3.1- Redox and standard electrode potential
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
What does a cell look like?
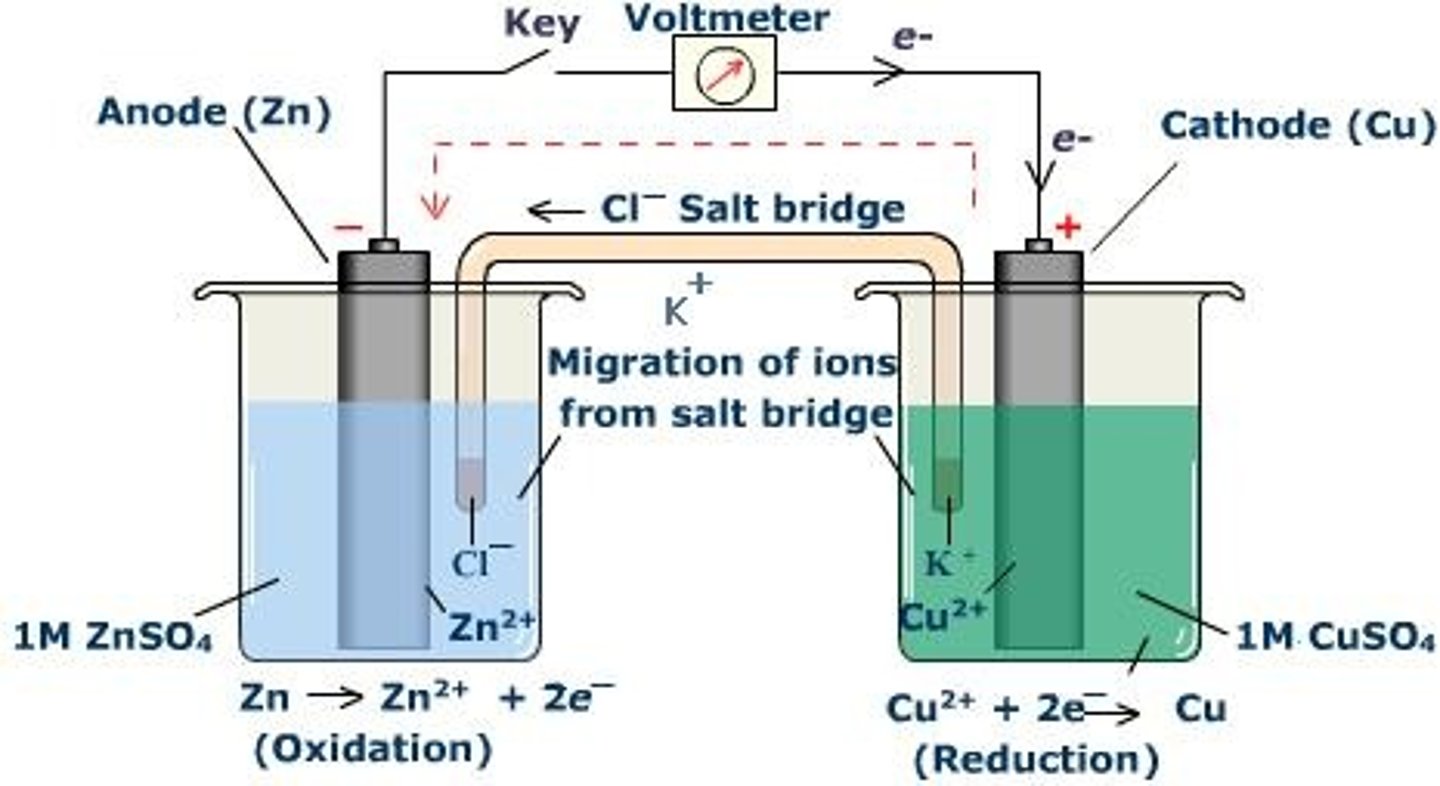
What is it possible to do to redox reactions?
To physically separate the two processes of oxidation and reduction, so they each occur at the half-cells
What does the wire allow for?
The flow of electrons from the half-cell where oxidation occurs to the half cell where reduction occurs
What do you include if you want to measure the potential difference?
A high resistance voltmeter
What does a salt bridge do?
Connects the solutions in the two half-cells so the circuit is complete and the current can flow without the solutions mixing
What is current?
A flow of charge
What is the current in a salt bridge?
A flow of ions
What is a salt bridge made of?
Filter paper soaked in potassium nitrate solution
What must a half cell contain?
Both the reactants and products for the half equation
Eg?
For copper, must contain Cu2+ ions and Cu atoms
What must it also contain?
A metal to allow electrons to flow into or out of the half cell
What are the 3 types of half cell?
1 Metal/metal ions
2. A gas in contact with a solution of non-metal ions, with an inert metal electrode
3. A solution containing ions of a metal in two different oxidation states, using an inert metal electrode
What are some examples of the metal/metal ions half cells?
Zn (s) with Zn2+ (aq)
Cu (s) with Cu2+ (aq)
What do you have in both cases?
A piece of metal as the electrode
A solution containing a 1 moldm-3 solution of the metal ions
What are some examples of the gas in contact with a solution of non-metal ions, with an inert metal electrode?
H2 in at 298K and 100kPa
Platinum electrode
1 moldm-3 H+ (aq)
Why must the platinum electrode be inert?
Since non-metals are not conductors, an inert electrode is needed to allow electrons to flow in or out of the half cell
What does this apparatus look like?
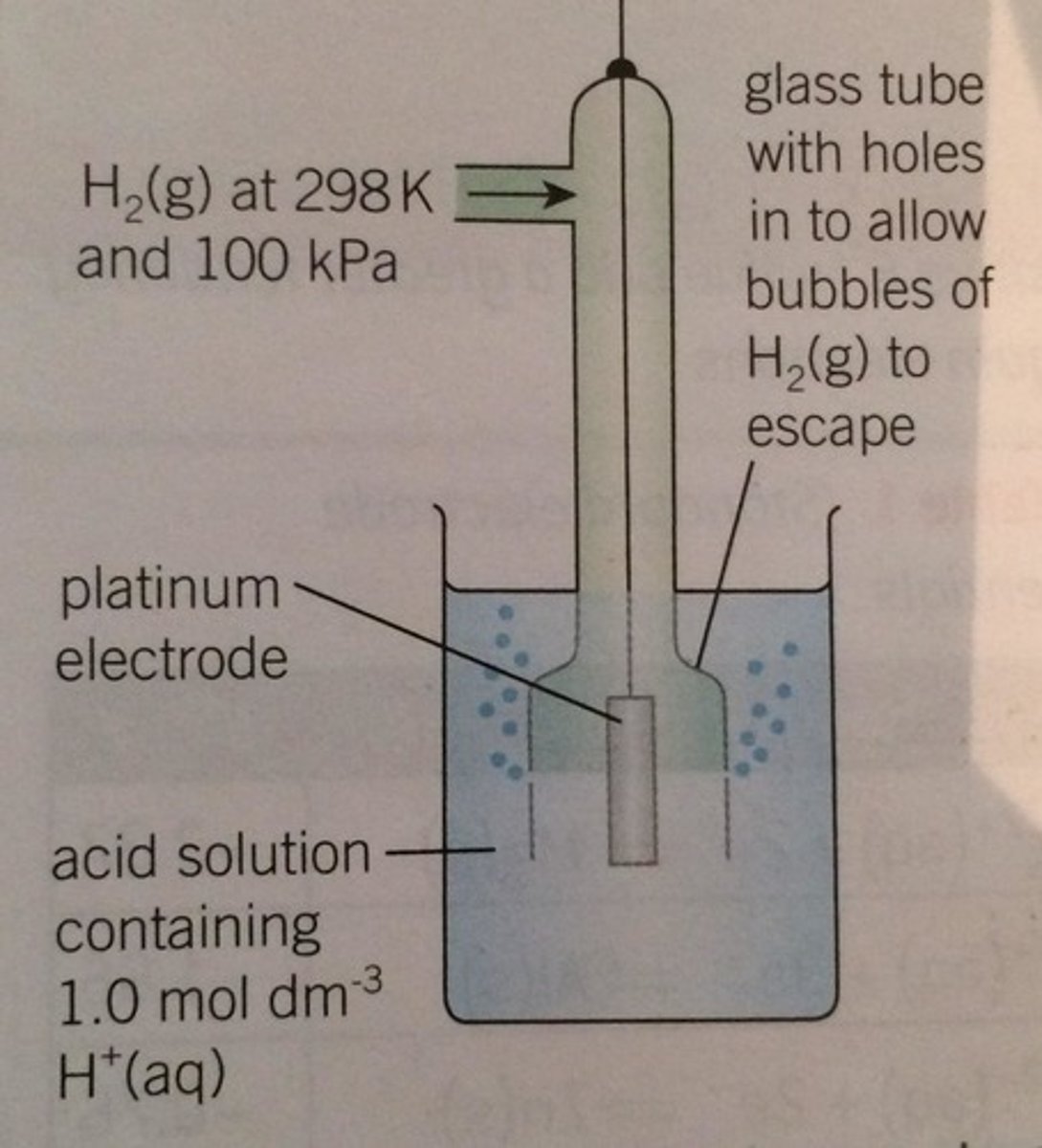
What is one example of the solution containing ions of a metal in two different oxidation states, using an inert metal electrode?
1 moldm-3 Fe2+ with 1 moldm-3 Fe3+
Inert Pt electrode
Why does it need to be inert?
To allow the flow of electrons in or out of the half-cell
Why is this typically used for the transition metals?
Because the metal may have several oxidation states
How can you represent a half cell?
Using a cell diagram
What do the lines represent?
A change of physical state
What do the double lines represent?
A salt bridge
What do you start with?
The metal used to conduct electrons into or out of the half cell (goes furthest at either end)
Write one for Mg2+(aq) with Mg(s)
Mg(s) | Mg2+(aq)
How would you write one for the gas/solution half cell?
Pt(s) | H2(g) | H+(aq)
For the mixed solution half cell?
Pt(s) | Mn2+(aq), MnO4-(aq)
For a mixed solution half cell with a metal/metal ion half cell?
Pt(s) | Fe2+(aq), Fe3+(aq) || Mg2+(aq) | Mg(s)
What's different for that one?
Put a comma between the mixed solutions
What is standard electrode potential a measure of?
The ability of a half-cell to gain or lose electrons
What is its definition?
The potential difference between any half-cell under standard conditions and the standard hydrogen electrode.
What are standard conditions for this?
A temperature of 298K, a pressure of 1atm and concentrations of 1moldm-3
What does the scale of standard electrode potential use hydrogen as?
Zero
What is the hydrogen half cell called?
The standard hydrogen electrode
What does the standard hydrogen electrode look like?
H2 (g) pumped in 1atm 298K
Pt electrode
H+ 1moldm-3 solution
What must you do to measure a standard electrode potential?
Set up a half cell that is connected to the standard hydrogen electrode
What is the standard electrode potential for the standard hydrogen electrode?
0V
What are all other values stated?
Relative to this
What will act as the positive electrode?
The half-cell with the most positive electrode potential, so is a stronger oxidising agent so becomes reduced
Which will act as the negative electrode?
The half cell with the least positive electrode potential, so is a reducing agent and becomes oxidised
In what direction will the electrons flow?
From negative to positive
What are the most reactive metals?
The strongest reducing agents so have the most negative standard electrode potential
What are the most reactive non-metals?
The strongest oxidising agents so have the most positive standard electrode potential
So if you were reacting:
Na+ + e- -> Na : -2.71 V
Fe3+ + e- -> Fe2+ : +0.77
Which would be the oxidising agent and which would be the reducing agent and what would be the equation?
Fe3+ would be the oxidising agent
Na would be the reducing agent
Fe3+ + Na -> Fe2+ + Na+
What does this show?
You reverse the direction of the equation of the reduction
What needs to happen for a successful reaction to occur?
The oxidising agent needs to be strong enough to oxidise the reducing agent
How do you show that a reaction is feasible?
By calculating the emf
By comparing standard electrode potentials
What would the emf of two standard electrode potentials -0.76 V and +0.34 V be?
0.34 - (-0.76) = +1.10 V
For
Mg2+ + 2e- -> Mg : -2.36
Zn2+ + 2e- -> Zn : -0.76
What is the equation for it to be feasible and the EMF?
Zn2+ + Mg -> Zn + Mg2+
-0.76 - (-2.36) = 1.60 V
What have fuel cells been developed as a method of?
Releasing energy very efficiently from fuels e.g.. hydrogen, methane or methanol
What do traditional engines do?
Burn the fuel, followed by using this heat to cause gas expansion which is used to move a motor
How does the fuel cell system work?
It passes fuel over the platinum metal which acts as a catalyst, but also as an electrode for the electrochemical system
What happens to the electrons?
They are removed from hydrogen atoms at one electrode:
H2 -> 2H+ + 2e-
What do the protons do?
Diffuse through a semi-permeable membrane to the other electrode where they receive electrons and oxygen molecules to form water molecules:
O2 + 4H+ + 4e- -> 2H2O
So what is the overall equation?
2H2 + O2 -> 2H2O
What are the benefits of fuel cells?
Convenient way of storing and releasing energy
More energy efficient than standard fuel systems
Emissions from fuel cells are less damaging than the CO2 from traditional engines
Disadvantages?
The gases needed are difficult to store compared to liquid fuels
The fuel cells operate at lower temperatures so need very efficient catalysts which use expensive metals
The hydrogen fuel must be generated elsewhere and this is likely to use fossil fuel energy sources which will cause their own CO2 emissions. There is also an energy loss here as the conversion process is not 100% efficient