chem 344 final
1/76
Earn XP
Description and Tags
pre and post labs
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
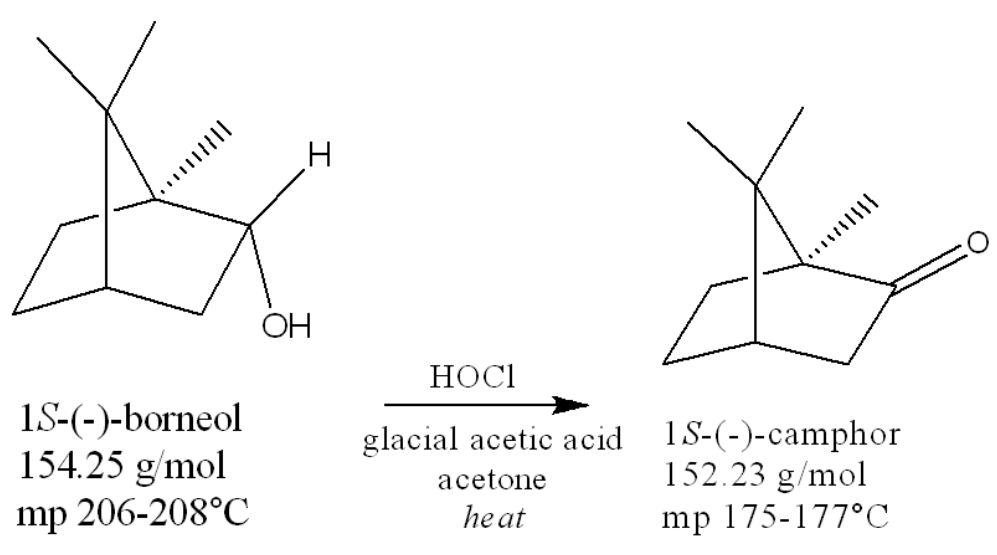
the functional group transformation taking place is __ to __
alcohol to ketone
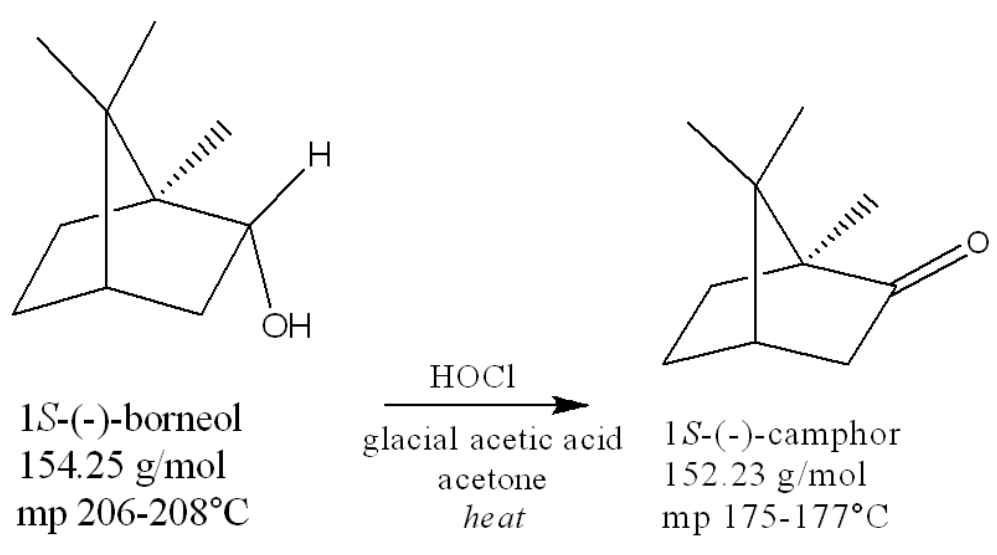
the atom being oxidized is ___. The atom being reduced is ___
carbon, chlorine
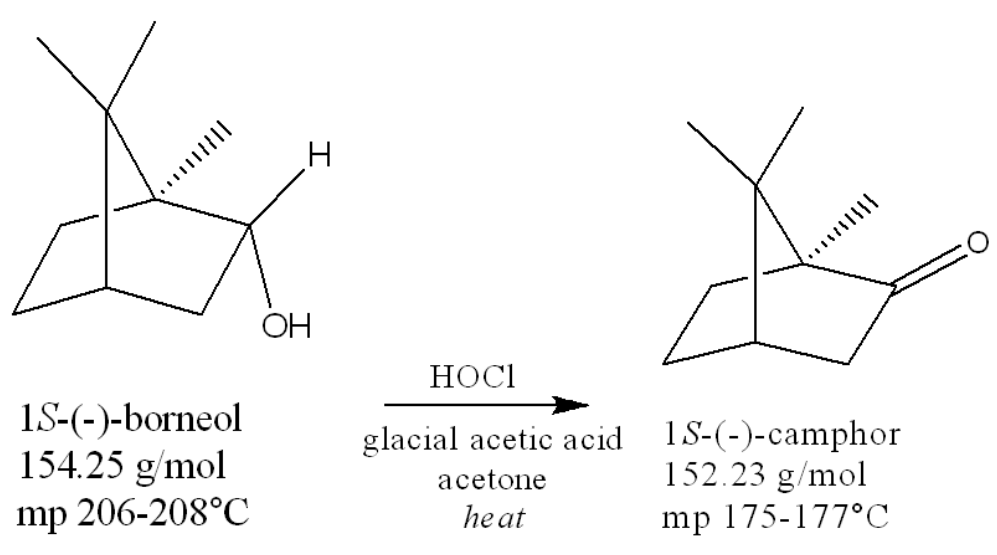
if you start with 0.35g borneol, what’s the theoretical yield of camphor?
0.35g
In the oxidation of borneol to camphor, You will be testing the reaction for completion with starch iodide paper. If excess hypochlorite is present, the paper will turn what color?
blue black, purple pink, red orange, it won’t change color
blue-black
During the work-up of the oxidation of borneol to camphor, you will perform a micro-extraction in a centrifuge tube. The organic solvent you will be using to perform the extraction is dichloromethane (DCM). Will the DCM be on the top layer or the bottom layer during the extraction? Why?
DCM will be on the bottom bc halogenated solvents are more dense than water
During the work-up of the borneol oxidation reaction you will perform a micro-extraction in a centrifuge tube. The organic solvent you will be using to perform the extraction is dichloromethane (DCM). Will your camphor product be more soluble in the aqueous layer or the organic layer?
organic
During the work-up, the dichloromethane layer is washed with sodium bicarbonate solution and there is bubbling. What is the point of the sodium bicarbonate wash? What is the identity of the bubbles?
Bubbles are CO2
sodium bicarb neutralizes remaining acids that would mess up the product purity
When you are instructed to add a drying agent (like sodium sulfate) to a solution at the end of an organic work-up, how can you tell when you have added enough?
You add just enough drying agent until some of it does not clump upon addition (i.e. you see "dry" drying agent in your flask).
At the end of some of the reaction work-ups this semester you are told to remove a solvent by evaporating under a vacuum and warming the flask in your hand and/or in a water bath. However, for just the oxidation of borneol to camphor lab, the solvent should be evaporated with MINIMAL or NO HEAT. Why is that?
Camphor has a very high vapor pressure, so it sublimes very easily. If we heat it, the vapor pressure could rise enough to where the camphor product sublimates, ruining our yield.
Where are we allowed to use DCM?
in the hood
What is the stereochemical relationship between the molecule (1S)-(+)-borneol (also called isoborneol) and (1R)-(+)-borneol?
enantiomers, diastereomers, same molecule, not related
diastereomers
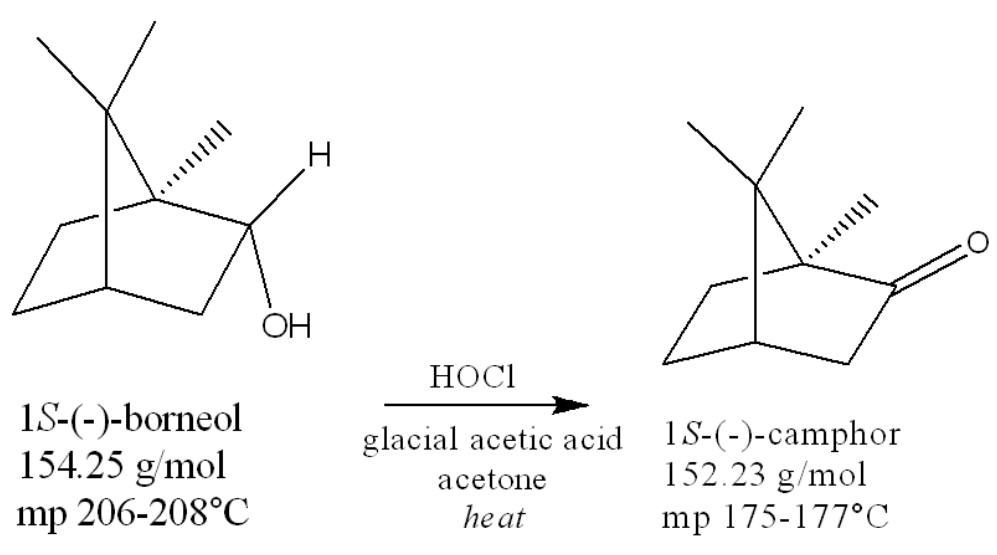
how many chiral centers in borneol
3
We used an enantiomerically-pure starting material for this reaction. Suppose we used racemic borneol instead. Propose an analytical technique that could be used to evaluate the stereochemistry of the new camphor product(s).
A polarimeter measures optical rotation, and a rotation of zero degrees would mean the product is a racemic mixture, as the enantiomers cancel each other out. If the product is a pure enantiomer, the rotation would be strongly positive or negative.
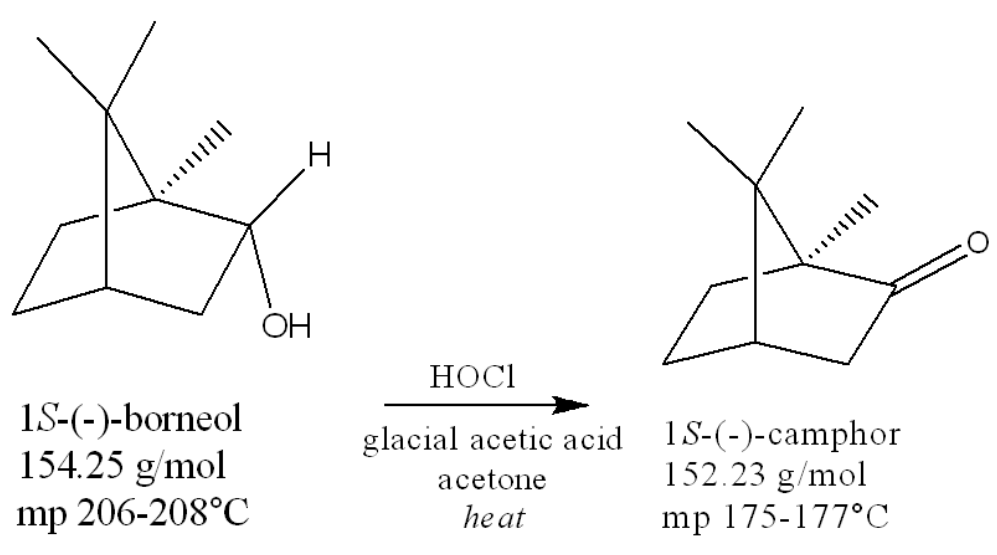
what is the role of acetic acid? select all
solvent, proton donor, oxidizing agent, reducing agent
solvent, proton donor
in the oxidation of borneol, What was the purpose of adding sodium bisulfite (NaHSO3) solution during the reaction workup?
neutralize xs acetic acid, neutralize xs HOCl, precipitate the product, reduce camphor
neutralize xs HOCl
If your solid camphor product has a melting point slightly lower than you expect, what can you conclude about the product? Select all that could apply.
The product cannot possibly be camphor
The product could be camphor contaminated with solvent (it is wet)
The product is actually borneol
The product could be camphor contaminated with starting material
contaminated, contaminated
Suppose you are monitoring the reaction progress by IR spectroscopy. What peaks would you expect to see appear/disappear as the reaction proceeds? Select all that apply.
Disappearance of strong, broad stretch (dog tongue) at 3200-3400 cm-1
Appearance of strong, broad stretch (dog tongue) at 3200-3400 cm-1
Disappearance of strong, very broad stretch (cow udder) at 3200-3600
Appearance of strong, very broad stretch (cow udder) at 3200-3600 cm-1
Disappearance of very strong, sharp stretch around 1700 cm-1
Appearance of very strong, sharp stretch around 1700 cm-1
1, 6
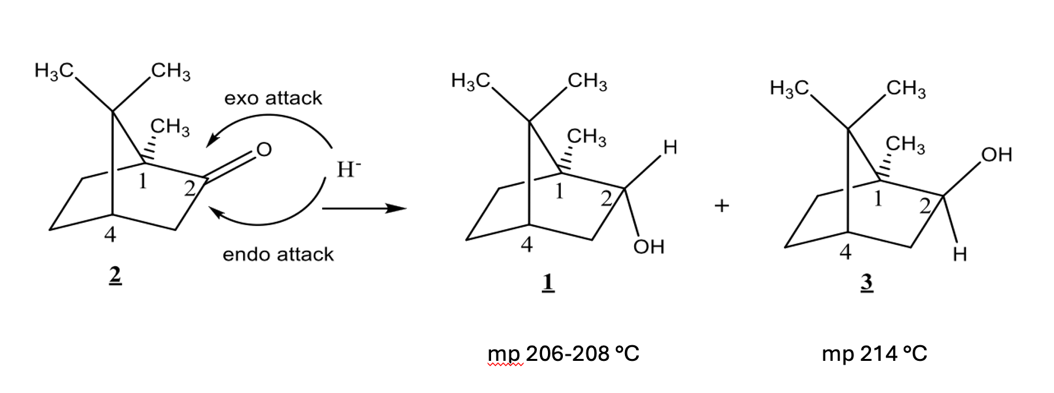
what is the stereochem relationship between 1 and 3?
same molecule, isomers, enantiomers, diastereomers, meso
diastereomers
NaBH4 is a reducing agent. What does it add to the camphor molecule to generate borneol/isoborneol?
proton (H+), Hydrogen atom, hydride (H-), neutron
hydride (H-)
NaBH4 is a reducing agent. Theoretically, how many moles of camphor can be reduced by ONE MOLE of NaBH4?
4
After the reaction mixture cools slightly, you are instructed to carefully add ice water. What is the purpose of this? (reduction of camphor using NaBH4)
water quenches xs NaBH4, forces (iso)borneol out of solution (organic, insoluble in water)
After isolating the crude organic product, you will dissolve it in ether and treat it with sodium sulfate. Why?
remove xs wetness from water. dry with NaSO4 in organic solvent so that the NaSO4 reacts only with water
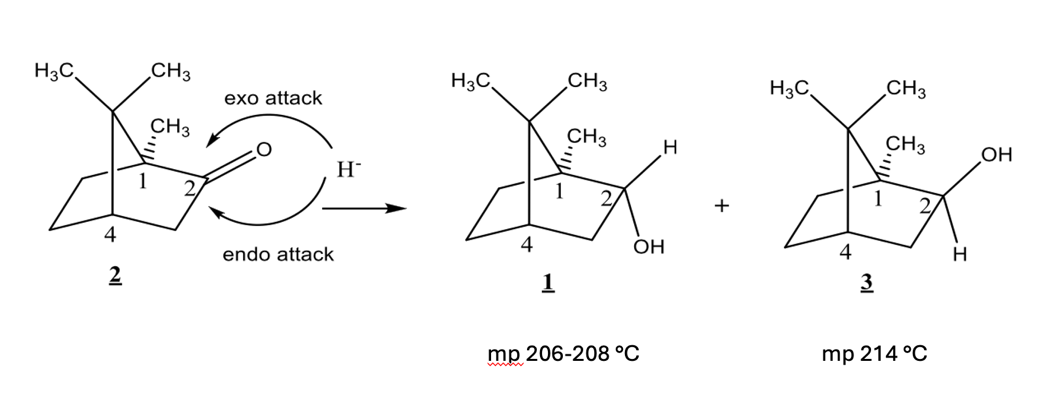
Camphor can be reduced from either the top face or the bottom face of the ring. An exo attack occurs from the top face. An endo attack occurs from the bottom face. Compound 1 is formed from an ___ attack, compound 3 formed by __ attack.
exo, endo
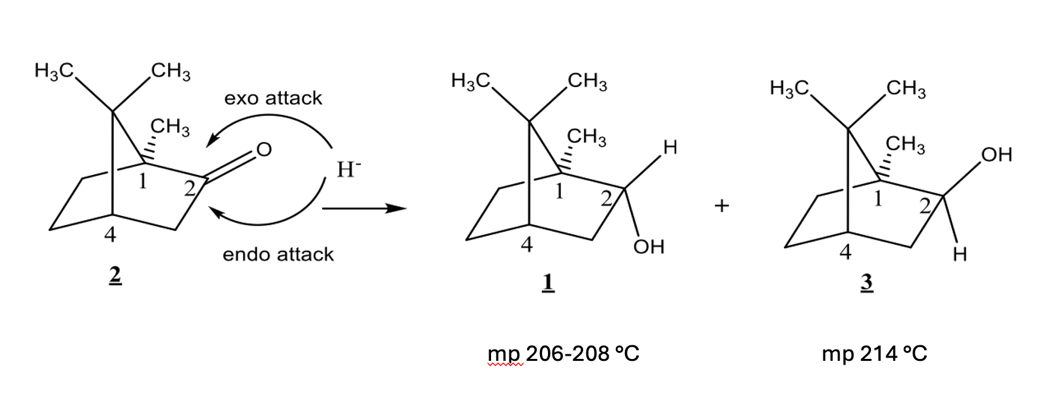
T/F: Compounds 1 and 3 have identical 1H NMR spectra
F
True or false: The aromatic ring of acetanilide is activated toward electrophilic aromatic substitution.
true
In the bromination of acetanilide, match reagents with classification.
Br2, pi e- of aromatic ring, acetic acid, HBr
solvent, Nu, ephile, byproduct
Br2 = ephile
pi e- = Nu
acetic acid = solvent
HBr = byproduct
In lecture, we use Br2 in the presence of FeBr3 (a Lewis acid catalyst) when we need to brominate a benzene ring. Here, we are just using bromine in acetic acid. Can you propose an explanation as to how our reaction still proceeds despite the absence of FeBr3?
catalyst isn’t necessary bc ring is activated by acetamido group (e- density makes it polar enough to polarize Br2 on its own)
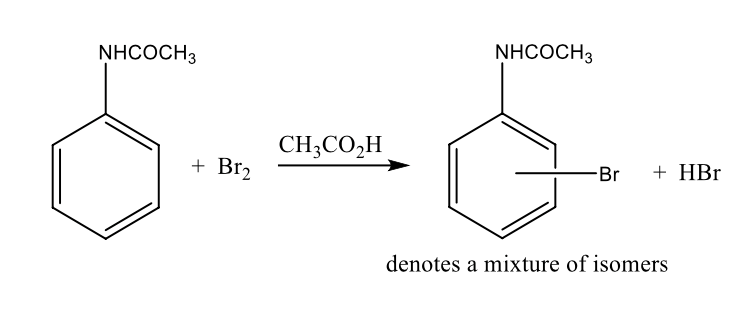
based on directing effect of amide group, which isomer(s) could theoretically be formed
OMP
ortho, para
You will be using bromine solution in this experiment. Which of the following measures will we take to ensure our safety while handling this nasty chemical? Select all that apply.
wear gloves
dispense bromine in hood only
cap or stopper my flask containing bromine before returning to the bench
neutralize excess bromine in our reaction using sodium thiosulfate solution before moving onto product collection/purification
all
bromination of acetanilide: I want to recrystallize my solid crude product using an ethanol/water mixed solvent system. This means that I should dissolve my product in (hot/cold) ethanol, then add (hot/cold) water dropwise until I reach the cloud point.
hot, hot
Suppose I am running a reaction, and I want to purify my solid crude product by recrystallization. After the reaction is complete, my crude yield is 1.789 g. After recrystallization, I am left with a final yield of 0.453 g. What is my percent recovery?
25.32
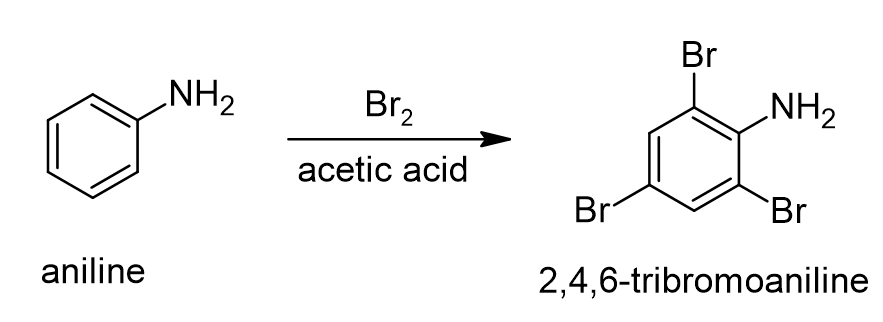
Note that in this reaction, we only formed the mono-brominated acetanilide product(s). However, the bromination of aniline under these same reaction conditions leads to the formation of 2,4,6-tribromoaniline (see below).
can you propose a reason for this difference in reactivity?
Aniline's amino group is a strong activator, and the nitrogen lone pair can resonate into the ring. This makes the ring very nucleophilic, so the ring can react over and over again until the ortho and para positions are filled.
Acetanilide is less activated. The oxygen in the amido group pulls electrons from the nitrogen, making the lone pair less available to the benzene ring. The reaction is slower and less favorable.
Based on the bromination mechanism and the predicted structure of the possible brominated products, which of the three isomers (o/m/p) do you expect to be your MAJOR product?
para
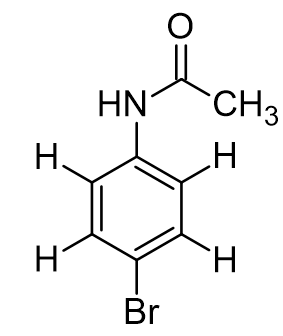
para-bromoacetanilide. how many total unique proton signals expected in 1H NMR?
4
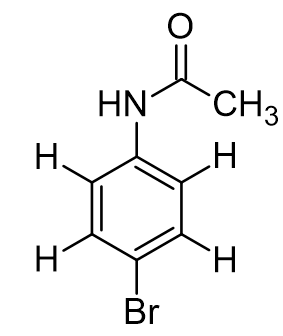
para-bromoacetanilide. which proton(s) would we expect to resonate the furthest upfield?
CH3, NH, aromatic ring next to amide, aromatic ring next to Br
CH3
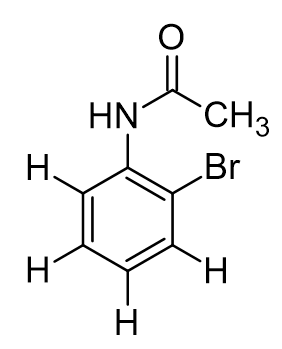
ortho-bromoacetanilide. how many total unique proton signals in 1H NMR
6
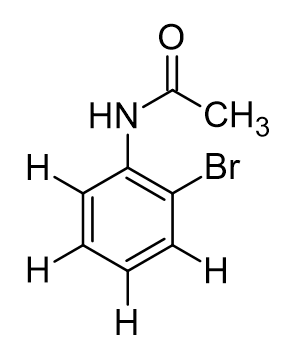
Suppose we exclusively generated and isolated the ortho-bromoacetanilide product and characterized it by IR spectroscopy. Assign each major signal to its characteristic bond.
3280 cm-1 (very strong but broad)
1662 cm-1 (very strong and sharp)
763 cm-1 (strong)
3032 cm-1 (medium strength, sharp)
C=O stretch, N-H stretch, C-Br stretch, Csp2-H stretch
3280 cm-1 (very strong but broad) N-H stretch
1662 cm-1 (very strong and sharp) C=O stretch
763 cm-1 (strong) C-Br stretch
3032 cm-1 (medium strength, sharp) Csp2-H stretch
The last step of the bromination of acetanilide reaction is the abstraction of a proton from the ring by a bromide anion (Br-). What is the driving force behind this step?
the formation of HBr, which is a strong acid
the bromide anion is a weak base, so it wants to react
restoration of aromaticity to the ring
removing the proton helps decrease unfavorable steric interactions in the product
restoration of aromaticity to the ring
bromination of acetanilide. When you visualize your TLC plate using the UV lamp, you'll notice that your spots for your reactant and product mixture will fluoresce (glow). Can you propose a chemical explanation for this phenomena? (hint: look at the end of Chap. 16 in the textbook)
Benzene rings have pi systems that can absorb UV light. The electrons in pi orbitals enter a higher energy state and release energy in the form of light
The IUPAC names of many of these compounds contain (+) and (-) notations, which indicate the direction each compound rotates plan-polarized light. Specifically, (+) compounds rotate light in the clockwise/counterclockwise direction, and (-) compounds rotate light in the clockwise/counterclockwise direction.
clockwise, counterclockwise
True or false: (+) and (-) notation directly tells you if a chiral center is R or S.
false
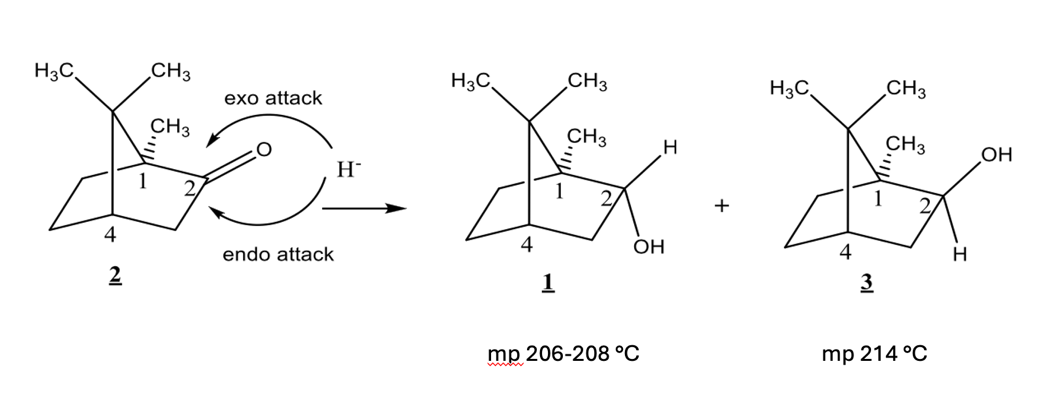
Borneol (Compound 1) has which of the following:
exo H
endo H
exo OH
endo OH
1H NMR signal at 4.0 ppm
1H NMR signal at 3.6 ppm
exo H
endo OH
1H NMR signal at 4.0
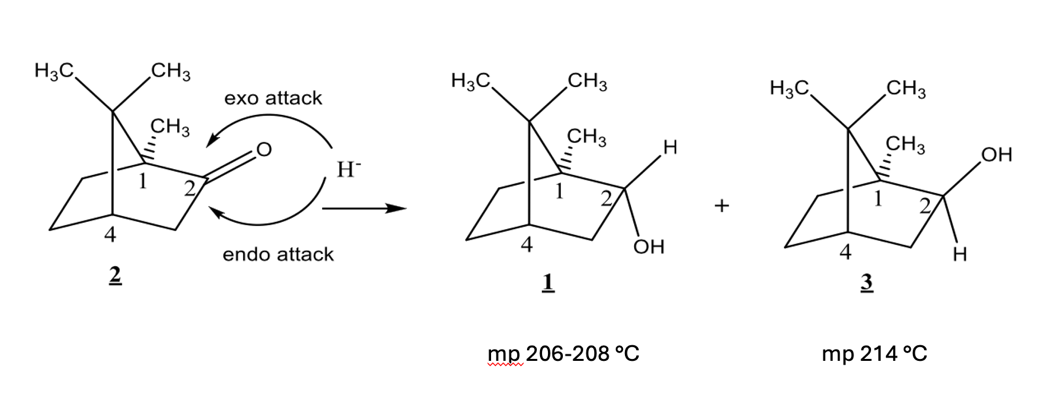
isoborneol (compound 3) has which of the following:
exo H
endo H
exo OH
endo OH
1H NMR signal at 4.0 ppm
1H NMR signal at 3.6 ppm
endo H
exo OH
1H NMR at 3.6
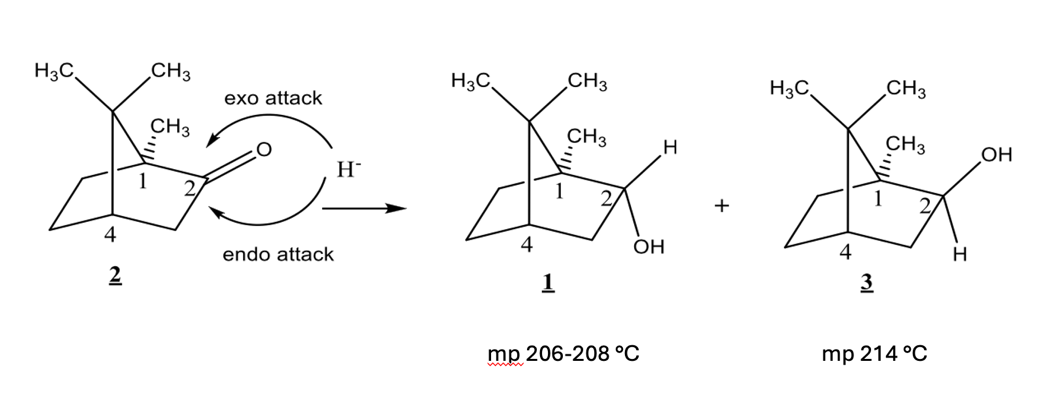
based on mechanism, which would you expect to be major?
compound 3 (isoborneol)
Consider the following 1H NMR spectral data:
Compound | Integration Value for Diagnostic Proton Signal |
Borneol | 1.00 |
Isoborneol | 3.14 |
What percentage of the product mixture is borneol? Round to the nearest whole number.
24
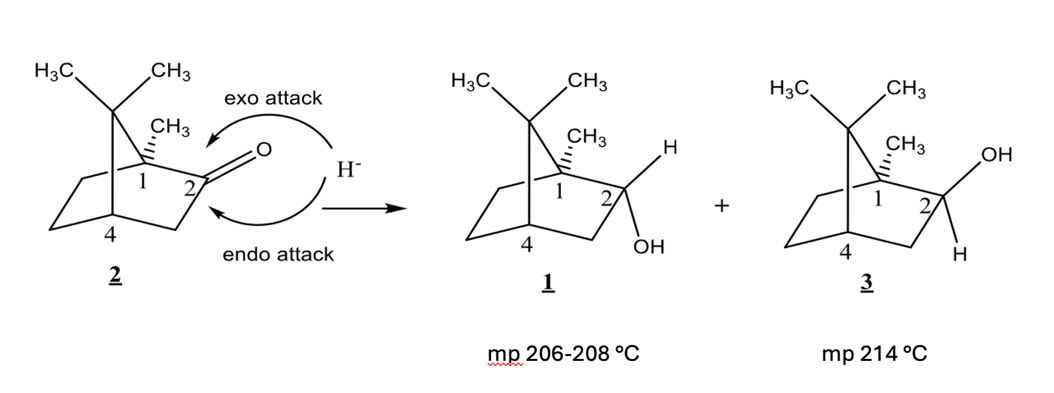
Would IR spectroscopy have been a useful technique to characterize the reaction product mixture? Why or why not?
no. diastereomers- contain same functional groups
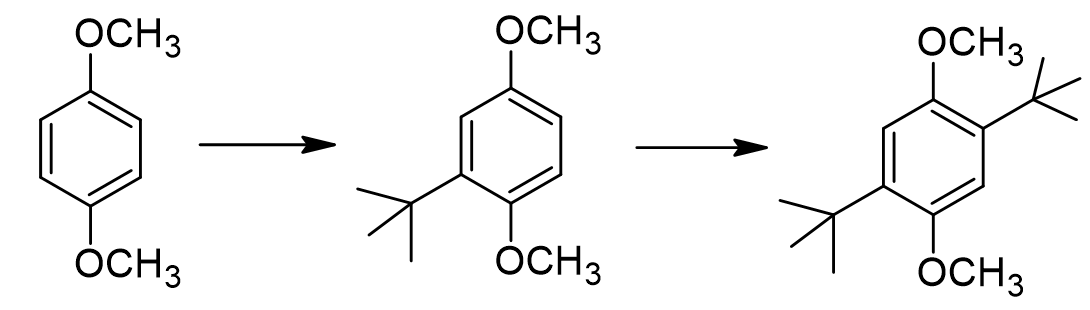
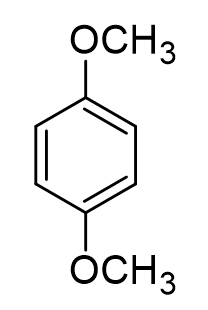
match:
1, 4 dimethoxybenzene
tert butanol
acetic acid
sulfuric acid
solvent, catalyst, Nu, ephile
1, 4 dimethoxybenzene Nu
tert butanol ephile
acetic acid solvent
sulfuric acid catalyst
The concentrated sulfuric acid you use in this experiment has a molarity of approximately 18 M. Assuming you use exactly 1.0 mL of sulfuric acid, how many millimoles (mmol) of sulfuric acid did you use?
18
tert-butanol has a density of 0.78 g/mL and a molar mass of 74.12 g/mol. Assuming you use exactly 0.25 mL of tert-butanol, how many millimoles (mmol) of tert-butanol did you use?
2.63
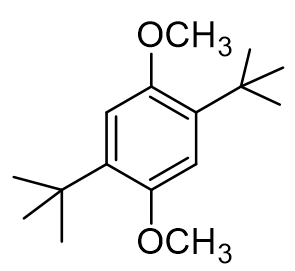
How many moles of t-butanol are required to form 1 mole of dialkylated product?
2
What is the relationship between the two methoxy substituents?
para
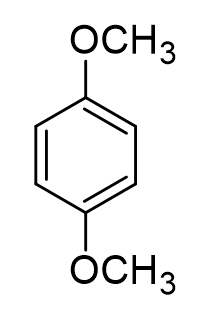
what type of directing effect do methoxy substituents do?
ortho/para, meta, none
ortho/para
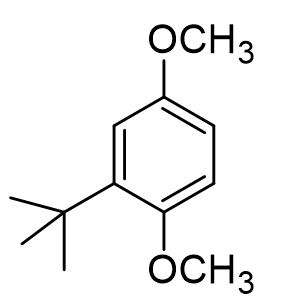
What is the directing group effect of the alkyl group on the benzene ring?
ortho/para, meta, none
ortho/para
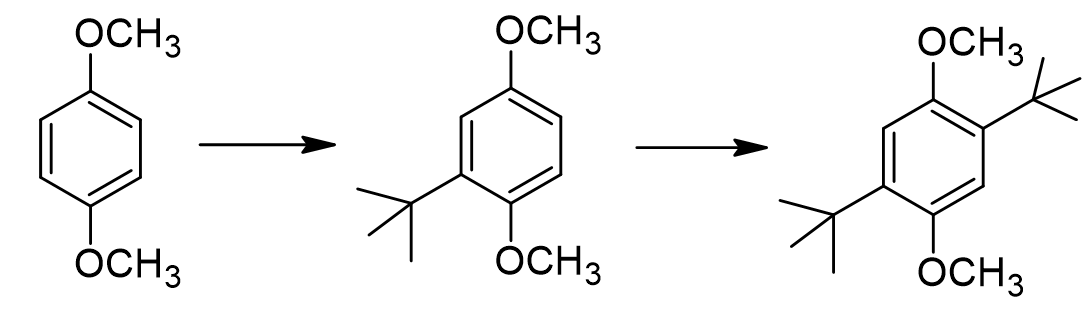
True or false: the monoalkylated intermediate demonstrates reinforced directing effects between at least one of the methoxy groups and the new tert-butyl group.
true
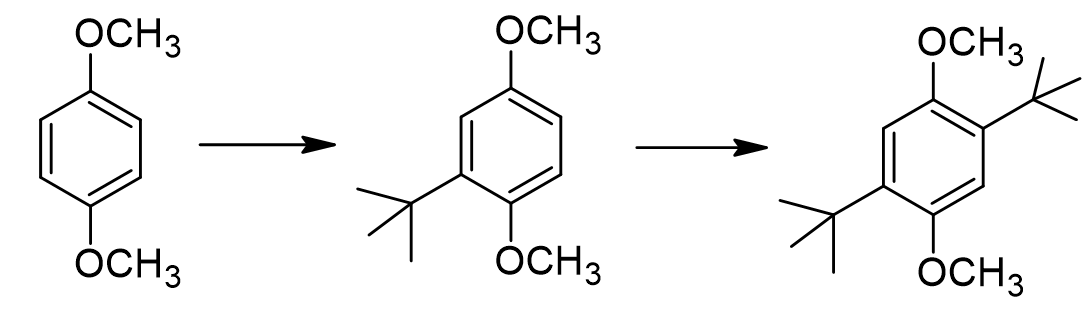
True or false: the monoalkylated intermediate contains a less activated/more deactivated benzene ring compared to the starting material.
false
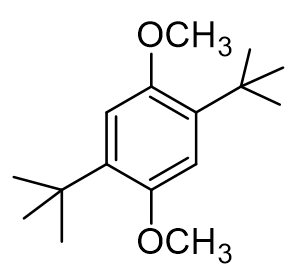
How many 1H NMR signals do we expect to see in the structure of our dialkylated product?
3
You are instructed to run melting point determination and TLC characterization on both the crude and pure products. Why?
We do both to compare how well the reaction has proceeded. The crude product melting point would probably be a little lower and broader because it hasn't been recrystallized / completely purified yet.
The TLC will show the extent of the reaction. The crude product would probably have two spots because the reaction hasn't fully proceeded yet- one for monoalkylated intermediate, and one for the dialkylated product. The final product is supposed to have one spot, proving that the recrystallization was successful in converting all of the intermediate.
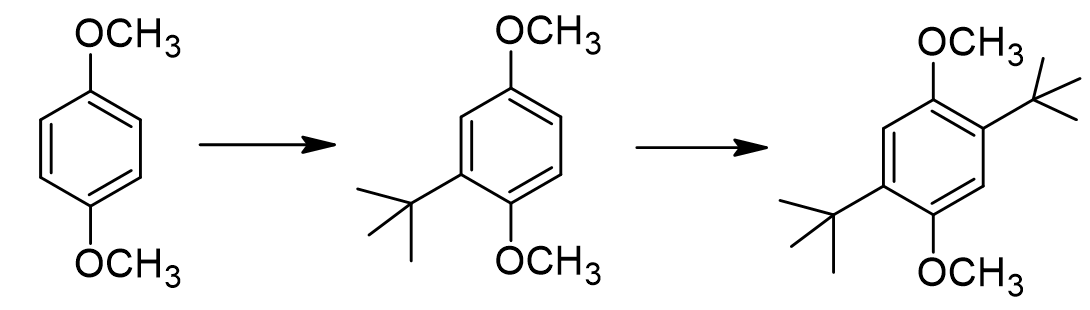
Why do we not form any tri-alkylated or tetra-alkylated product, even when we use a gross excess of tert-butanol?
steric hindrance
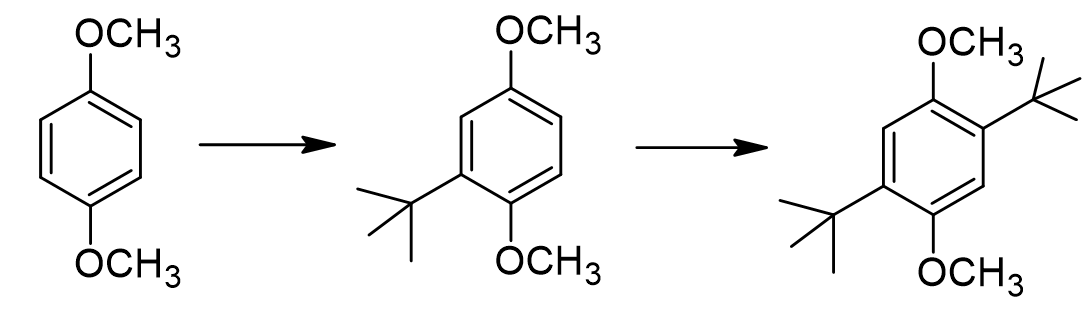
Normally, we need a Lewis acid catalyst like AlCl3 for Friedel-Crafts reactions. However, this specific reaction proceeds in the presence of just a small amount of sulfuric acid. Can you propose a reason why?
ephile is stable carbocation, and substrate has strong activating methoxy groups. so it’s already pretty reactive
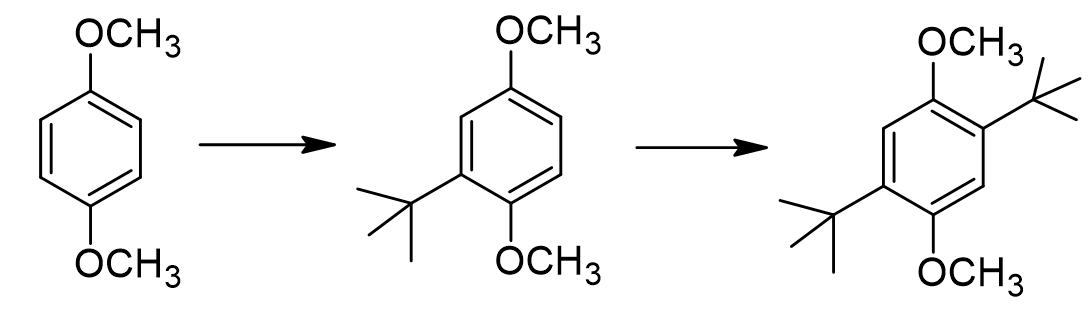
Normally when we do a Friedel-Crafts alkylation, we stop at the mono-alkylated product. Why does this substrate continue reacting to form the dialkylated product?
alkyl groups are strongly activating, so more alkyl groups make it more reactive (donate e- thru resonance to increase ring e- density)
What is the advantage of using an electrophile like tert-butanol for Friedel-Crafts alkylation over a different alcohol like 2-propanol or 2-butanol?
The carbocation formed from tert-butanol is tertiary, so it's pretty stable. 2-propanol or 2-butanol would be dehydrated to form secondary carbocations, which are less stable. It's easier (and faster) to create a stable carbocation in the rate determining step.
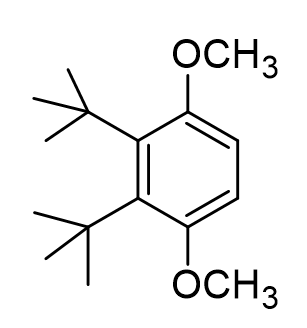
Theoretically, it could be possible for us to form a different di-alkylated product (one option shown below). Can you propose a reason we do NOT form this product?
cause ts ugly twin
jk The tert butyl groups are super bulky, so this product would have tons of steric strain esp between methoxy groups
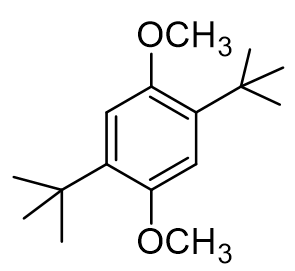
True or false: all of my 1H NMR signals of the dialkylated product should appear as singlets.
true
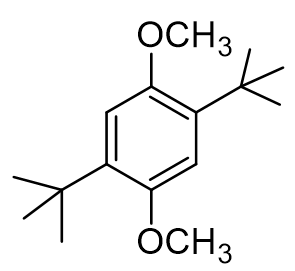
True or false: the methoxy protons (OCH3) are shielded compared to the tert-butyl protons.
false
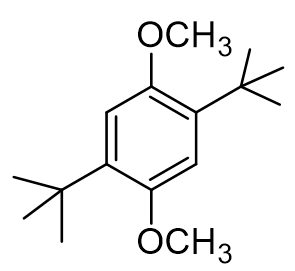
how many 13C NMR peaks
6
This week, we used a 1:1 mixture of EtOAc:hexanes by volume to develop our TLC plates. This is VERY different from the bromination experiment, where we used approximately a 7:1 mixture of EtOAc:hexanes by volume. What does this tell us?
my dialkylated product is more polar than our previous bromination product
my dialkylated product is less polar than our previous bromination product
my dialkylated product is roughly the same polarity as our previous bromination product
less
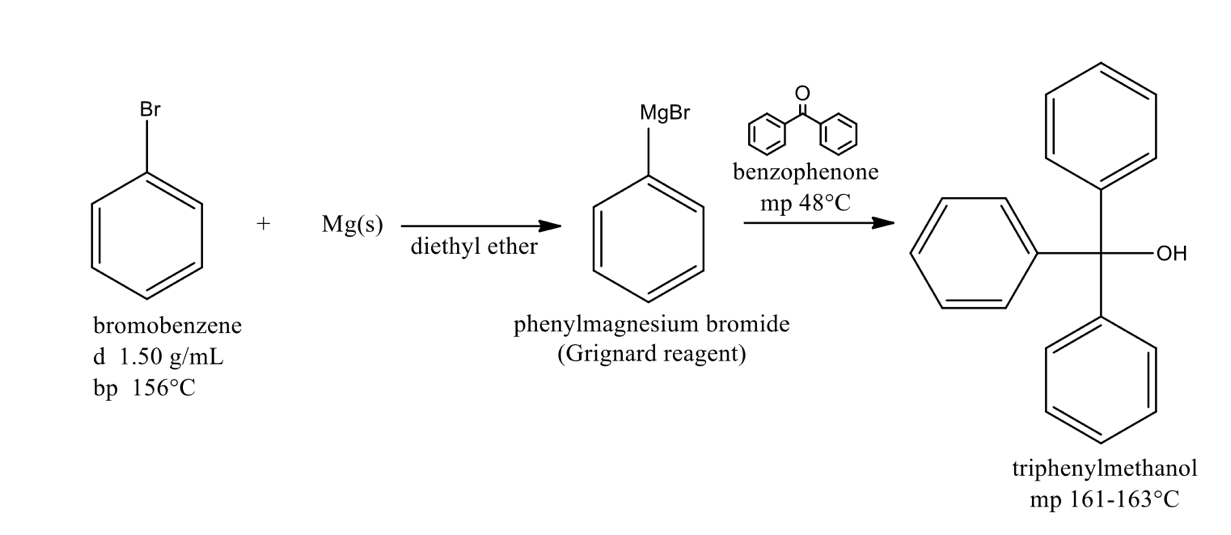
The overall result of this Grignard synthesis is the conversion of a ketone (benzophenone) to triphenylmethanol, which is a: primary, secondary, tertiary, quaternary alcohol
tertiary
True or false: Grignard reagents are highly sensitive to water.
true
What measures will we take in grignard to prevent the introduction of ambient water into our reaction? Select all that apply.
flame-dry or oven-dry our glassware before starting
attaching a drying tube filled with drying agent to our reaction flask
using anhydrous diethyl ether for our reaction
all
True or false: We can isolate and store the Grignard reagent for later use.
false
Suppose the synthesis of your Grignard reagent is not going well, and you're having issues getting the reaction started. What steps can I take to try and make it work?
add a crystal of iodine to the flask and heat gently
use a glass stir rod to grind the magnesium mixture against the flask walls while gently heating
find a friend whose reaction is working, then take a few drops of their reaction liquid and add it to your flask
yell angrily at your reaction until it decides to start working
talk nicely to your reaction until it decides to start working
1, 2, 3
How will you know when the in-situ synthesis of your Grignard reagent (phenylmagnesium bromide) is fully completed?
appearance of magnesium metal
disappearance of magnesium metal
evolution of gas
color change
disappearance of magnesium metal
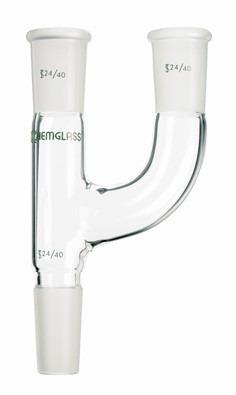
claisen adapter

microscale drying tube

centrifuge tube

rubber septum