Hexose Monophosphate Shunt
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
introduction to hexose manophosphate
the hexose monophosphate shunt is a metabolic pathway parallel to glycolysis that serves two main functions:
production of NADPH: essential for reductive photosynthesis and maintaining redox balance.
synthesis of ribose-5-phosphate: precursor for nucleotide and nucleic acid synthesis
the hexose monophosphate pathway occurs in the cytoplasm of the cells and is especially active in tissues that require high levels of NADPH, such as:
-liver
-adipose tissue
-adrenal glands
-red blood cells
*the HMP pathway is active in tissues that require high levels of NADPH because the HMP pathway produces NADPH.

phases of hexose monophosphate shunt pathway
-the hexose monophosphate can be divided into two major phases:
-oxidative phases (irreversible): the NADPH is produced by oxidizing glucose-6-phosphate (the oxidative phase is irreversible due to the large negative ΔG (Highly Exergonic)
The oxidative step:
Transfers a hydride (H⁻) → Reduces NADP⁺ → NADPH, Oxidizes an alcohol group to a lactone
Oxidation + reduction of NADP⁺ releases significant free energy.
That makes: ΔG≪0
Large negative free energy change = reaction strongly favors products = irreversible under physiological conditions
-non-oxidative phases (reversible): this phase involves the rearrangement of ribulose-5-phosphate into various sugar needed for biosynthesis.

Phases of hexose monophosphate shunt (in biochemistry notebook with “HMS” on the page)
as mentioned previously, the hexose monophosphate shunt is a pathway that will occur parallel to glycolysis.
here is an overview of both pathways for your reference.

step by step of the oxidative phase (in biochemistry notebook with “HMS” on the page)
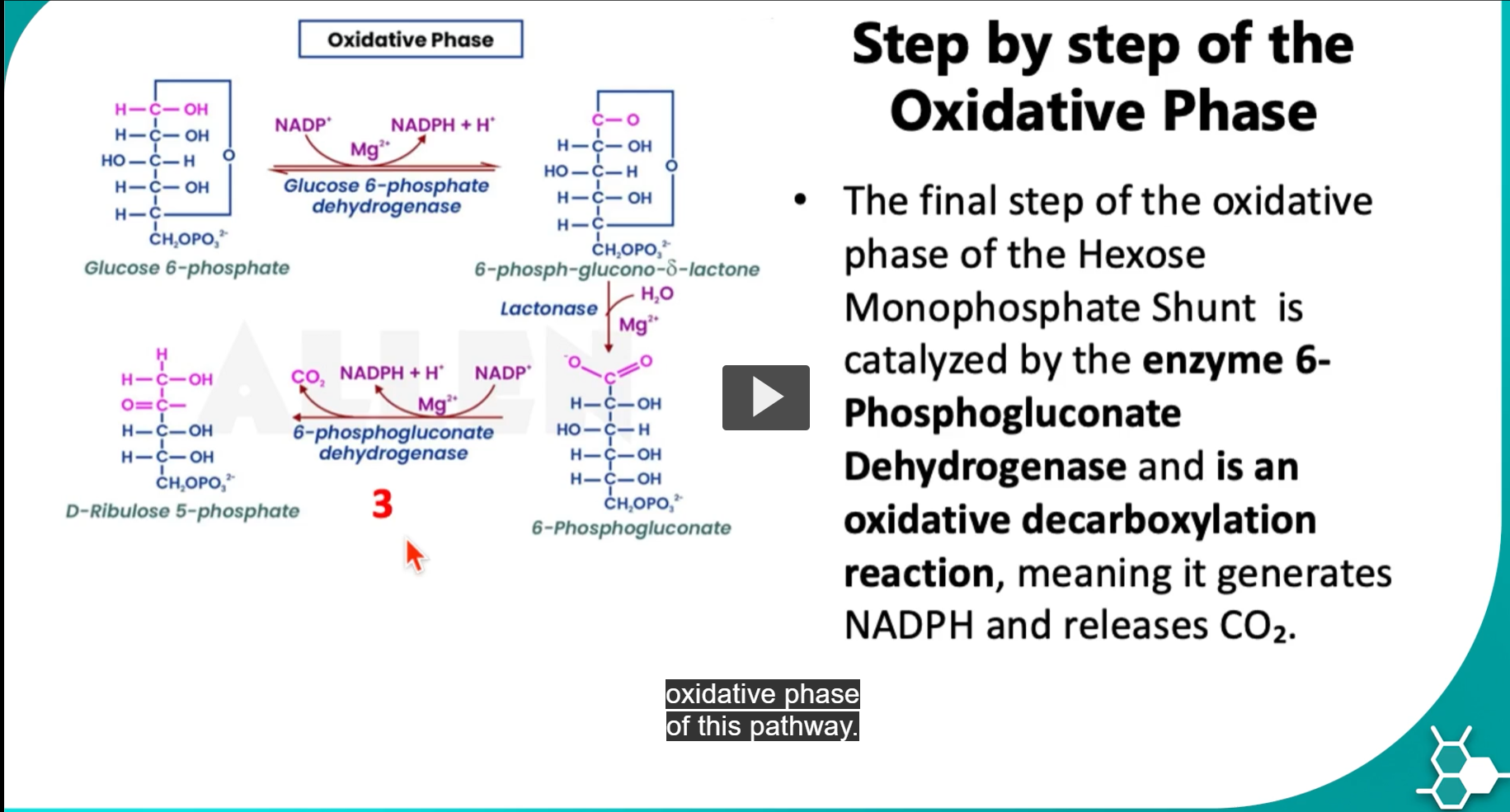
oxidative phase of HMP: breaks down glucose-6-phosphate (oxidizes glucose-6-phosphate) → NADPH + H.
the first step of the hexose monophosphate shunt is catalyzed by the enzyme Glucose-6-phosphate dehydrogenase (G6PD)
At C1: There is an aldehyde in the open-chain form,
1. G6PD removes a hydride (H⁻),
2. NADP⁺ accepts that hydride → becomes NADPH, there is another proton (hydride is H-, proton is H+), the other proton is released into the aqueous environment.
So chemically: Alcohol/aldehyde → more oxidized carbonyl derivative
This forms: 6-phosphoglucono-δ-lactone (a cyclic ester)
That immediately cyclizes into a lactone glucose-6-phosphate at carbon 1 forming a lactone
-the reaction reduces NADP→ NADPH, generating the first molecule of NADPH in the pathway.
-Mg2+ is a cofactor that is used to stabilize the negatively charged phosphate group on glucose-6-phosphate. the phosphate has a negative charge that repels nearby negative residues, makes precise positioning harder, need stabilization inside the enzyme active site. Mg2+ is there to neutralize the charge and reduce electrostatic repulsion.
-this is the RATE LIMITING STEP of the pathway and is highly regulated:
Why Is This the Rate-Limiting Step (The slowest step in a metabolic pathway that determines the overall speed (flux) of the pathway.)?
It is the first committed step (The first irreversible reaction that sends a metabolite down one pathway and prevents it from easily returning to competing pathways.)
It controls NADPH production
It has the largest negative ΔG in the pathway
It is tightly regulated by NADP⁺/NADPH ratio
Regulation of G6PD (Very High Yield) Primary Regulator: NADP⁺ Availability
High NADP⁺ | → Activates G6PD |
|---|---|
High NADPH | → Inhibits G6PD |
This makes perfect physiological sense:
If the cell needs reducing power → NADPH gets used
NADP⁺ rises
Pathway turns ON
This is elegant redox control.
what is NADPH used for?
NADH → ATP production (energy extraction)
NADPH → biosynthesis + protection (reduction reactions)
1⃣ Fatty Acid Synthesis (Very High Yield): In liver, adipose, lactating mammary gland: NADPH provides electrons to reduce carbon chains during fatty acid elongation, convert acetyl-CoA into palmitate.
Without NADPH: You cannot convert acetyl-CoA into palmitate.
This is one of the biggest consumers of NADPH.
Why This Step Is Irreversible
1. Strongly Exergonic (Large Negative ΔG)
Redox reactions involving NADP⁺ release substantial free energy.
2. NADPH Is Rapidly Consumed in:
Fatty acid synthesis
Cholesterol synthesis
Glutathione reduction (RBC survival)
Cytochrome P450 detox
Product removal pulls reaction forward.
3. It Commits G6P Away From Glycolysis
Once oxidized at C1, glucose cannot easily return to glycolysis as G6P.

Lactone comes from:
Latin: lac, lactis = “milk”
-one = suffix in organic chemistry indicating a ketone or carbonyl-containing compound
“milk ketone”
Why “Milk”? In the 1800s, chemists first discovered these compounds while studying lactic acid (from sour milk).
They noticed that certain hydroxy acids could: Internally react, Form a cyclic ester, Lose water in the process
Because this was discovered in lactic acid chemistry, the cyclic product was named a lactone.
step by step of the oxidative phase (step 2) (in biochemistry notebook with “HMS” on the page)
in step 2,
6-phosphoglucono-delta-lactone binds to the active site of lactonase. (there is a double bond to the non-lactone oxygen, but it’s not shown in the picture)
a water molecule is positioned near the lactone ring, making the oxygen in water perform a nucleophilic attack to the lactone ring carbonyl groups.
-The carbonyl carbon is electrophilic because the C=O bond is polarized.
lactonase then releases 6-phosphogluconate (, which is now an open chain form and ready for the next step.
-the Mg2+ is also used here to stabilize the phosphate group.
-lactonase: uses the nucleophilic oxygen in water to destroy the bond between the oxygen in the lactone ring and C5.
Gluconic acid (deprotonated) = Gluconate (“ate”: deprotonated due to physiological pH 7.4, so there is a negative charge on the oxygen, -COO-)

step by step of the oxidative phase (Final step of oxidative phase) (in biochemistry notebook with “HMS” on the page)
in the third step and final step,
6-phosphogluconate, binds to enzyme six-phosphogluconate dehydrogonase
once this binding happens, NADP extracts a hydride from C3, oxidizing 6-phosphogluconate at carbon 3 forming NADPH.
• A hydride (H⁻) is transferred from C3 to NADP⁺, That hydride contains 2 electrons + 1 proton = NADP⁺ → NADPH
So only one hydride is transferred.
what happens at C3?
• The hydrogen on C3 leaves as a hydride (H⁻)
• That hydride goes to NADP⁺ → NADPH
• The OH is converted into a carbonyl.
C2?
No hydrogen from C3 OH migrates to C2, instead, Because after decarboxylation, there is:
• An enol intermediate
• Protonation from solvent
• Tautomerization
That proton comes from water, not from C3.
So yes — C3 loses one hydride (2e⁻ + H⁺ equivalent).
It does not lose two separate hydrogens.
Only one hydride is transferred.
An extra proton appears in solution with NADPH to get NADPH + H.
Oxidation of 6-phosphogluconate at C3
Formation of ribulose-5-phosphate after CO₂ leaves
An additional H⁺ appears in solution.
this will form an unstable intermediate that will release CO2 (the CO2 that had the negative charge on top of 6-phosphogluconate) and form ribulose-5-phosphate. (because the carbon was lost)
-this D-ribulose-5-phosphate will now be used for the non-oxidative phase of this pathway.
(in biochemistry notebook with “HMS” on the page)
the produced ribulose-5-phosphate will undergo either an isomerization OR epimerization process, which will produce different products.
-the most important difference between these products is that both reactions rely on acid-base catalysis to form each product, but isomerization changes a ketone → aldehyde, while epimerization alters stoichiometry at C3.
-iso “equal” mer “part”: isomer: “equal parts”, Isomers are: molecules that have the same molecular formula but different arrangements of atoms.
epi: “upon”, mer: “part”, epimer: “different at one part”, Epimers are: stereoisomers that differ in configuration at only ONE chiral carbon.
epimers are stereoisomers.
Functions of the hexose monophosphate pathway
the hexose monophosphate pathway has several key functions in cellular metabolism. Due its metabolic flexibility, it helps the cell produce energy, nucleotides, antioxidants, and lipids depending on its needs.
In this table, we can see a detailed summary of all the key functions of this pathway and where it is going to be important in the body.


Functions of the hexose monophosphate pathway: NADH Production
1. NADPH Production What the pathway produces
The oxidative phase of the PPP generates NADPH.
Reaction example:
Glucose-6-phosphate+NADP+→NADPH
NADPH is used for:
• Redox balance – maintaining proper electron balance
• Biosynthesis – building molecules like fatty acids
• Detoxification – cytochrome P450 reactions in the liver
NADPH is essentially a source of reducing power (electrons).
Where this matters most
Liver
detoxification
lipid synthesis
Red blood cells
antioxidant defense
Adipose tissue
fatty acid synthesis


Functions of the hexose monophosphate pathway: Ribose-5-phosphate Synthesis
2. Ribose-5-Phosphate Synthesis What the pathway produces
The PPP generates:
Ribose-5-phosphate\textbf{Ribose-5-phosphate}Ribose-5-phosphate
This is a 5-carbon sugar used to build nucleotides.
Key role
Ribose-5-phosphate is needed for:
• DNA synthesis
• RNA synthesis
• ATP production
• NAD⁺ / FAD synthesis
It becomes PRPP (phosphoribosyl pyrophosphate), which starts nucleotide synthesis.
Where this is important
Cells that divide rapidly:
Bone marrow
Skin
Tumor cells
These tissues need large amounts of DNA and RNA.


Functions of the hexose monophosphate pathway: Sugar-rearrangement
3. Sugar Rearrangement What happens
The non-oxidative phase rearranges sugars.
Pentose sugars (5 carbons) are converted into glycolysis intermediates.
Example:
3 pentoses (5C)→2 fructose-6-P+1 glyceraldehyde-3-P3
Enzymes responsible:
• Transketolase (2-carbon transfer)
• Transaldolase (3-carbon transfer)
Key role
This allows the cell to:
• recycle pentose sugars
• reconnect the PPP with glycolysis
Where this is important
All cells
Because every cell uses glycolysis.


Functions of the hexose monophosphate pathway: Xylolose-5-phosphate
4. Xylulose-5-Phosphate What it is
Xylulose-5-phosphate is an intermediate produced in the non-oxidative PPP.
Key role
It acts as a metabolic signal molecule.
In the liver it activates:
ChREBP (carbohydrate response element binding protein)
This transcription factor increases expression of genes involved in:
• glycolysis
• fatty acid synthesis
So it helps the body convert excess glucose into fat.
Where this matters
• Liver
• Adipose tissue


Functions of the hexose monophosphate pathway: Oxidative Stress Defense
5. Oxidative Stress Defense Key role
The PPP protects cells from reactive oxygen species (ROS).
NADPH produced by the pathway keeps glutathione reduced.
Reaction:
GSSG+NADPH→2GSH
Reduced glutathione (GSH) neutralizes:
hydrogen peroxide
free radicals
oxidants
Where this is critical
Red blood cells (RBCs)
RBCs cannot make NADPH any other way because they lack mitochondria.
If PPP fails → oxidative damage → hemolysis.
Example:
G6PD deficiency


Functions of the hexose monophosphate pathway: Fatty Acid & Steroid Synthesis Key Role
6. Fatty Acid & Steroid Synthesis Key role
NADPH is required for reductive biosynthesis.
Examples:
• Fatty acid synthesis
• Cholesterol synthesis
• Steroid hormone synthesis
Each step requires reducing equivalents from NADPH.
Where this is important
• Liver
• Adipose tissue
• Adrenal glands
Adrenal glands use NADPH to synthesize:
cortisol
aldosterone
sex steroids

the hexose monophosphate pathway is tightly regulated to balance NADPH production, nucleotide synthesis, and sugar metabolism on the cellular needs.
The key regulatory mechanisms include
allosteric control: (This means molecules bind to an enzyme and change its activity, the main allosteric regulator is: NADPH, If NADPH binds to G6PD → enzyme slows.)
substrate availability (This means the reaction rate depends on how much substrate or cofactor is present, Here the important molecule is: NADP⁺, G6PD requires NADP⁺ to accept electrons. If NADP⁺ levels rise → the enzyme becomes more active.
hormonal regulation: Hormones change enzyme expression, Insulin
Insulin increases production of G6PD enzyme in certain tissues.
during the oxidative phase, we will see different ways that glucose-6-phosphate will be regulated, mainly, due to HIGH or LOW LEVELS of NADPH, but we will also see insulin and gluthathione, which can regulate the action of this enzyme.
3. High NADPH → Inhibits G6PD
Effect: High NADPH inhibits the enzyme.
Why: This is negative feedback inhibition.
If NADPH is already abundant, the cell does not need more.
4. Low NADPH / High NADP⁺ → Activates G6PD
Effect
Low NADPH or high NADP⁺ activates the enzyme.
5. Insulin → Activates G6PD
Effect
Insulin increases the expression of the G6PD enzyme.
Why
Insulin signals the fed state.
In the fed state:
glucose levels are high
excess glucose is converted to fat
Fatty acid synthesis requires large amounts of NADPH.
So insulin stimulates the PPP to provide that NADPH.
6. Glutathione Demand (Oxidative Stress) → Activates G6PD
Effect
When cells experience oxidative stress, G6PD activity increases.
Why: Cells use glutathione (GSH) to neutralize reactive oxygen species.
Reaction: 2GSH+H2O2→GSSG+2H2O
But once glutathione becomes oxidized (GSSG), it must be regenerated:
GSSG+NADPH→2GSH
So oxidative stress increases NADPH consumption.
That raises NADP⁺ levels, which activates G6PD.

Regulation of the hexose-monophosphate pathways: non-oxidative phase regulation
during the non-oxidative phase, we will also see different forms of regulation. mainly, this phase will be regulated by substrate availability and cellular demands for ribose-5-phosphate or glycolytic intermediates.

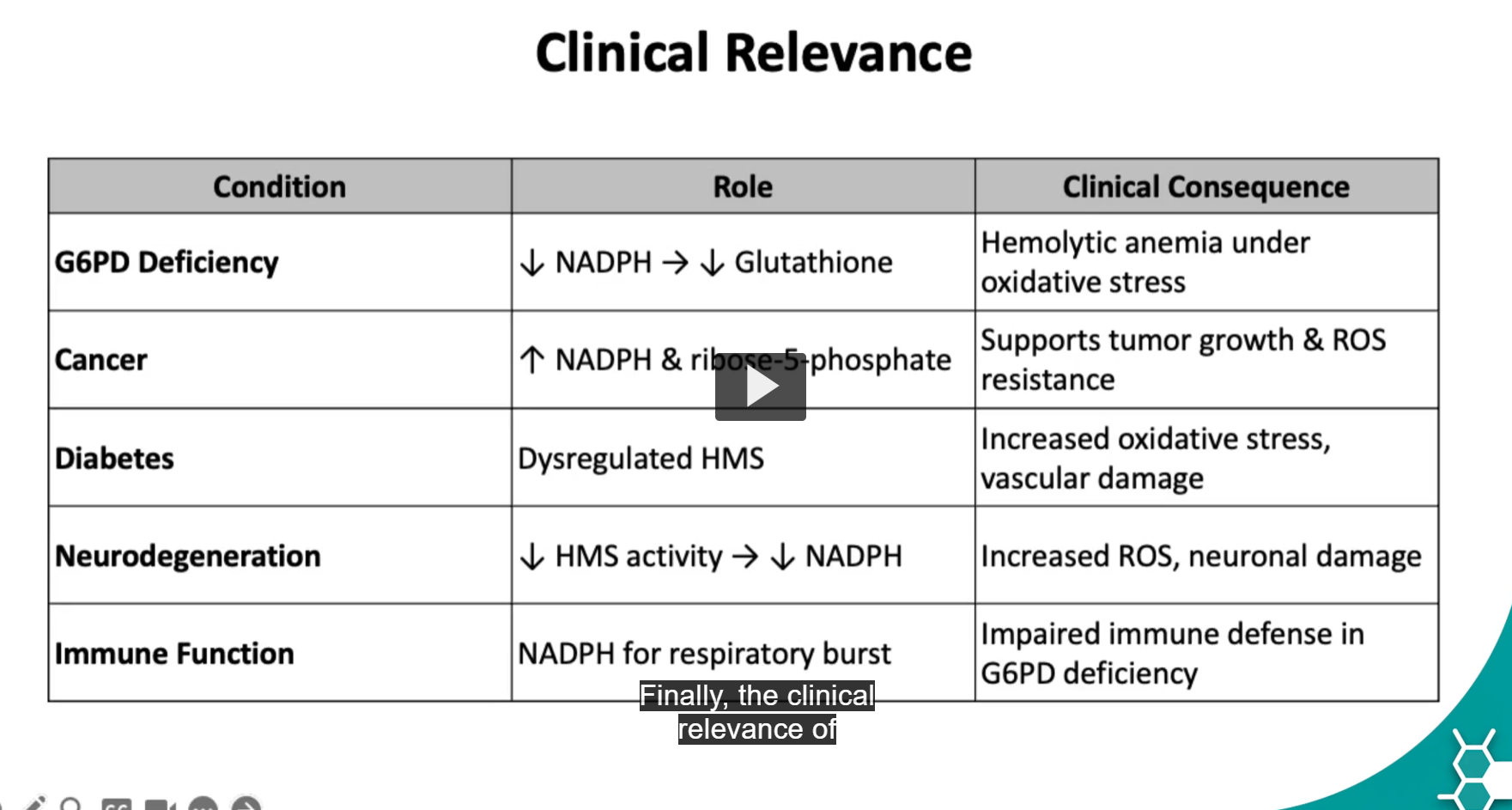
Clinical Relevance of HMS
Finally, the clinical relevance of the hexose monophosphate shunt is mostly observed in conditions related to
1. oxidative stress (a harmful imbalance where reactive oxygen molecules accumulate faster than the cell can neutralize them.)
2. metabolic disorders
3. cancers.
in glucose-6-phosphate dehydrogenase deficiency cases, we will see that this could lead to hemolytic anemia
in the case of some cancers, we will see that increases levels of NADPH and ribose-5-phosphate will support tumor growth and reactive oxygen species
in diabetes, deregulation of the hexose monophosphate shunt will lead to increased oxidative stress and vascular damage.
For neurodegenerative conditions, like Alzheimer’s, we will see that decrease of hexose monophosphate pathway activity will lead to decreased levels of NADPH, which ultimately leads to increase levels of reactive oxygen species and neural damage.
Finally, there are also conditions that can affect the immune system, mostly, when there is glucose-6-phosphate dehydrogenase deficiency, which leads to impair or decrease immune response.
1. G6PD Deficiency (hemolytic anemia)
What happens in the pathway
Glucose-6-phosphate dehydrogenase (G6PD) is the rate-limiting enzyme of the PPP.
If it is deficient: 1. decreased G6PD → 2. decreased NADPH → 3. decreased glutathione (GSH)
Why this matters: Glutathione protects cells from oxidative damage.
Reaction:
2GSH+H2O2→GSSG+2H2O
But glutathione must be regenerated:
GSSG+NADPH→2GSH
The –SH (sulfhydryl) group is what neutralizes oxidants by donating electrons to the Reactive Oxygen Species (ROS).
the ROS wants to steal electrons from cellular molecules, damaging: lipids, proteins, DNA. This damage is called oxidative stress. Instead, the sulfhydryl group on GSH gives the electrons so there is no damage.
Without NADPH:
glutathione cannot be regenerated
oxidative damage accumulates
Clinical consequence
Red blood cells are most affected because they:
1. lack mitochondria
2. rely almost entirely on PPP for NADPH
Result: Hemolytic anemia during oxidative stress (Hemolytic anemia = anemia caused by destruction of red blood cells.)
2. What Hemolytic Anemia Actually Is
Normally, red blood cells (RBCs) live about:
120 days
Then they are removed by the spleen.
In hemolytic anemia (hemo: blood, lytic: “destroying” Anemia = “lack of blood.”, RBCs are destroyed faster than the bone marrow can replace them.
So the body develops:
Too few functional RBCs (Red blood cells break inside blood vessels.) → This reduces oxygen delivery to tissues.
Intravascular hemolysis
Red blood cells break inside blood vessels.
This releases hemoglobin directly into the blood.
Common causes:
G6PD deficiency
mechanical damage (prosthetic heart valves)
severe infections
4. What Happens When RBCs Break Down
When RBCs rupture:
Hemoglobin is released
Hemoglobin breaks into heme + globin
Heme becomes bilirubin
So hemolysis leads to:
↑ bilirubin
This causes jaundice.
Triggers include:
infections
fava beans
sulfa drugs
antimalarials
2. Cancer
2. Cancer What happens in the pathway: Many cancers increase PPP activity.
This leads to: 1. increased NADPH 2. Ribose-5-phoshphate
Why cancer cells need this
Ribose-5-phosphate
→ used for DNA and RNA synthesis
Cancer cells divide rapidly and need lots of nucleotides.
NADPH
→ protects tumor cells from oxidative stress
→ supports fatty acid synthesis
Cancer cells produce many membranes for new cells.
Clinical consequence
PPP activation helps tumors:
1. grow rapidly
2. resist oxidative stress (ROS)
3. Diabetes
3. Diabetes
What happens: In diabetes, metabolism becomes dysregulated (insulin).
The PPP may not produce sufficient NADPH in some tissues.
Why this matters
Less NADPH means:
less glutathione
more reactive oxygen species
ROS damage:
endothelial cells
blood vessels
tissues
Clinical consequence
This contributes to diabetic complications:
retinopathy
nephropathy
neuropathy
vascular disease
4. Neurodegeneration
Examples:
Alzheimer’s disease (The protein amyloid-β accumulates in neurons and can enter mitochondria, inhibit mitochondrial enzymes, decrease ATP production and increase ROS)
Parkinson’s disease (In Parkinson’s disease, mitochondrial damage is central to the disease.
The neurons most affected are dopamine neurons in the substantia nigra.
Many Parkinson’s patients show problems in Complex I of the electron transport chain.
Result:
↓ ATP
↑ ROS
Dopaminergic neurons are especially sensitive to oxidative stress.
What happens: PPP activity may decrease in neurons.
Result:
decreased NADPH
decreased antioxidant capacity
decreased ROS
Why neurons are vulnerable
Neurons:
consume large amounts of oxygen
generate many reactive oxygen species
Without enough NADPH to maintain glutathione:
oxidative stress → mitochondrial damage → neuronal death.
3. Connection to Alzheimer’s Disease
In Alzheimer’s disease, mitochondrial dysfunction occurs early.