Unit 4 Chem
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Define Metallic Bonding
Electrons are delocalized / mobile (they are free to move throughout the solid)
results from the electrostatic attraction between positive metal ions and the surrounding mobile valence electrons.
Properties of Metallic Bonding
Malleable - can be hammered into thin sheets
Ductile - can be pulled into wires
Solid (except for Hg - Mercury)
Luster / Shiny (light reflects or bounces off the outer electrons)
Good conductors of heat/electricity
electrons are able to slide into new positions without breaking the bonds
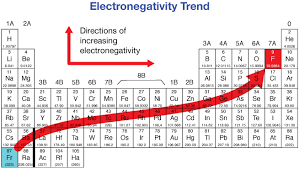
Define Electronegativity
How much an atom attracts shared electrons in a chemical bond
Define Alloy
mixtures of 2 or more elements and at least one is a metal
Why would we use alloys like Brass, Pewter, Carbon Steel, or Bronze instead of pure metals?
Properties are often superior to those of their component elements.
Define Dipole
Occurs when there is a separation of electrical charge within a bond or a molecule—meaning one part is slightly positive and another part is slightly negative
A nonpolar covalent bond involves ______ of electrons.
equal sharing
A polar covalent bond involves ______ of electrons.
unequal sharing
An ionic bond involves ______ of electrons.
transfer
Define Percent Ionic Character
the degree to which a chemical bond between 2 atoms exhibits ionic character
What is the relationship between the percent ionic character of single bonds and the electronegativity difference?
As the electronegativity difference increases, the percent ionic character increases
Define ionic bonding
In ionic bonding, electrons are transferred between a metal and nonmetal to achieve 8 valence electrons.
Properties of Ionic Compounds:
Hard but brittle
Good conductors when dissolved or molten
Soluble in water
High melting/boiling points
Crystalline solids (ions arrange in a repeating 3-D pattern)
When do Ions form?
Ions form when neutral atoms loses (+ charged Cation) or gains (- charged Anion) electrons
Dot Diagram for Lithium fluoride
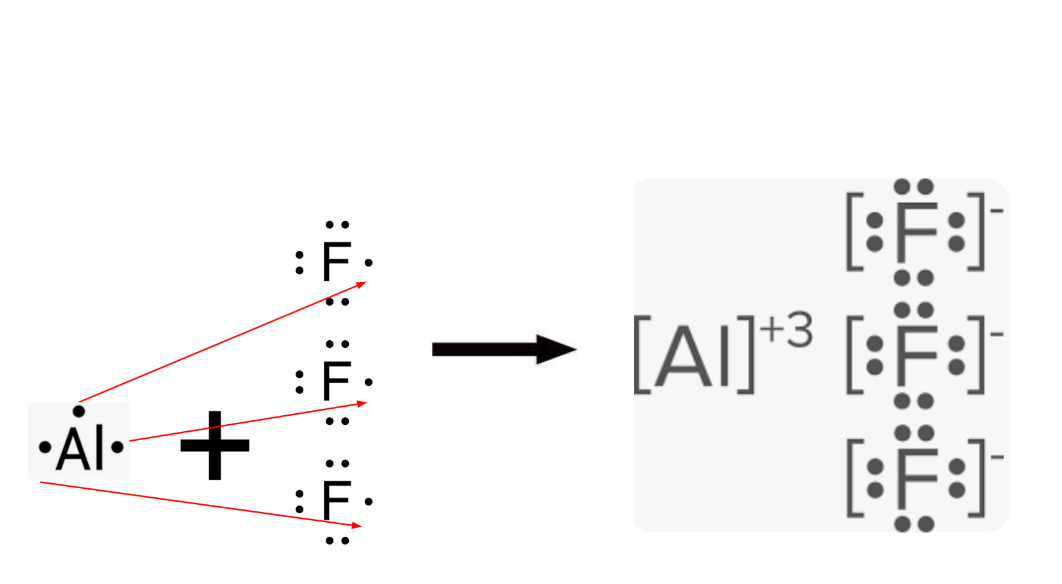
Oppositely charged particles ____ one another; particles with the same charge ___ one another.
Oppositely charged particles attract one another; particles with the same charge repel one another.
What is an ionic compound?
neutral compound consisting of positive and negative ions.
The electrostatic attraction that holds ions together in an ionic compound is
ionic bond
What can be used to illustrate electron transfer and the formation of ions?
Dot diagrams
What can be used to illustrate electron sharing?
Lewis Structures
Define Covalent Bonding
In covalent bonding, electrons are shared between nonmetal elements.
When 2 electrons are shared, it is a ____covalent bond
When 4 electrons are shared, it is a ____ covalent bond
When 6electrons are shared, it is a ____ covalent bond
When 2 electrons are shared, it is a single covalent bond
When 4 electrons are shared, it is a double covalent bond
When 6 electrons are shared, it is a triple covalent bond
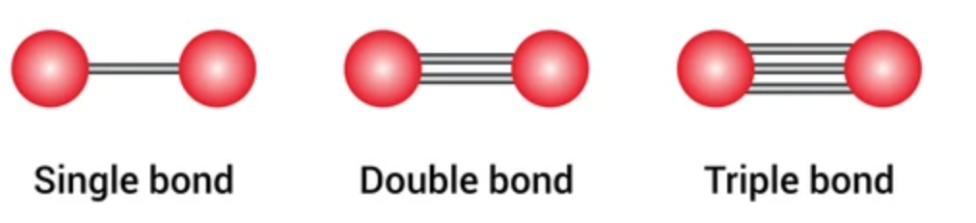
Covalent Bond properties
Poor conductors of heat/electricity
Lower melting and boiling points
Most nonmetal elements are gases, some solids, and one is a liquid (Br)
Covalent Nomenclature
Only add prefix to first element if there is more than 1 atom and ALWAYS add prefix to second element
ex:
N2O5 = Dinitrogen pentoxide
NH3 = Nitrogen trihydride (ammonia)
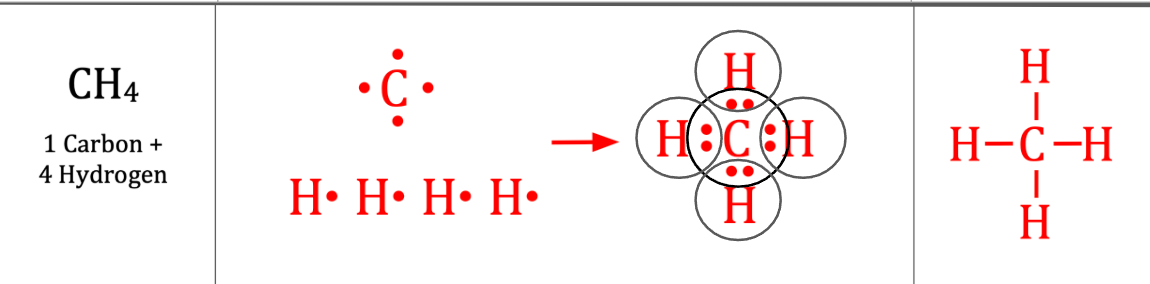
Complete Lewis Structure of:
Ch4
Complete Lewis Structure of:
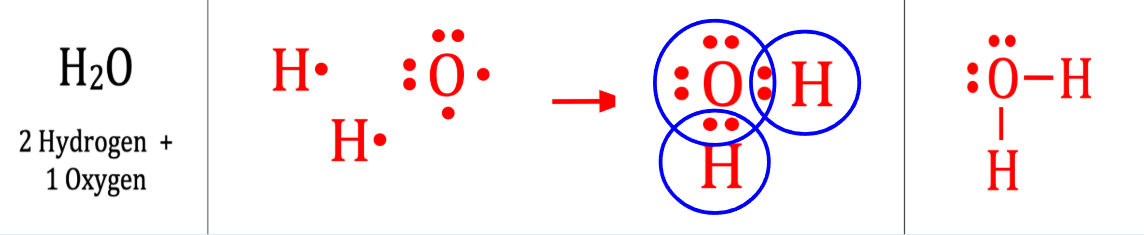
H20
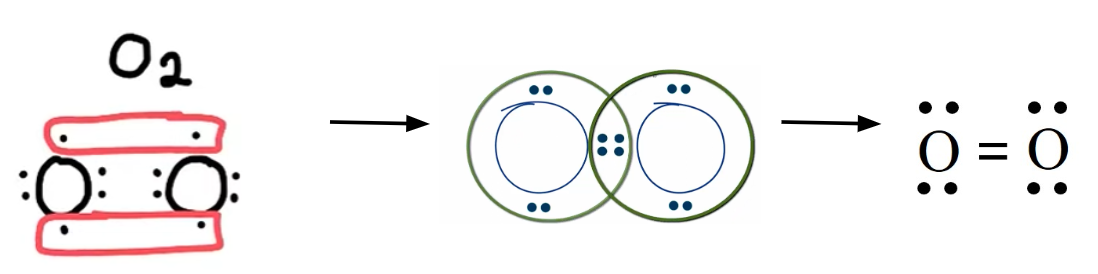
Complete Lewis Structure of:
O2
double bond
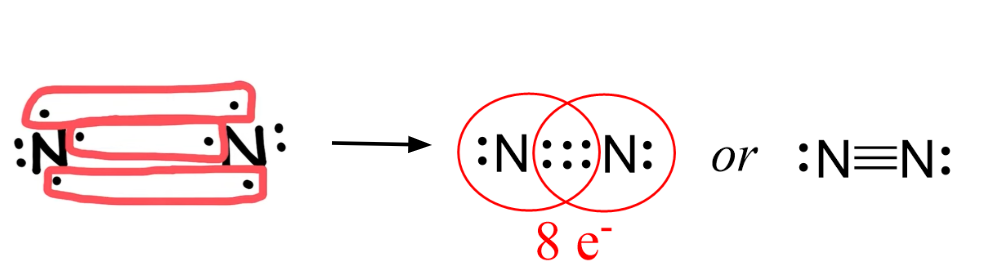
Complete Lewis Structure of and what kind of covalent bond it is:
N2
triple bond
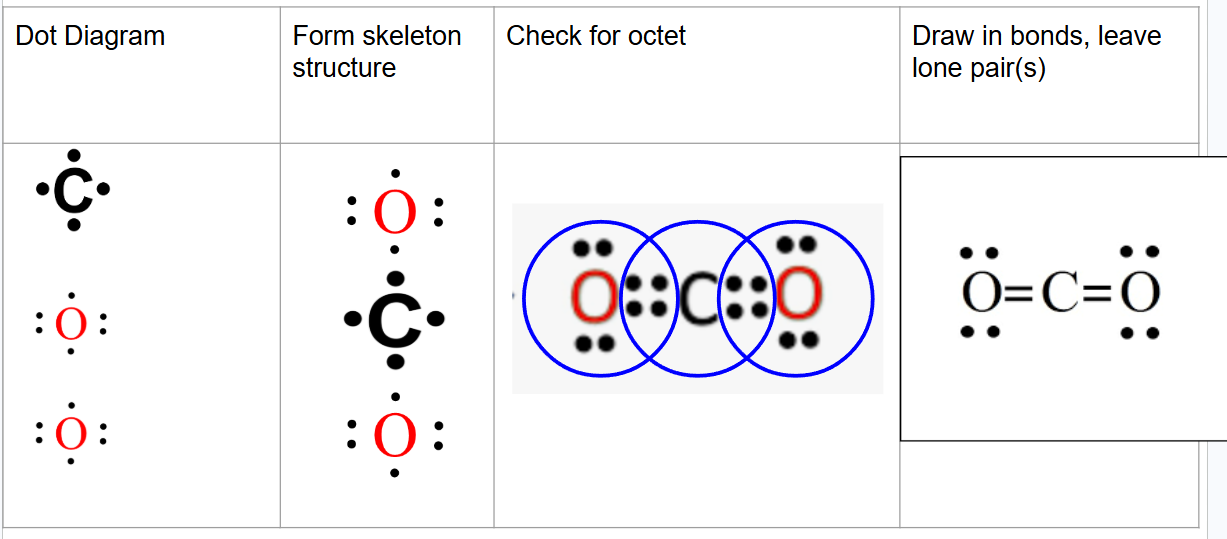
Complete Lewis Structure of and what kind of covalent bond it is:
CO2
double bond
When metallic elements become ions they:
become positively charged and lose electrons (Cation)
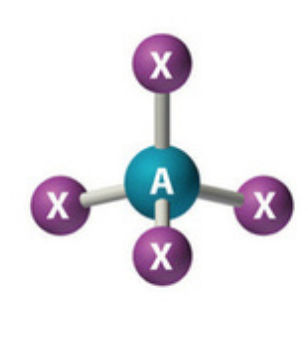
What is the VSEPR theory used to predict?
Molecular Shape
According to VSEPR, molecules adjust their shapes to keep which of the following as far away as possible?
Pairs of valence electrons
VSEPR stands for ________ theory.
Valence Shell Electron Pair Repulsion
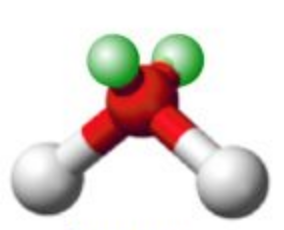
Name this molecular shape
Bent
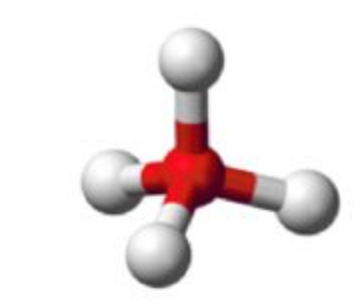
Name this molecular shape
Tetrahedral
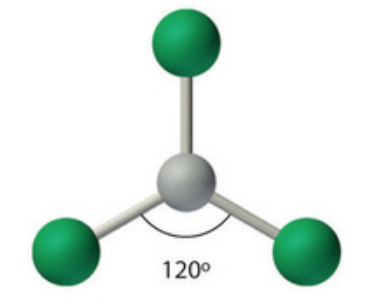
Name this molecular shape
Trigonal Planar
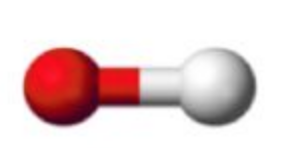
Name this molecular shape
Linear
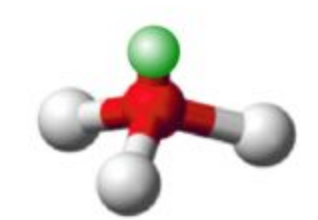
Name this molecular shape
Trigonal Pyramidal
Name this molecular shape
Tetrahedral
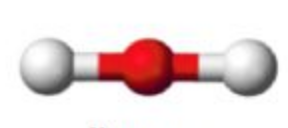
Name this molecular shape
Linear
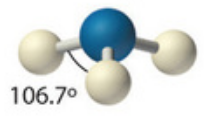
Name this molecular shape
Trigonal Pyramidal
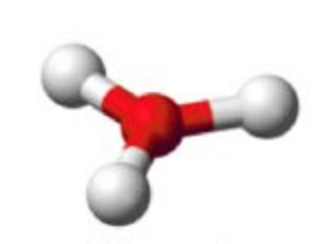
Name this molecular shape
Trigonal Planar
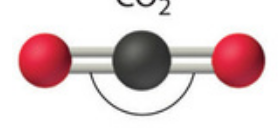
Name this molecular shape
Linear
Define Bond polarity
- based on electronegativity difference
- distribution of charge within a bond
Define Molecular polarity
- distribution of charge within a molecule
- based on bond polarities and shape
Molecular Polarity
Polar:
- Has lone pairs OR no lone pairs and asymmetrical
Nonpolar:
- No lone pairs and symmetrical
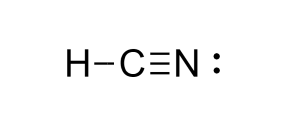
Determine Bond polarity and Molecular Polarity of:
Hydrogen Cyanide or HCN
Bond polarity
H = 2.1 C = 2.5 N = 3.0
C—H bond 2.5 - 2.1 = 0.4
C—N bond 3.0 - 2.5 = 0.5
Polar Bonds
Molecular Polarity
VSEPR shape = Linear
no lone pairs
asymmetrical
Polar Molecule

VSEPR
