OCHEM EXAM THREE
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
What Does NABH4 Do?
-selective
reduces only aldehydes (primary alchol) and ketones to secondary alchols
What is Wittig’s Reaction, what does it do?
-converts aldehydes and ketones to alkenes
1.) R-BR 2.) Sn2 3.) PPH3—-R
What does LAH/LiAL4H do?
carboxylic acids and esters to primary alchols
reduces amides, to amines
Reduces nitriles to primary amines
What does PCC do?
-convert primary alcohols to aldehydes
- secondary alcohols to ketones.
WHat does Jones do?
Converts primary alcohols directly to carboxylic acid
Aldehydes to Carboxylic Acids
Cro3h2so4,h20/acetone
Decarboxylation
-works on beta keto acids
Acetoacetic Ester Synthesis
preparing ketones having one or two alkyl groups on the ɑ position:
1.) NaoET/ R-X H30+, Heat
Enamine Alkylation
Formation of an enamaine using NH ring and aco; with h, h20 work up forms a Ketone
HVZ
br3/pbr3
replaces the H atom on a carboxylic acid (not on the OH) with a bromien atom
with H20, it adds on a secondary br on another alpha H
DiBAL-H , -78
-reduces nitriles (-C triple bond N) to aldehydes at low temps
-reduces esters to aldehydes
LiALH(Ot)Bu3
reduces acid chlorides to aldehydes
organometallics
R-MGX
Best bases Ever
NOT COMPATABLE with CARB. ACIDS
Ketones: Tertiary OH
Acid Chlorides + Anhydrides: two equivalents of to form tertiary alcohols (adds on two groups)
Grignards
form ketones from C triple bond N
R-MGBR/H30
Ozonolysis
cleavage of alkenes to carbonyl compounds
POCL3
converts primary amides into nitriles
Micheals Addition
1,4-addition
NAOCH3, CH3OH + R-X
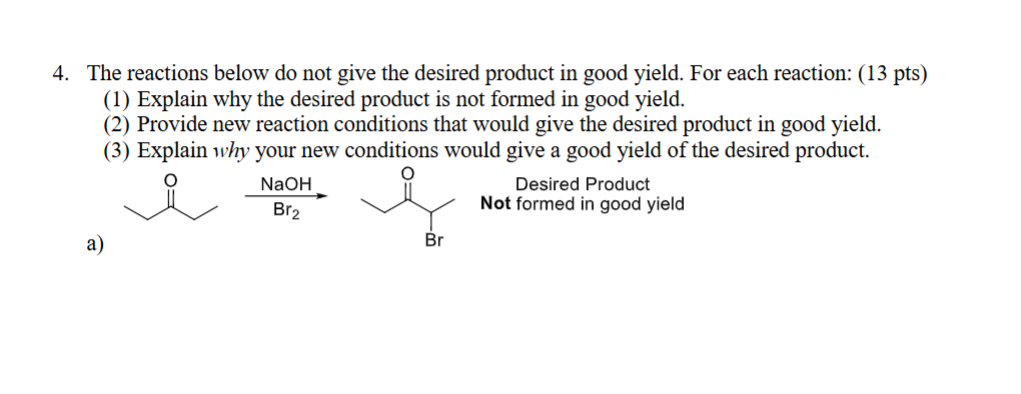
The end product is much more acidic than the starting material. Therefore, this will result in mixed end over halogenated materials due to further deprotonation.
Better option would be using TSOH due to the fact that it is not strong enough to deprotonate a mono-halogenated product.
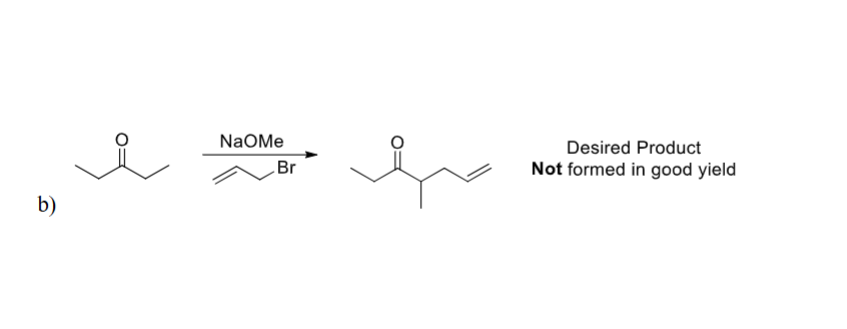
NaOMe is a reversiable base and alot of it remains in the solution in comparsion to the enolate. THereofre, it will result in an SN2 reaction. What would be better would be adding an irrev. Base such as LDA, -78 as it react fully without reversing and the enolate can attack the electrophile.
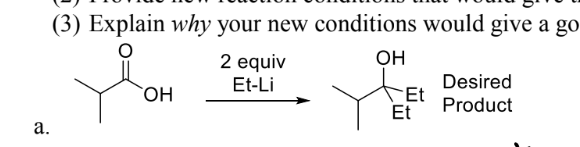
R-Li or R-MGBR are awseome bases that will deprotonate the carboxylic acid (oh) proton, resulting in the carboxylic acid forming again in work up. Change the carbox into an ester or an acid chloride as both are more E+ than the starting material