TRIPLE: Carboxylic acids
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
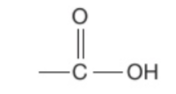
What are carboxylic acids?
A homologous series of compounds which all contain a —COOH functional group attached to a hydrocarbon chain
Vinegar definition (it is an objective on the spec)
An aqueous solution containing ethanoic acid
How are carboxylic acids formed?
They are formed by oxidation of an alcohol
Ethanoic acid is formed when ethanol is left in the air or oxidised using potassium dichromate(VI) in dilute sulfuric acid
Complete the table (si te da pereza al menos haz uno, a lo mejor te sorprende)
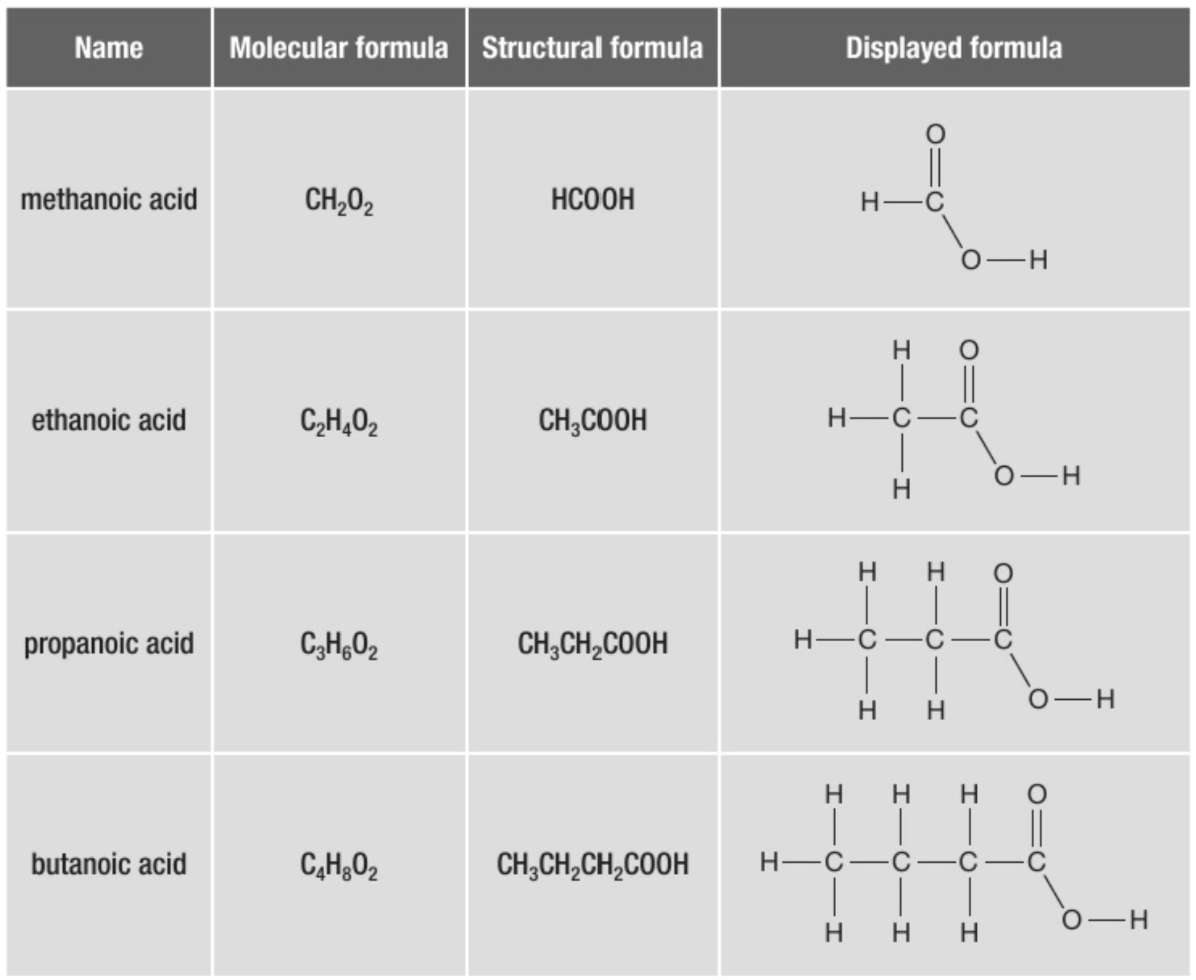

For displayed show all the bonding. If you just want to write the formula in an equation it is normal just to use the e. g CH3COOH
What is the range of pH for carboxylic acids? How can you show they are acidic?
They are weak acids with a pH of about 3-5 depending on the concentration of the solution.
They will turn damp blue litmus paper red and react with all the things you expect acids to react with
What happens when you dissolved ethanoic acid (in aqueous)?
What are you left with?
Hydrogen dissociates
You are left with ethanoate ions and hydrogen ions
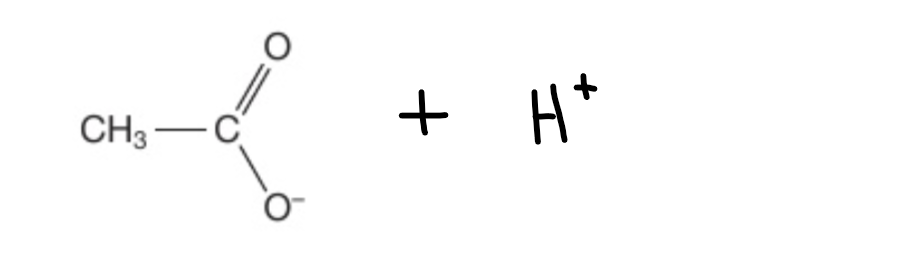
What is the charge on a carboxylic acid ion?
Negative 1 (-1)
All of these carboxylic acids will behave similarly in these reactions.
How does the reaction between dilute ethanoic acid with metals compare to the reaction between other dilute acids like HCl with metals?
What can you see + why?
Word and chemical equation for reaction between ethanoic acid and magnesium?
They react the same way as other dilute acids but MORE SLOWLY.
There is fizzing because hydrogen is produced and a colourless solution is formed because the salt formed is magnesium ethanoate which is soluble in water.
Magnesium + ethanoic acid → magnesium ethanoate + hydrogen
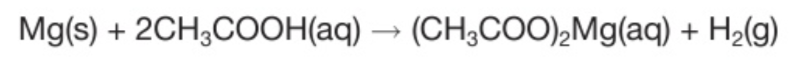
Endings of the salts produced from reactions of carboxylic acids and metals/metal carbonates
-oate
methanoate
butanoate
ethanoate
What kind of compounds are the ones formed?
Ionic compounds (they are salts)
Therefore, REMEMBER to balance the charges when doing the equations
How does the reaction between dilute ethanoic acid with metal carbonates compare to the reaction between other dilute acids like HCl with metal carbonates?
What will you see and why?
Word and chemical equation between reaction of sodium carbonate and ethanoic acid
They are the same. Carboxylic acids will behave like any other acid
Fizzing because CO2 is released (+ colourless solution formed)
Calcium carbonate + ethanoic acid → calcium ethanoate + carbon dioxide + water
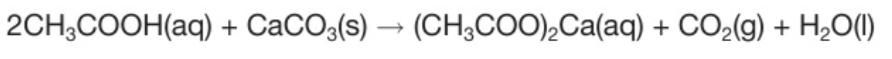
Word equation for reaction between butanoic acid and sodium hydroxide?
Butanoic acid + sodium hydroxide → sodium butanoate + water
Will copper react with ethanoic acid? Will copper carbonate react? Why?
Copper won’t react because it is lower in the reactivity series than hydrogen so it cannot displace it
Copper carbonate will react. It is a neutralisation reaction.
REMEMBER: Reactions with these acids with metals follow the same rules as with other acids (sodium very vigorous reaction, below H in reactivity series = no reaction)