Unit 3 Chem 2
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
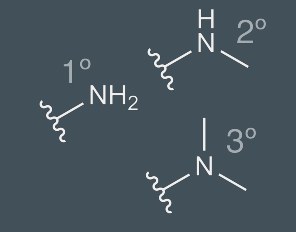
Amines
Functional group containing nitrogen atoms; primary, secondary, teritiary; basic, and polar forming hydrogen bonds
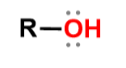
Alcohols
R-OH; suffix of -ol, hydroxyl; properties of both water and R group, larger R group properties more reflect R group
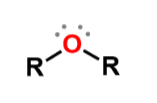
Ethers
formula R-O-R where R is an alkyl group or an aromatic ring; slightly polar molecules; water solubility similar to alcohols w/ similar mass; melting point similar to alkynes w/ similar mass; name by just adding ether to end
Carbonyl Group
Carbon atom double bonded to oxygen atom
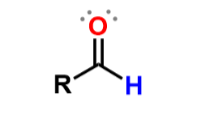
Aldehydes
carbonyl group w/ 1 R group and 1 H atom; add suffix -al
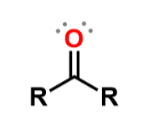
Ketones
carbonyl group bonded to 2 R groups; add -one to ending; similar to ethers in water solubility
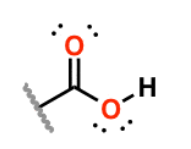
Carboxylic Acid
carbonyl w/ R group and -OH; organic compound that are weak acid proton donors; add -oic acid; always first, highest priority;
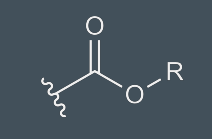
Esters
carboxylic acid w/ OR’ instead of OH; hydrogen bonding; weak acid proton donor; formed by condensation reaction between carboxylic acid and alcohol; named w/ group attached to O first, then acid part w/ -oate
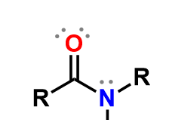
Amides
carboxylic acid w/ amine instead of -OH; forms hydrogen bonds; formed by condensation reaction between carboxylic acid and amines; named by replacing -oic acid w/ -amide
Condensation Reaction
two molecules combine to form 1 larger molecule & a smaller molecule is formed (usually water)
Electrophilic Addition
reaction where atoms are added to regions of high electron density
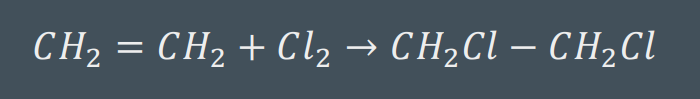
Hydrohalogenation Reaction
used to convert alkenes and alkynes into alkanes by adding either H2 molecules or halides

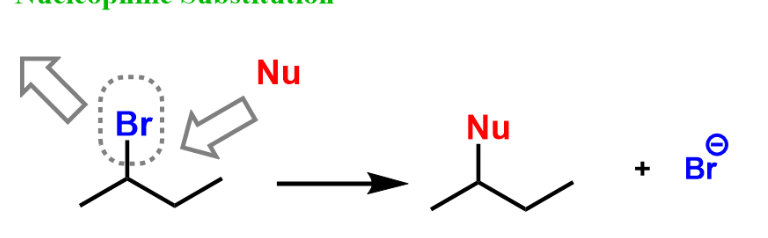
Nucleophilic Substitution
breakage in weak C-X(halide) bond where both electrons go w/ halide
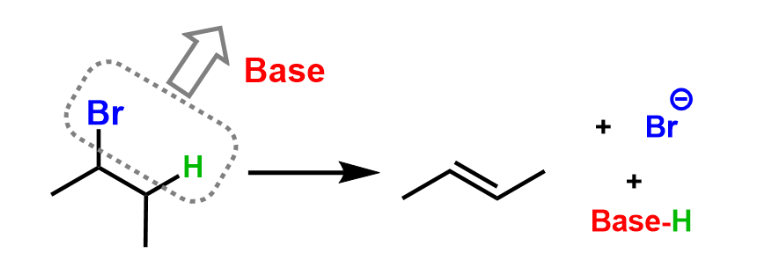
Elimination Reaction
halogen and neighboring hydrogen removed by a base