Chem 239 MT 1
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
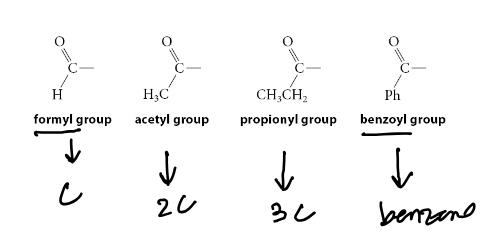
Structure of: Formyl Group, Acetyl Group, Propionyl Group, Benzoyl Group
Formyl: formaldehyde, aldehyde-like group
Acetyl: acetone, ketone-like group
Propionyl: 3 total carbons, so propionyl
Benzoyl: attaching a benzene

How do you form acetals from ketones
Need 2 equivalents of alcohol and a catalytic acid.
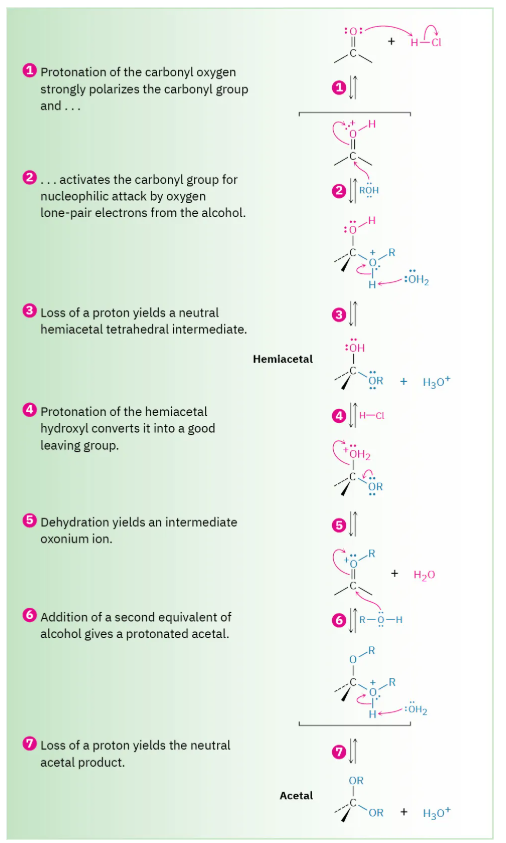
Mechanism not on exam
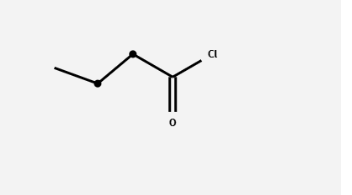
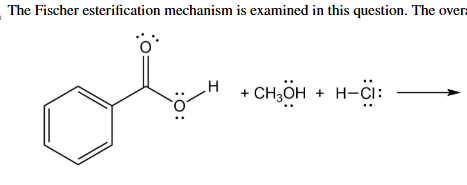
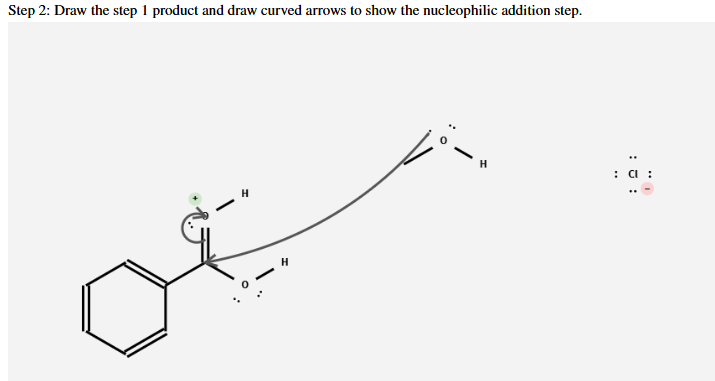
What is the product and what is the mechanism
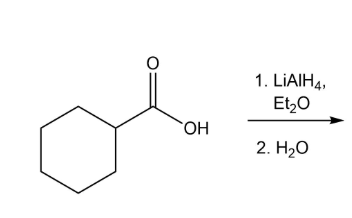
What are 3 reducing reagents, what does it mean to reduce a carboxylic acid, and what are their relative strengths?
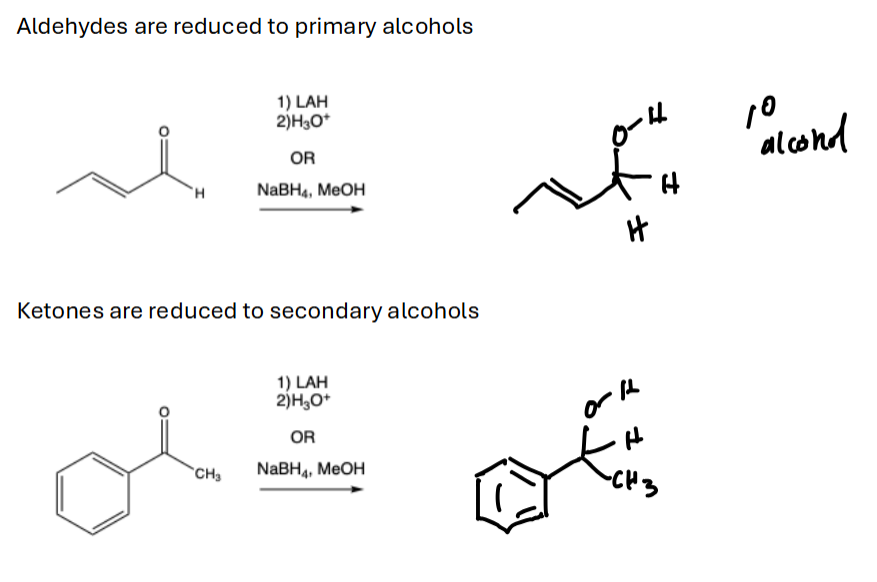
LAH > NaBH4 > H2.
LAH will reduce C.A., ketone, aldehyde
NaBH4 will reduce ketone, aldehyde
H2 will reduce just aldehydes

Aldehyde Nomenclature: suffix, carbon numbering, multiple aldehydes, ring
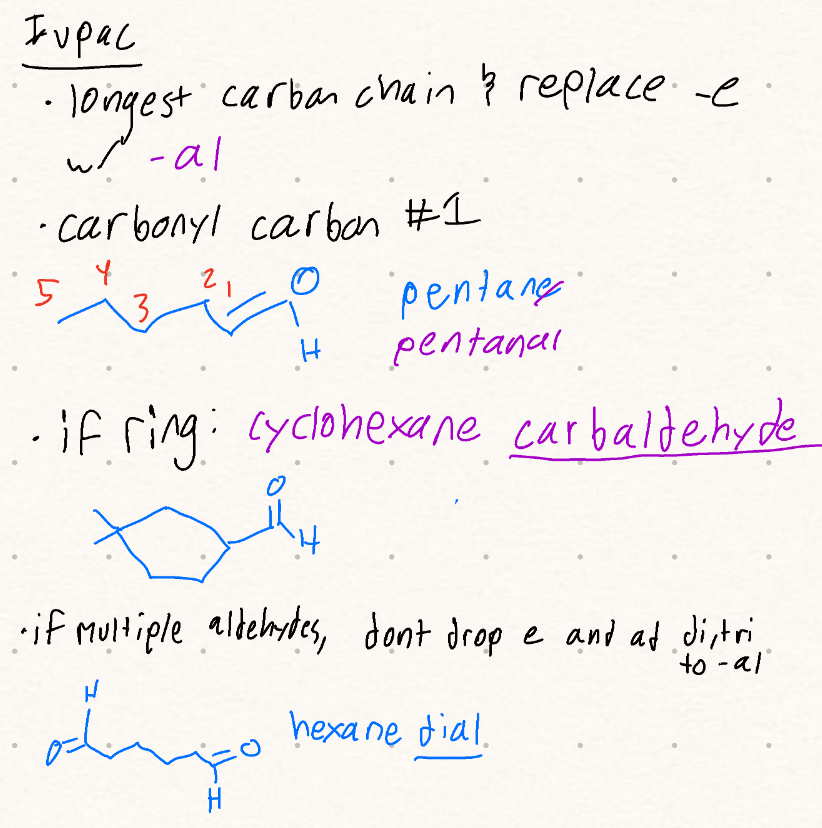
Ketone Nomenclature:

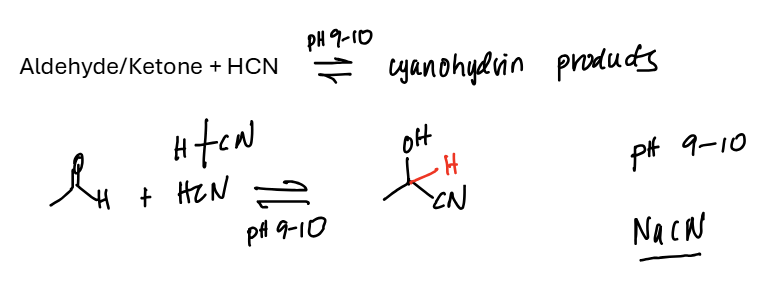
Aldehyde / Ketone + HCN = add -C—N & -H to the central carbon

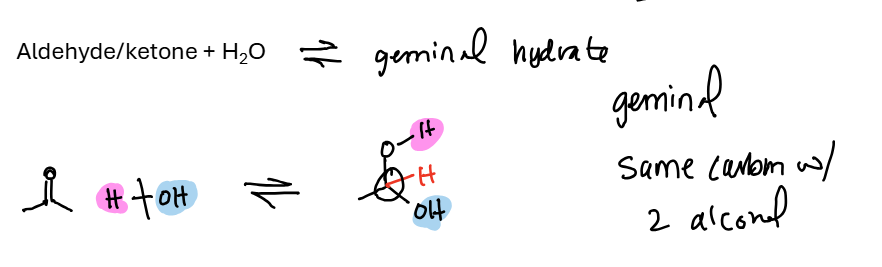
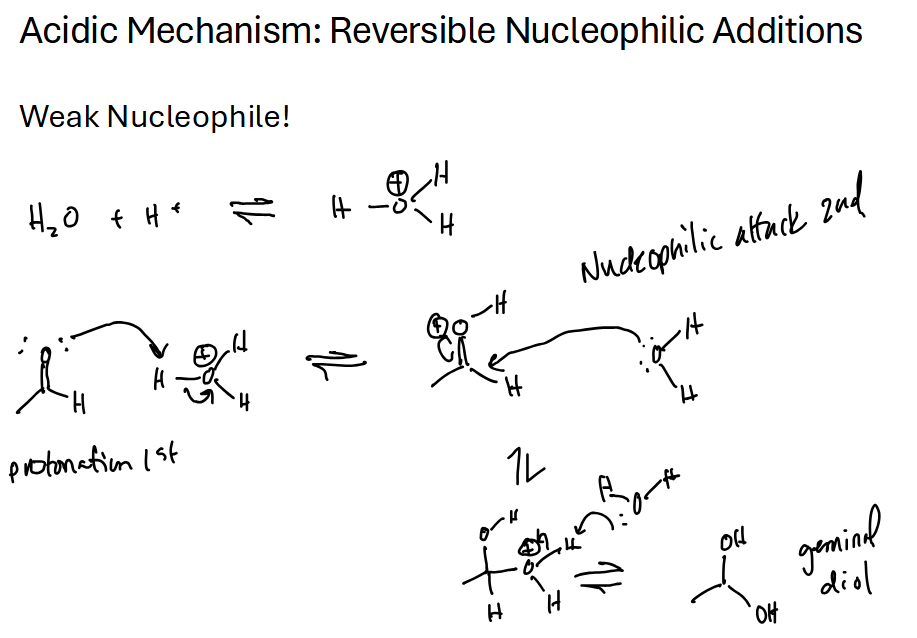
Aldehyde / Ketone + H2O = geminal hydrate (twin alcohols)
Ketones equilibrium lies mostly to the left, less reactive. Formaldehyde lies mostly to the right as a geminal hydrate

And what are examples of strong nucleophiles
The strong nucleophile here is CN-, attacking the basic carbonyl carbon and then getting protonated in an acid workup step
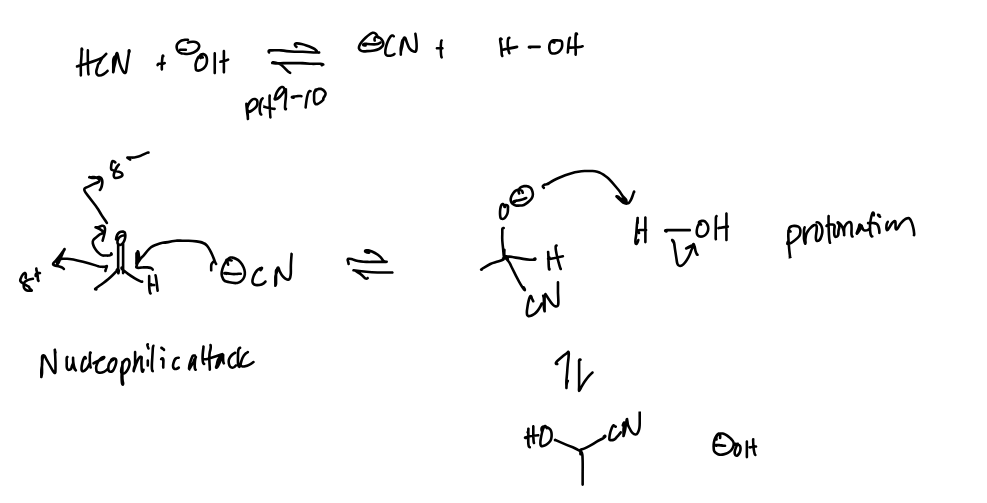

And what’s an example of a weak nucleophile
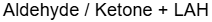


Sodium Borohydride Mechanism
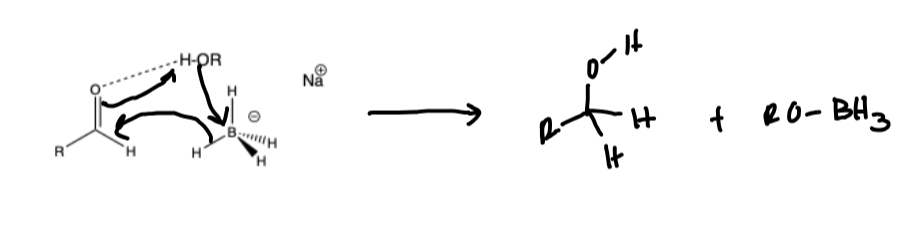
LAH Mechanism
Regular strong nucleophile mechanism,
Lithium cation bonds to O
H- attacks carbonyl carbon
Acidic workup protonates O and gets rid of Li
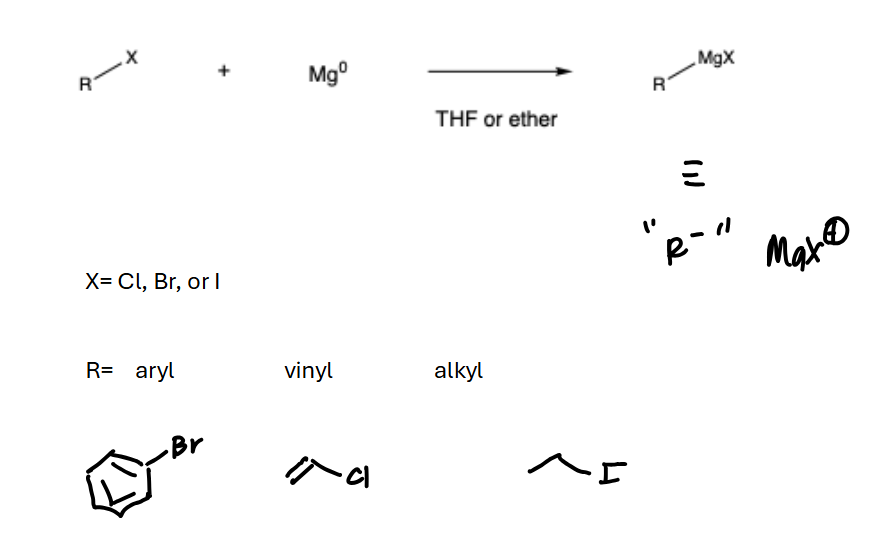
Grignard synthesis


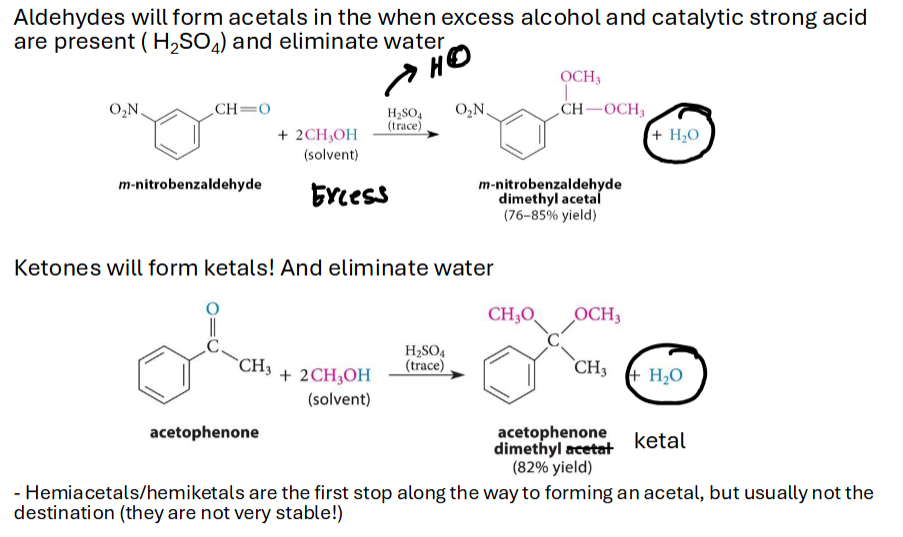
What are Acetals & Ketals (normal and Hemi)
Acetals & Ketals - a carbon bonded to two O-R groups. If its bonded to a hydrogen too its an acetal, two C groups is a ketal
Hemi means one O-R & one O-H
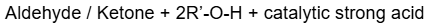
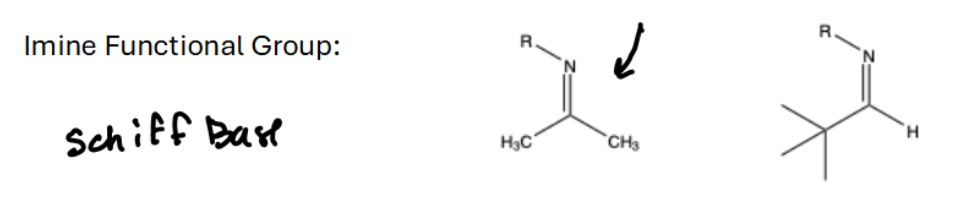
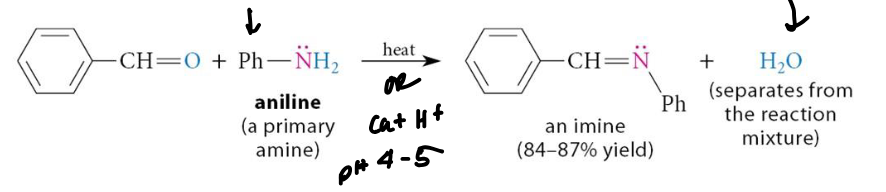
What is an Imine functional group
N=O

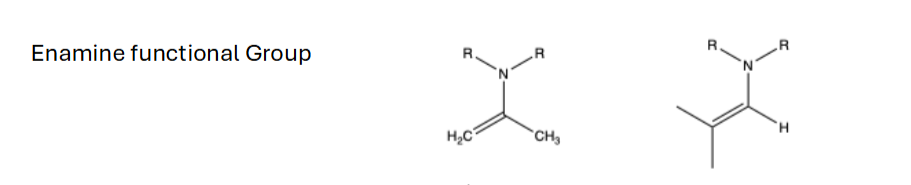
What is an Enamine
R’-N-C=R the N-C=C
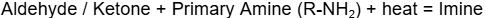
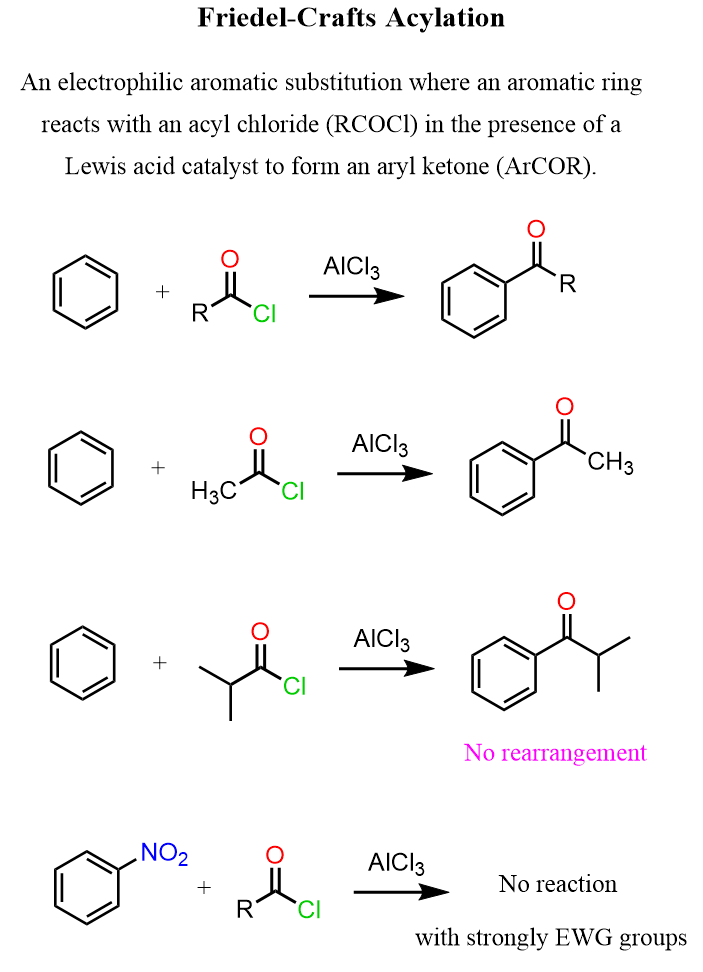
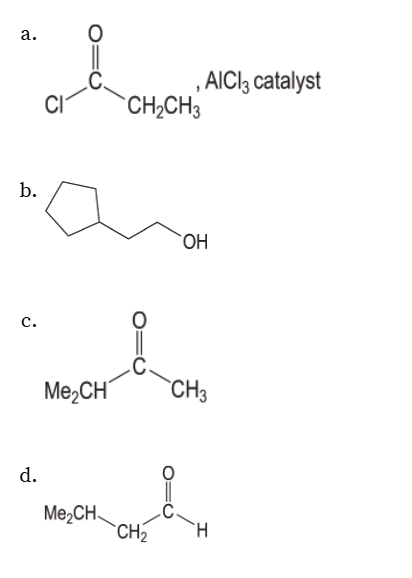
Four ways to synthesize carboxylic acids
You oxidize primary alcohols, alkyl benzenes, and aldehydes. For CO2 you use a grignard reagent to attack the partially positive middle carbon
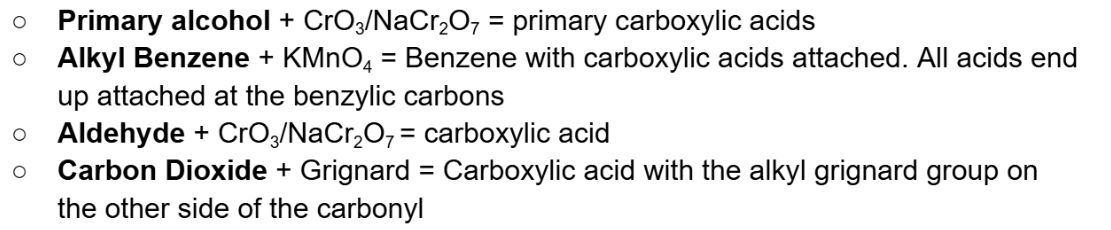
Friedel Crafts Acylation
Ester Nomenclature

Reactions of Carboxylic Acids: at the C, O, and decarboxylyation
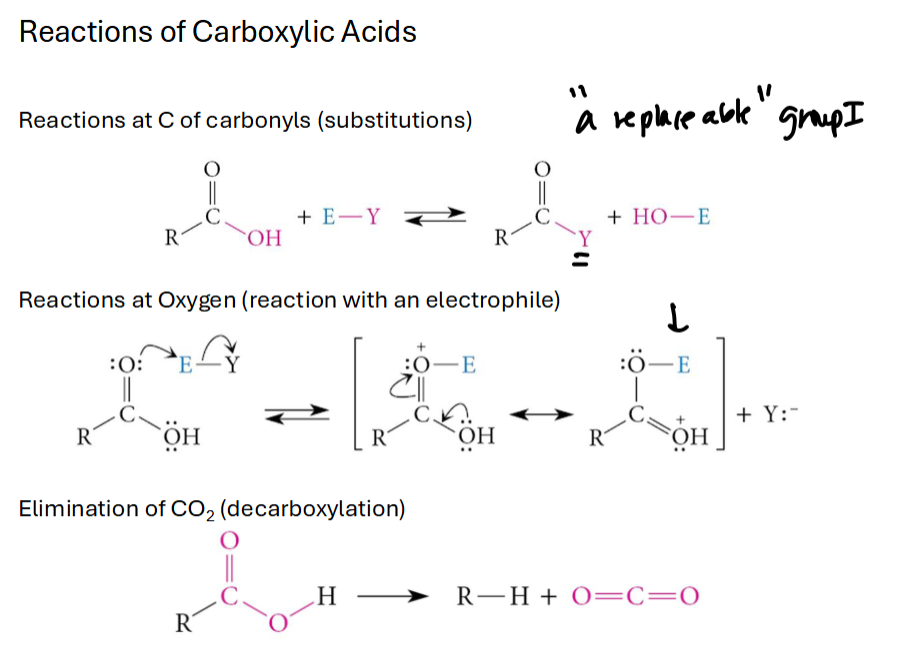
Fisher Esterification

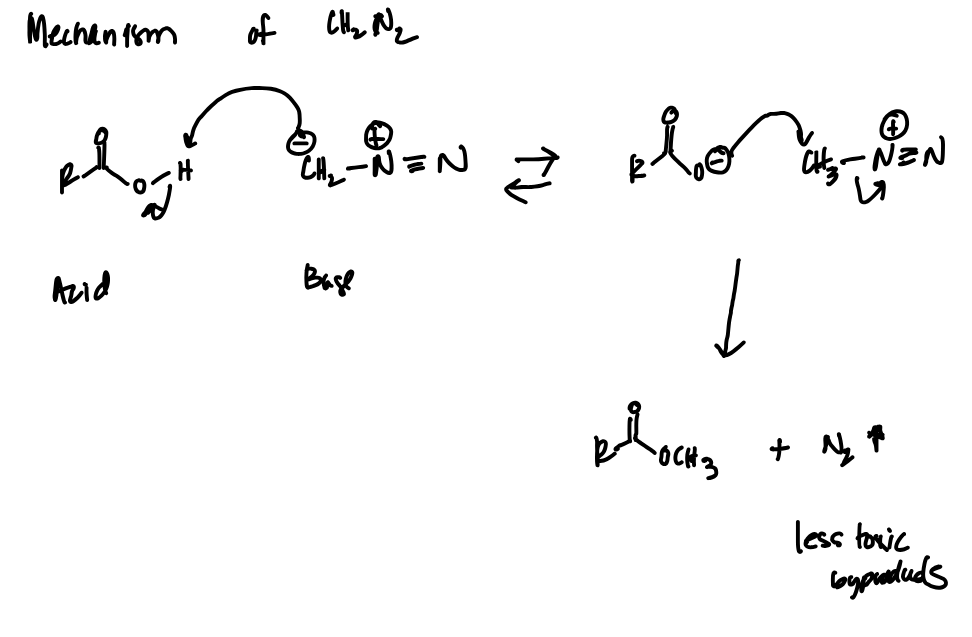
CH2N2 Reagent Reaction
Reducing Agents & what they do to Ketones & Carboxylic Acids & Relative Strengths
LAH, reduces aldehydes to primary alcohols, ketones to secondary alcohols, C.A.s to alcohols to primary alcohols
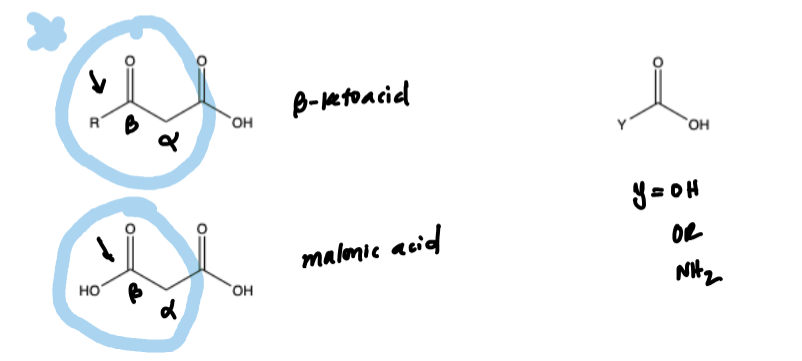
What carboxylic acids can be de-carboxylated & how do you do it
Beta Ketoic Acid (double carbonyl with carboyl at Beta carbon)
Maleic acid (double carbonyl with C.A. carbonyl at Beta carbon)
Y-COOH where Y = OR, OH, NH2
You decarboxylate by reacting with heat & acid. Chops off the right CO2 and the H adds to the alpha carbon.
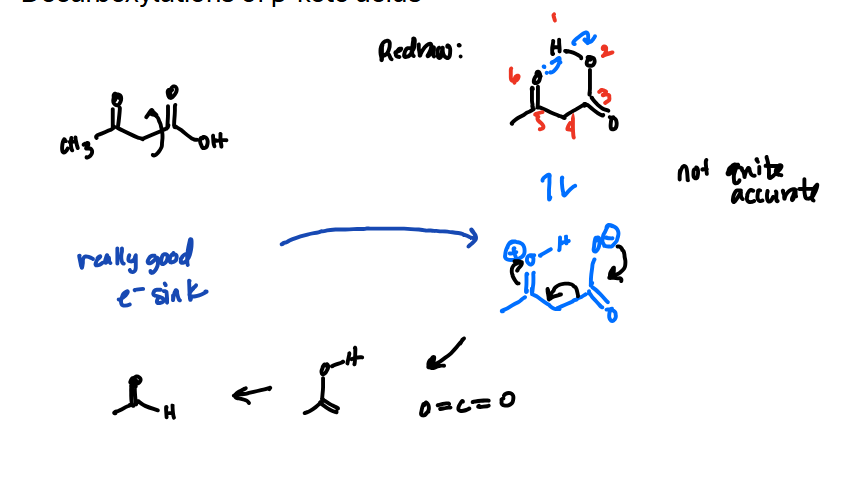
Decarboxylation of Beta-Keto acids mechanism
Forms the cyclohexane looking intermediate by rotating central carbon
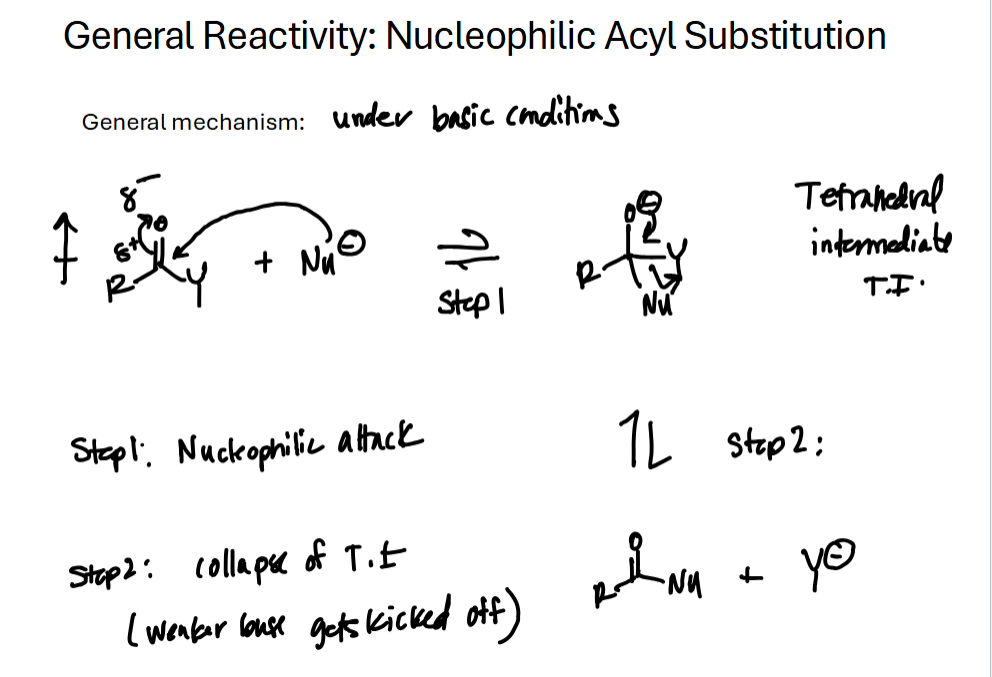
Have an idea of the general reactivity of C.A. derivatives
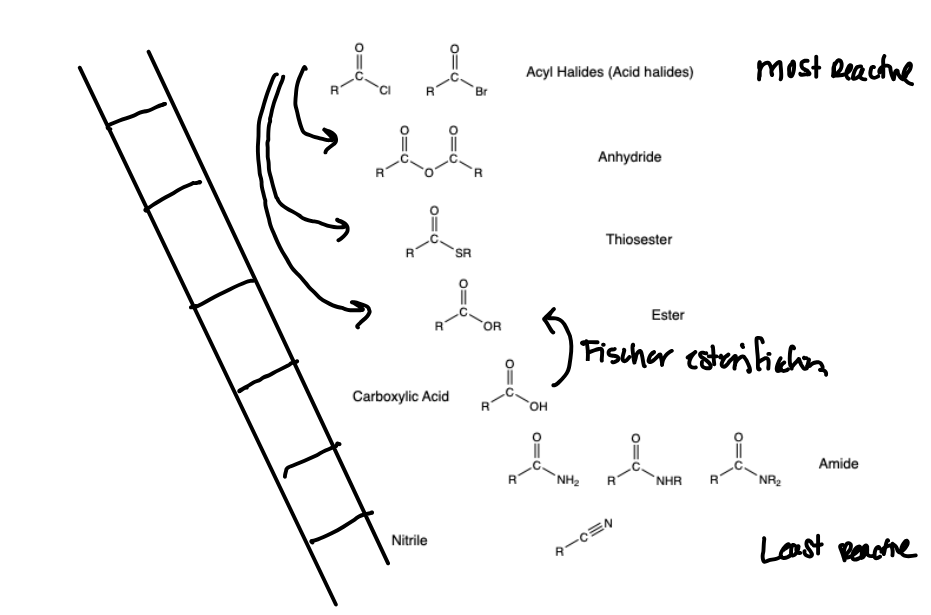
Steps of C.A. Derivative Nucleophilic Acyl Substitution
Nucleophile attacks carbonyl carbon, pi-bond electrons kick up to oxygen.
Tetrahedral intermediate is formed, strongest leaving group leaves