form them?
4 x C5 units
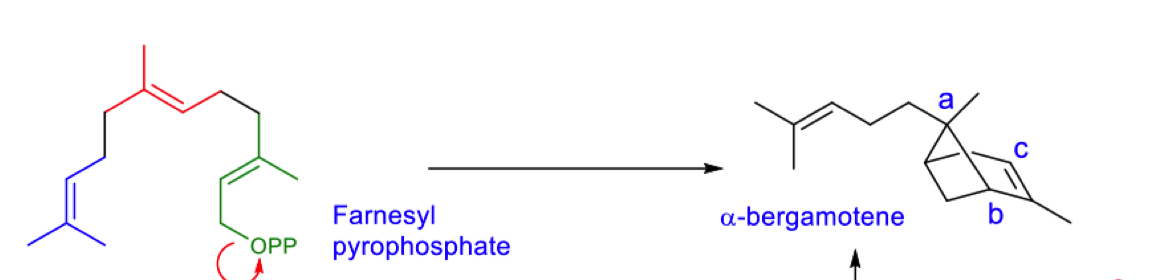
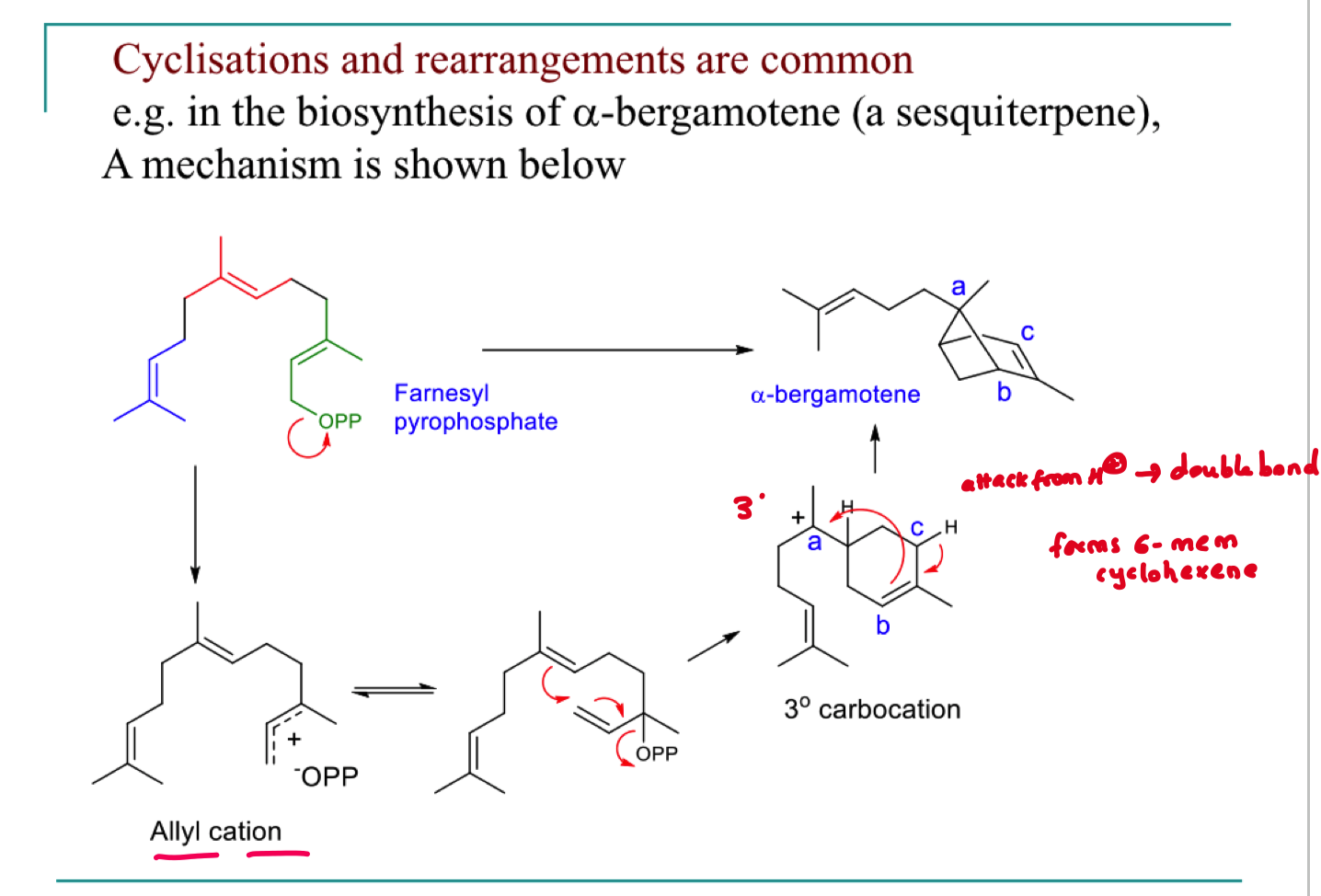
describe the rearrangements this undergoes to form the product
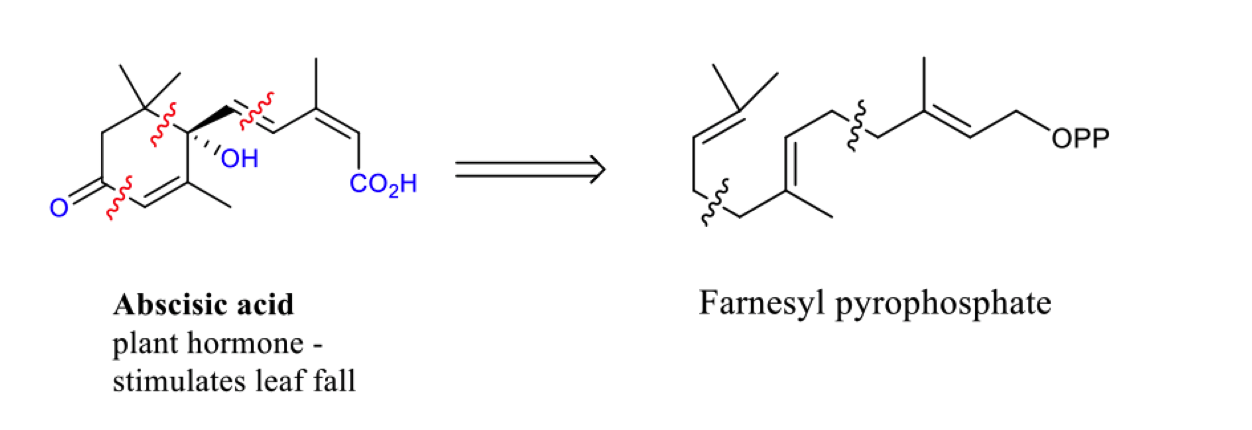
describe the biosynthesis of the sesquiterpene absicisic acid
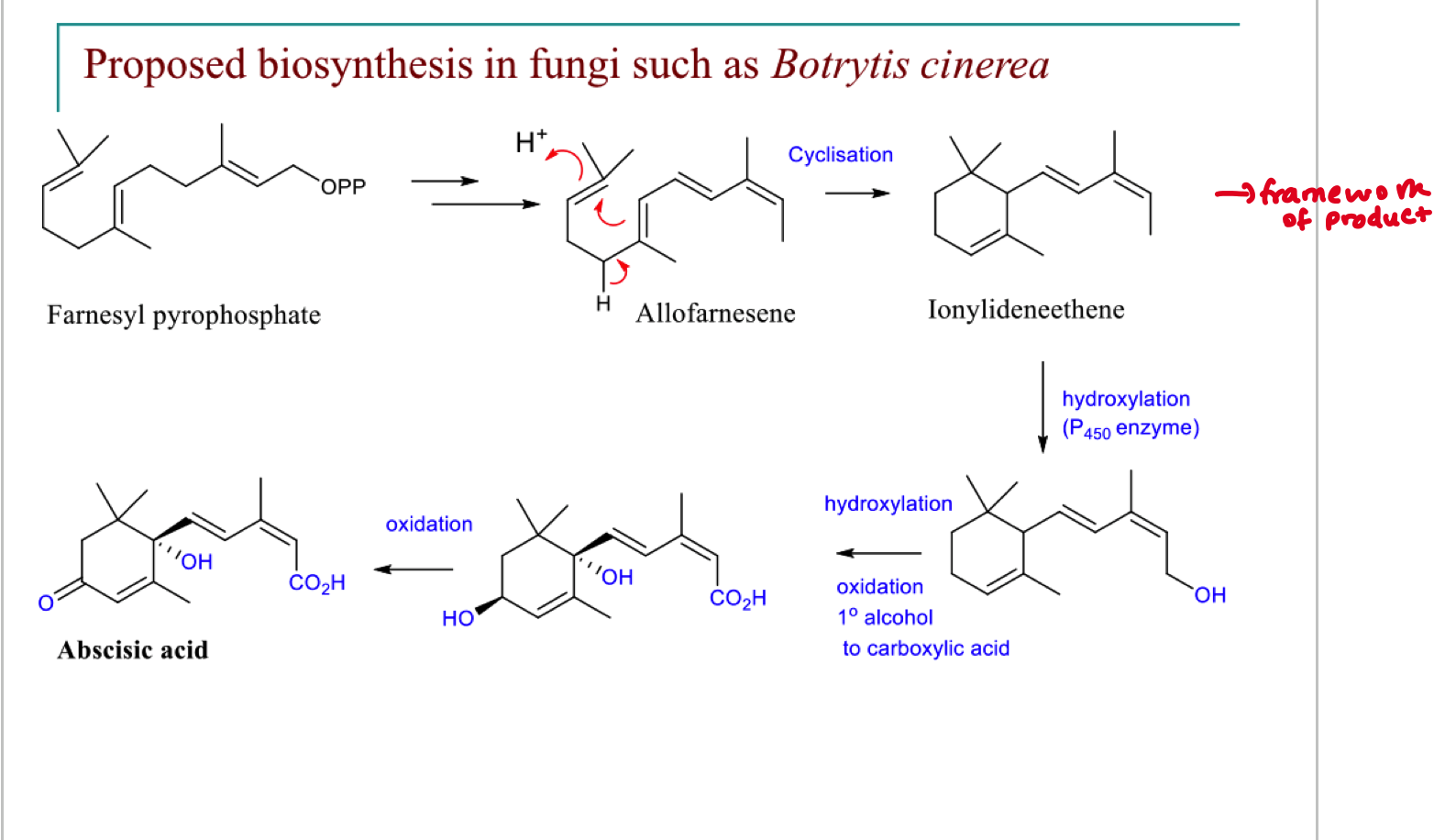
what are the two ways of introducing hydroxyl groups
what are primary metabolites ?
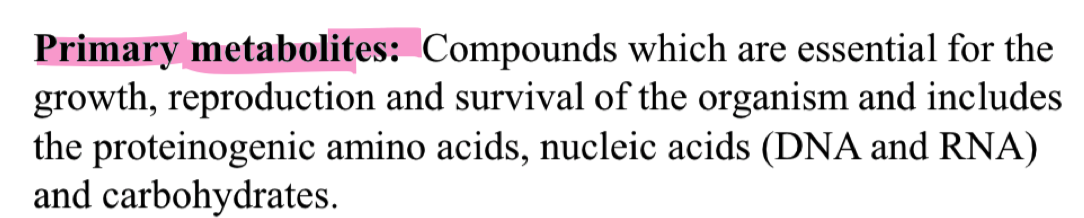
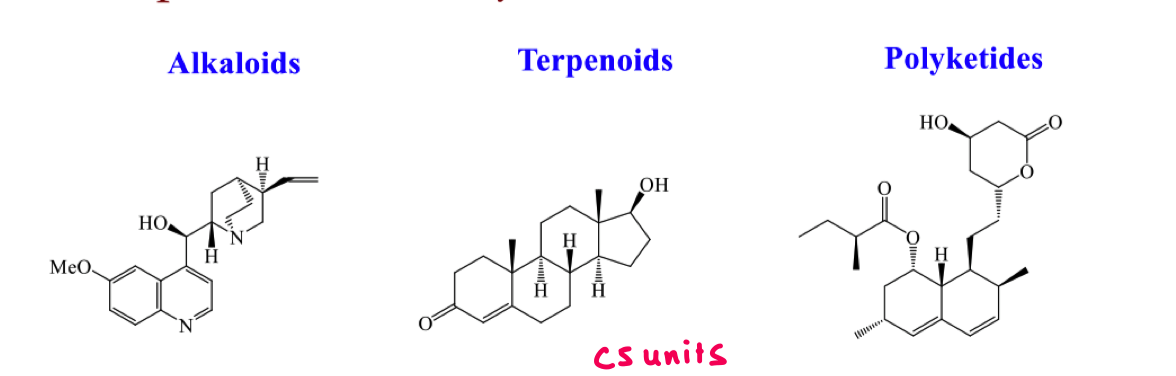
what are secondary metabolites?

what are the 3 different classes of secondary metabolites?
what two things are commonly involved in biosynthesis? and where does this biosynthesis occur?
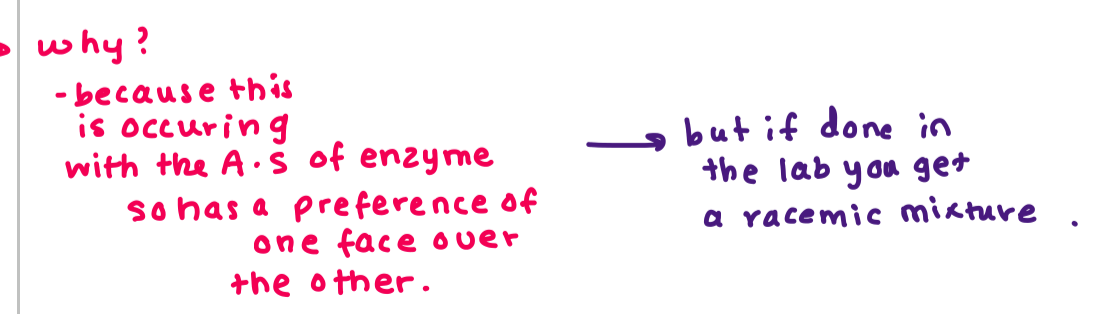
biosynthesis occurs in the active sites of enzymes and allows us to do things which otherwise would be difficult in the lab

what is the role of NADH/NADPH? Draw the structure of NADPH
acts as natures reducing agent by delivering a hydride and enabling oxidation to occur forming NAD+
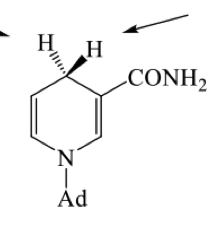
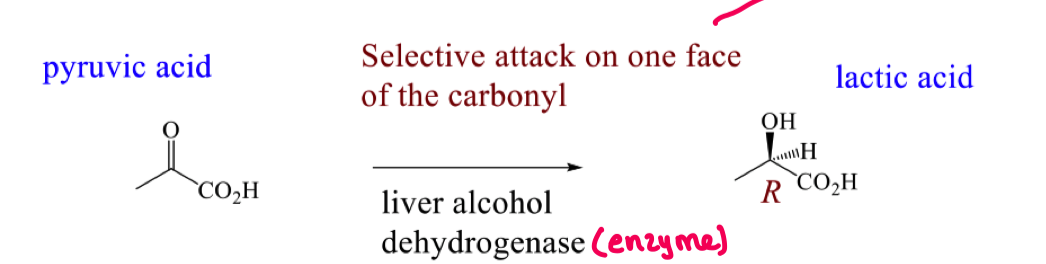
For this reaction why do we see selective attack on one face of the carbonyl?
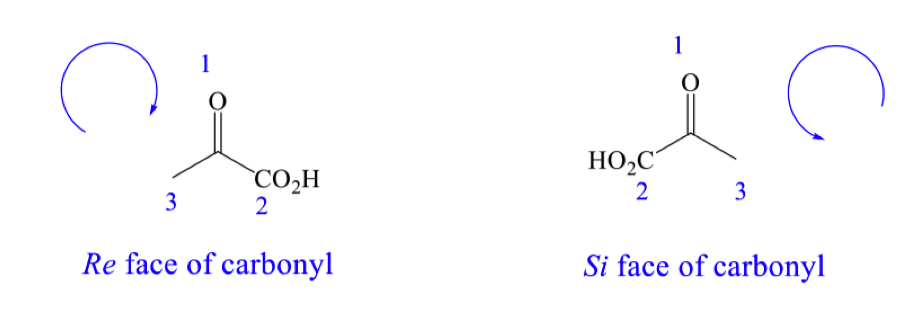
is this the Re or Si face ? draw the other face and label
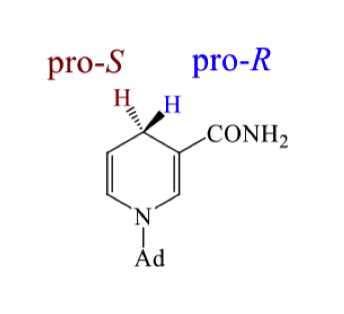
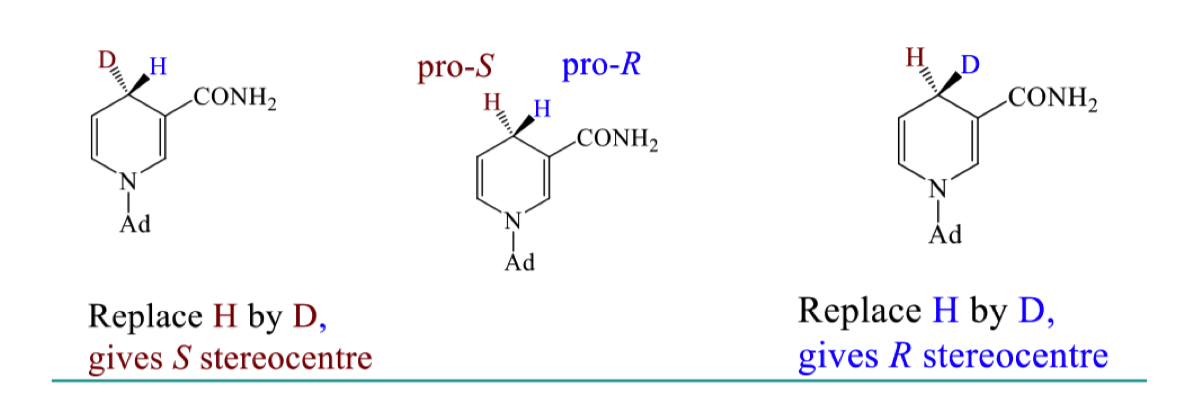
how can we determine which is Pro S and which is Pro R? determine it
we can replace one H by deuterium and use cahn ingold prelog rules to find which stereochemistry we get
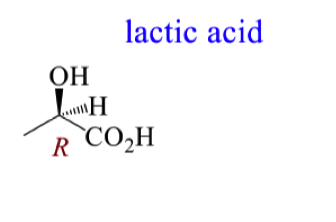
Draw the mechanism for the attack of NADH on pyruvic acid to form lactic acid
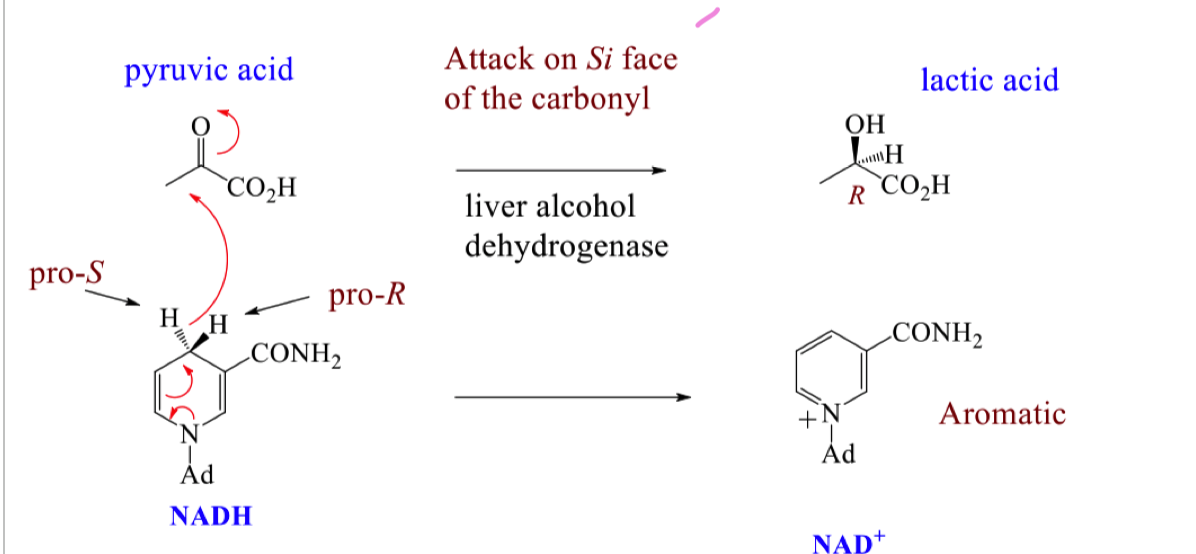
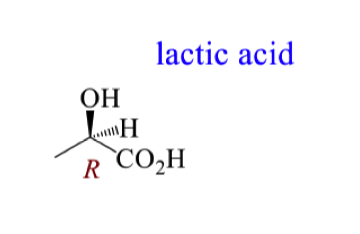
For the mechanism of attack on pyruvic acid, how do we know the si face is attacked by looking at the stereochemistry of the product?
In the product the hydride is going backwards and the OH is coming forwards
this means the NADH must have delivered the hydride from the backside of the pyruvic acid (si face)
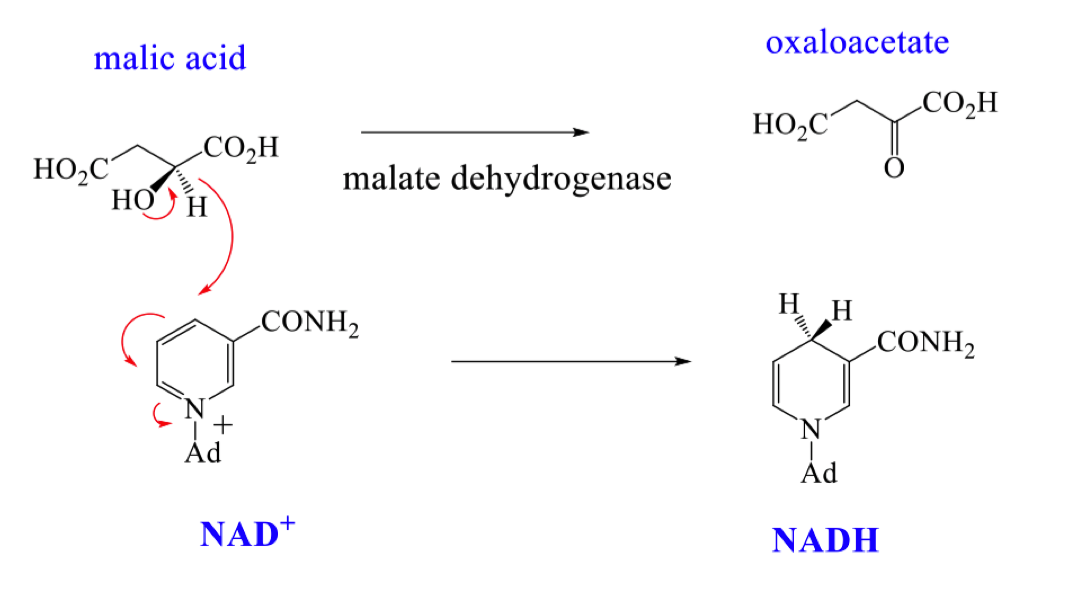
explain how the reverse reaction works and draw the mechanism for enzyme catalysed oxidation with NAD+
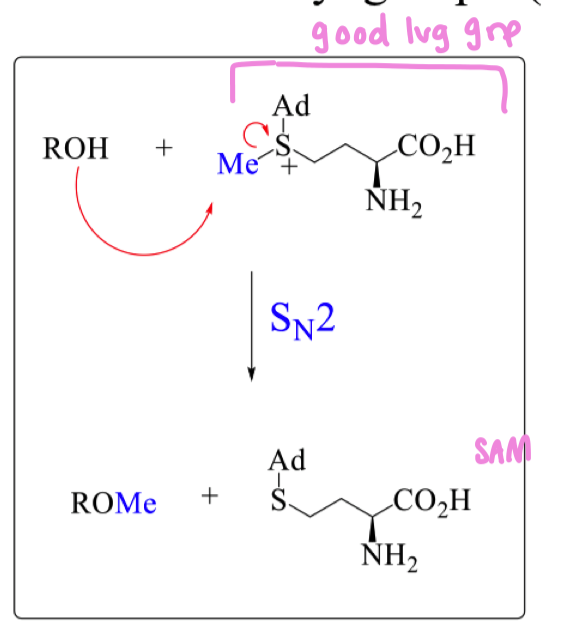
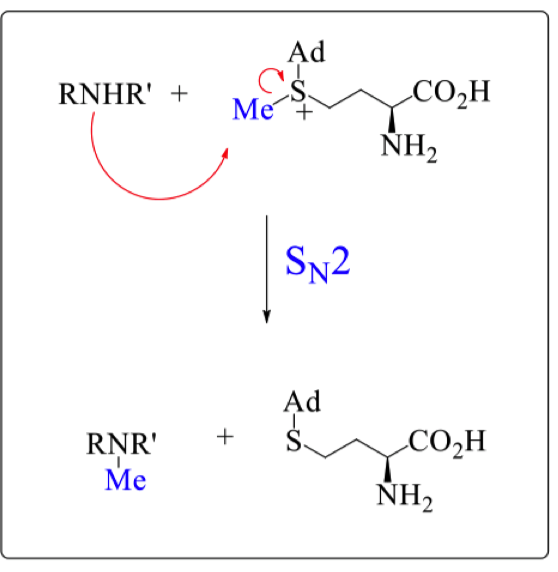
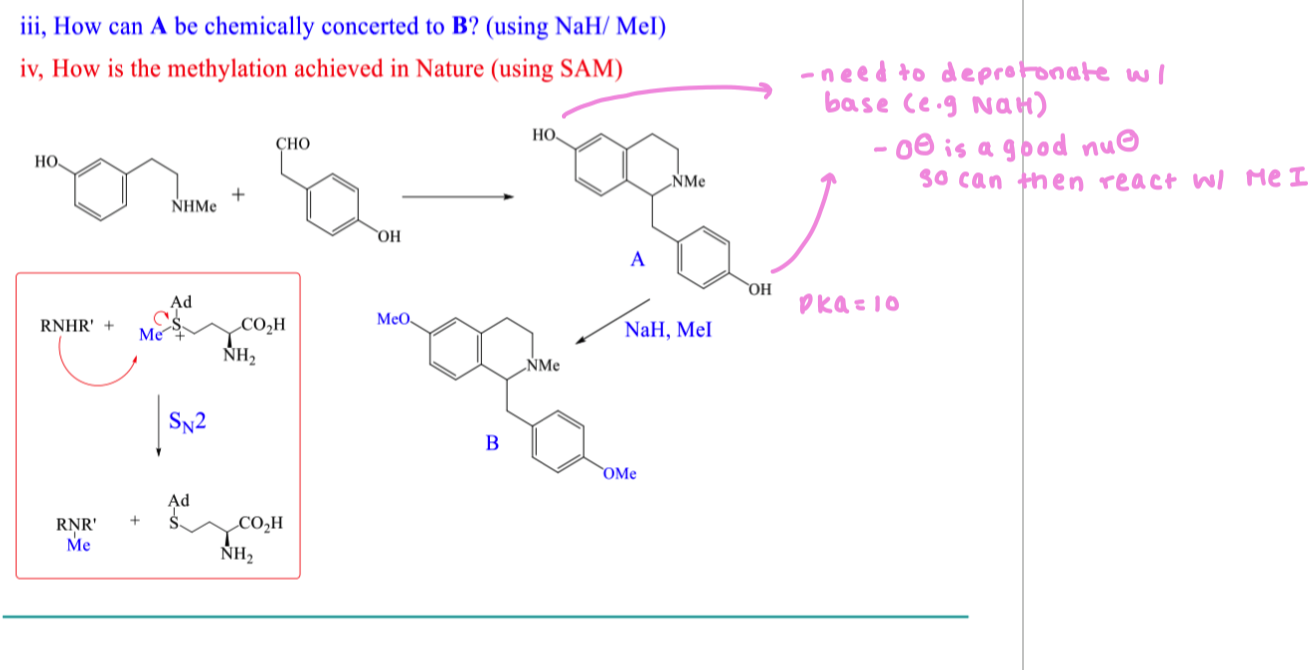
what is SAM (S-Adenosyl methionine) used for in nature?
Used to introduce methyl groups
draw the mechanism of ROH with SAM
draw the mechanism of RNHR’ with SAM
What is an alkaloid
Alkaloids are non-peptidic and non-nucleosidic natural products containing nitrogen
How do we classify alkaloids?
they are classified according to the amino acid from which they are derived
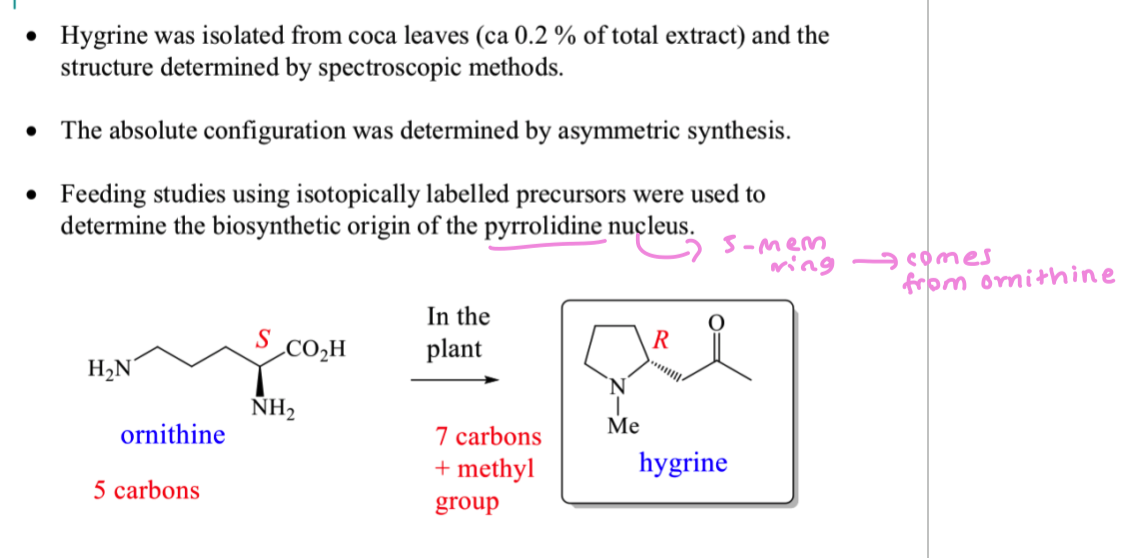
where do pyrrolidine alkaloids come from?
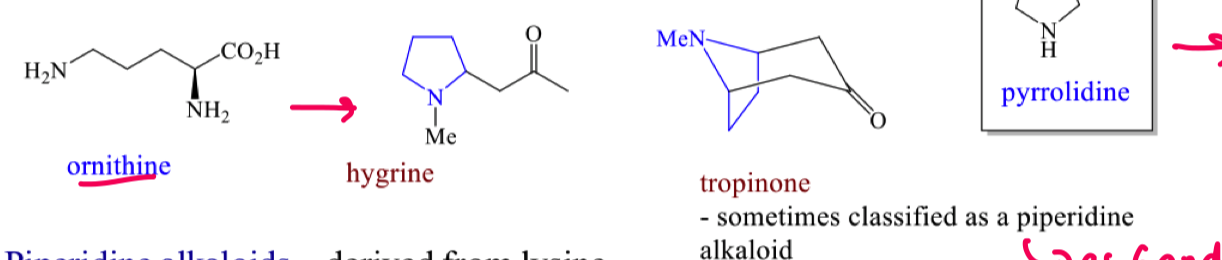
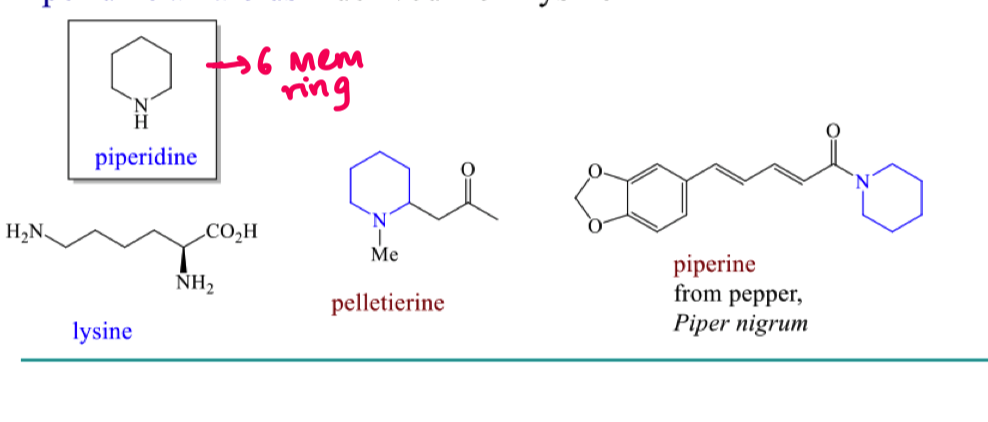
Where do piperdine alkaloids come from?
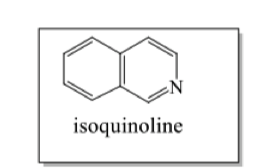
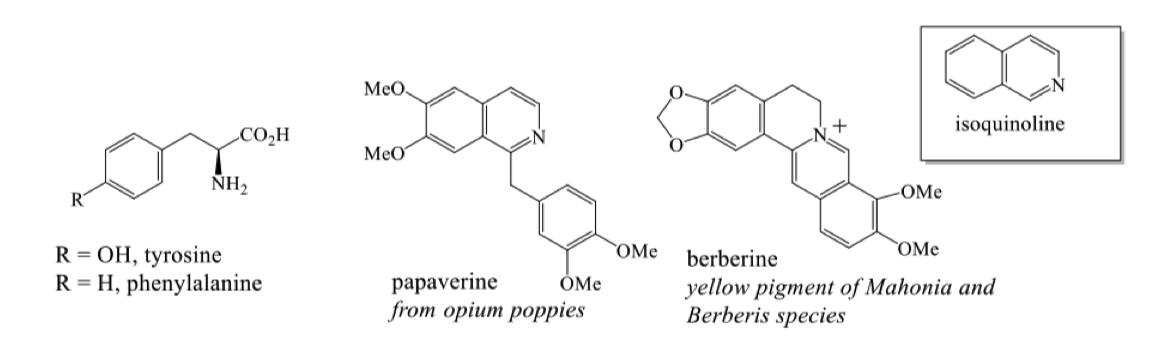
where do benzyl isoquinoline alkaloids come from?
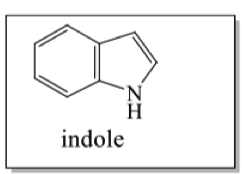
where do indole alkaloids come from?
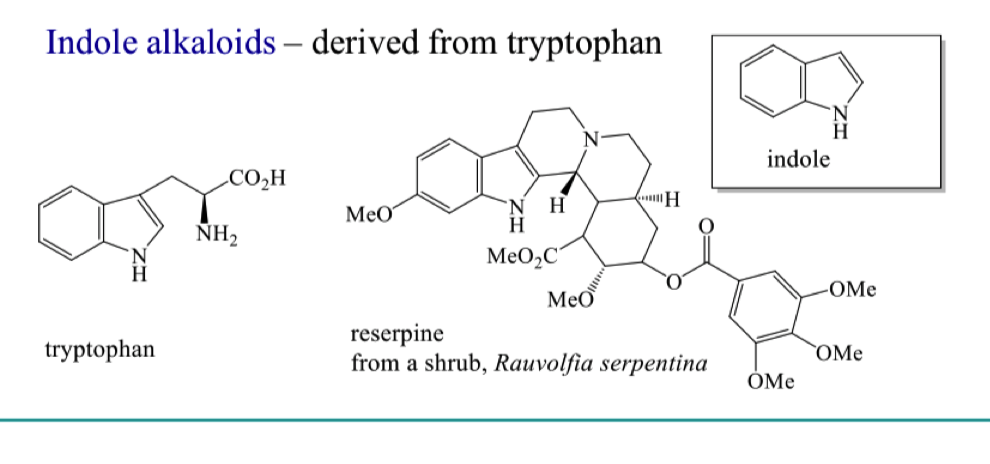
why is this classification system not the best?

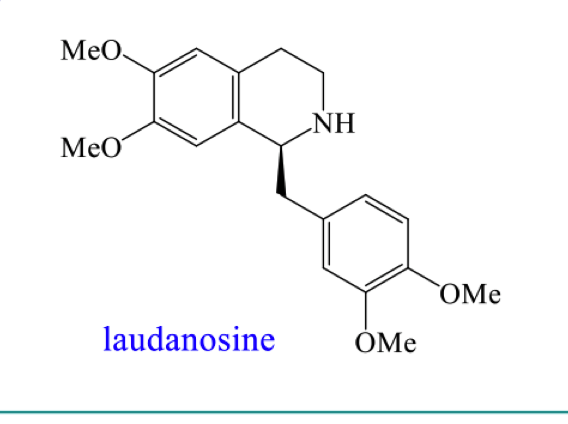
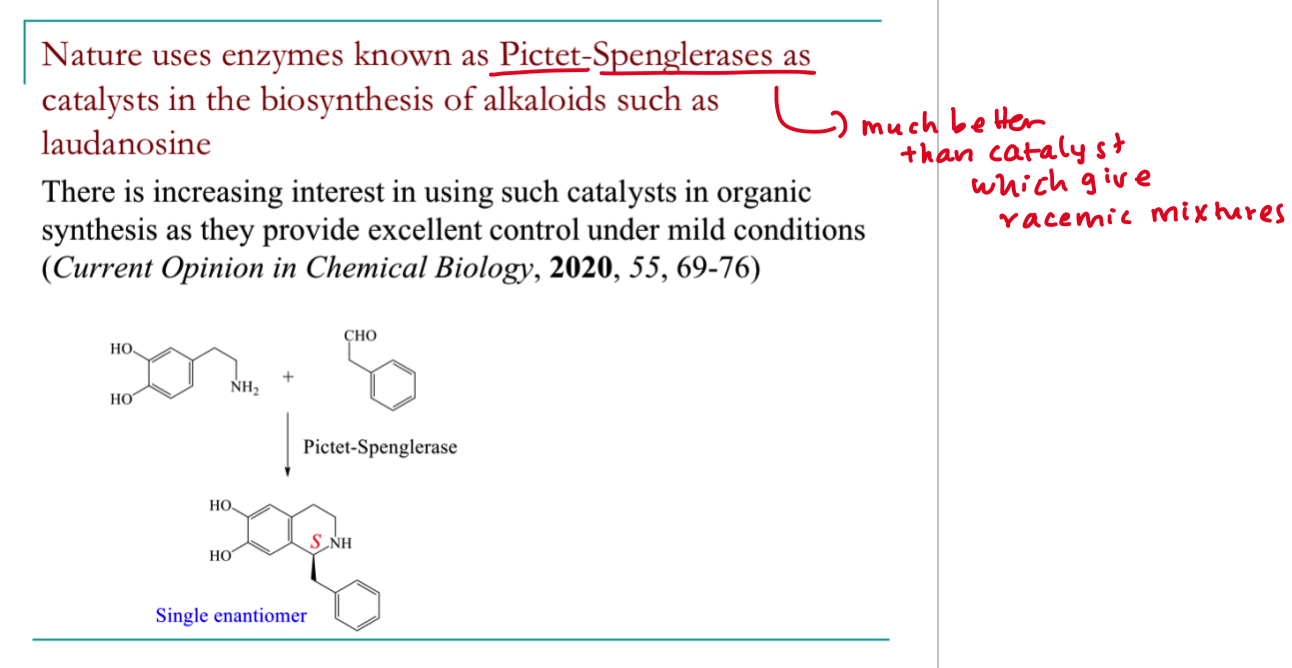
what is laundanosine?
its a type of tetrahyroisoquinoline alkaloid
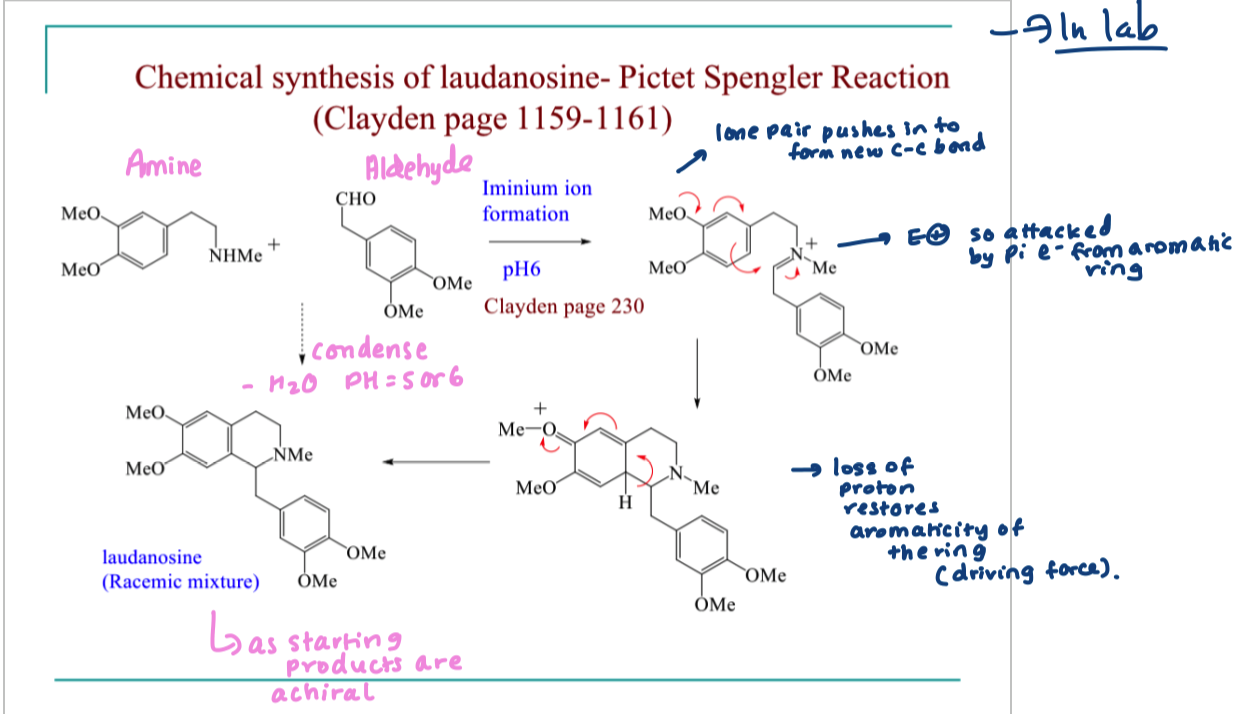
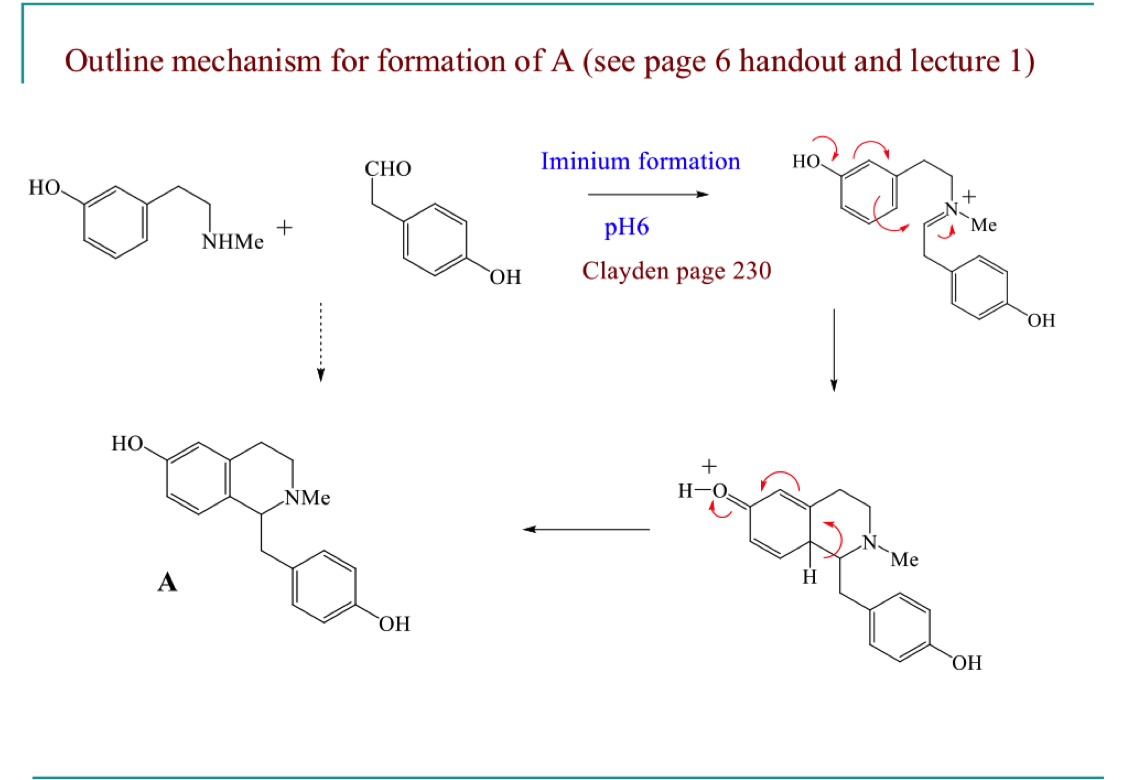
describe the in lab synthesis (pictet spengler reaction) of laundanosine
involves amine reacting with an aldehyde
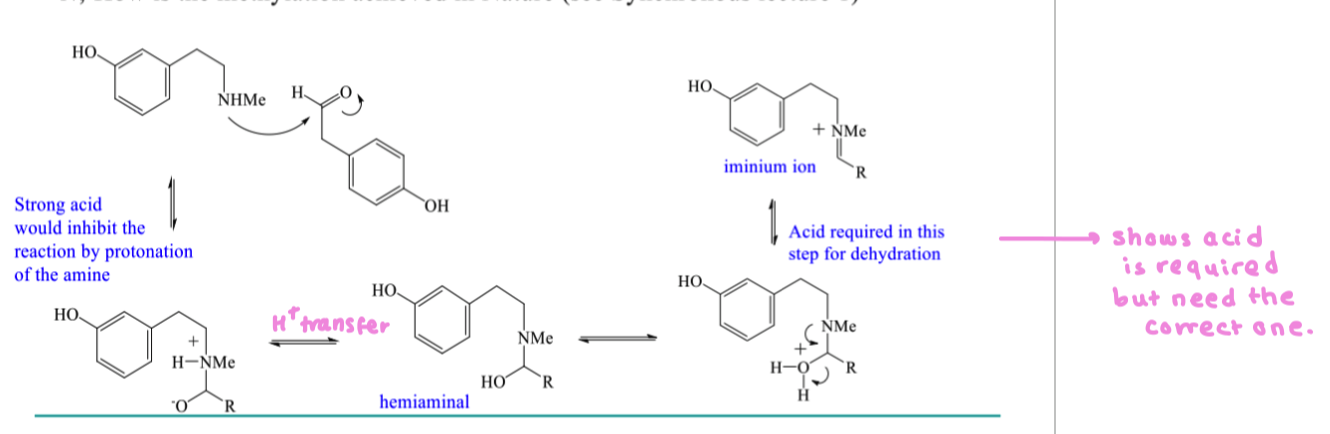
condense at pH 5/6
loss of water
forms a racemic mixture as staring materials are achiral
draw the mechanism of pictet spengler reaction
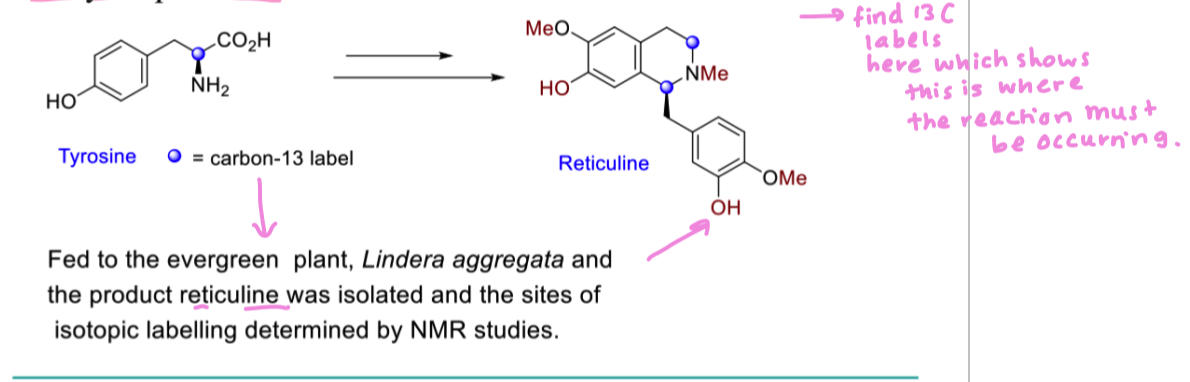
how can we isotopic labels to see how alkaloids are produced in nature
isotopic labels act as tracers to monitor the enzyme catalysed process
which enzyme does nature use in the pictet spenglerase as a catalyst and why?
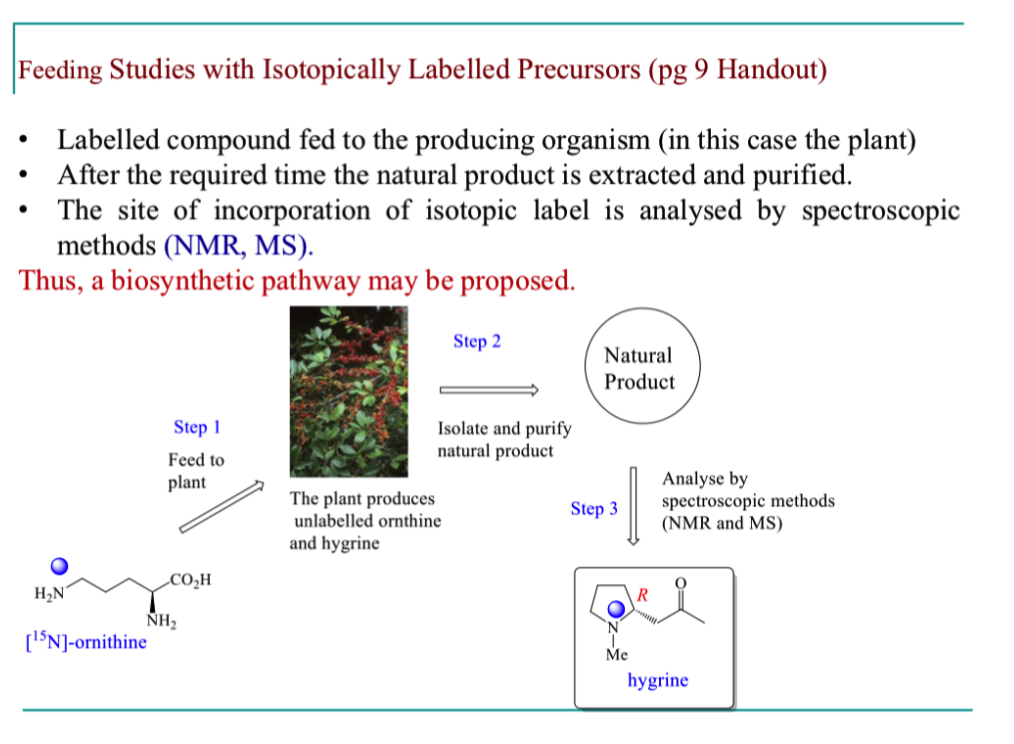
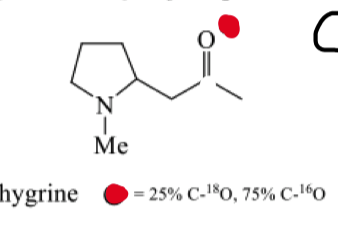
How can we invetsigate the biosynthesis of hygrine?
explain how feeding studies with isotopically labelled precursors work
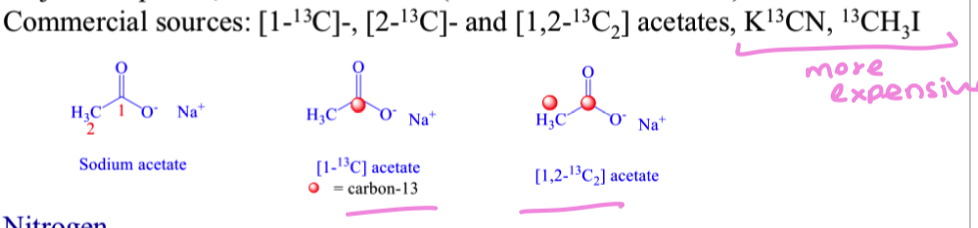
what is a stable isotope of carbon to be used in biosynthetic studies?
13C (1.1% natural abundance)
what are stable isotope labels of nitrogen
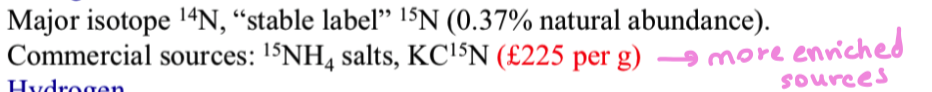
what is a stable isotope label of hydrogen?

what is a stable isotope label of oxygen?
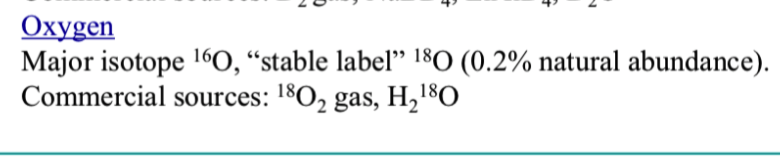
why can we not mass spec to analyse stable isotopes for biosynthesis?
natural product contains a relatively small amount of our fed precursor

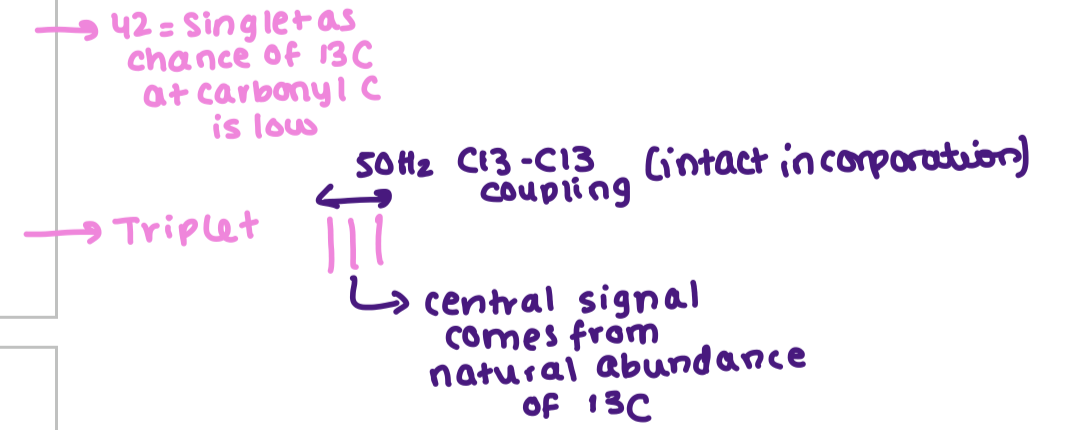
how does 13C NMR analyse stable isotopes? what percentage of incorporation does it work at?
Sites enriched with 13C will have more intense signals
has to be minimum 2%
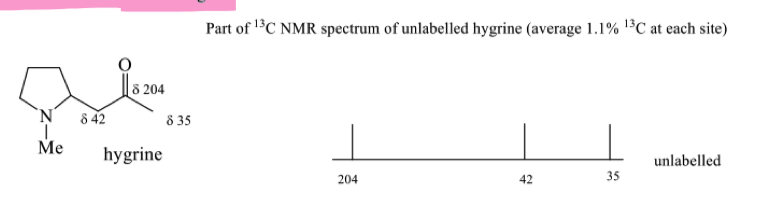
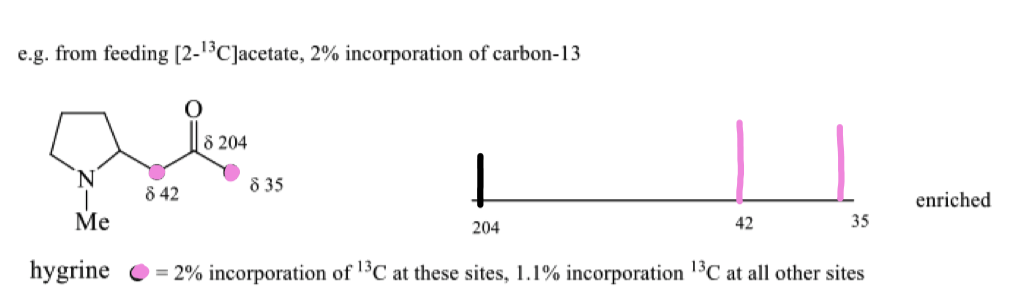
draw the peaks of enriched hygrine
what is a more sensitive technique to use for 13C?
as long as the bond isnt broken we will see coupling of the carbons

draw the nmr of hygrine
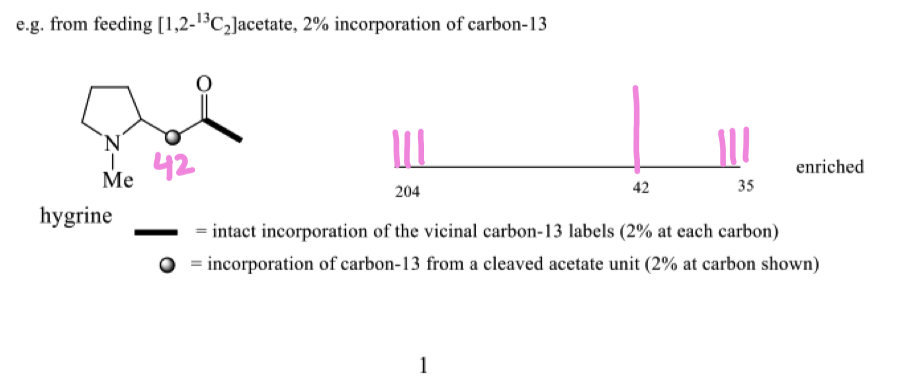
from the nmr of hygrine explain why 42 is a singlet? and the triplet
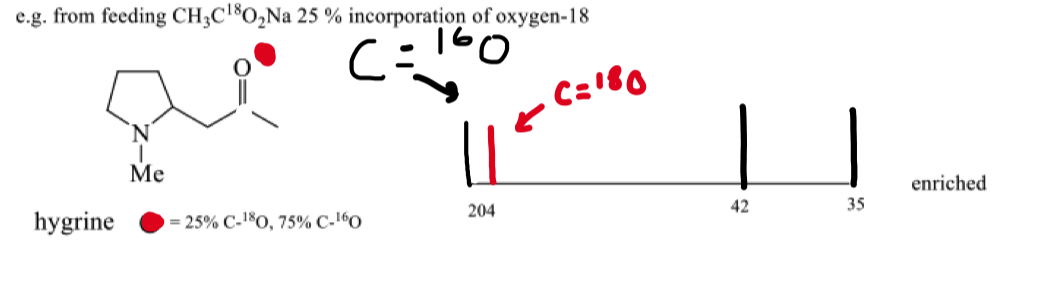
how can oxygen 18 enriched compounds be analysed ?

draw the 18O nmr for hygrine
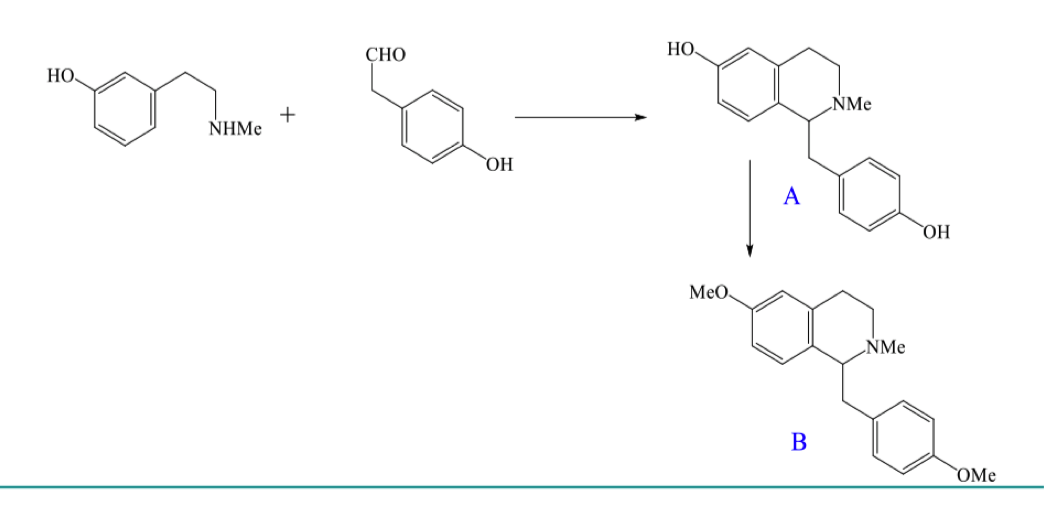
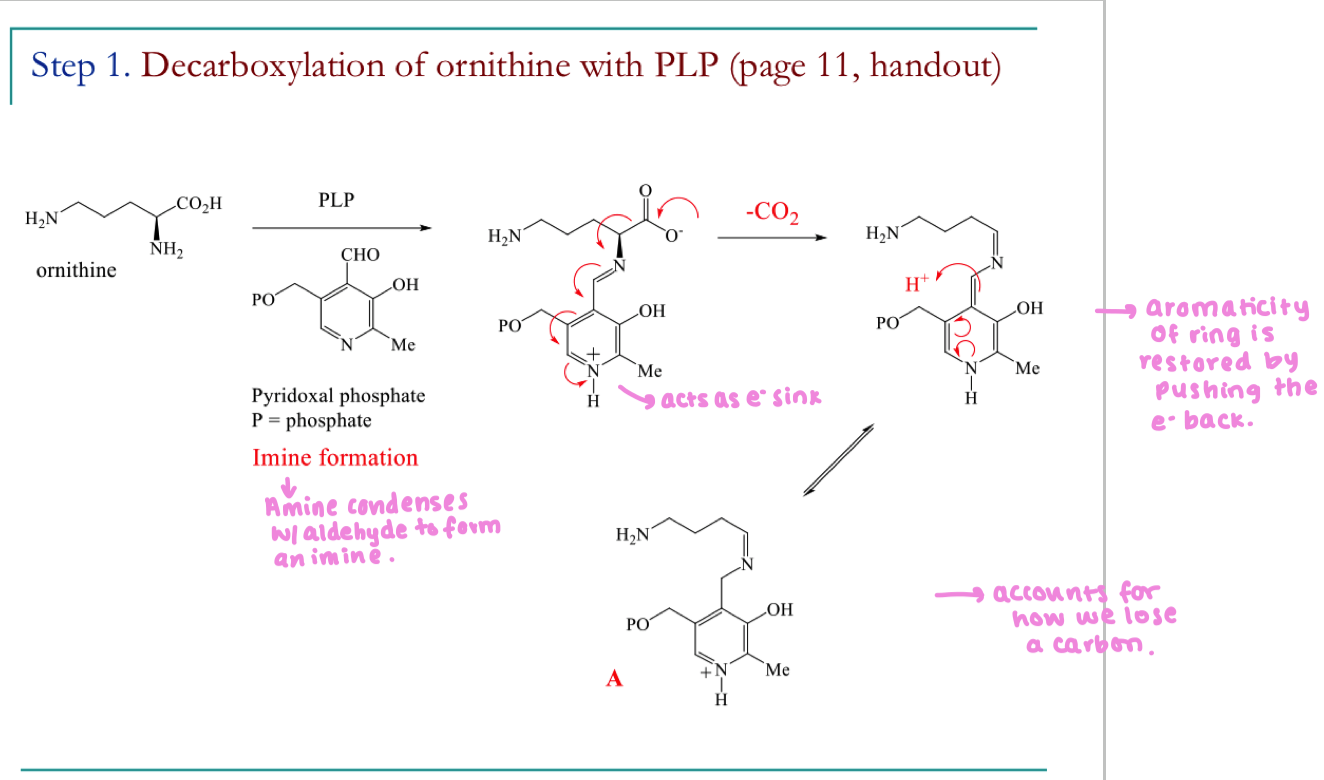
Give the full mechanism for the formation of A


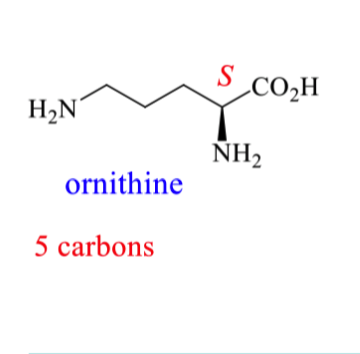
How can we prepare ornithine incorporating isotopic labels?
Add 15N to the 2 NH2 groups
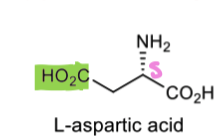
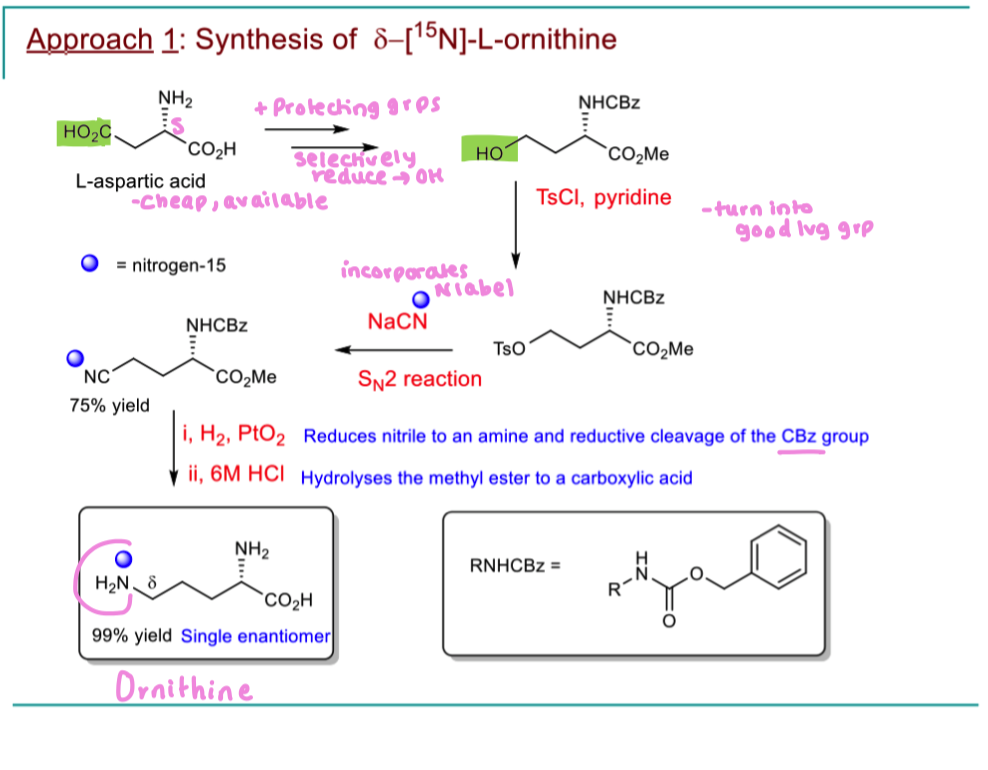
How can we put the 15N isotopic label on L ornithine (starts with aspartic acid)
what are the advantages of the 15N on L-ornithine pathway?
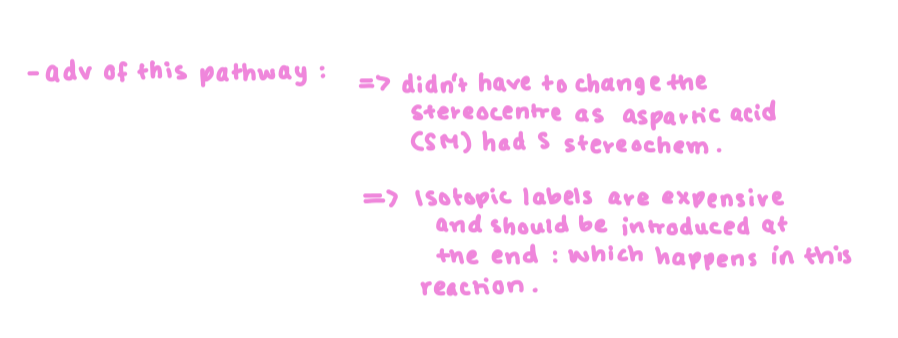
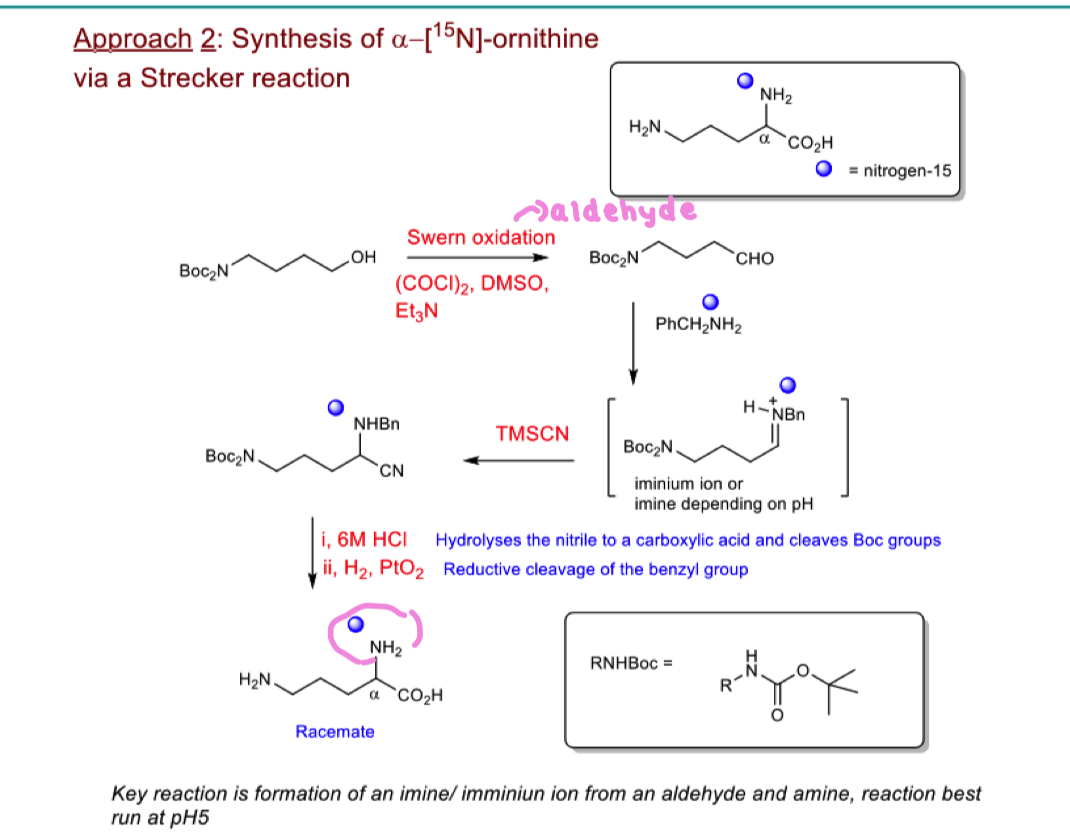
How to put 15N isotopic label onto alpha ornithine C
how can we separate the racemic mixture to get a single enantiomer?

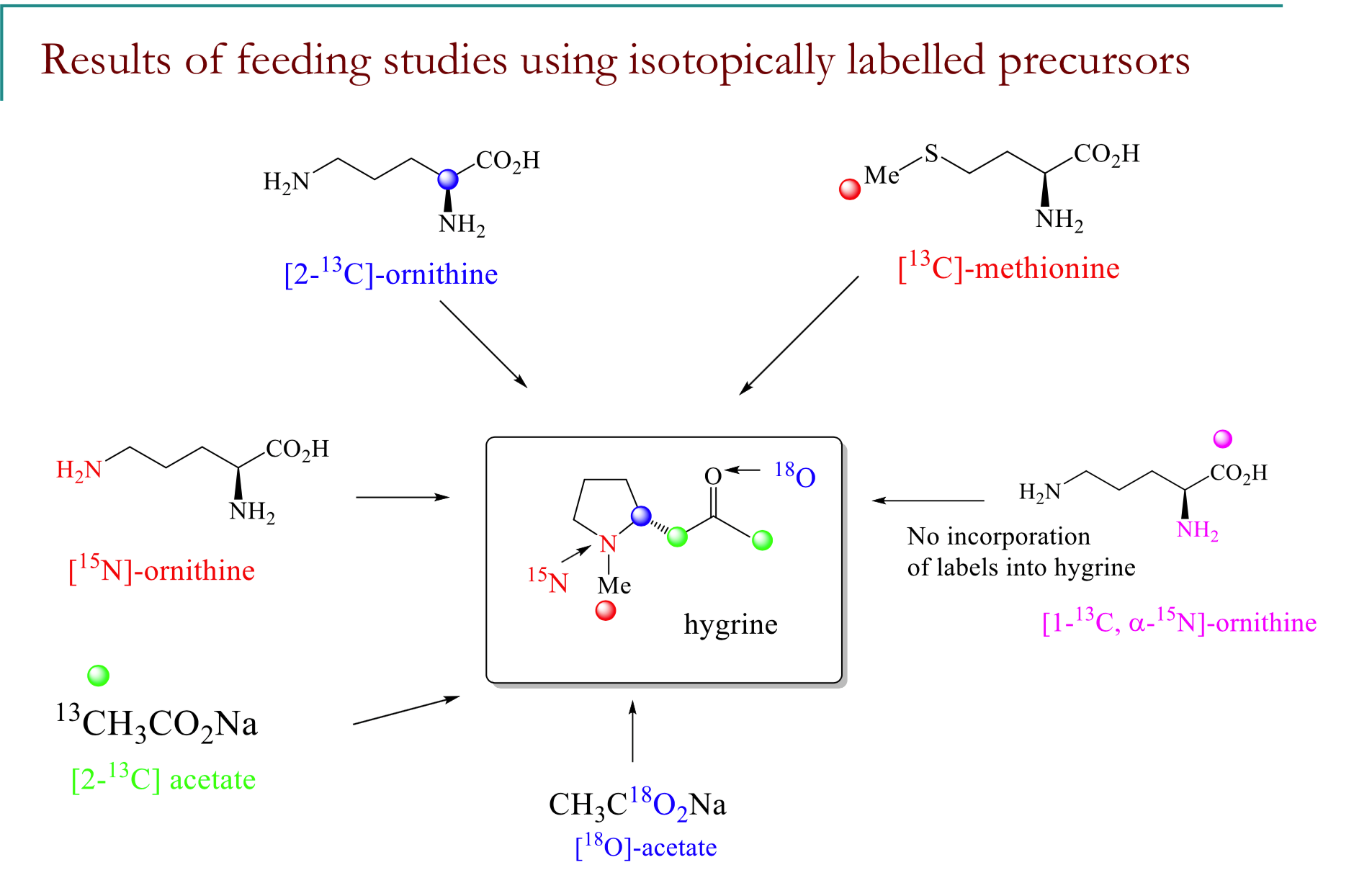
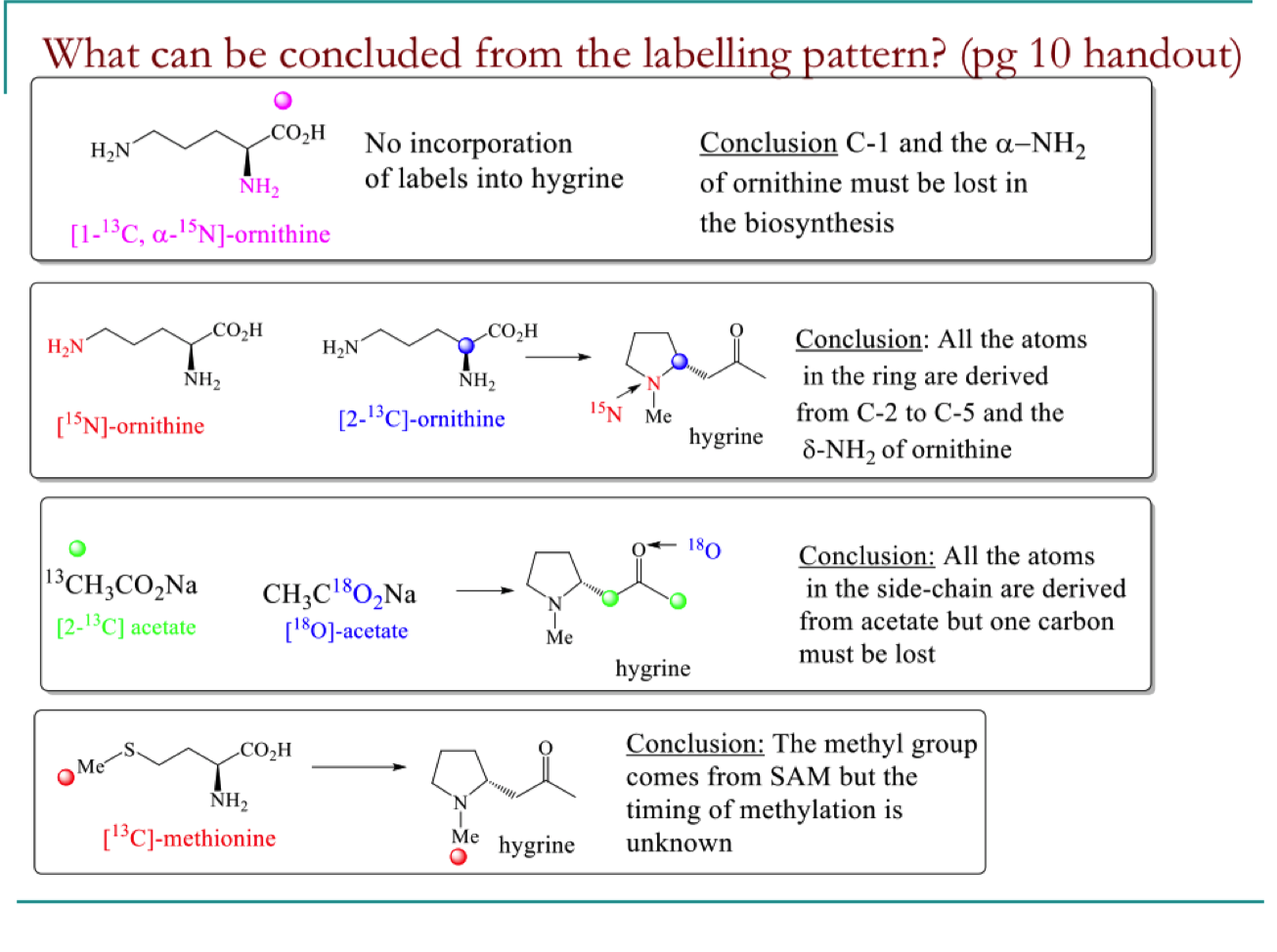
Explain the results of the feeding study
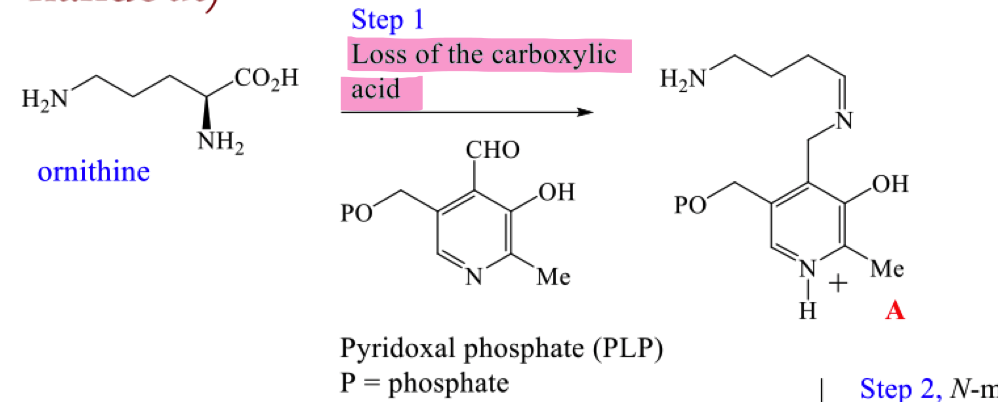
Draw the mechanism for the first step of the biosynthetic pathway to hygrine
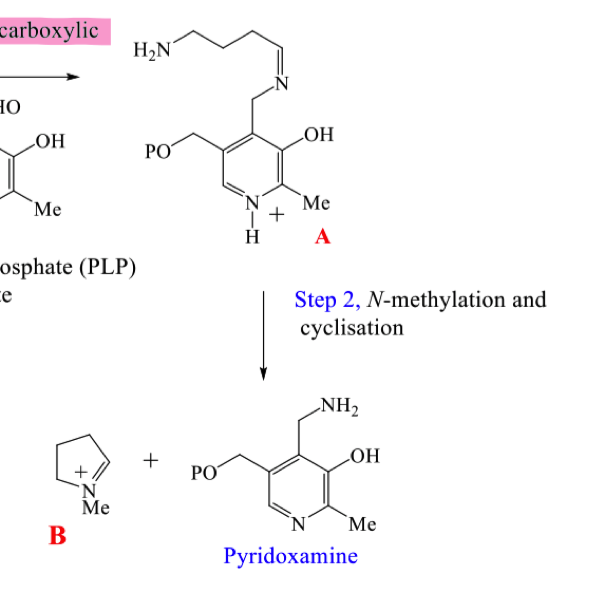
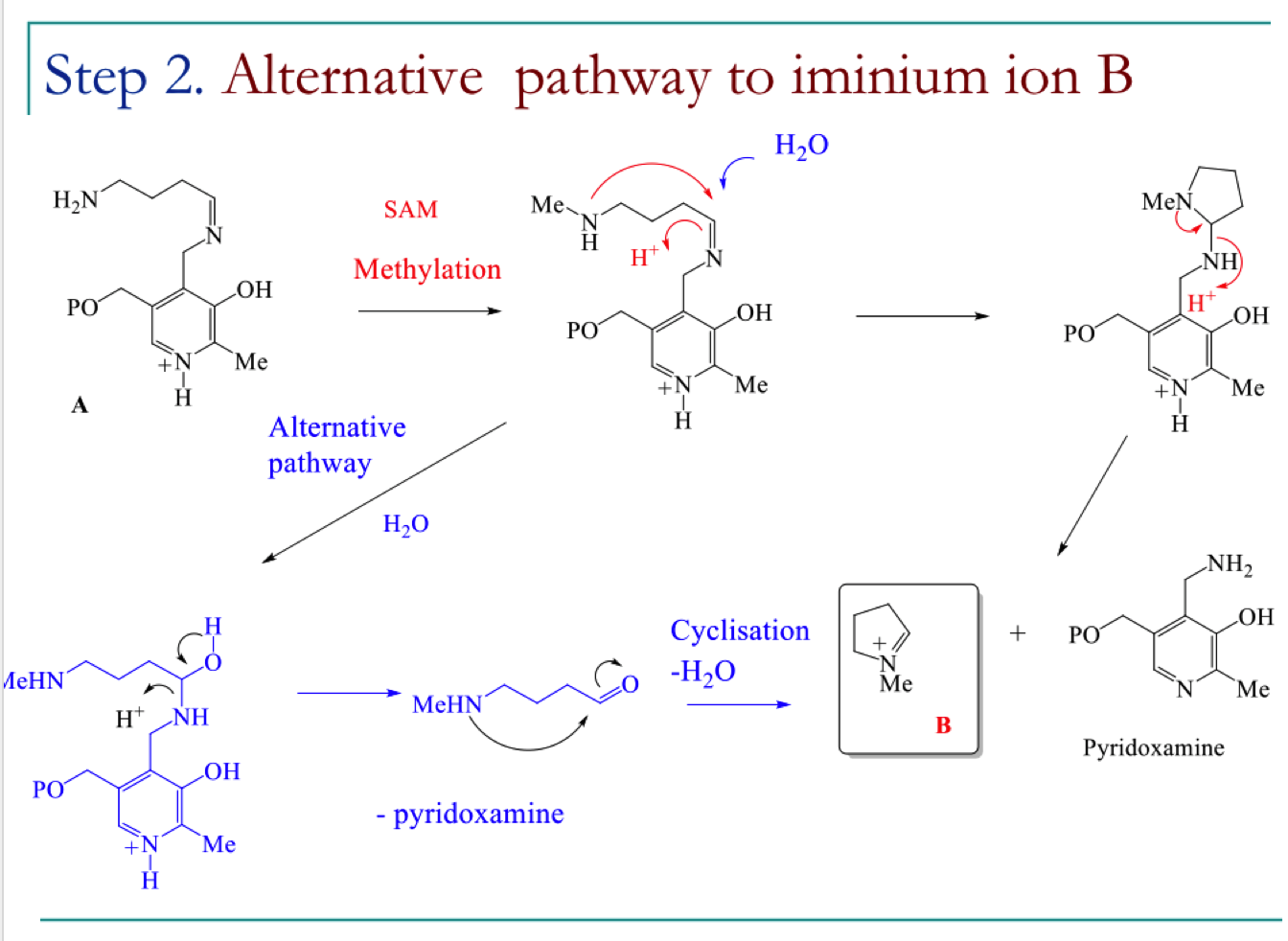
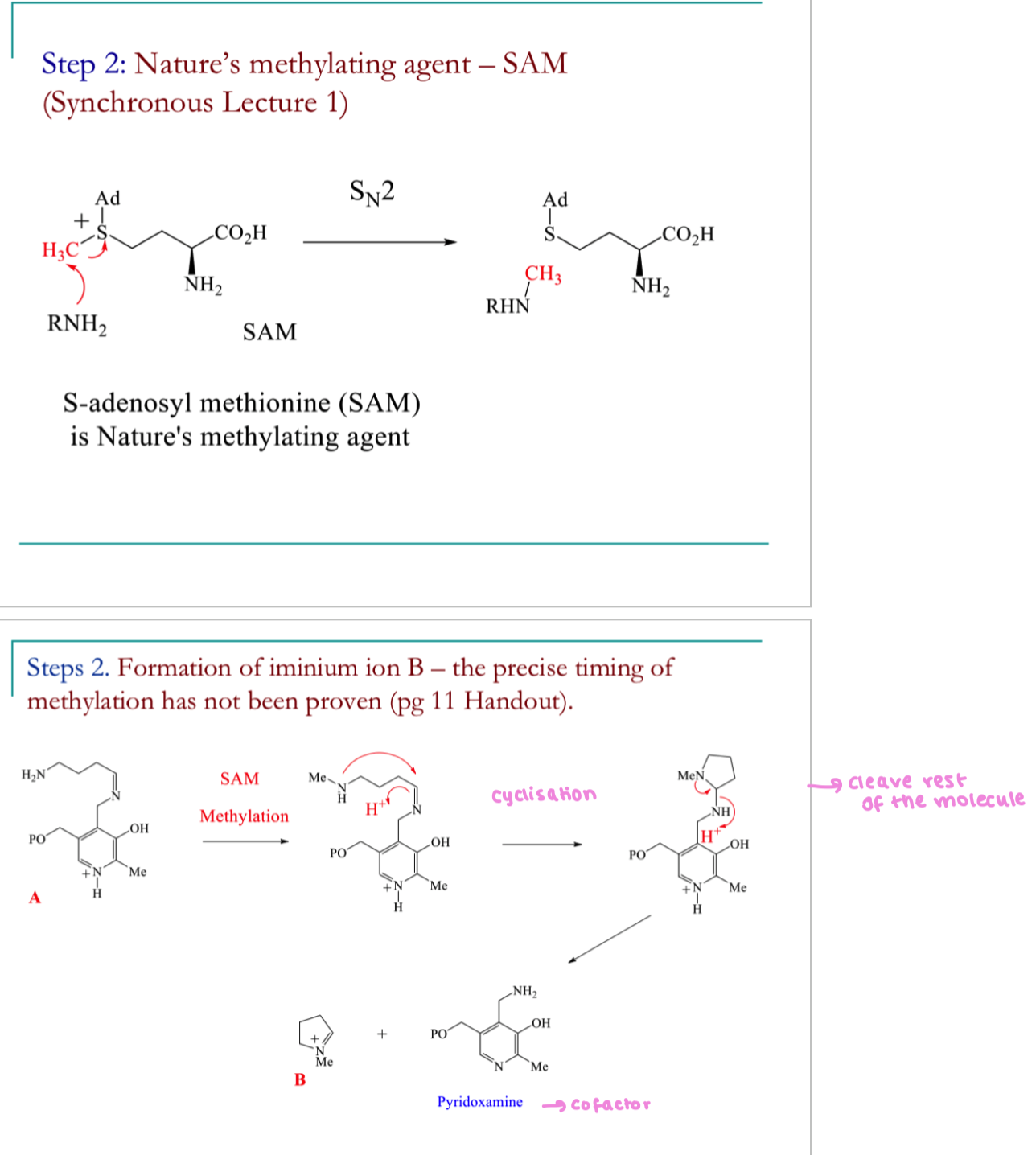
Draw the mechanism for step 2 of the biosynthetic pathway to hygrine
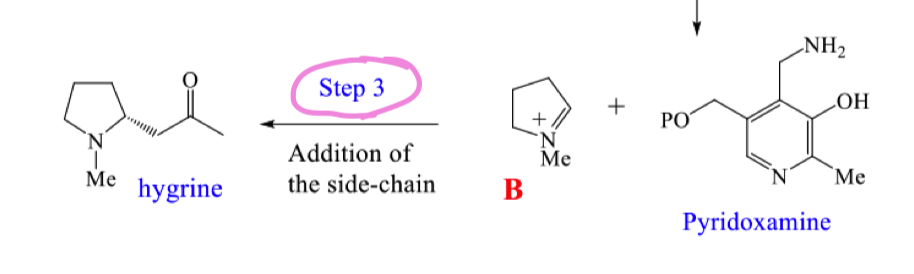
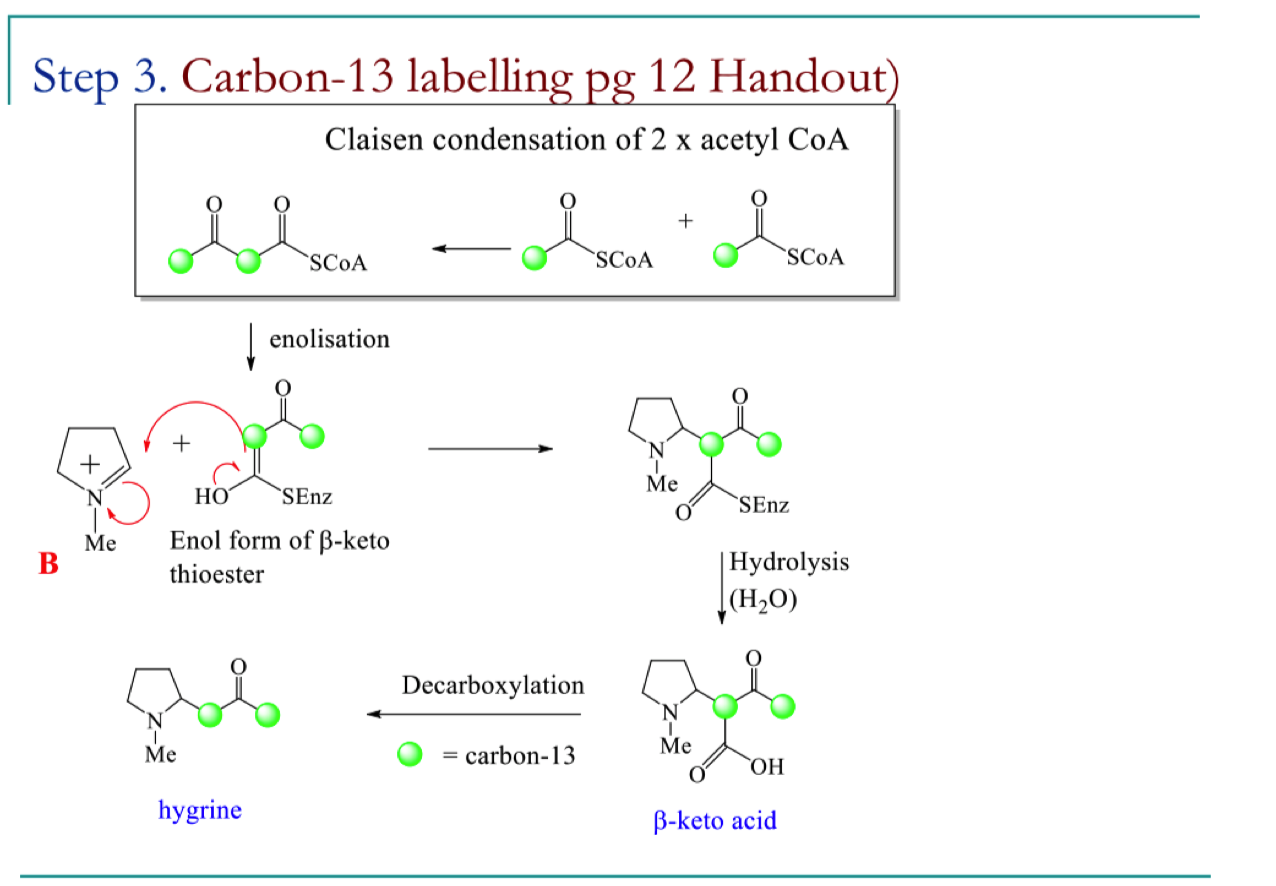
draw the mechansim for step 3 of the biosynthetic pathway to hygrine
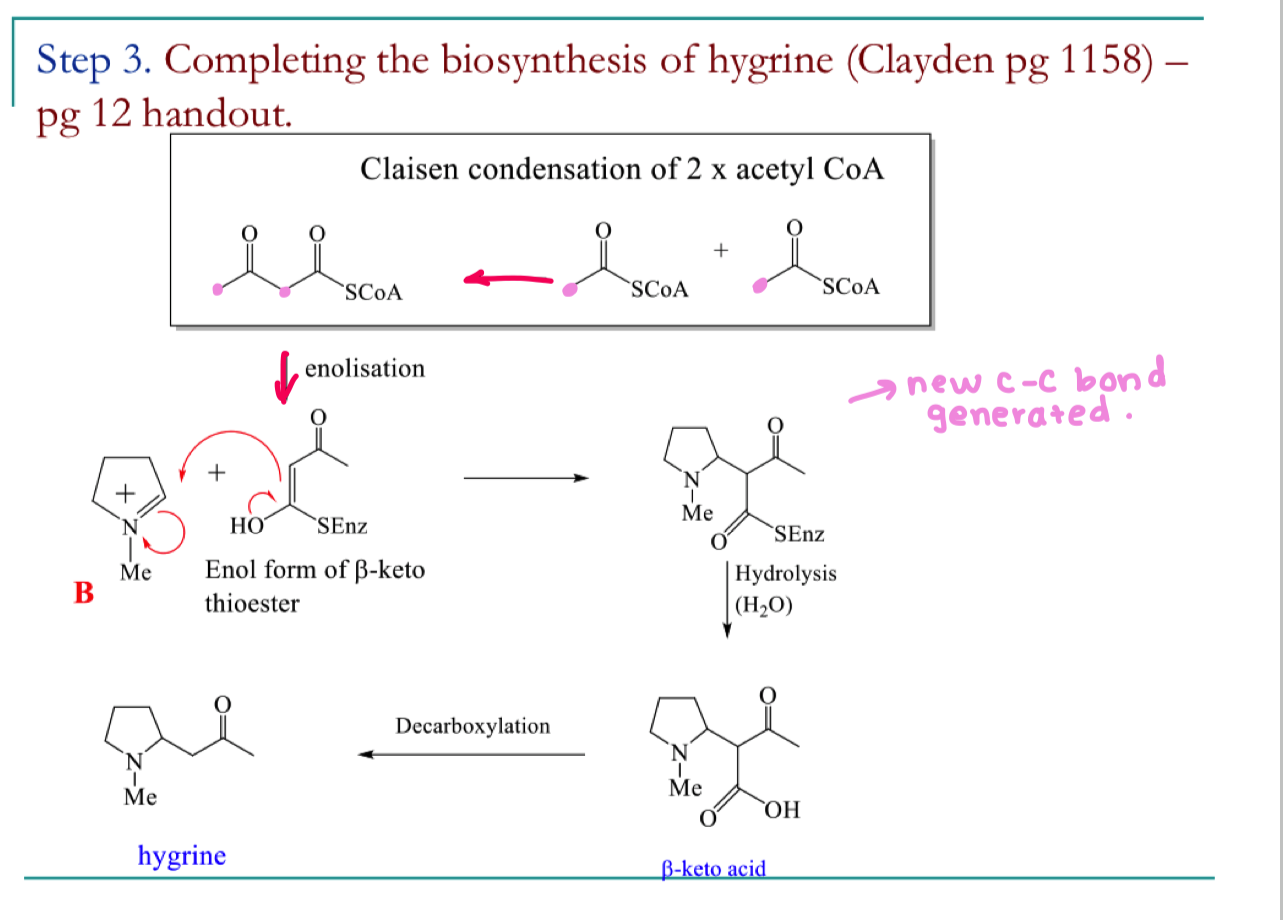
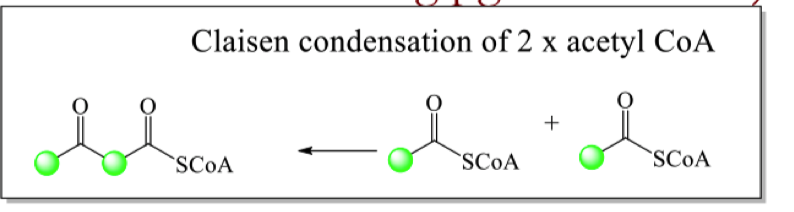
how can isotopic labelling of the acetyl CoA and the mechanism show that hygrine is made up of acetate
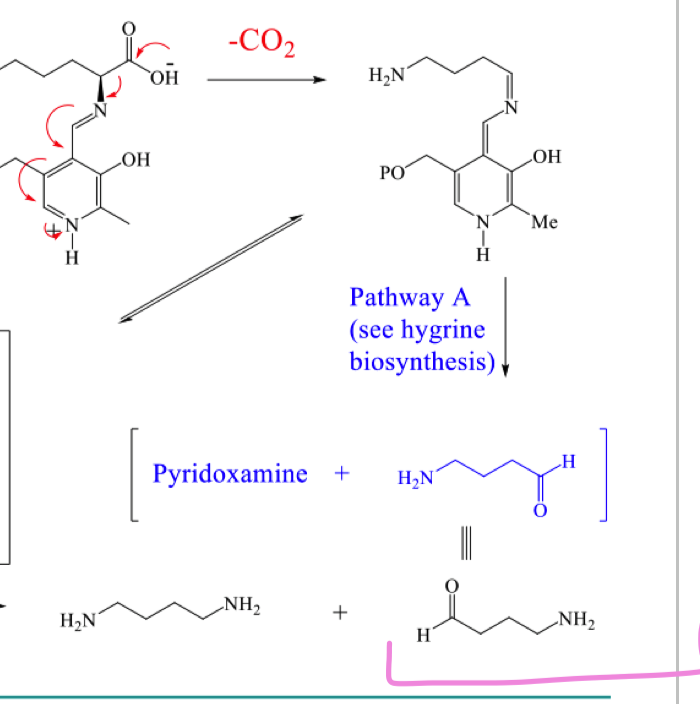
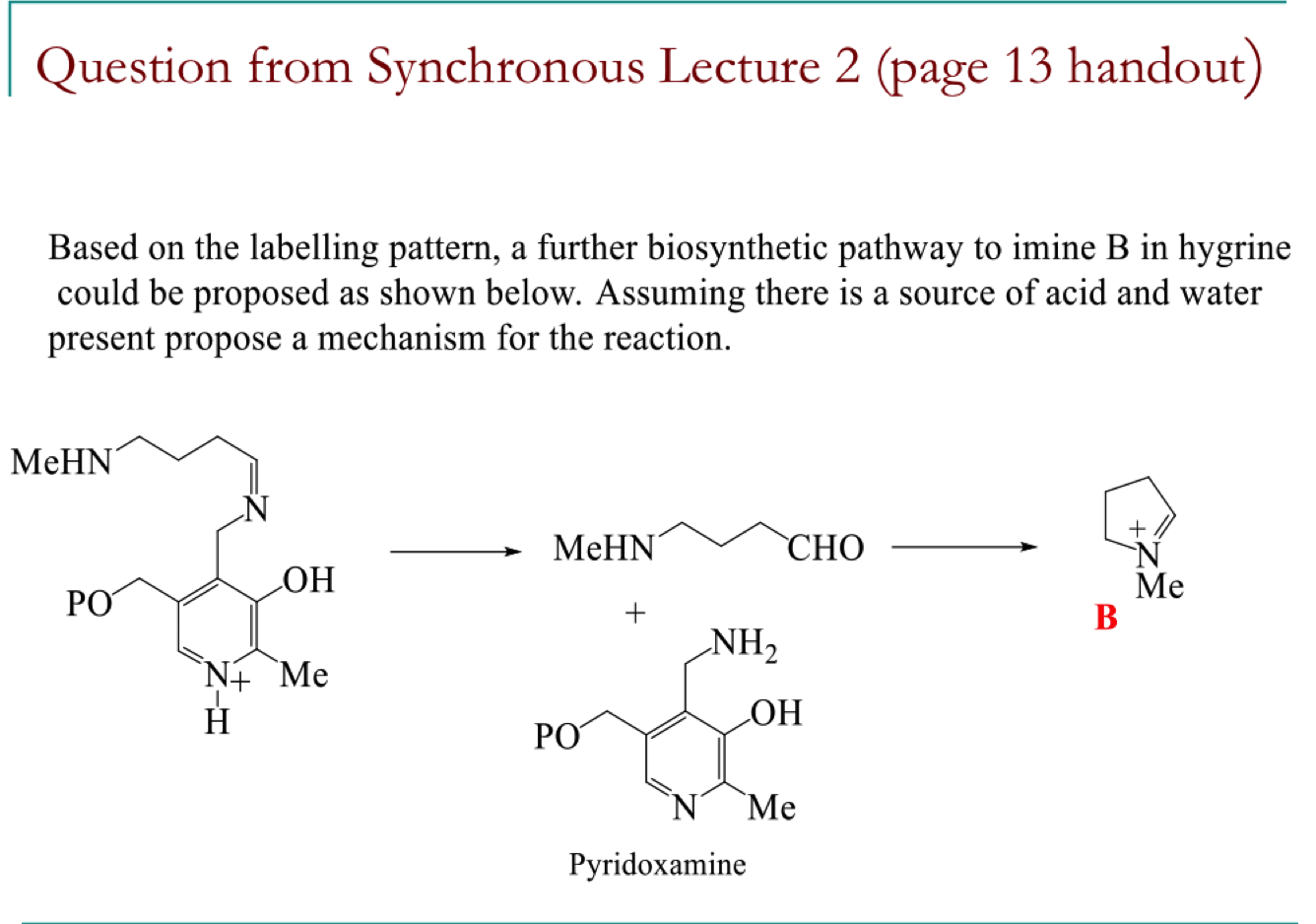
draw the mechanism of pathway B to form the 2 black products
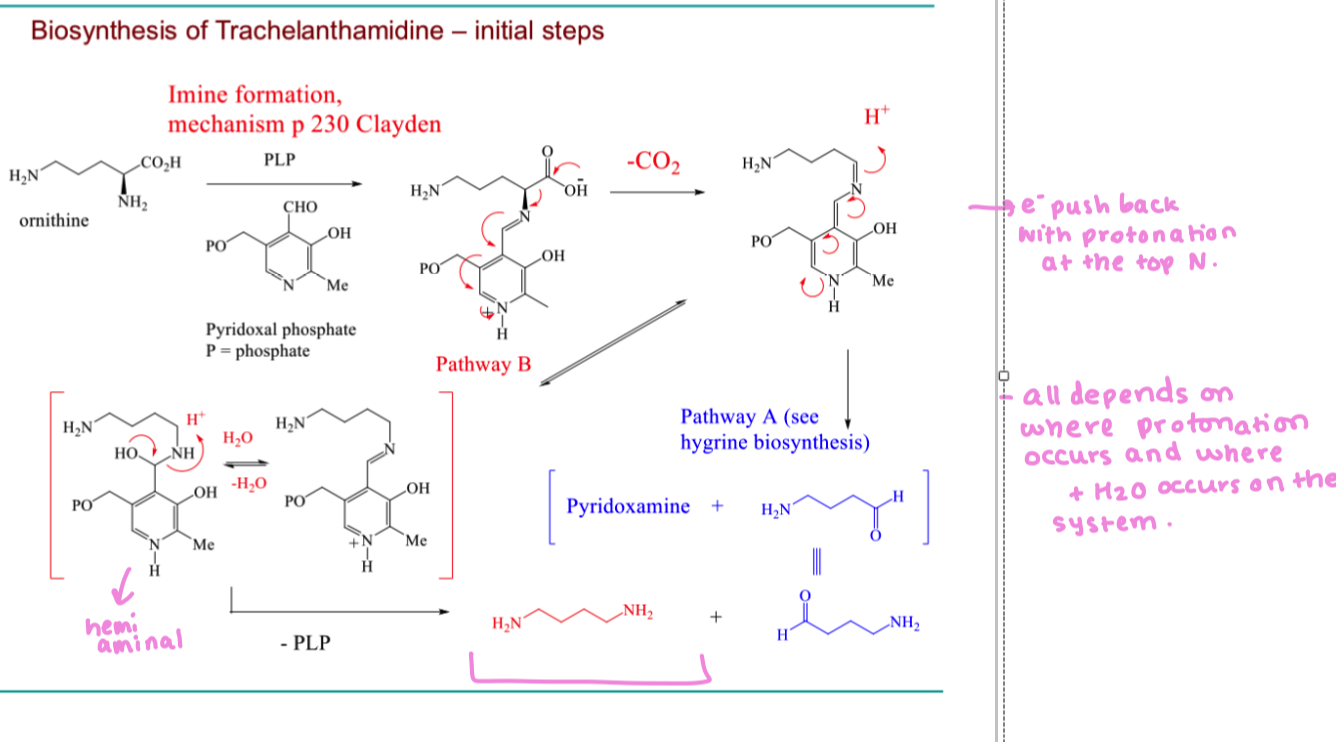
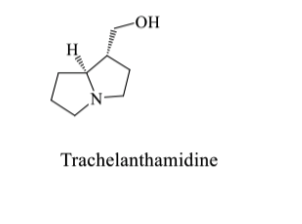
show the mechanism to form this cyclic product when two black products obtain react
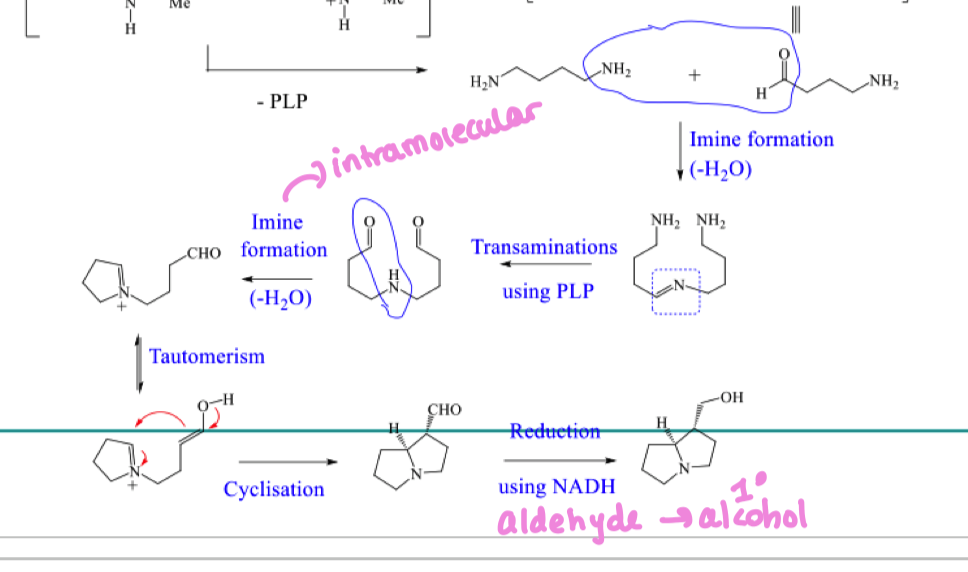
draw alternate mechanism to form the straight chain aldehyde