L1 - BB rad - Introductory Spectroscopy
1/8
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
The period of an oscillation is …. and how is it calculated
period of an oscillation (time for 1 full cycle) is the reciprocal of the frequency, 1/ν. Use this c = λν Units are ms^-1
What is a wavenumber and how is it caluclated
wavenumber (v ~) is the reciprocal of the wavelength and can be interpreted as the number of wavelengths in a given distance. wavenumber = 1/λ in cm^-1.
Length: 1 m = 100 cm Reciprocal length: 1 m-1 = 0.01 cm-1
how does a spectrometer work (pic of slide)
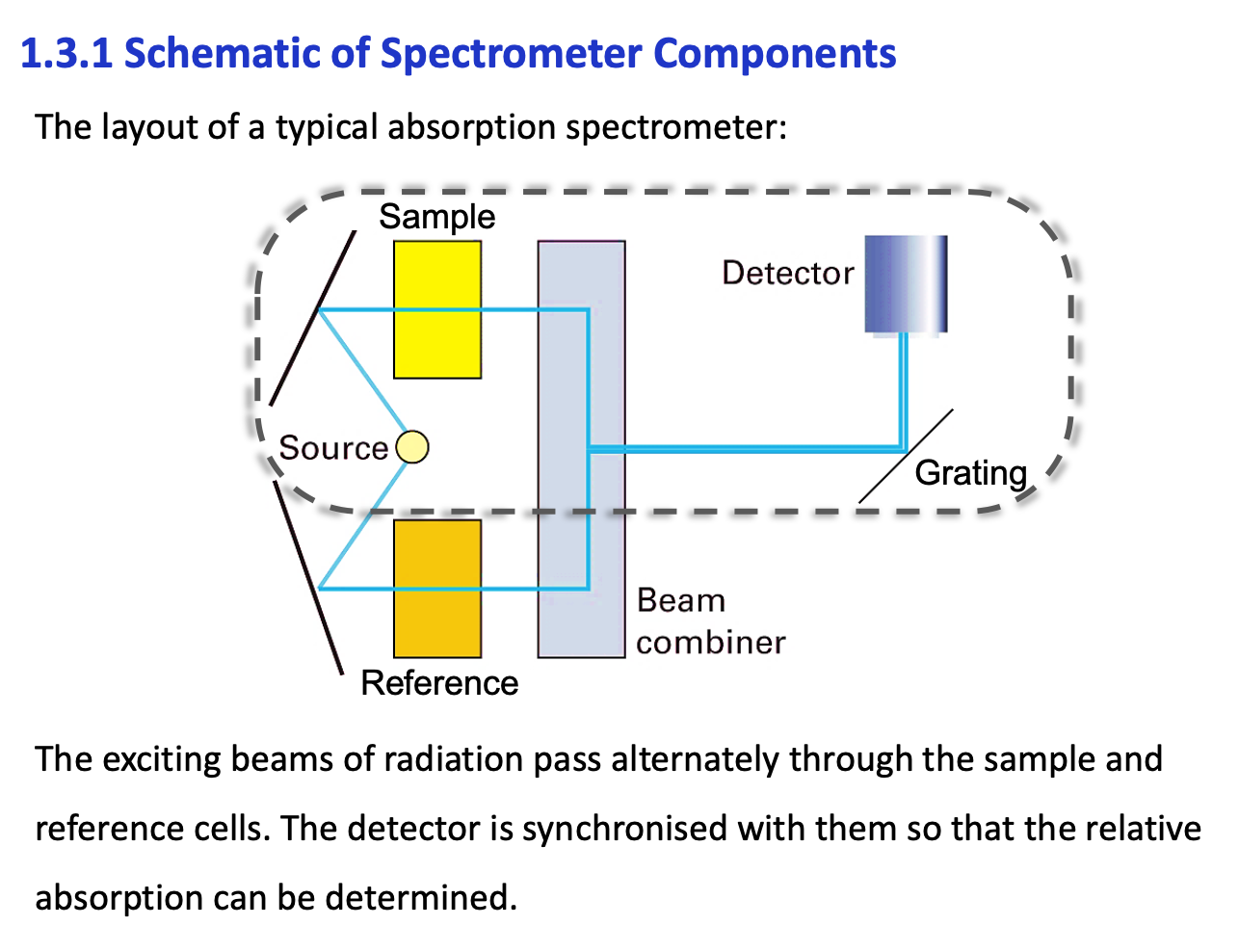
Why is it better to use monochromatic radiation to probe a sample rather than polychromatic. What can be done if polychromatic
Monochromatic (radiation of a single wavelength or frequency) will give a muich simipler and easy to interpret result.
If in case only a polychromatic source is available (e.g. light from the sun) a dispersing element can be used to select wavelengths. Dispersing elements = glass prism and diff grating
how else can we seperate wavelength from a beam of radiation from polychromatic source?
Diffraction grating diva
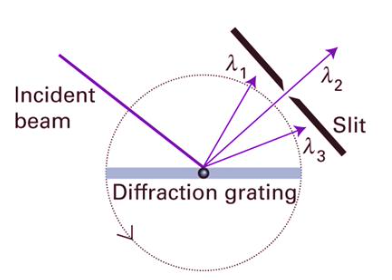
A diffraction grating consists of many equally spaced slits that act as coherent sources of light; when light passes through, the waves interfere to produce bright maxima at specific angles where the path difference between adjacent slits is an integer multiple of the wavelength. This condition is given by dsinθ=nλd. cant explain any more i dont get it
continuous vs absorption vs emission spectra (colours, black parts, produced by)
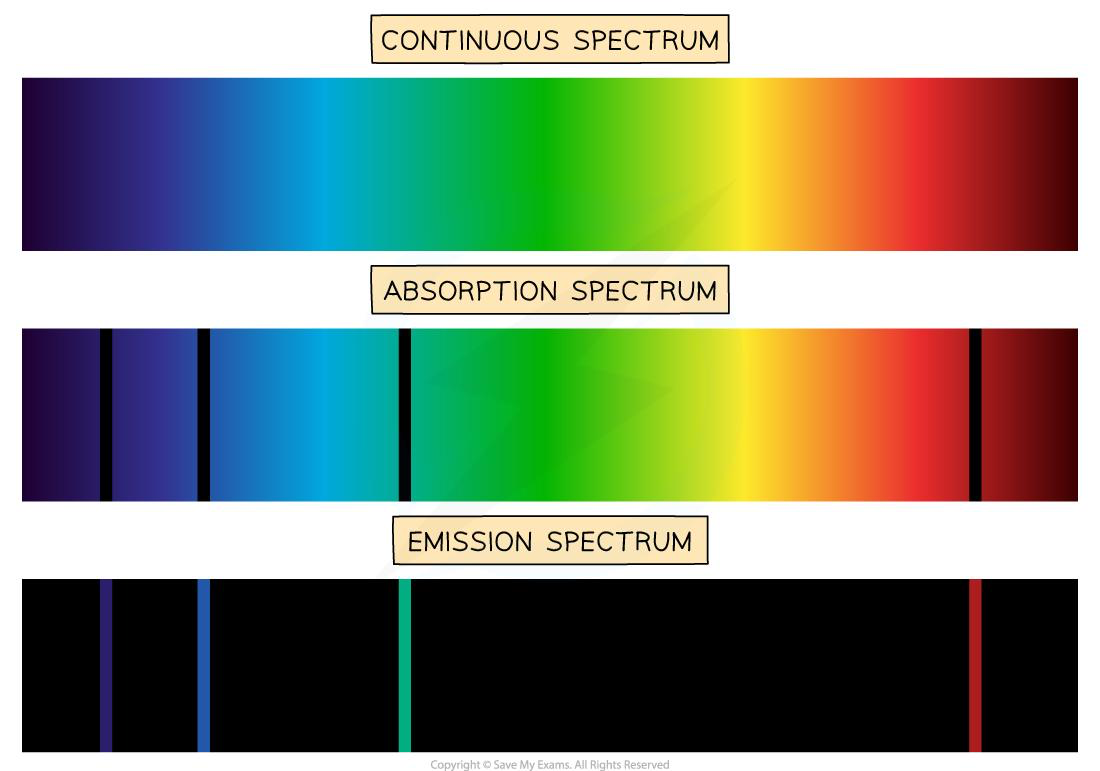
CONTINUOUS = Colours: All wavelengths present - No black - Produced by: Hot, dense objects (e.g. solids, liquids, high-pressure gases) All objects are composed of atoms and molecules, and these are moving in discorded random motion. This disordered translational molecular motion is considered kinetic energy which = temp of object. This gives rise to continuous spectra (thermal/black body spectra). This is emitted by any object that radiates heat.
The light is spread out into a continuous band with every wavelength having some amount of electric and magnetic radiation.
ABSORPTION = Colours: Continuous rainbow background Black parts: Dark lines where specific wavelengths are absorbed by electrons jumping to higher energy levels
Produced by: Cool gas in front of a continuous source
EMISSION = Colours: Discrete bright lines (specific wavelengths only)
Black parts: Everything else is dark (no light emitted there)
Produced by: Excited low-density gas (electrons falling to lower energy levels)
what is a black body
A ‘black-body’ is an (ideal) object capable of emitting and absorbing (no reflection) all frequencies of radiation uniformly. Basically a sponge that absorbs everything but then emits it based on T. Hotter → emits more light and shifts toward shorter wavelengths (bluer). Cooler → emits less light, mostly longer wavelengths (red/infrared)
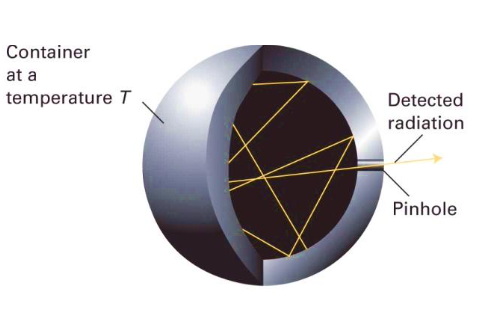
Example: a pinhole in an empty container maintained at a constant temperature T. This is showing that inside a blackbody cavity, radiation is continuously absorbed and re-emitted by the walls until (Energy absorbed = Energy emitted. The radiation inside becomes stable) thermal equilibrium is reached (absorption and emission cancel eachother out bc they are the same), producing a spectrum that depends only on T.

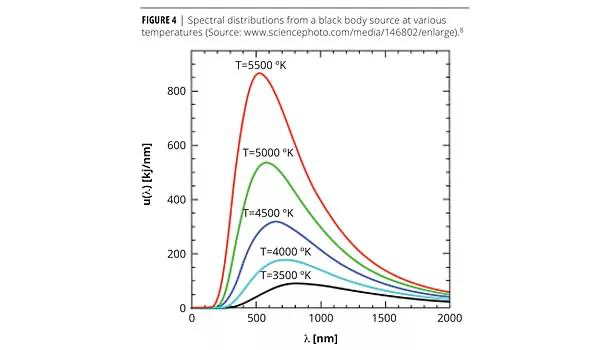
What is energy spectral density (p)
It is essentially the amount of energy emitted per unit wavelength at a given wavelength and temperature (i.e. how energy is distributed across wavelengths).
As the temperature increases, the maximum in the p moves to shorter wavelengths and increases in intensity overall. area under curve is total energy
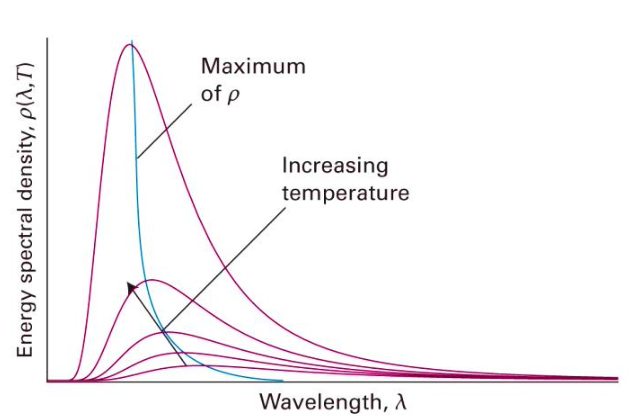
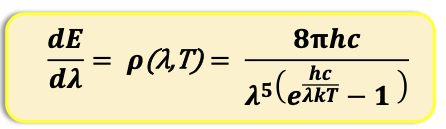
star application of bb radiation
Blackbody radiation allows temperature to be determined from emitted light, with hotter objects emitting more intense, shorter-wavelength radiation. This explains stellar classification (O–M), where colour and spectral features correspond directly to surface temperature.