Chemistry Chapter 1: Keys Studying Chemistry
1/39
Earn XP
Description and Tags
These are only for the terms listed within the slides. Remember to study commonly known conversion rates, calculation of sig figs, celsius to kelvin conversions, and rounding of sig figs
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
The study of matter, it’s properties, the changes that matter undergoes and the energy associated with these changes is what?
Chemistry
What is the definition of Matter?
Anything that has both mass and volume
Aka: how much “stuff” there is
What is the definition of Composition?
The types and amounts of simpler substances that make up a sample of matter. Like how eggs and milk are within the matter of a cake
What is the definition of Properties?
The characteristics that give each substance a unique identity
What state of matter has a fixed shape and volume?
A solid, solids may be soft, flexible, or rigid.
What state of matter has a varying shape that conforms to the shape of its container, and has a fixed volume?
A liquid. A liquid has an upper surface and that’s it.
What state of matter has no fixed shape or volume?
A gas - since it has neither, it does not have a surface.
Physical Properties are…
Things a substance does WITHOUT interacting with another substance
What are some examples of Physical Properties?
Color, melting point, boiling point, and density are some examples.
Chemical properties are…
Properties a substance shows as it interacts with or transforms into, other substances
What are some examples of Chemical Properties?
Flammability and corrosiveness are some examples.
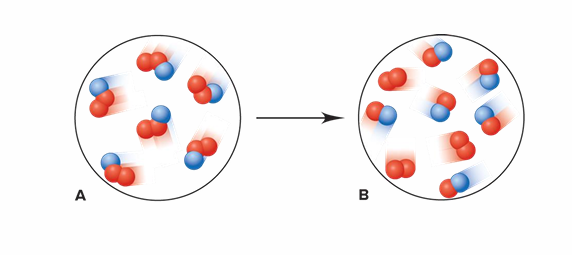
Is this an example of a chemical change or a physical change?
It is a chemical change:
Each particle of substance A is composed of one blue and two red spheres. Sample B is composed of two different types of particles—some have two red spheres, while some have one red and one blue. The composition has changed; therefore, it is a chemical change
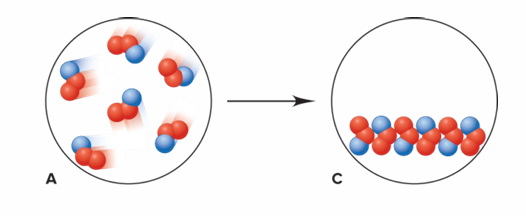
Is this an example of a chemical change or a physical change?
It is a physical change.
Each particle of C is still composed of one blue and two red spheres, but the particles are closer together and are arranged in a regular pattern. The composition remains unchanged, but the physical form is different. Therefore, it is a physical change.
Is a change of state (solid → liquid → gas) a physical or chemical change?
And are they reversible?
These changes are a physical change. Since the composition itself does not change, only the physical form.
Yes, it is reversible, since it is able to be changed by the temperature
What is the ability to do work?
Energy
What type of energy is due to the position of the object?
Potential energy
What is the energy due to the movement of an object?
Kinetic Energy
What is the equation for Total Energy?
Potential Energy + Kinetic Energy = Total Energy
What type of energy state is more stable?
Lower energy states are more stable and are favored over higher energy states
Can energy be created or destroyed?
NO!!!!! Energy cannot do either of these things
It is conserved
It can be converted from one form to another
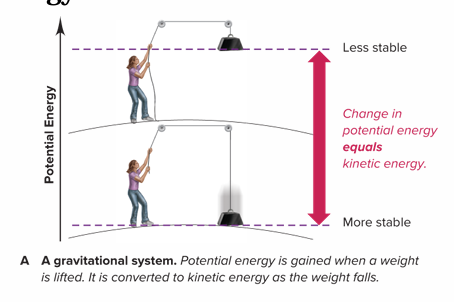
What is a gravitational system?
It is the potential energy gained when a weight is lifted is converted to kinetic energy as the weight falls. A lower energy state is more stable; therefore, the weight falls down.
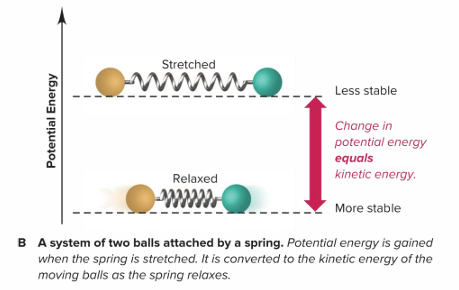
What is an example of how energy is conserved when it is transformed?
An example would be a system of two balls attached by a spring.
The potential energy gained by a stretched spring is converted to kinetic energy when the moving balls are released.
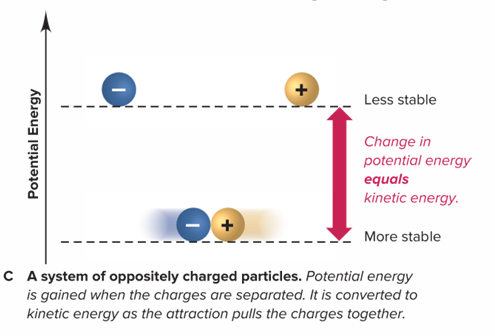
How does a system of oppositely charged particles operate?
The potential energy gained when the charges are separated is converted to kinetic energy as the attraction pulls these charges together.
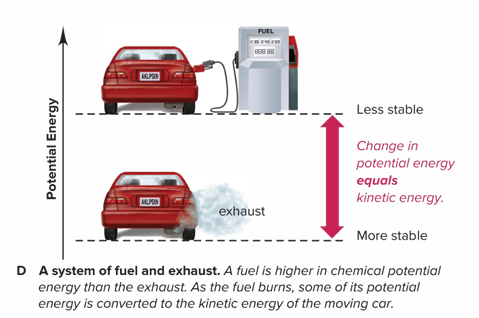
How does a system of fuel and exhaust operate?
A fuel is higher in chemical potential energy than the exhaust. As the fuel burns, some of its potential energy is converted to the kinetic energy of the moving car
What are the steps of the scientific approach?
Observations:
Natural phenomena and measured events →
Hypothesis:
A tentative proposal that explains said observations →
Experiment:
The procedure to test the hypothesis. Tests one variable at a time. (If the experiment results do not support the hypothesis, the hypothesis is revised) →
Model (Theory):
Set of conceptional assumptions that explains data from accumulated experiments: predicts related phenomena (Model is altered if predicted events do not support it) →
Further Experiment:
Tests predictions based on models.
What is a measure of how hot or cold one object is relative to another?
This measure is Temperature
What is the energy that flows from an object with a higher temperature to to an object with a lower temperature?
This energy is Heat
What temperature scale is based on the freezing point and boiling point of water?
Celsius (°C) it is also the temperature scale used most commonly around the world.
What temperature is known as the “absolute temperature scale” It begins at absolute zero and has only positive values.
Kelvin (K) - The Celsius and Kelvin scales use the same size degree although their starting points differ (It also doesn’t use the sign (°)!!)
What temperature scale is commonly used in the U.S?
Fahrenheit (°F) – The Fahrenheit scale has a different degree size and different zero points than both the Celsius and Kelvin scales.
How do you convert °C to K?
°C + 273.15 = K
is the equation
Do exact numbers limit the number of significant digits in a calculation?
NO
Exact numbers do not limit the #, since they have no uncertainty associated with them. Don’t worry!!!!
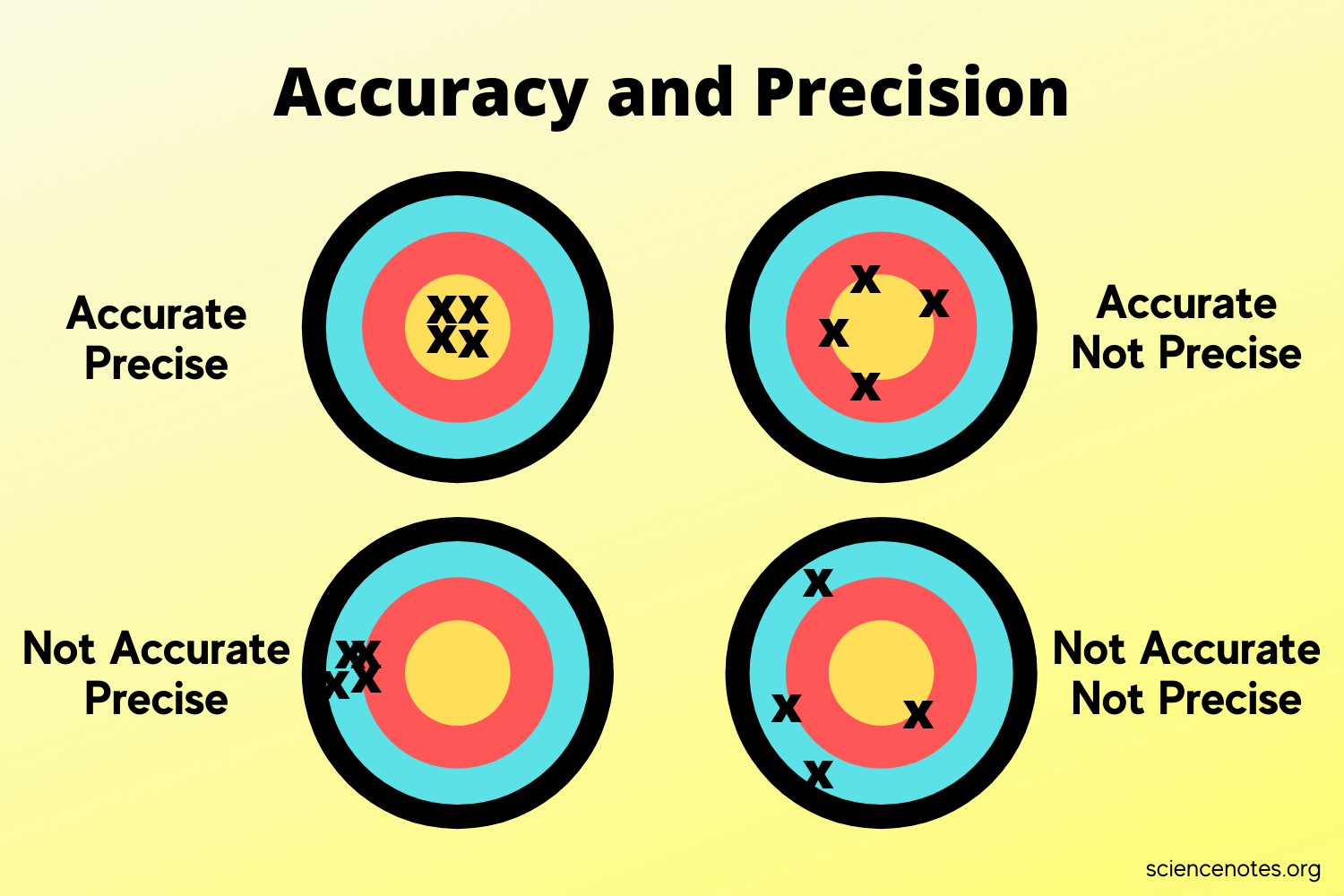
What is the difference between Precision and Accuracy?
Precision refers to how close the measurements in a series are to each other.
Accuracy refers to how close each measurement is to the actual value
What type of error produces values that are either all higher or all
lower than the actual value? (your fault)
It is a Systematic Error - This error is part of the experimental system.
What type of error produces values that are both higher and lower
than the actual value.
It is a Random Error - Random error always occurs
All measured quantities consist of a…
They consist of a number and a unit.
A conversion factor is a…
it is a ratio of equivalent quantities used to express a quantity in different units
Ex: 1 mi = 5280 ft
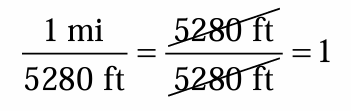
What is the mass of a sample divided by its volume?
(Mass/Volume) = _______
Density
At a given temperature and pressure, the density of a substance is a characteristic physical property and has a specific value
What properties are dependent on the amount of substance present?
Extensive Properties - some examples include mass and volume
What properties are independent on the amount of substance present?
Intensive Properties - density is an intensive property