Chapter 16 Chemistry: Amino Acids, Proteins, and Enzymes
1/81
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
Proteins
in the body are polymers made from 20 different amino acids
differ in characteristics and functions that depend on the order of amino acids that make up the protein
what do proteins form?
Form structural components such as cartilage, muscles, hair, and nails
How do proteins function?
Function as enzymes to regulate biological reactions such as digestion and cellular metabolism
Such as hemoglobin and myoglobin transport oxygen in the blood
Amino acids
the molecular building blocks of proteins
What do amino acids have?
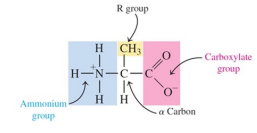
Have a central carbon atom called the a-carbon bonded to two functional groups: an ammonium group (— NH3+ ) and a carboxylate group (—COO- )
Have a central carbon atom bonded to a hydrogen atom and R group or side chain in addition to the carboxylate and ammonium groups
How are amino acids classified?
as
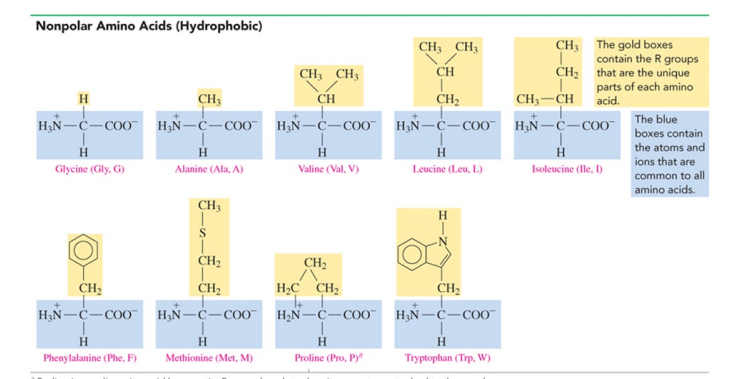
nonpolar (hydrophobic ) with hydrocarbon side chains
polar (hydrophilic) with polar or ionic side chains

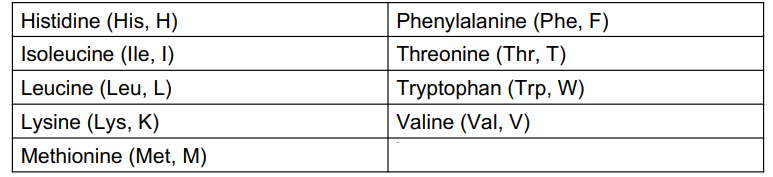
How are amino acids nonpolar?
when the R group is H, alkyl, or aromatic
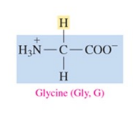
Glycine (Gly, G)
nonpolar amino acids with H as R group
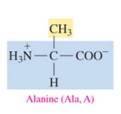
Alanine (Ala, A)
non polar amino acid with alkyl as R group
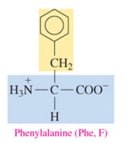
Valine (Val, V)
nonpolar amino acid with alkyl as R group
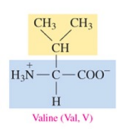
Leucine (Leu, L)
nonpolar amino acid with alkyl as R group
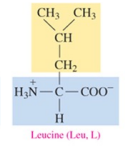
Isoleucine (Ile, I)
nonpolar amino acid with alkyl as R group
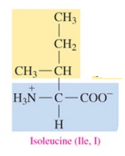
Phenylalanine (Phe, F)
nonpolar amino acid with aromatic as R group
Methionine (Met, M)
nonpolar amino acid with alkyl as R group
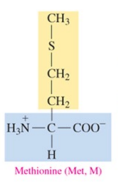
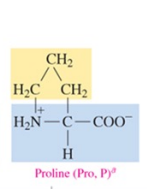
Proline (Pro, P)
nonpolar amino acid with alkyl as R group
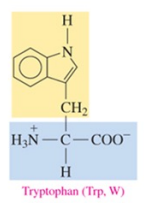
Tryptophan (Trp, W)
nonpolar amino acid with aromatic as R group
How is an amino acid polar?
when the R group is hydroxyl, a thiol, or an amide
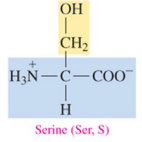
Serine (Ser, S)
polar amino acid with hydroxyl as R group
Threonine (Thr, T)
polar amino acid with hydroxyl as R group
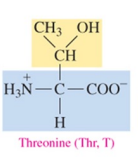
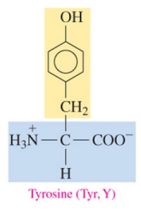
Tyrosine (Tyr, Y)
polar amino acid with hydroxyl as R group
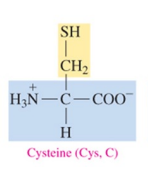
Cysteine (Cys, C)
polar amino acid with thiol as R group
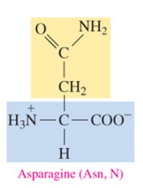
Asparagine (Asn, N)
polar amino acid with amide as R group
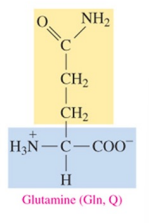
Glutamine (Gln, Q)
polar amino acid with amide as R group
How is an amino acid acidic?
When the R group is a carboxylate
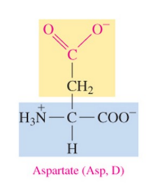
Aspartate (Asp, D)
polar acidic amino acid with carboxylate as R group
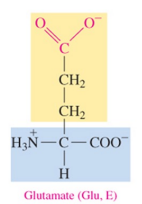
Glutamate (Glu, E)
polar acidic amino acid with carboxylate as R group
How is an amino acid basic?
When the R group is an amine, which ionizes to give an ammonium ion
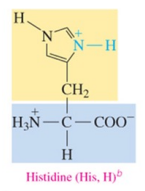
Histidine (His, H)b
polar basic amino acid with amine as R group
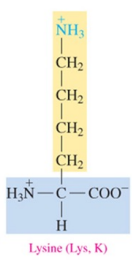
Lysine (Lys, K)
polar basic amino acid with amine as R group
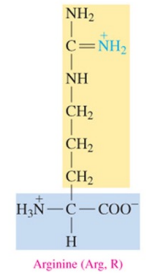
Arginine (Arg, R)
polar basic amino acid with amine as R group
Structural Formulas of Amino Acids
has
an a-carbon atom that is attached to three components:
— NH3+ , — COO- , and —H
a fourth component, an R group that differs for each particular amino acid
a three-letter or one-letter abbreviation derived from its name
Proteins: Primary structure
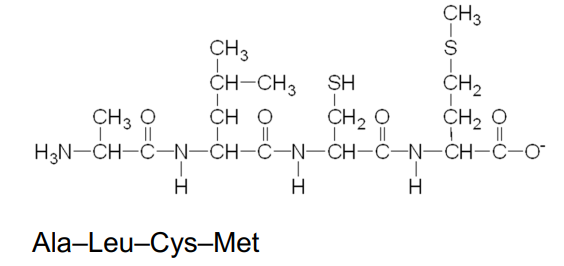
A peptide bond is an amide bond that forms when the —COO- group of one amino acid reacts with the —NH3 + group of the next amino acid.
The linking of two or more amino acids by peptide bonds forms a peptide
Peptides formed from
Two amino acids are called dipeptides
Three amino acids are called tripeptides
four amino acids are called tetrapeptides
A peptide bond
is an amide bond
forms between the —COO- group of one amino acid and the —NH3 + group of the next amino acid
Formation if dipeptide
A peptide bond between glycine and alanine forms the dipeptide glycylalanine
Naming Peptides
With the exception of the C-terminal amino acid, the names of all the other amino acids in a peptide end with yl.
Essential Amino Acids
Of the 20 amino acid used to build the proteins in the body, only 11 can be synthesized in the body
only 11 can be synthesized in the body
the other 9 amino acids are essential amino acids that must be obtained from the proteins in the diet
Primary structure of Proteins
A protein is a polypeptide of 50 or more amino acids that has biological activity.
The primary structure of a protein is the particular sequence of amino acids held together by peptide bonds.
Insulin
was the first protein to have its primary structure determined
Insulin structure
has a primary structure of two polypeptide chains linked by disulfide bonds
has a chain A with 21 amino acids and a chain B with 30 amino acids
Enkephalins and endorphins
are natural painkillers produced in the body. They are polypeptides that bind to receptors in the brain to give relief from pain
Enkephalins
are found in the thalamus and the spinal cord, are pentapeptides, the smallest molecules with opiate activity
What are two hormones produced by the pituitary gland?
nonapeptides oxytocin and vasopressin
Oxytocin
stimulates uterine contractions in labor
Vasopressin
is an antidiuretic hormone that regulates blood pressure by adjusting the amount of water reabsorbed by the kidneys.
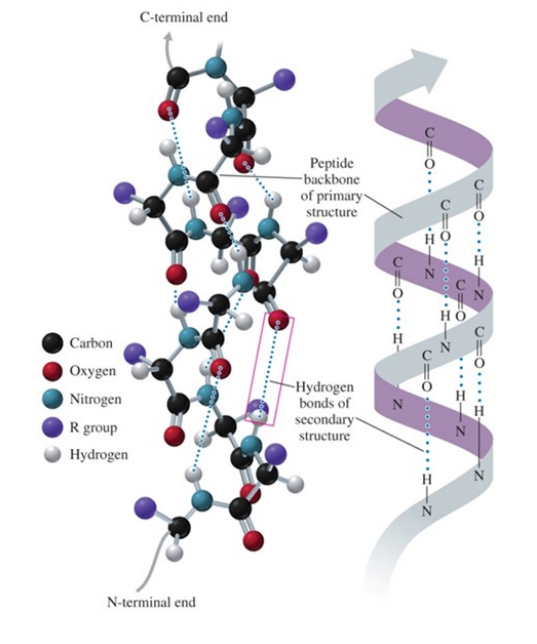
Alpha helix structure
is similar to that of a spiral staircase
acquires a coiled shape from hydrogen bonds between the oxygen of the C=O group and the hydrogen of the N—H group in the next turn.

Secondary structure of alpha helix
hydrogen bonds form between the oxygen of the C=O groups and the hydrogen of N—H groups of the amide bonds in the next turn of the a helix
the formation of many hydrogen bonds along the polypeptide chain gives the helical shape of a spiral staircase
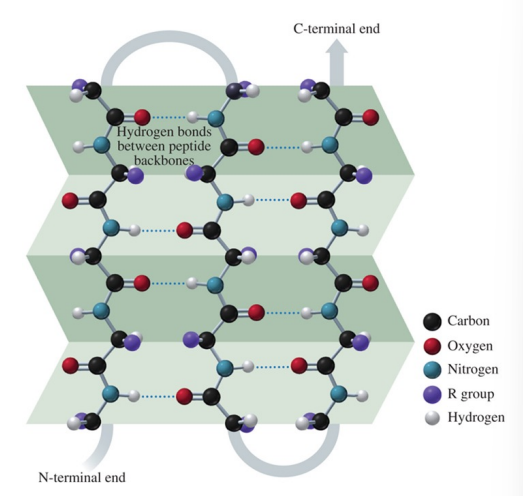
secondary structure of a beta-pleated sheet
hydrogen bonds form between the carbonyl oxygen atoms and hydrogen atoms in the amide groups bending the polypeptide chain into a sheet
Secondary structure of a triple helix
Three polypeptide chains are woven together
hydrogen bonds hold the chains together, giving the polypeptide the added strength typical of collagen, connective tissue, skin, tendons, and cartilage
Collagen fibers are triple helices of polypeptide chains held together by hydrogen bonds
In an Alzheimer’s brain..
beta-amyloid plaques and neurofibrillary tangles damage the neurons and interfere with nerve signals
Tertiary structure of a protein
is an overall three-dimensional shape caused by interactions of different parts of the chain, causing it to bend and twist
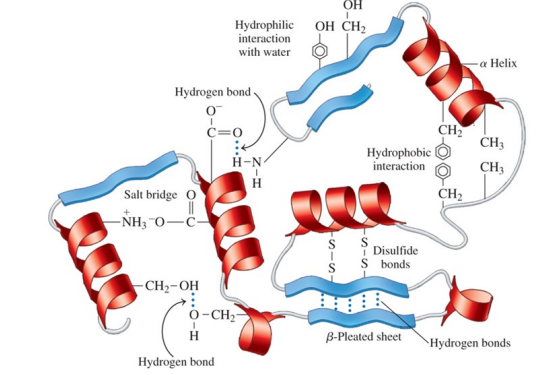
How is the tertiary structure of a protein determined?
by cross-links, the attractions and repulsions between the side chains (R groups) of the amino acids in a peptide chain
Sections of a protein interact to create the tertiary structure of a protein due to
hydrophobic interactions between two nonpolar amino acids
hydrophilic interactions between the external aqueous environment and the R groups of polar amino acids
salt bridges, ionic bonds between ionized R groups of basic and acidic amino acids
hydrogen bonds between H of a polar R group and the O or N of another amino acid
disulfide bonds —S—S— between the —SH groups of cysteine amino acids
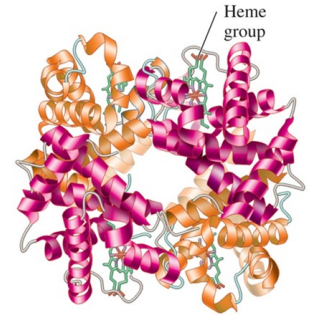
Quaternary Structure
is the combination of two or more protein units
consist of four polypeptide chains as subunits in hemoglobin
is stabilized by the same interaction found in tertiary structures
Denaturation of proteins
involves the disruption of bonds in the secondary, tertiary, and quaternary protein structures
Heat and organic compounds in denaturation
break apart H bonds and disrupt hydrophobic interaction
Acids and bases in denaturation
break H bonds between polar R groups and disrupt ionic bonds
Heavy metal ion in denaturation
react with S — S bonds to form solids
Agitation in denaturation
whipping, that stretches peptide chains until bonds break
Enzymes
are proteins that act as biological catalysts. On the surface of an enzyme, a small region called an active site binds a substrate and catalyzes a specific reaction for that substrate
increase the rate of reaction by lowering the energy of activation
The name of an enzyme
is derived by replacing the end of the name of the reaction or reacting compound with the suffix ase
could be a common name, particularly for the digestion enzymes, such as pepsin and trypsin
What does the name of an enzyme identify?
the reacting substance—for example, sucrase catalyzes the reaction of sucrose
What does the name of an enzyme describe?
the compound or the reaction that is catalyzed— for example, oxidase catalyzes an oxidation reaction
Active site
is a region within an enzyme that fits the shape of the reacting molecule called a substrate
releases products when the reaction is complete
What does the active site contain?
amino acid R groups that bind the substrate
Enzyme-catalyzed reaction
a substrate attaches to the active site
an enzyme-substate (ES) complex forms
reaction occurs and products are released
an enzyme is used over and over
Lock and key model
active site has a rigid, nonflexible shape
enzyme binds only substrates that exactly fit the active site like a lock
substrate is the key that fits the lock
induced-fit model
enzyme structure is flexible, not rigid, and adjusts to the shape of the active site in order to bind to substrate
the range of substrate specificity increases
shape changes improve catalysis during reaction- lowers the activation energy of the reaction
Isoenzymes
are different forms of an enzyme that catalyze the same reaction in different cells or tissues of the body
what do isoenzymes consist of?
quaternary structures with slight variations in the amino acids in the polypeptide subunits
The activity of an enzyme
describes how fast an enzyme catalyzes the reaction that converts a substrate to product
What affects the activity of an enzyme?
temperature, pH, and the presence of inhibitors
When are enzymes most active?
optimum temperature (usually 37 C in humans)
show little activity at low temperatures
At what temperature do enzymes lose activity?
above 50 C as denaturation occurs with loss of catalytic activity
What pH are enzymes most active?
at optimum pH
contain R groups of amino acids with proper charges at optimum pH
At what pH do enzymes lose activity?
in low or high pH as tertiary structure is disrupted
What is the optimum pH for enzymes in the body?
about 7.4
Enzyme inhibitors
are molecules that cause a loss of catalytic activity
prevent substrates from fitting into the active sites
Competitive inhibitor
Has a structure that is similar to that of the substrate
competes with the substrate for the active site
has its effect reversed by increasing substrate concentration
Noncompetitive inhibitor
Has a structure that is much different than that of a substrate
binds to an enzyme at a site other than the active site and distorts the shape of the enzyme by altering the shape of the active site
prevents the binding of the substrate
cannot have its effect reversed by adding more substrate
Irreversible inhibitor
is a molecule that causes the enzyme to lose all activity
is often a toxic substance that destroys enzymes
usually forms a covalent bond with an amino acid side chain preventing catalytic activity
may be a nerve gas, an insecticide, or an antibiotic