anionic and cationic polymerisation
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
what are the requirements of monomers for anionic polymerisation?4
EWG to stabilise anion by resonance
no acidic protons
no electrophilic groups
anion formed on monomer must react with another monomer
what are the two types of initiators in anionic polymerisation?
organometallic
radical anions from electron transfer
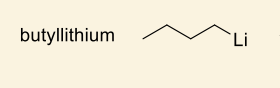
show how this makes anionic monomer
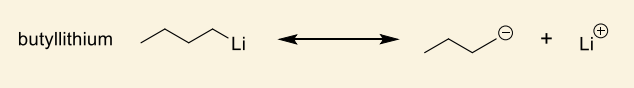

show how this makes anionic monomer

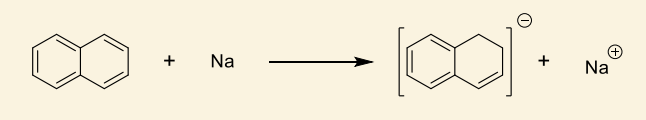
show how this makes radical anion?
what must be favourable for successful initiation?
free energy of initiation step must be favourable
how can the first addition be energetically favourable?
match the monomer with the right strength of initiator
what can you do if the propagating anion is not strongly stabilised?
powerful nucleophile needed as initiator
what solvents do you need for anionic polymerisation? give examples
solvents must be aprotic
e.g. saturated hydrocarbons - aromatic (benzene) or aliphatic (hexane)
why must solvents be dilute for anionic polymerisation?
how else must solvents be selected?
high conc polymers are often insoluble
to minimise chain transfer reactions
what temps are used for anionic polymerisation? why?
instability
low temps (-100 to +50C)
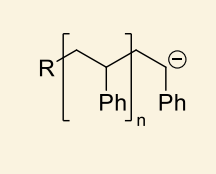
what happens if a protic solvent is used for anionic polymerisation?
show example with ammonia
can react with the active site
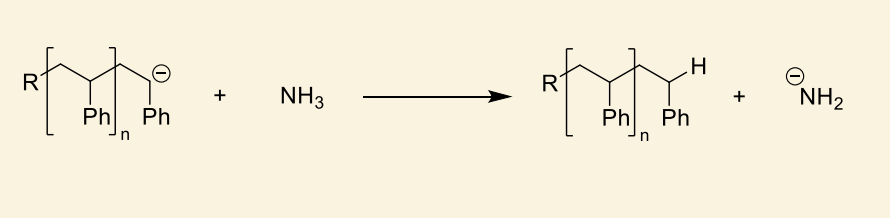
what is the significance of NH2-?
can initiate polymerisation of new chain
what is living polymerisation?
where a chain transfer or termination are absent
what are the 4 consequences of living polymerisation?
active, functionalisation, initation, synthesis?
chains are active indefinitely
chain end functionalisation can be carried out quantitatively
all chains initiated at similar time
synthesis highly controlled (molecular weight and distribution)
what is the conversion like for living polymerisation?
100%
what is the Mn proportional to in living polymerisation?
what is Mn controlled by?
directly prop to conversion
controlled by stoichiometry
what is the criteria for initiators of living polymerisation?
each initiator makes one chain
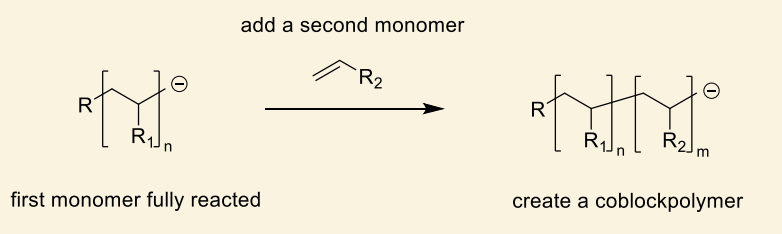
how are coblockpolymers made?
there is nothing for the anion to react with so there is no termination
after first reacted, second monomer added etc
what is glass transition temperature (Tg)?
temperature at which amorphous materials transform form hard and brittle state into molten or rubber like state
what happens to chains at the glass transition temp?
molecular chains slide past each other when force applied
how do chemical groups affect Tg?
stiff groups (e.g. benzene) interfere with flowing process and increase Tg
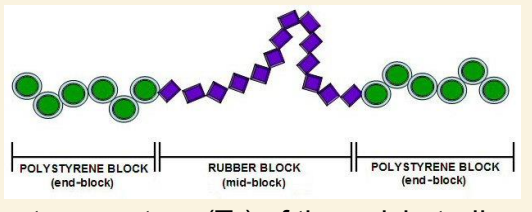
if the glass transition temp of polybutadiene block is -90, and of polystyrene is +100, what happens between these temps?
what happens above Tg of styrene blocks? what does this mean about the material’s properties?
it behaves as a physically crosslinked elastomer
physical cross links change from rigid glassy regions to flowable melt regions
flows and therefore can be cast, folded or extruded into desired form
what can be incorporated into a polymer to decrease Tg?
plasticisers
in cationic polymerisation, where do electrons come from?
what must monomers be like?
electrons come from monomer not initiator
monomers must be electron rich
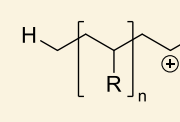
show cationic polymerisation of this polymer
is cationic or anionic polymerisation faster?
cationic rates are faster
what temperatures is cationic polymerisation done at?
typically -40 to -80
to slow rxn down
are cations reactive or stable? what does this mean about polymerisation?
very reactive and hard to control and stabilise
more chain transfers and side reactions
how can nucleophiles affect cationic polymerisation?
react with cationic active centre and shut down polymerisation
what can be said about cationic polymerisation and dispersity? block copolymers?
it is difficult to make polymers with low dispersity
tend to be different chain lengths
difficult to make block copolymers
which monomers can undergo cationic polymerisation?
stabilised by substitution (+I) or resonance (+M EDG)
what are the two types of initiators for cationic polymerisation?
protic acids
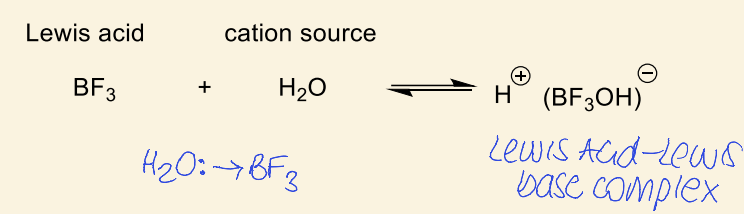
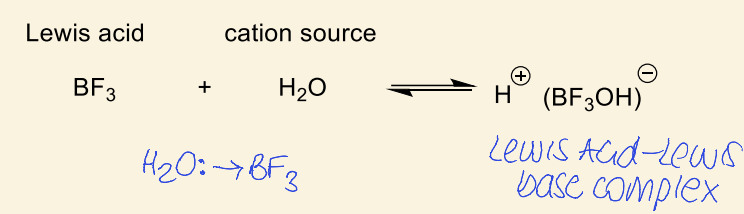
Lewis acids and cation source
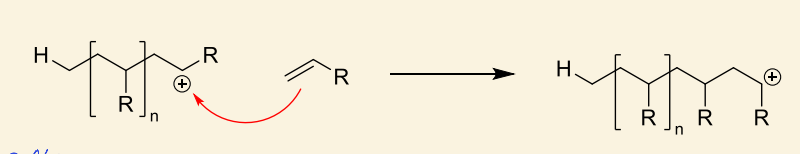
show how HA acts as initiator for alkene monomer
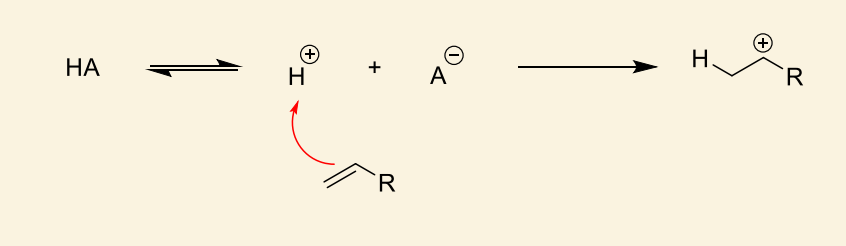
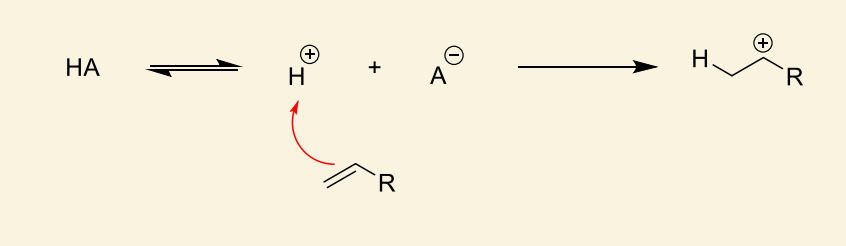
which side of the alkene reacts?
reacts to form most stable cation (i.e. more substituted carbocation)
show example of lewis acid and cation source and how it makes a complex
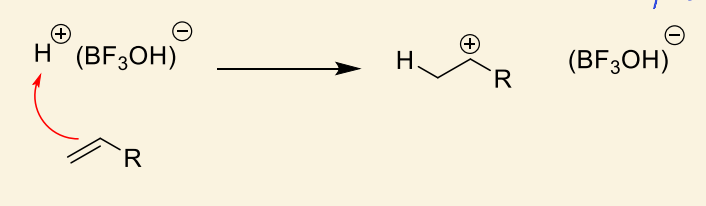
how does this complex react with alkene monomer?
what are 3 suitable cation sources?
water
alcohols
ester
what are 3 suitable lewis acids?
SnCl4
BF3
TiCl4
what can cationic polymers be terminated by?
counter ions
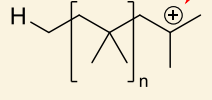
what is unimolecular rearrangement?
show with anion
anion acts as nucleophile
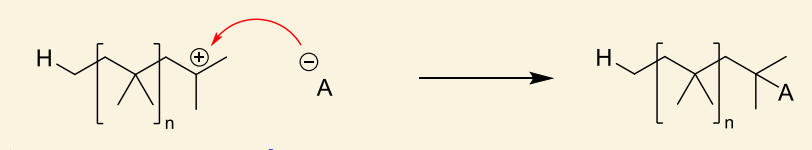
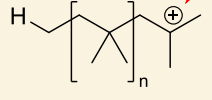
what is hydrogen abstraction?
show with anion
anion acting as base
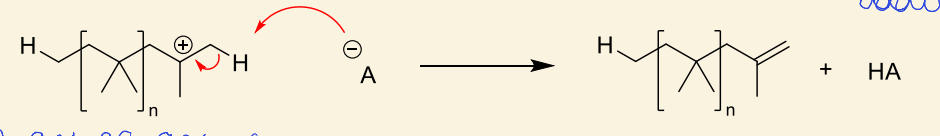
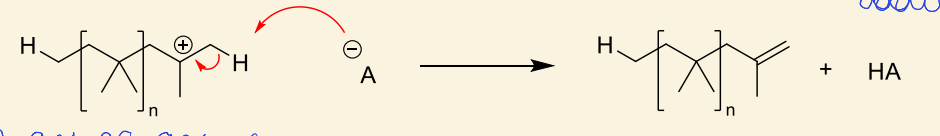
why can this also be considered chain transfer?
HA is regenerated for initiation again
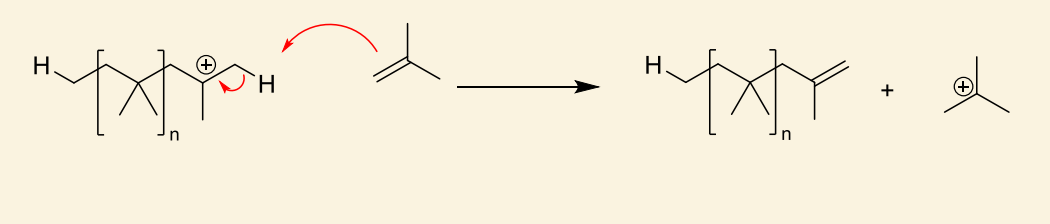
show example of chain transfer by hydrogen abstraction using alkene
cationic monomer regenerated
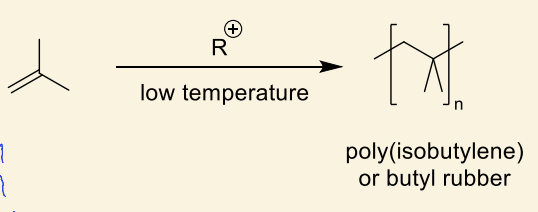
what are the properties of polyisobutylene?
high impermeability, chemical stability, flexibility and tackiness
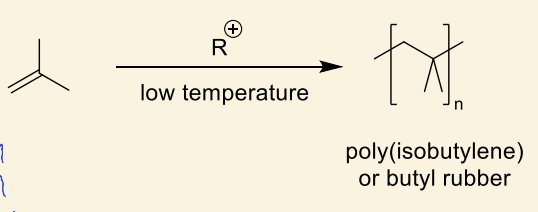
what are the 3 applications of polyisobutylene?
rubber (gloves/tires)
fuel additives
chewing gum
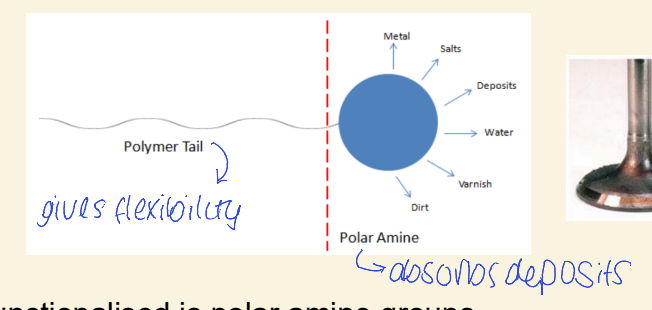
why is polyisobutylene used as a fuel additive?
how is it functionalised?
forms hydrocarbon soluble backbone
polymer is functionalised with polar amine groups
keeps engines clean as absorbs carbonaceous deposits that build up on metal surface
how is gum made?3
gum base melted (polymer becomes flexible and malleable)
sweeteners and flavours added. mixing
scored and moved to temp controlled environment to cool
what is injection moulding?3
molten material/melted polymer is injected into mould
takes form of desired plastic item
solidifies on cooling to give plastic item