LAB 1: Thin-Layer Chromatography : Analyzing Analgesics
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Chromotography
the separation of compounds based on physical properties
--------------
REQUIREMENTS:
- need a mobile phase
- need a stationary phase
Thin Layer Chromatography (TLC)
a sensitive, fast, simple, and inexpensive analytical technique used measure the separation of two or more compounds
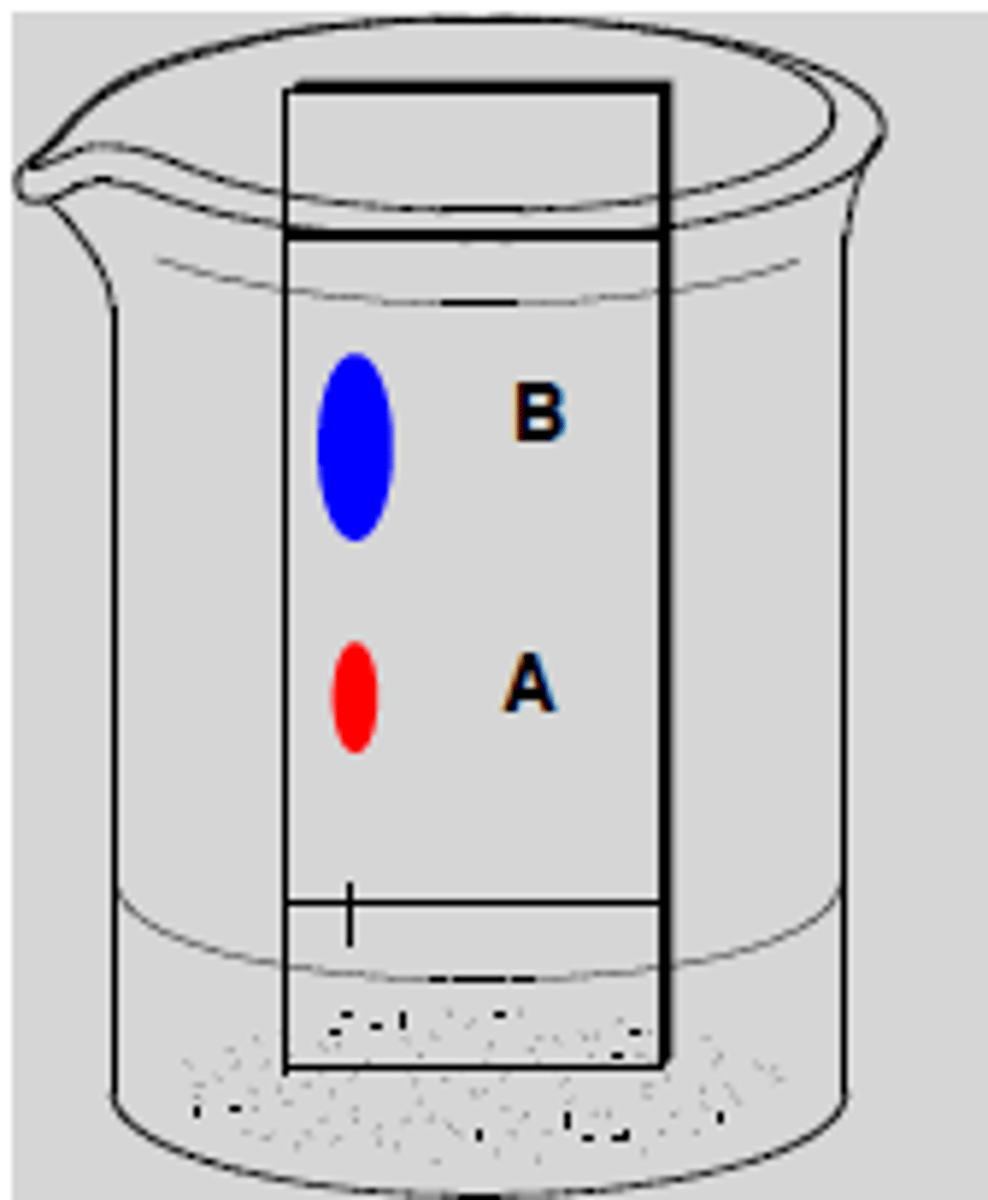
What does TLC determine
- number of components in a mixture
- identity of two or more substances
- monitors the progress of a reaction
- determine the effectiveness of purification
- determine the appropriate conditions for a column chromatographic separation
- monitor the column chromatography
mobile phase of TLC
liquid solvent
- can range from nonpolar to increasing in polarity by mixing solvents
stationary phase of TLC
solid solvent
- is always the most polar in the system (plates is usually silica or alumina)
when choosing stationary and mobile phase solutions
determine how molecules would act with the two phases
- free state
- absorbed state
- in constant equilibrium
Free state (TLC)
dissolved in the liquid or gaseous mobile phase
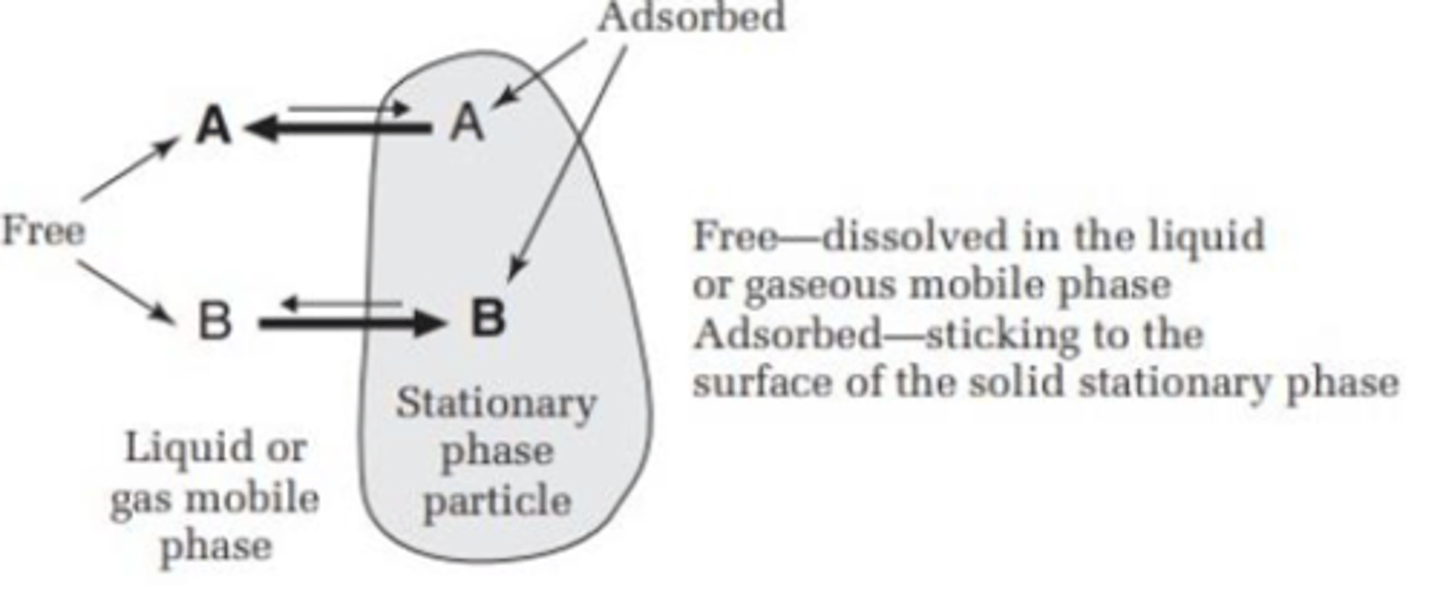
Absorbed state (TLC)
sticking to the surface of the solid stationary phase
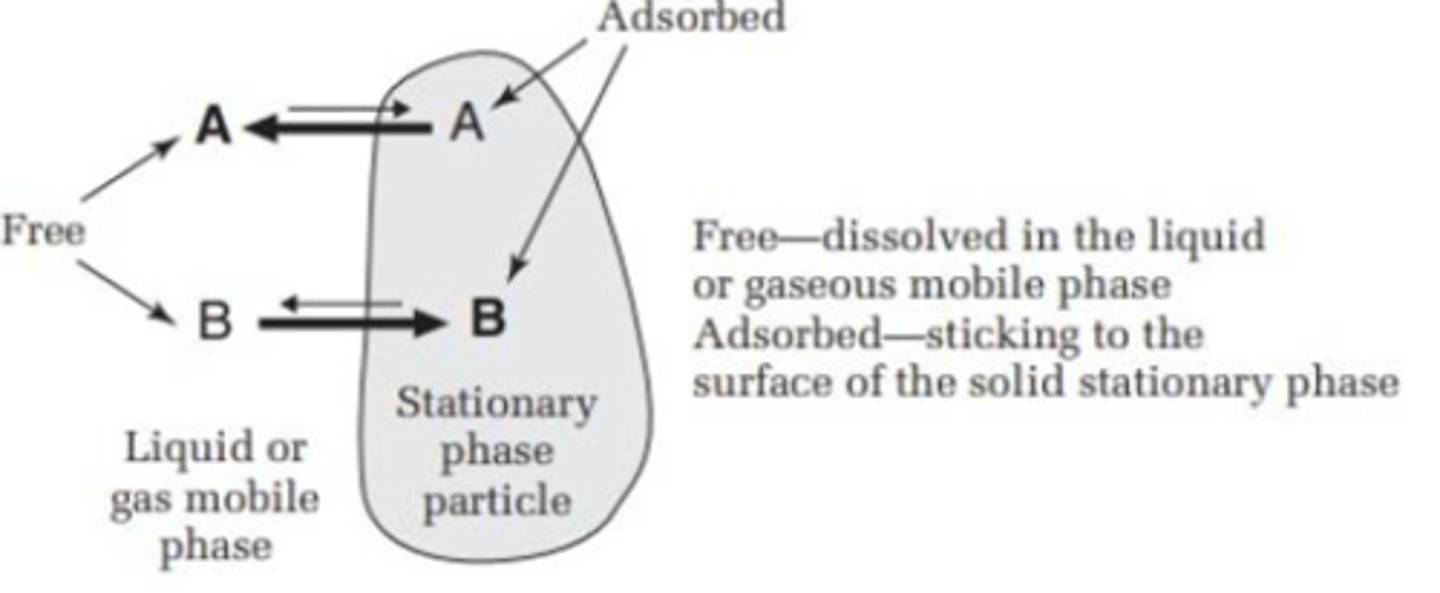
the difference in the strength of attraction (TLC)
strength of attraction between the molecules and the mobile/stationary phase causes the progress at different rates and therefore separation
Factors that effect separation in TLC
- size
- polarity
- hydrogen bonding
Trend for polarity and travel distance on TLC
TREND:
- increasing the polarity makes compounds run faster
How can you get the best separation on a TLC experiment?
you need to find the correct mixture of solvents
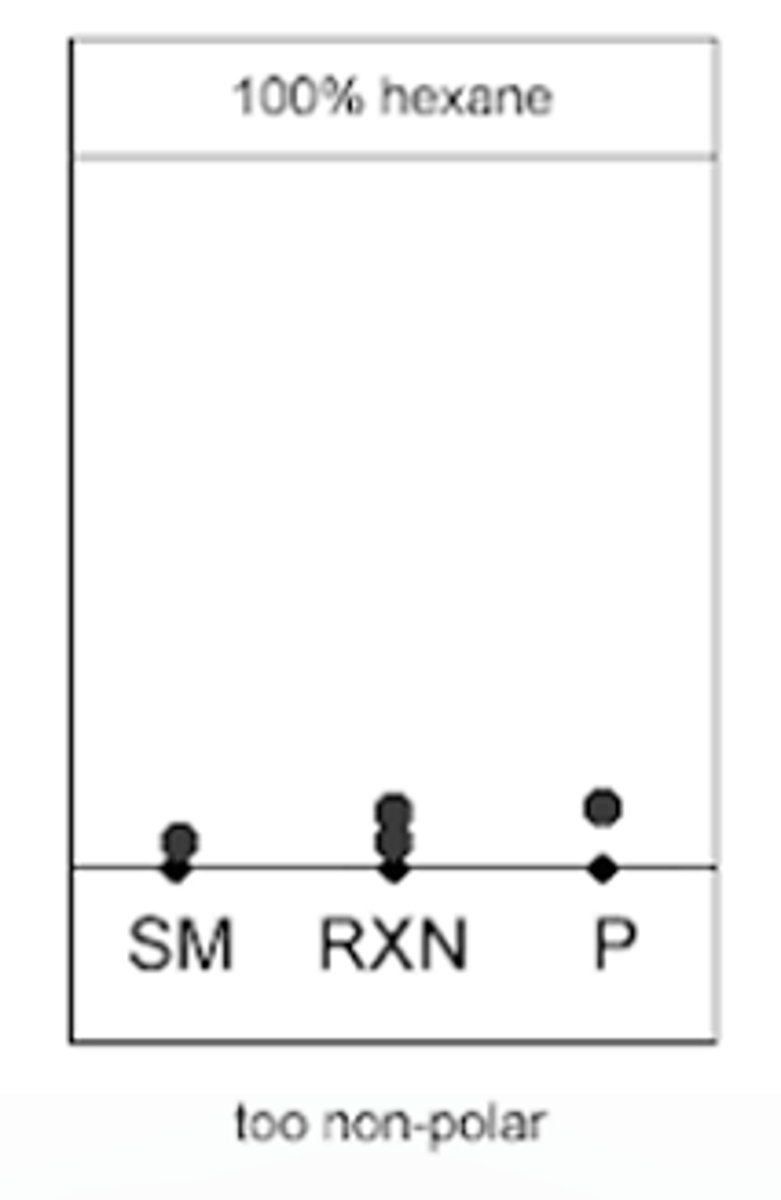
100% hexane (TLC EXAMPLE)
starting material, reaction progress and the product will barley move from the starting point
--------------
WHAT DOES THIS MEAN:
- the mixture of solvents was too non polar
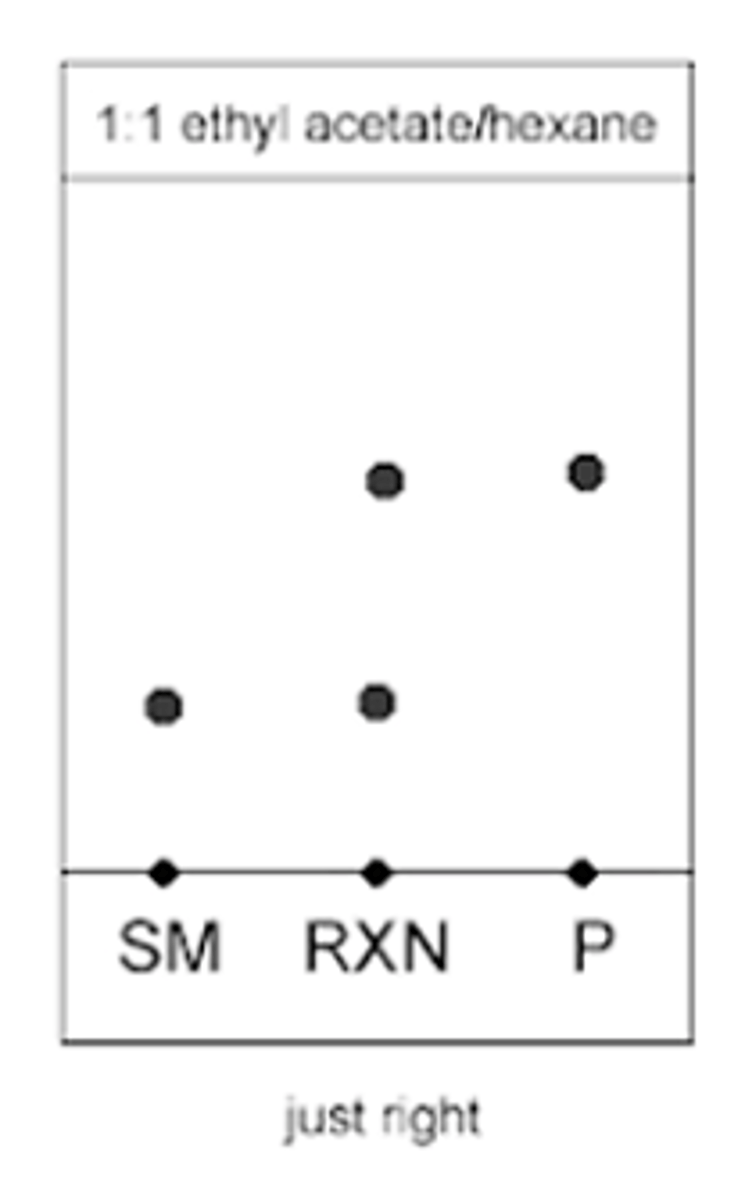
1:1 ethyl acetate/hexane (TLC EXAMPLE)
Starting material, reaction, and product will move towards the middle of the TLC plate
--------------
WHAT DOES THIS MEAN:
- the mixture of solvents was correct, "was just right"
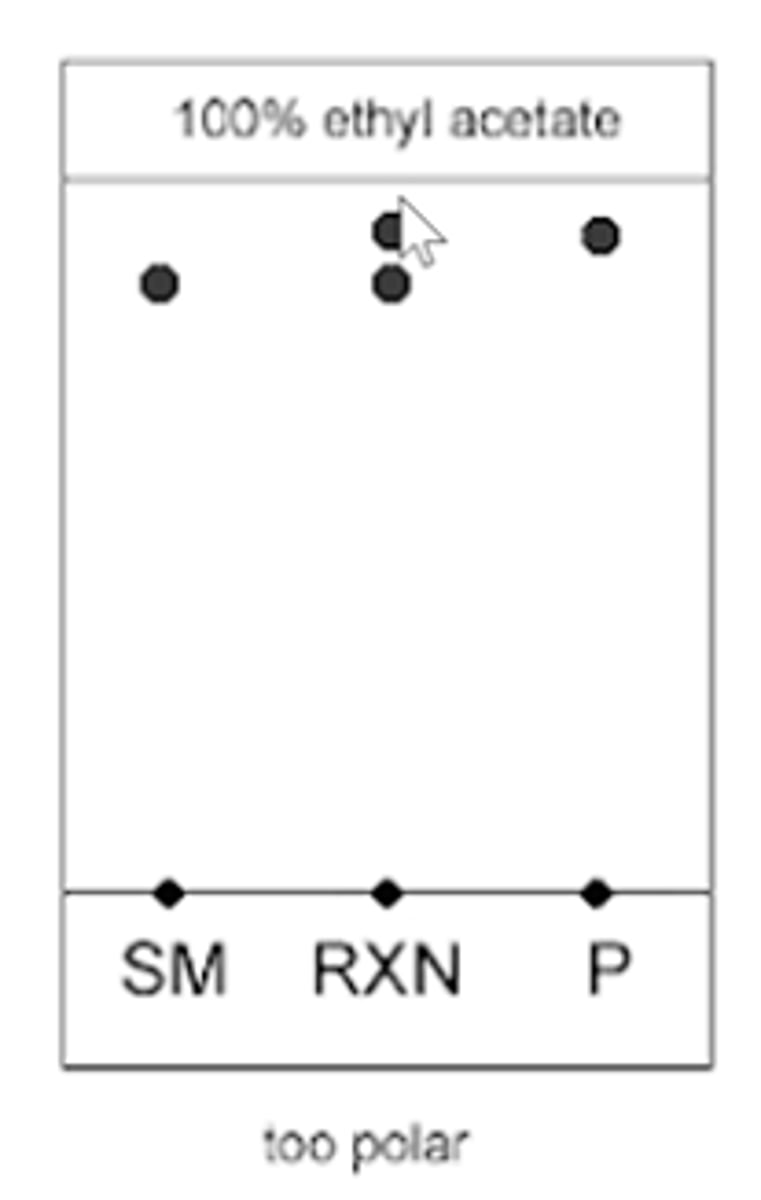
100% ethyl acetate (TLC EXAMPLE)
starting material, reaction, and product will move very fast and will reach near the top of the TLC plate
- risk of reagents running off the plate
--------------
WHAT DOES THIS MEAN:
- the mixture of solvents was too polar
How to run TLC plate
1. obtain a small beaker and watch glass
2. draw a straight line 1 cm from the bottom (WITH A PENCIL)
3) make small/narrow spots on the straight line (let the solvent evaporate on the plate)
4) check the spots under UV
5) create a developing chamber
6) once the solvent is reaching the near top of the TLC plate immediately stop and mark the solvent line (WITH A PENCIL) and let it dry
7. lightly circle spots under UV then calculate Rf
why do we use a pencil when labeling TLC plates
- it prevents smugging (unlike a pen)
- lead is inert meaning it wont react with the compounds or the solvent

why mark the 1 cm line?
when the plate is immersed in a development chamber the spots don't wash off because they are not touching the solvents
Development chamber
WHATS INSIDE THE BEAKER:
- 10 ml of solvent
- saturated filter paper (used to saturate the entire development chamber)
- a standing TLC plate
--------------
WHATS ON TOP OF THE BEAKER:
- The beaker will be covered with foil (used to make sure that solvent doesn't evaporate out)
- watch glass
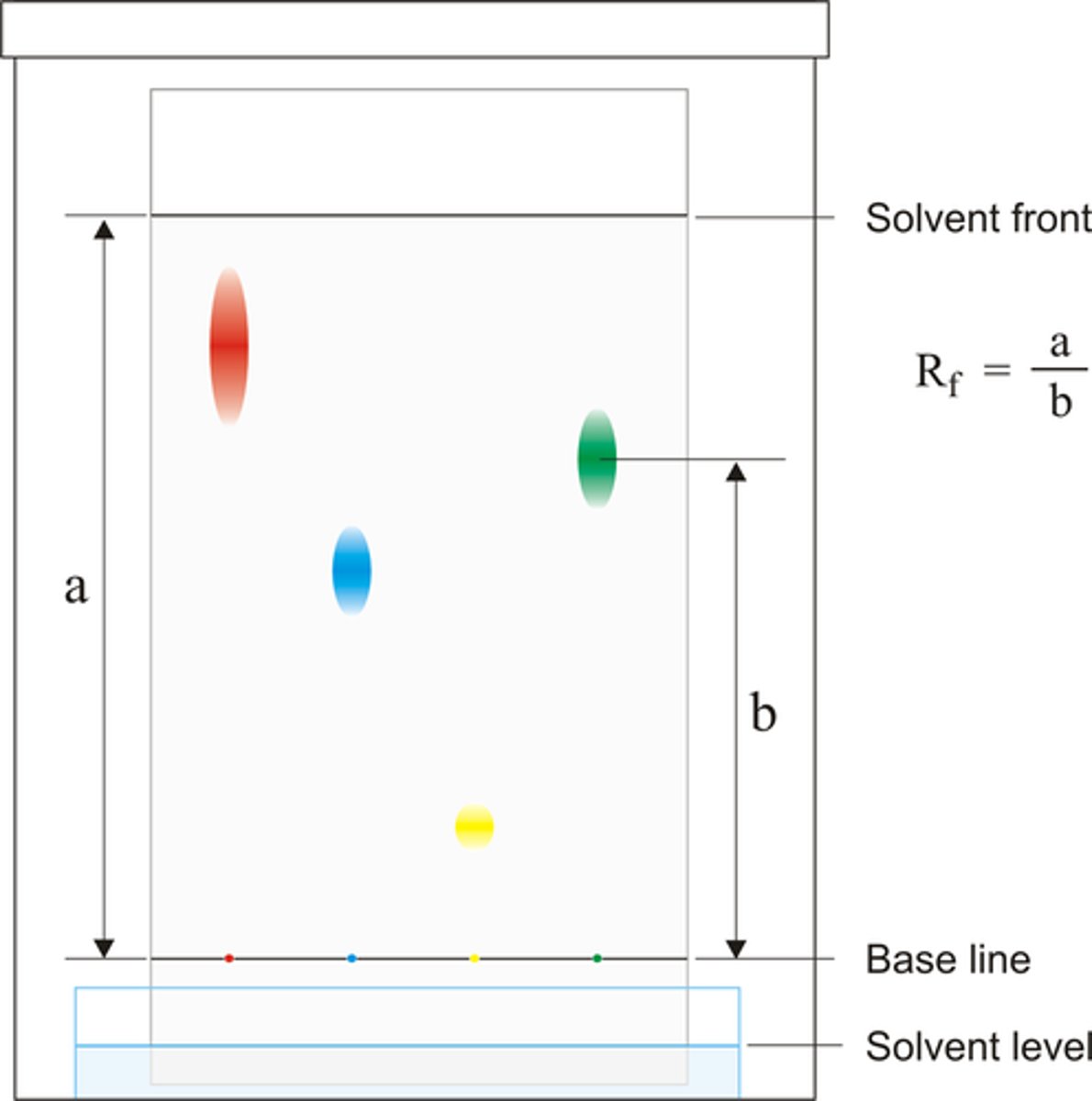
solvent front
the furthest point reached by the solvent
common mobile phase listed by increasing polarity
LOOK AT PICTURE
--------------
KEY SUMMARY
- its from least polar (TOP) to most polar (BOTOOM)

UV light
used to observe the seperation of spots on a TLC plate
--------------
- fluorescent indicator will mark the plate green which will yield a dark purple or bluish spots on plate
--------------
WAVELENGTH OF UV LIGHT:
- 254 nm (nanometers)

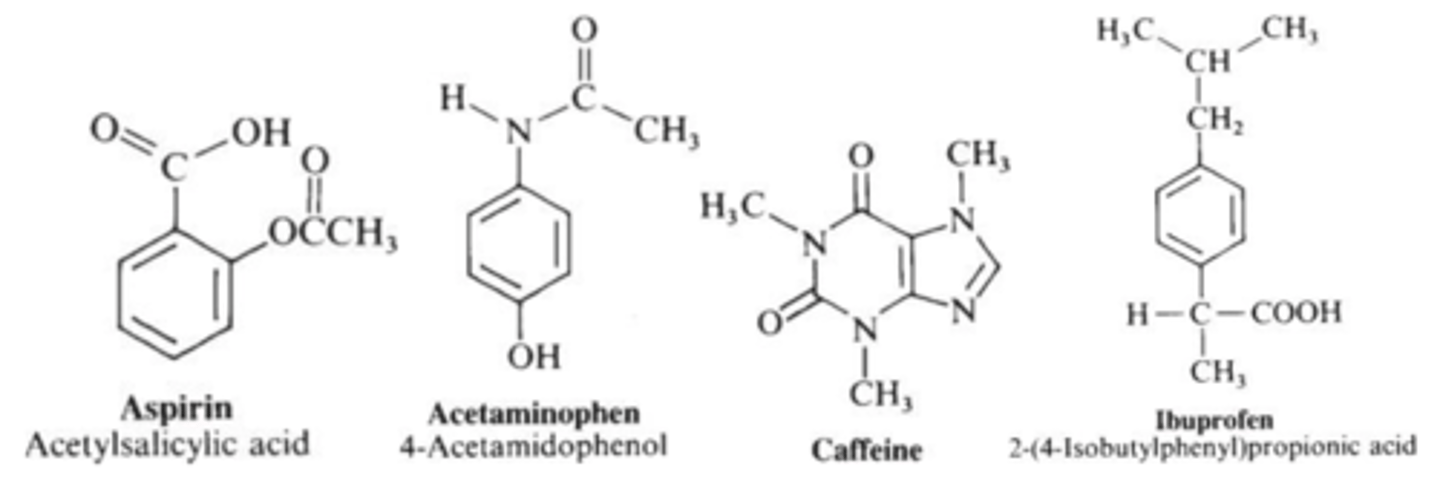
Compounds used in lab 3
- acetylsalicylic acid (aspirin)
- acetaminophen
- caffeine
- ibuprofen
what are the two states that molecules on TLC plates are in equilibrium in between
Free state and absorbed state
sort the following mobile phases by increasing polarity: (hexane, acetic acid, ethyl acetate, and ethanol)
FROM WEAKEST TO STRONGEST
1.) hexane
2.) ethyl acetate
3.) ethanol
4.) acetic acid
If you get a streaking spot on a TLC plate, what was wrong with the sample?
it means the the samples that were spotted were heavily concentrated which will result in streaks
Why is filter paper used in a TLC chamber?
POURPOSE:
- used to ensure that that the air inside the TLC chamber is saturated with solvent vapor
What are two factors that affect how far a compound moves on a TLC plate?
1.) polarity
- if the sample is non-polar it will rise to the top of the plate
- if the sample is polar it will move only a little bit up or not at all
--------------
2.) solvent
- the more polar the solvent the move distance it will travel up the plate
- the less polar the solvent the less distance it will travel up the plate
order the compounds based on increasing distance traveled on a TLC plate (aspirin, acetaminophen, caffeine)
FROM NO TRAVEL TO FURTHEST TRAVEL
1.) caffeine
2.) acetaminophen
3.) aspirin
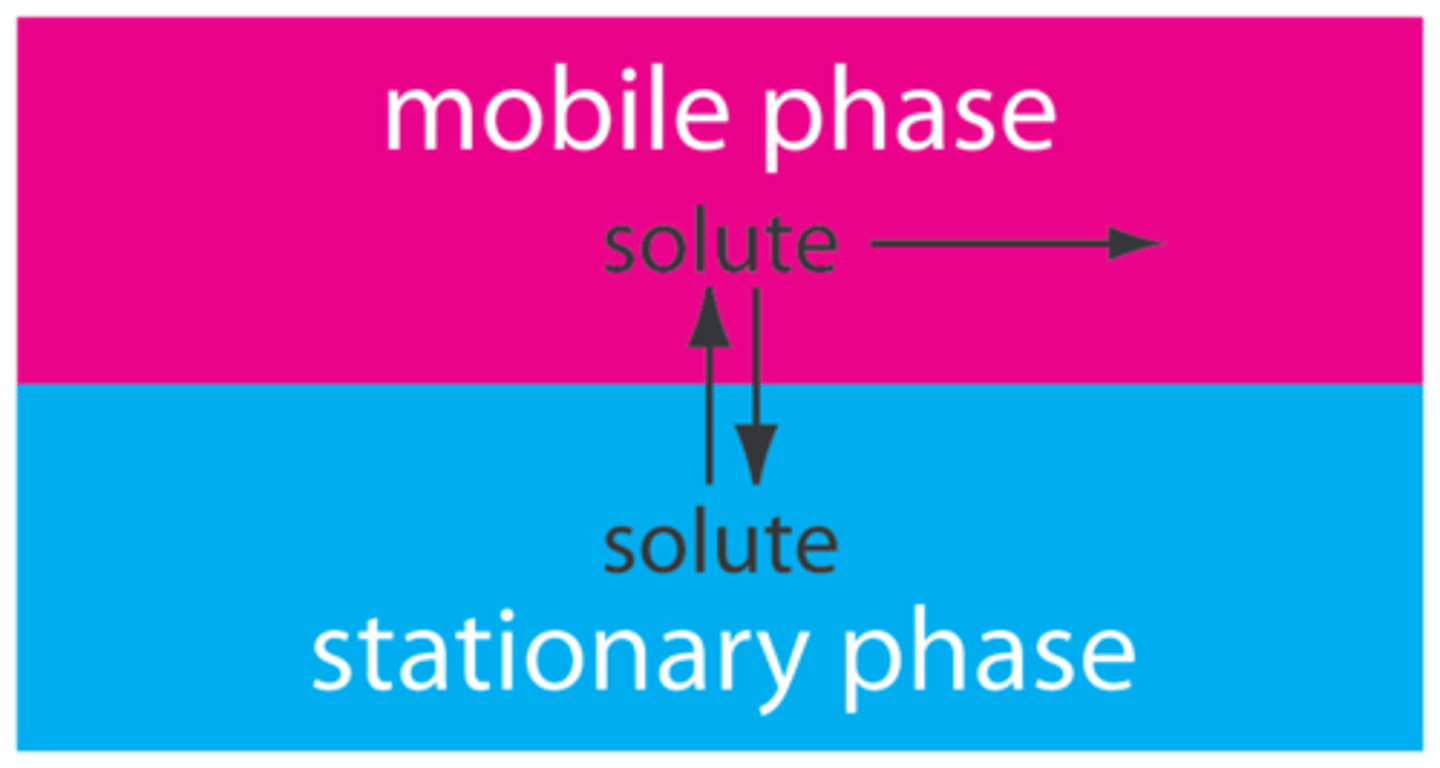
What is meant by the Stationary Phase in Chromatography ?
The component which will absorb the solutes/components in the mixture
It may be a solid or a liquid (held by solid support)
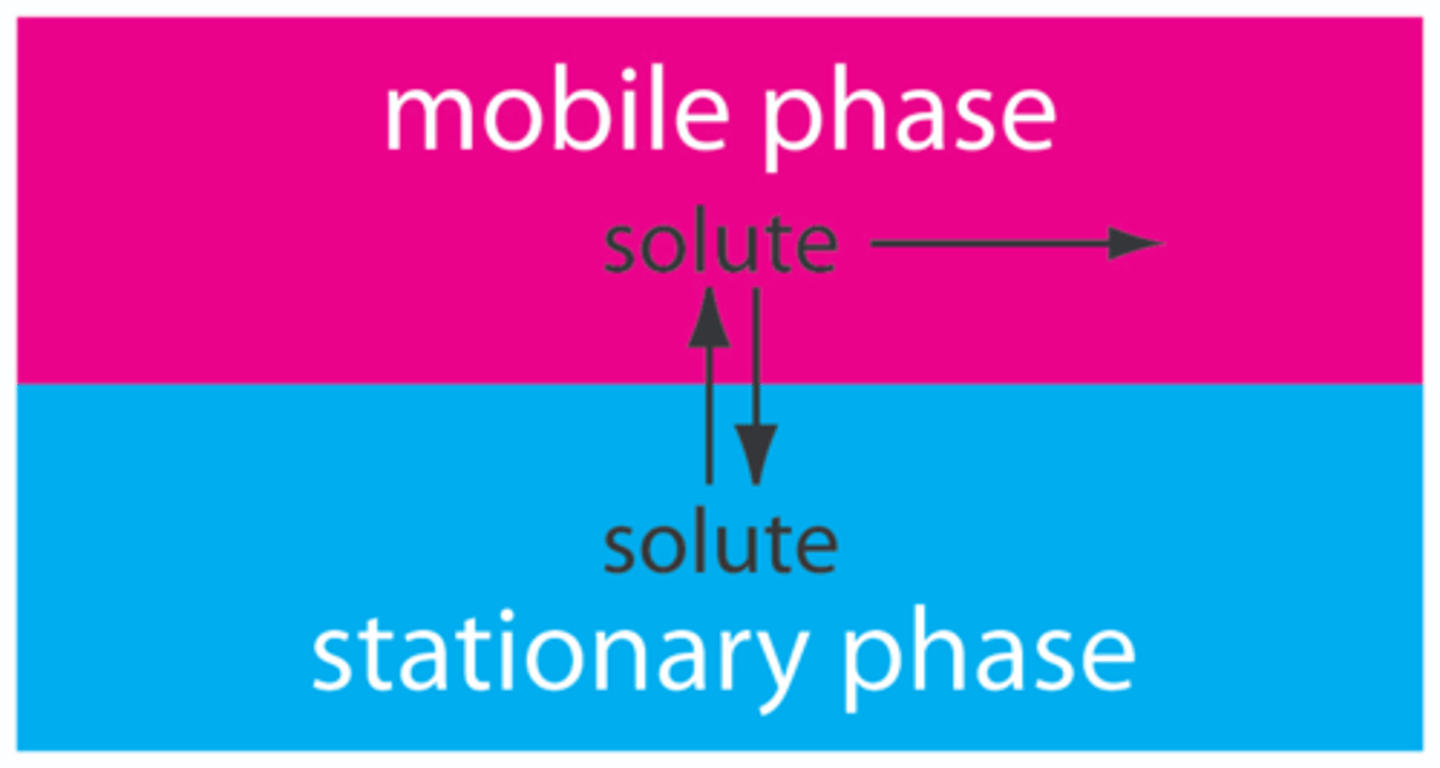
What is meant by the Mobile Phase in Chromatography?
The moving phase, which usually is the solvent which carries the soluble components of the mixture with it.
It moves through the stationary phase and may be a liquid or a gas
How is TLC Chromatography carried out ?
1. Draw a pencil line 1 cm from the bottom of the TLC plate and place 2 pencil crosses on the line
2. Place a drop of the purified solid on a watch glass and dissolve in a few drops of a solvent such as ethanol. Use a capillary tube to place a spot of the solvent on tp a pencil cross. Allow the spot to dry and repeat 3-4 times to produce a concentrated spot
3. Repeat this for the pure solid
4. Place solvent in a beaker/developing tank to a depth of 1 cm
5. Place the TLC plate in the beaker/developing tank and cover with a lid
6. Allow the solvent to run up the chromatography plate for about 30 minutes. When it has almost reached the tip of the plate, remove from the beaker and mark the line of the solvent front with a pencil
7. Place the plate in a fume cupboard until all of the solvents have evaporated and the plate is dry. If the spots are colourless then there is a different way to view them
- Place the plate under a UV lamp and mark the locations of the substances using a pencil
-spray with a developing agent which colours the spot
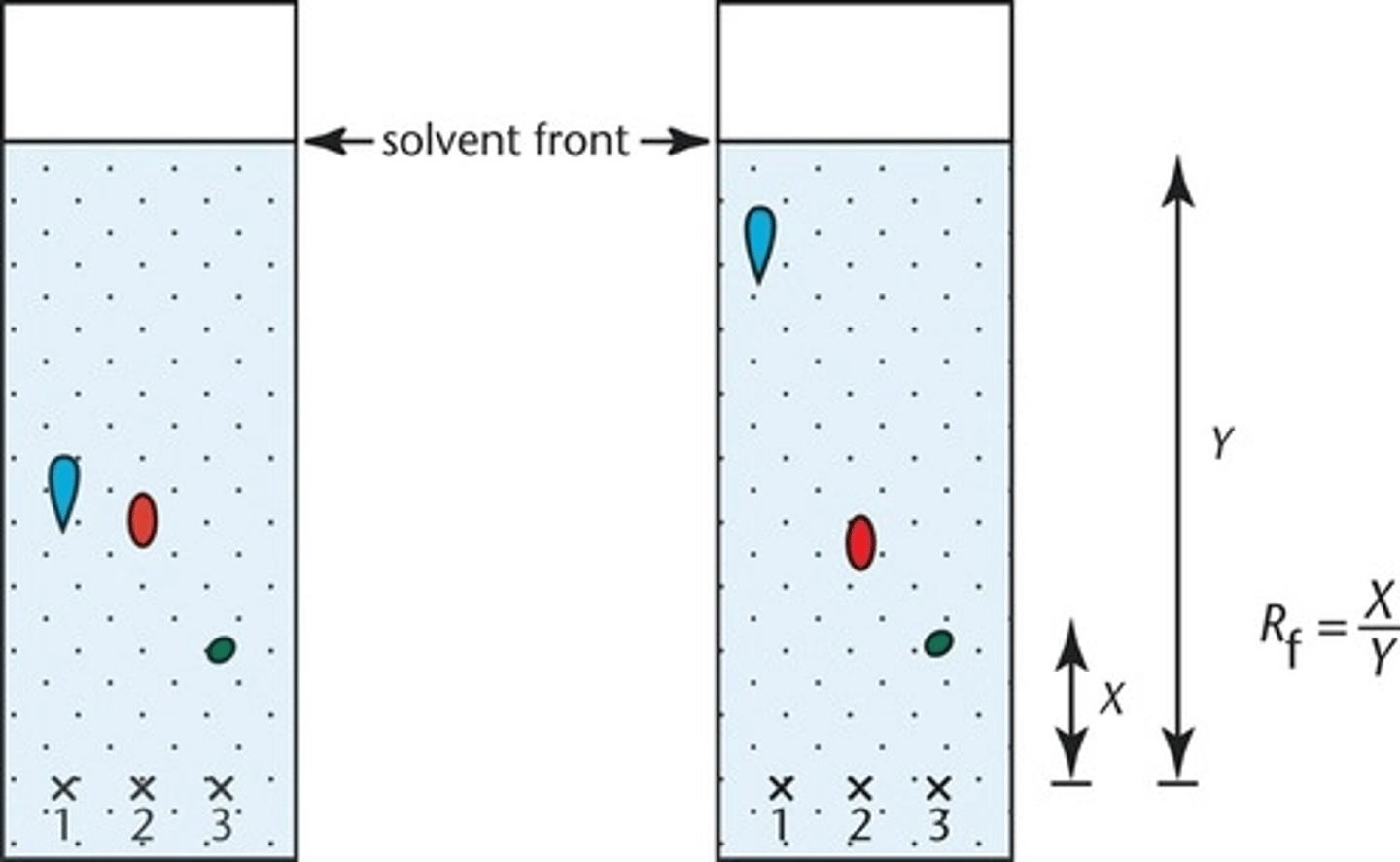
8. Measure the distance from the pencil line to the spot, and measure the distance from the pencil line to the solvent front, Calculate the Rf value of each substance visible on the plate and compare to the data book
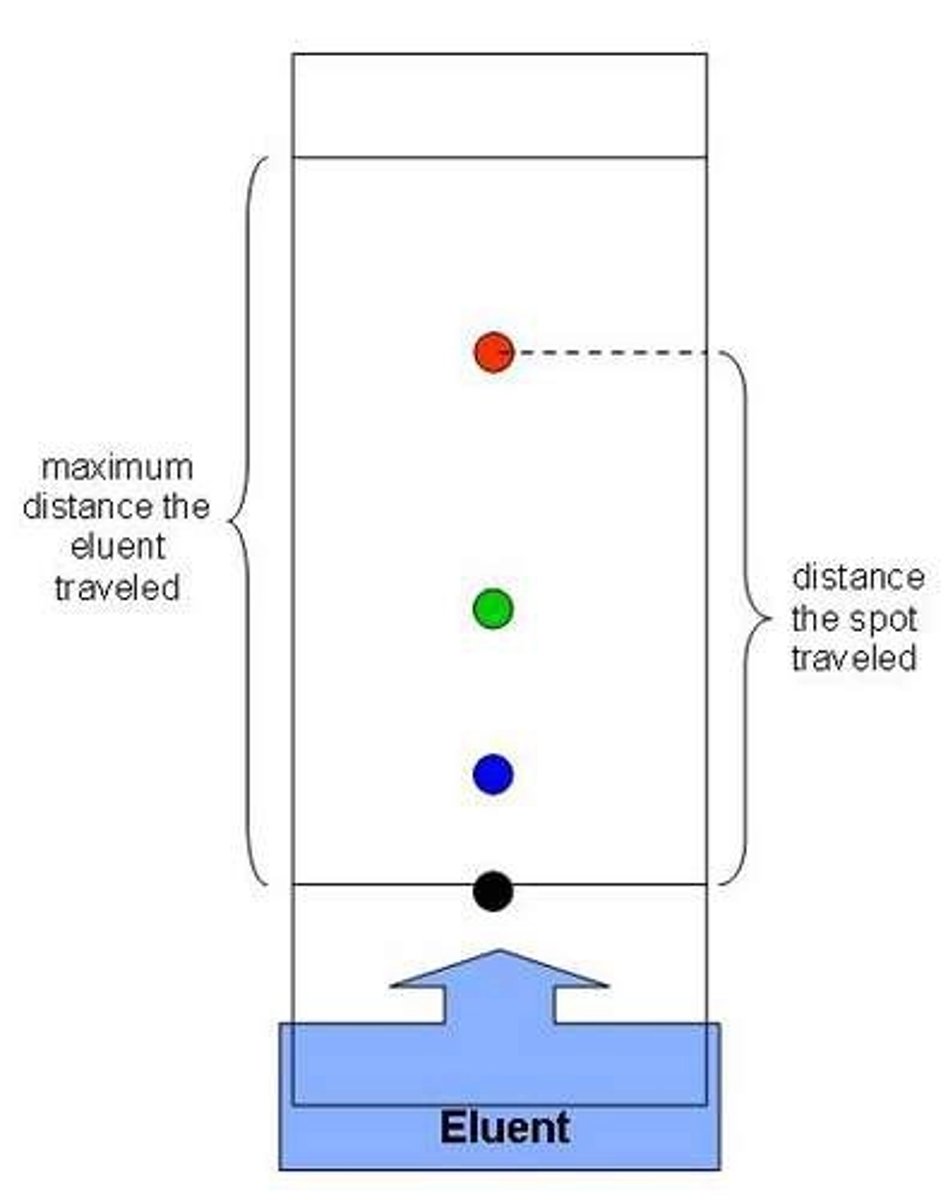
Ex: Why is it necessary to draw a pencil base line 1.5 cm from the bottom of the TLC plate ?
The pencil line is insoluble and will not move with the solvent or interfere with results
Ex: Explain why tiny drops of the sample are added to the TLC plate with a capillary tube?
too big a drop will cause different spots to merge
Ex: Explain why the developing tank/beaker is sealed with a lid when the TLC plate is placed in it ?
If the tank is open, the solvent may evaporate and not advance up the plate. Having a sealed environment also allows the solvent to saturate the atmosphere inside
