Phase Diagrams + Lever Rule + Other Rule
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Alloy
A mixture of 1 metal with 1 or more other materials
Component
An element within an alloy
Phase
A region within a material having uniform physical and chemical properties - i.e. must all be the same state but could be any state.
Composition
Mass of each component present in an ALLOY OR PHASE
Constitution
List of all phases, their masses and each of the phases composition
Phase diagram
Graph showing the physical state of a material system at different temperatures and compositions
Solvent
The majority component in a solution
Solute
Ute .:. Acute .:. The minority component of a solution
Solution
A mixture of homogenous composition in which the solute has fully mixed in with the solvent at all points in the sample
Solid solution
solid state + solution definition
Complete solid solubility
Means that in the solid state AT ANY COMPOSITION, the solute atoms are fully soluble in the solvent and therefore A SECOND PHASE WILL NOT FORM.
Limited solid solubility
Same as solid solubility except for not all compositions - must be below a certain solute concentration
Grain
A piece of material where every single atom is perfectly lined up in the same lattice pattern.

is this an example of complete solid solubility, limited, or no solid solubility?
Complete - to identify: you must look at all solid phase regions on the graph (usually split by solvus lines) - for this one there is only one and its a single phase solution that spans across any composition of the material - hence complete ss.

Name of the lines between phase regions
Phase boundaries
Solvus line
Line that marks the maximum SOLID solubility of one component in another (wrt temp) on a phase diagram.
(It therefore separates phase regions of the same state on a phase diagram (NOT DEFINITION THO))
Liquidus Line
Line above which whole system is liquid
Solidus Line
Line BELOW which all of system is solid
Invariant Point
Point at which the LIQUIDUS lines meet. (More on this pt later)
Lever rule purpose + method (NOT GIVEN)
P: to calculate the FRACTIONS of weights of each phase in a 2 phase region. (Decimal value)
M:
Draw a tieline within the 2 phase region at the specified temperature
Mark the specified composition (X) onto the tieline. (Note: composition tells you how much of each material is in the system - lever rule uses this to tell you how much of each PHASE is in the system)
Sub into equations:
M_alpha = (x_beta - X)/(x_beta - x_alpha)
where x_… = point of intersection of tieline and corresponding … boundary line between phase regions (could be liquidus, solvus etc).
NOTE: in the M equation for alpha you look at the beta side of the tie line (and vv for M_beta- not what you’d expect.)

How to find the compositions of each phase in a two phase region
Plot given co-ordinate (temp and TOTAL composition value)
Draw tie line in corresponding phase region
Draw down at the points of intersection between tieline lines and phase region boundaries
Each x/composition value is the composition of that phase in the two phase system.
NOTE: here it IS the sides you’d expect for alpha and beta.
Rules for solid solubility (in metals) (all rules met = completely solid soluble, some met = limited, none = insoluble)
Atomic size - (if within 15% size range of eachother they can replace eachother in each others lattice at any ratio)
Crystal structure - both lattice structures must be the same for this interchangability of atoms at different ratios
Electronic charge - they should both have similar electric charge to form a solid solution
Valences - lower valence is better at being solvent for high valence solute than vv.
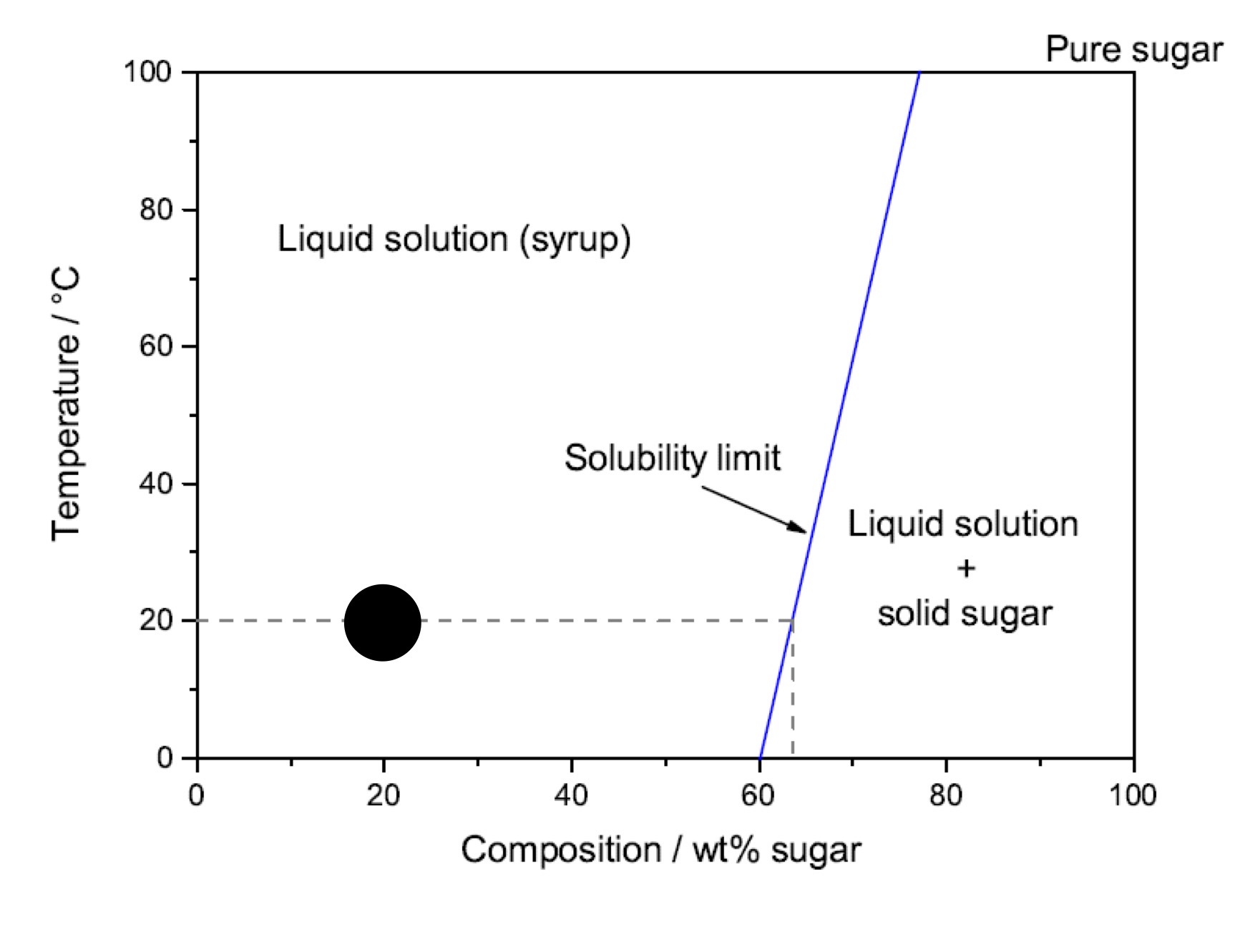
Is this an example of complete, limited or insoluble solution
Limited
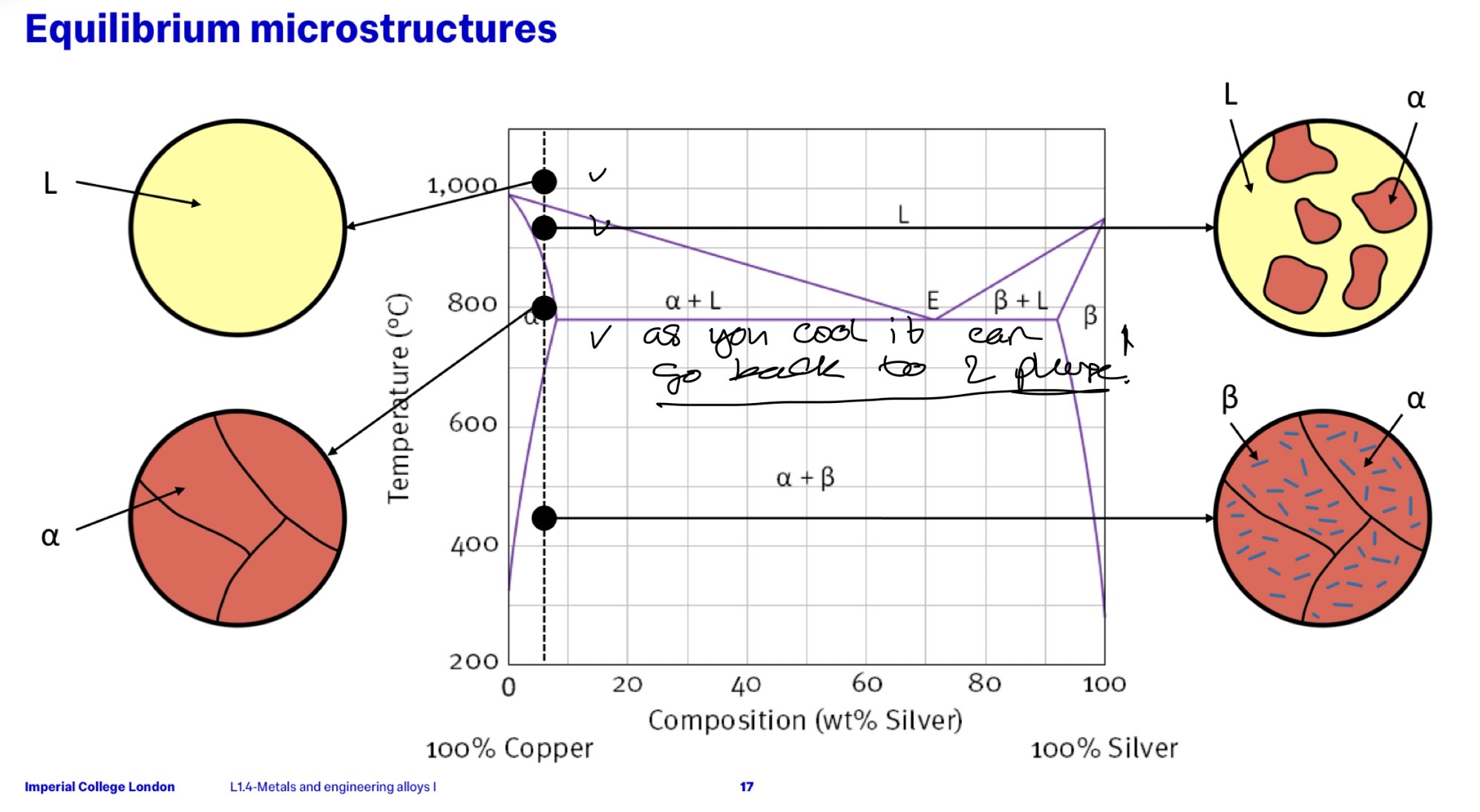
What does L represent in this image?
What does L + alpha …?
A THIRD phase where both metals are in liquid state mixed together completely.
Alpha phase solute in L phase solvent
NOTE: the best way to tell the states of these phases is identifying the liquidus and solidus line through the y intercepts of said lines at compositions 0 and 100% i.e. each materials melting points. + Liquidus lines always furthest up.
Derive the lever rule

What do you do when they ask:
“Find mass/weight fractions of each phase”
“Find volume fractions of each phase”
Lever rule
Lever rule for weight fractions + other phase diagram trick to get concentration of each material in each phase (C_mat in wt%) to then get absolute densities of phases + volume fraction = volume of phase/ total volume of both phases together.
(HOW density equation WORKS: rho_alpha = (mass_mat1+mass_mat2)/(v_mat1+v_mat2). V_mat1 = (Conc% of total weight)/material density IF total mass at top = 1 (or x both by 100)!!!!)

How to tell if your density of phases is incorrect
PHASE DENSITY SHOULD ALWAYS BE BETWEEN THE TWO MATERIAL DENSITIES!