Orgo (Everything)
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
Reduces ketones and aldehydes, turning carbonyl into a hydroxyl and adding the chain to that carbon. Also does 1,2 addition to an enone (makes that carbonyl an alcohol and adds the chain there)
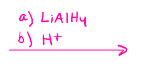
Reduction of ketones/aldehydes, turning that carbonyl into a hydroxyl
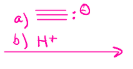
Reduces ketones/aldehydes. Adds onto that most substituted carbon
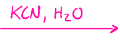
Adds to the carbon with the carbonyl and turns the carbonyl into a hydroxyl
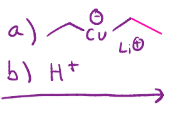
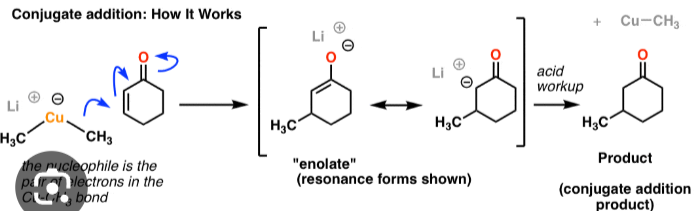
1,4 addition to an enone, negatively charged cu reaches out to double bond, as those electrons move up towards the carbonyl, and the co double bond becomes a single bond, then acid workup (more stable when o is double bonded)
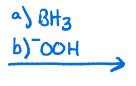
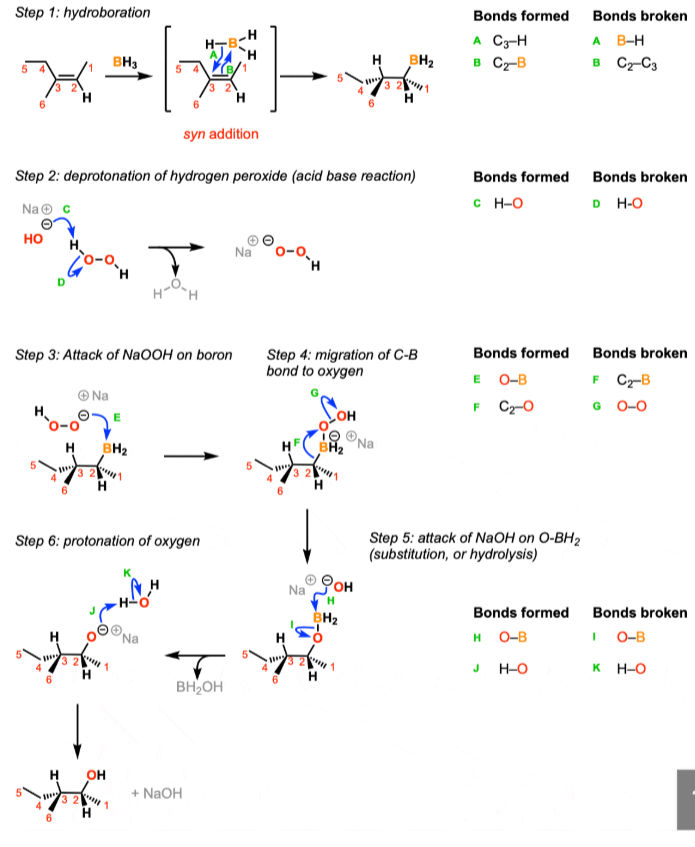
Hydroboration of alkenes, adds alcohol on least substituted carbon
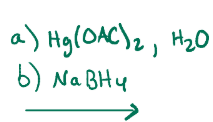
Oxymercuration of alkenes, adds an alcohol on most substituted carbon, does not do carbocation rearrangement, unlike H+, H2O
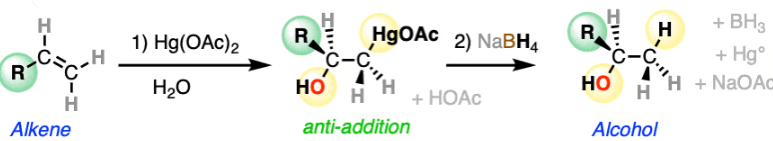

Adds alcohols (syn addition) to both sides of the alkene, will be racemic if chiral

Adds alcohols (syn addition) to both sides of the alkene, will be racemic if chiral
mCPBA
Epoxidation of alkenes (racemic if chiral, because it can come from either side)

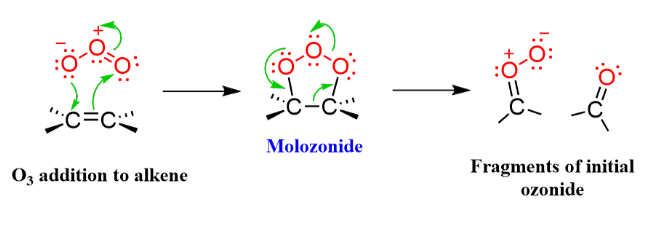
Splits alkene in half and makes an aldehyde in its place, puts a double bonded oxygen at both ends
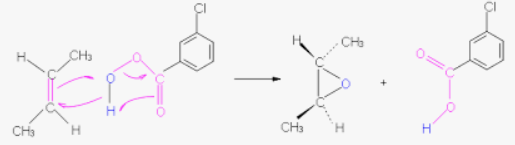
CH2N2
Takes an alkene and makes it into a “carbon epoxide” (racemic if chiral)
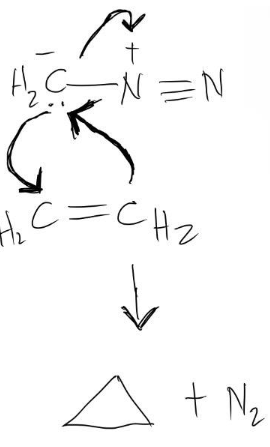

Alkyne into a ketone
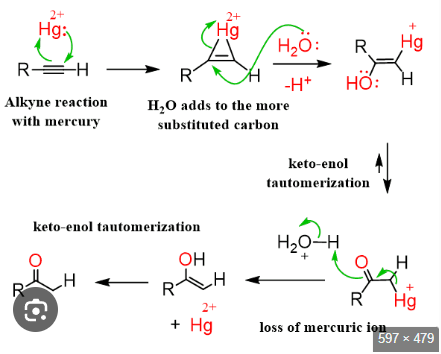
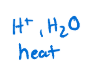
Alkyne to ketone
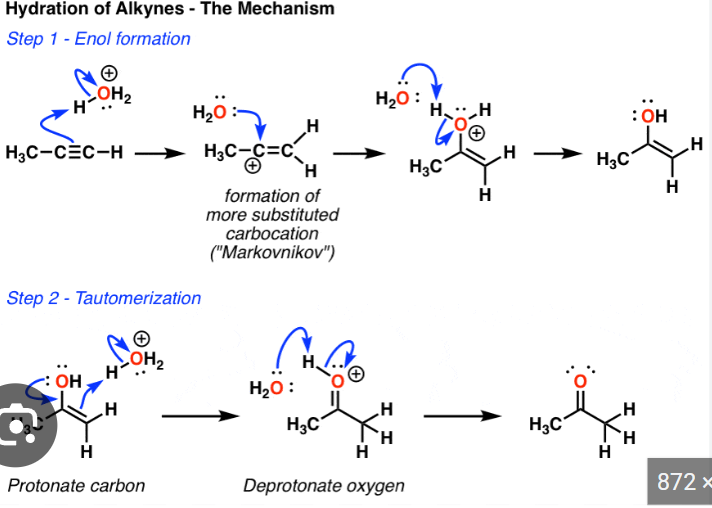
Alkyne to aldehyde
H2, Lindlars
Alkyne to Z alkene, will nor react with alkenes
Li, NH3(I)
Alkyne to E alkene, will not react with alkenes
H2, Pd
Alkynes and Alkenes to Alkanes
Br2, Light
Bromine on most substituted carbon
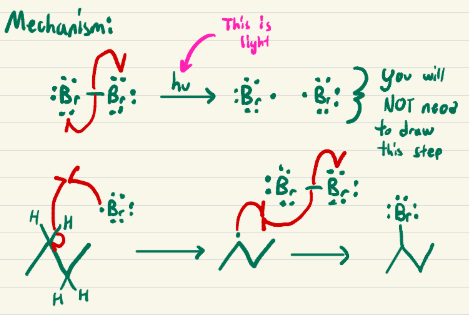

Bromine on least substituted carbon of a double bond
Cl2, light
Puts Cl on one carbon, but there are multiple options, not regiospecific, so there is a mixture
LDA
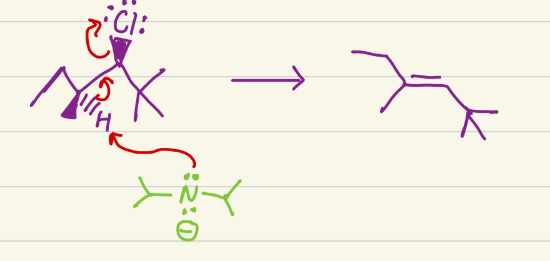
E2. Needs a good leaving group, and makes alkane into alkene. If stereochem of LG and H are the same, its a Z alkene, if they are different, its an E alkene.
HCl (or any other mineral acid) and heat
Does SN1 and can do it multiple times. Replaces alcohol with the mineral.
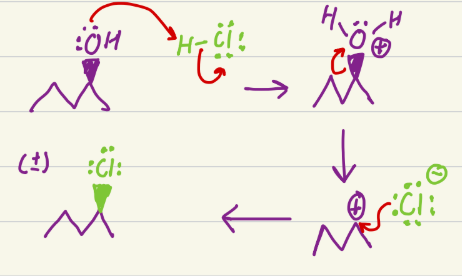
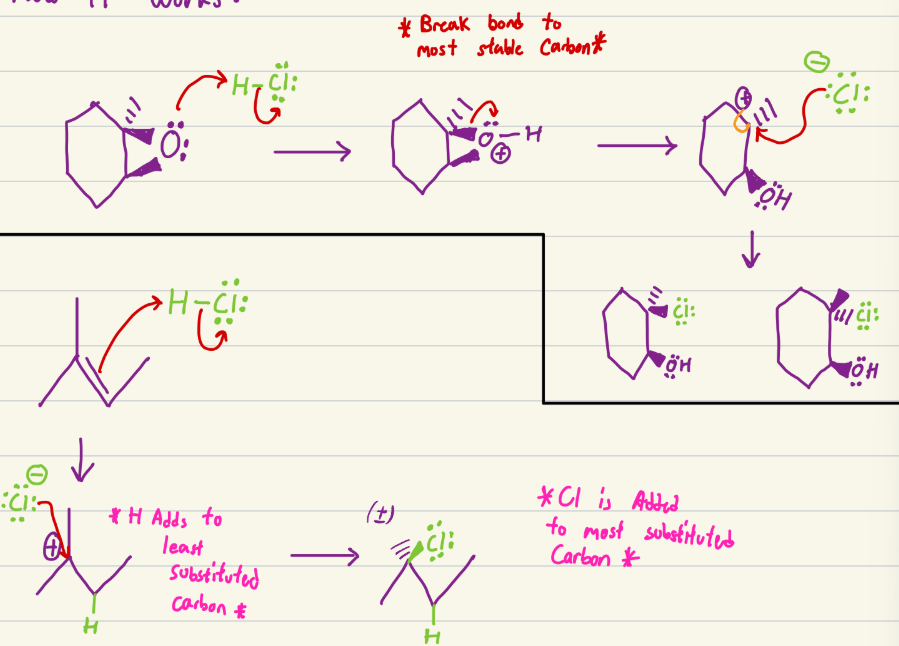
HCl
Addition and epoxide opening. For epoxide opening, OH ends on least substituted, and mineral ends on most substituted. OH stereochem is not racemic, so two products for epoxide opening. For addition, H is added to least substituted, and the mineral to the most substituted.
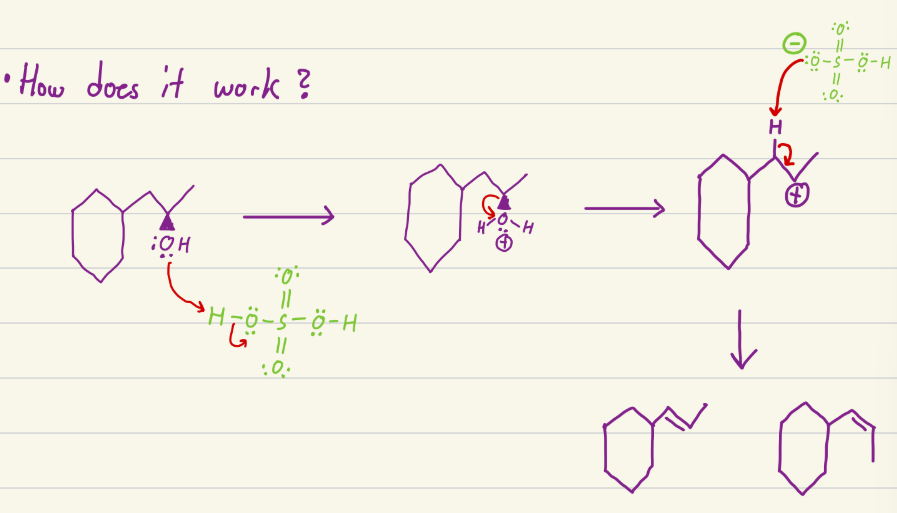
conc. H2SO4 and Heat
Does an elimination reaction with OH and makes both E and Z products, so alcohol to alkane. OH becomes a double bond between the carbon with the OH and the most stable carbon.
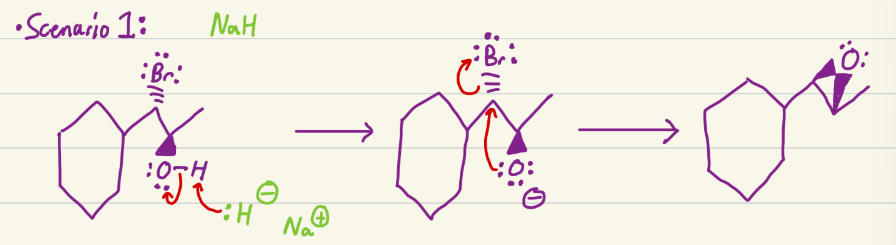
NaH
Needs an electrophile with an OH and good LG. H leaves hydroxyl so that it is now negative, and leaving group goes, so that carbon is positive, oxygen bonds to that carbocation to make an epoxide.
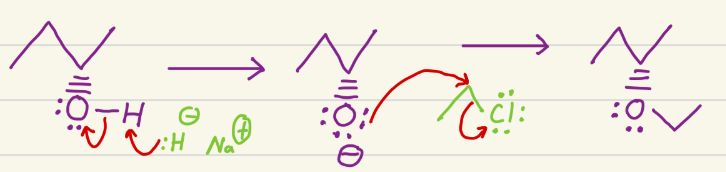
Electrophile has OH, and this adds a carbon chain to the oxygen.
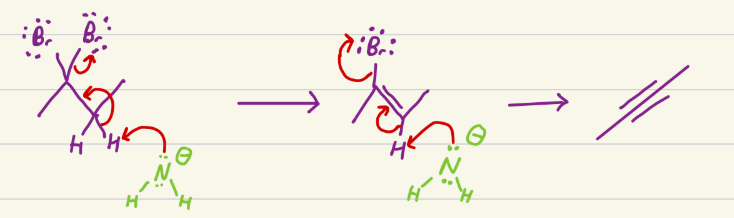
Excess NaNH2, heat
Elimination reaction. Na leaves, so the now negative N reaches out to one of the hydrogens, creatine an alkene, as a leaving group leaves. Then repeats as other h leaves, and other LG leaves, creating an alkyne.
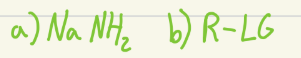
Adds a carbon chain to terminal alkyne
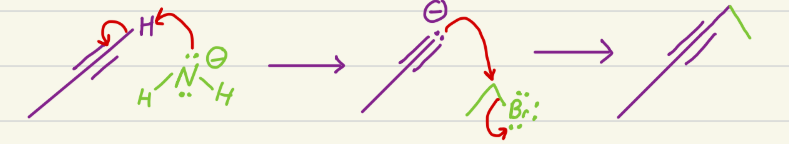
Br2 (others work, like Cl2, I2)
Adds Br (or other halogen) to both carbons on an alkene, with opposite stereochem
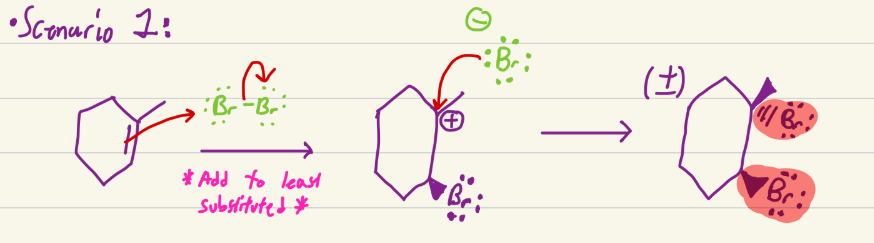
Br2, H2O
Adds Br on least substituted and hydroxyl to most substituted, with opposite stereochem
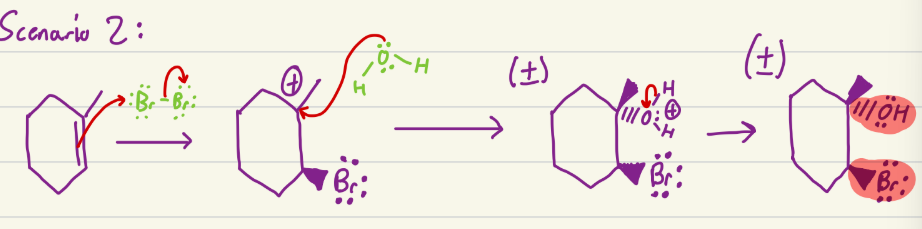

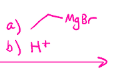
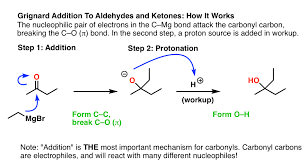
Grignards
Can do Sn2 and E2 based on conditions. Add carbon chain to molecule, and can do epoxide opening (a b steps)
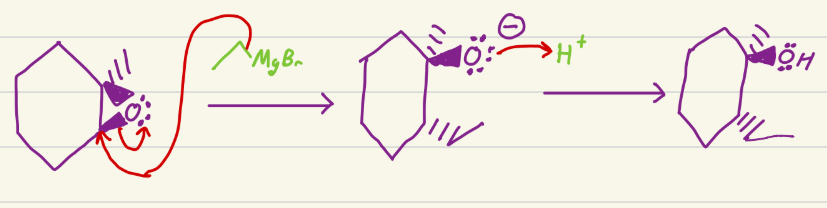
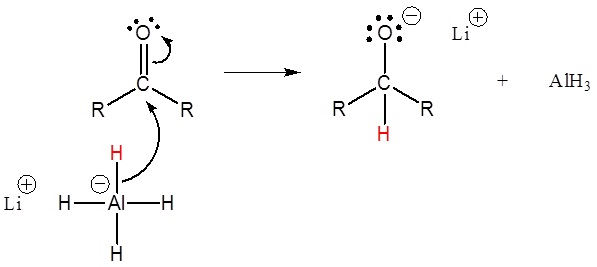
LiAlH4
Aldehyde to primary alcohol
Ketone to secondary alcohol
Carboxylic acid to primary alcohol
Esters to primary alcohol
Amides to amines
Nitrile to amines
Epoxides to alcohol
Alkyl halides to alkanes

Turns aldehydes and ketones into alcohols (can be racemic for ketone reaction)
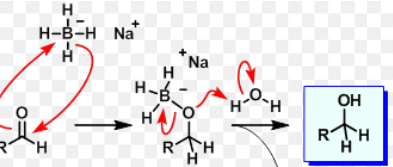
Mg and Li
Make grignard with carbon chains that have leaving groups Br, Cl, or I
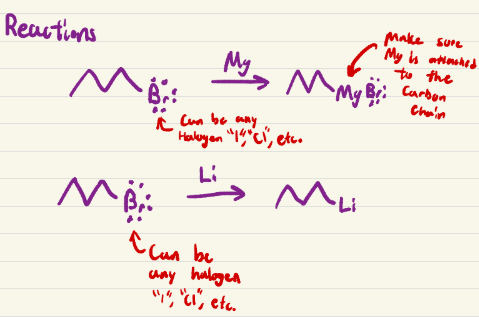

SN2, adds onto that carbon with a carbonyl, turning it into a hydroxyl, cyanohydrin formation
Ortho para directors
Usually electron donating groups (lone pairs adjacent to a pi system), and halogens. Ortho para will always be together, because of resonance
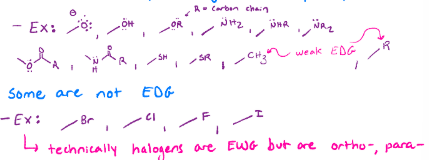
Meta directors
Usually electron withdrawing groups (pi orbital adjacent to pi system)
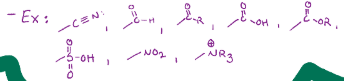

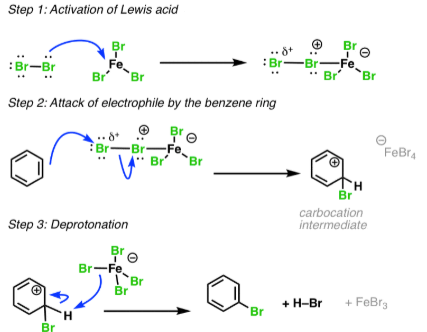
Bromination, adding on a bromine

Chlorination, adding a Cl on
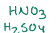
Nitration, adding on an NO2

Sulfonation, adds SO3H, but only meta or para

Desulfonation, removes an SO3H group
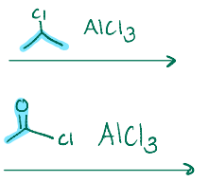
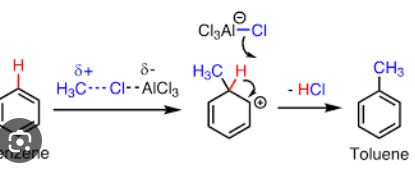
Cl leaves, and that carbon becomes charged and then attaches
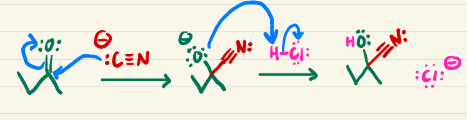
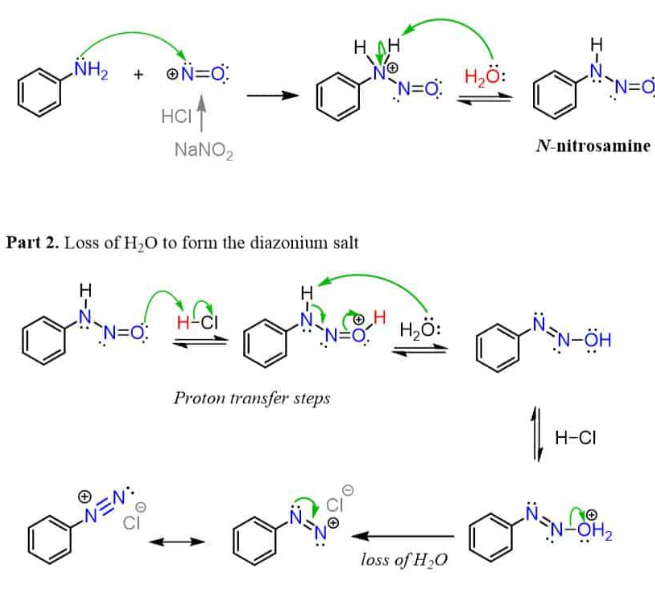
HNO2 HCl (diazonium salt)
Converts NH2 into N triple bonded to N
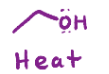
(diazonium salt)
Converts the n triple bonded to another n into an o with a chain
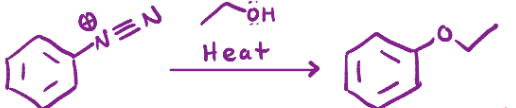
H2O, heat (diazonium salt)
Converts the n triple bonded to an n into an alcohol

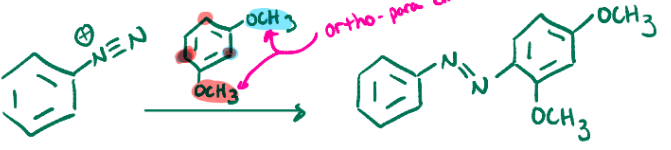
Diazonium salt example
OCH3 groups are ortho para directors, can’t add in between them because of sterics
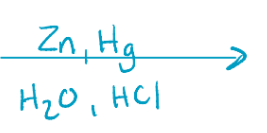
Clemmenson reduction, removes ketones/aldehydes
H2, Pt or Zn, HCl
NItro reduction, turning NO2 group into NH2
PCC
turns primary alcohols into aldehydes, and secondary into ketones, but doesn’t react with tertiary
H2CrO4
Turns primary alcohols into carboxylic acids, secondaries into ketones, and doesn’t react with tertiary
H2O, H+
Hydrate formation, turns that carbonyl into a diol
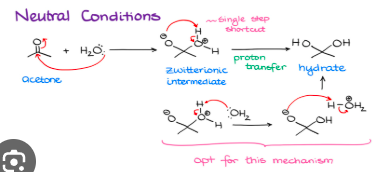
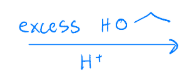
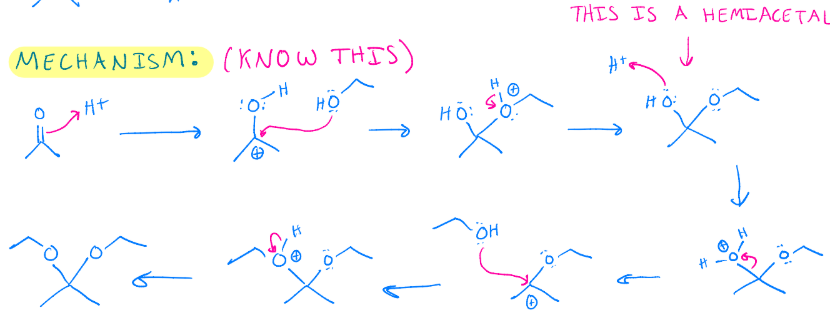
Acetal formation
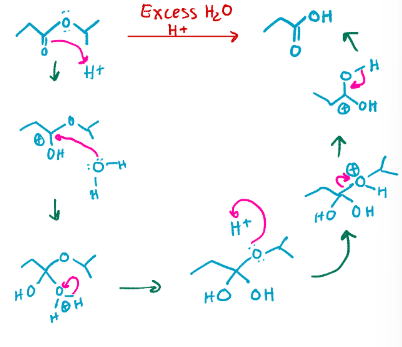
excess H2O, H+
Acetal hydrolysis
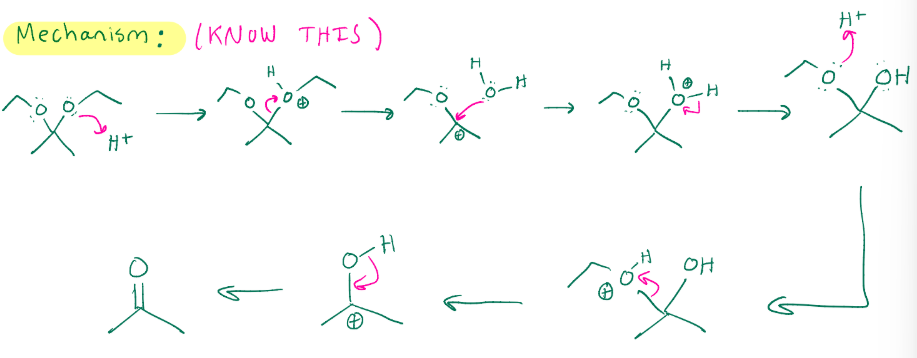
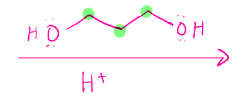
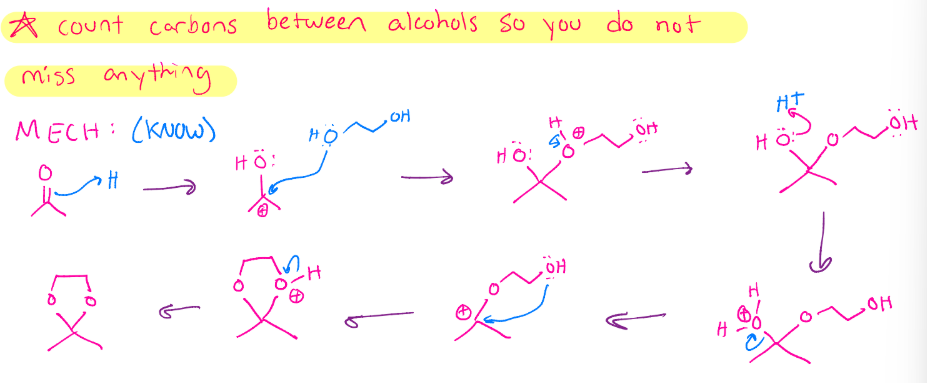
Cyclic acetal formation
H+ H2O
Cyclic acetal hydrolysis, don’t need excess H2O like in regular acetal hydrolysis because its just the one piece, also does imine hydrolysis
H+
Intramolecular hemiacetal. Need an aldehyde an alcohol
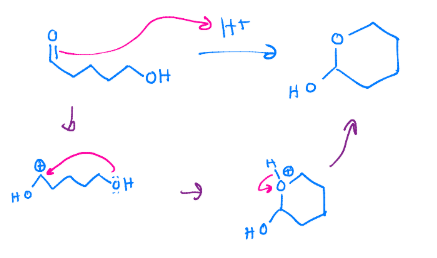
Hemiacetal formation
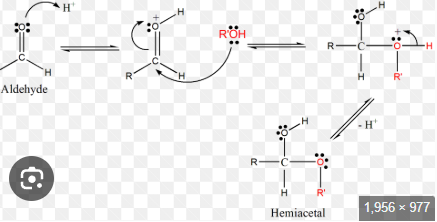
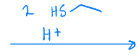
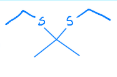
Creates a thioacetal from a ketone
HgCl2, H2O or Hg(ClO4)2, H2O
Thioacetal Hydrolysis
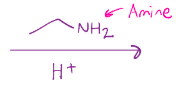
Imine formation
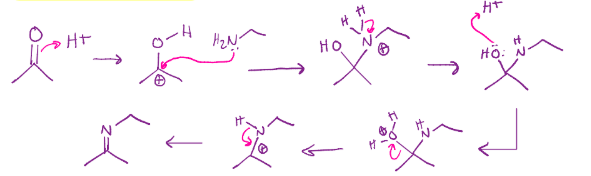
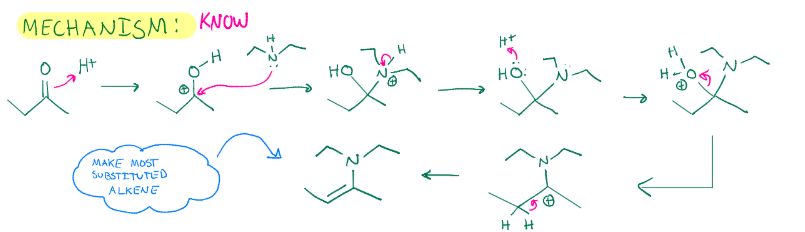
Enamine formation
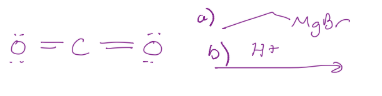
Grignard reaction, so it adds the chain and converts it into a carboxylic acid
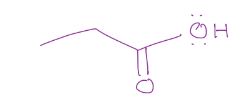
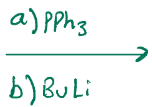
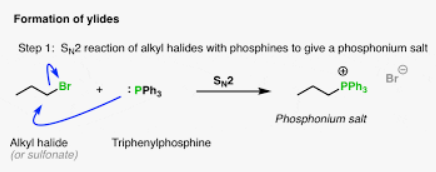
Ylide formation, need a leaving group, and adds the
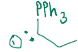
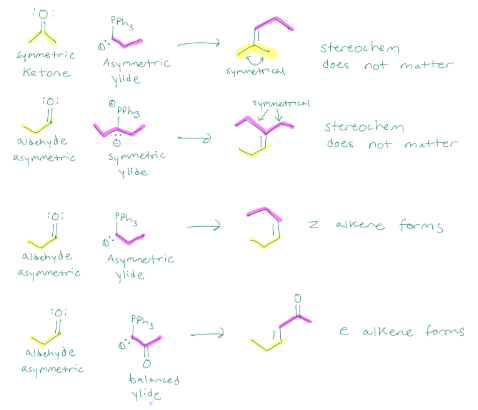
Wittig reaction, concerted addition to create an alkene from a carbonyl. If the ketone is symmetric, but the ylide is asymmetric, the stereochem doesn’t matter. If the aldehyde is asymmetric, but the ylide is symmetric, sterochem doesn’t matter. If you have an asymmetric aldehyde, and an asymmetric ylide, z alkene forms, but if aldehyde is asymmetric and ylide is balanced, e alkene forms.
H2NNH2, KOH, heat
Wolf-kischner, removes carbonyl
mCPBA (aldehyde)
Turns an aldehyde into a carboxylic acid
H2CrO4
Till turn aldehydes into carboxylic acids, and alcohols into ketones
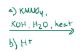
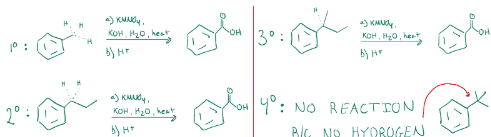
Forms aromatic carboxylic acids, and requires a hydrogen to react. Reacts with the carbon directly adjacent to the ring, so it removes all carbons behind it
Makes an ester into a carboxylic acid
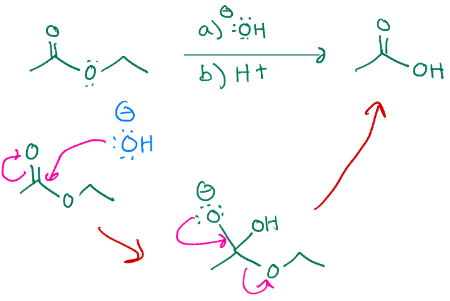
C.A derivative
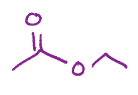
Turning ester into carboxylic acid
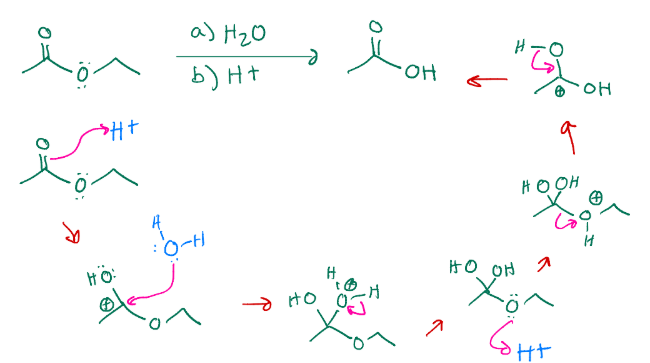

Fishcher esterification, turns a carboxylic acid into an ester with that chain added onto it
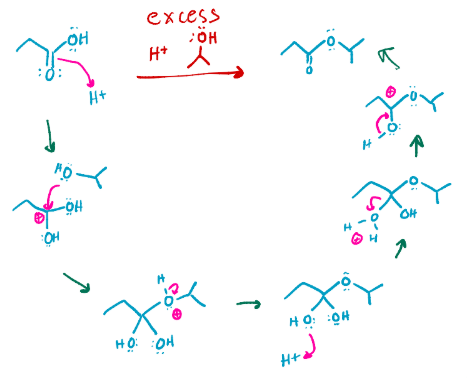

Reverse fischer esterification, turns that ester with a chain into a carboxylic acid
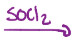
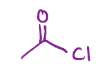
Carboxylic acid into a carbonyl with a Cl, an acid chloride
P2O5
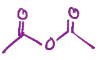
Turns carboxylic acid into a anhydride (two carbonyls with an o in between)

Carboxylic acid to thioester
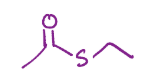

Thioester, same as fischer esterification

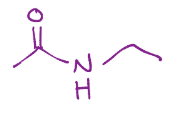
Amide formation from a carboxylic acid
Heat
Decarboxylation, going to remove a CO2 (usually a carboxylic acid)
H2O
Use to convert acid chloride and anhydrides back to a carboxylic acid
H2O, H+
Used to convert thioesters and esters back to carboxylic acids
H2O, H+, heat
used to convert amides back into carboxylic acids

Transesterification, changes out the oxygen that was in the ester, so you can now add a new chain
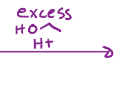
Transesterification, changes out the oxygen that was in the ester, so you can now add a new chain
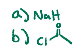
Interconversion, adds that ester onto the hydroxylZ
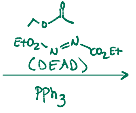
Adds onto an alcohol, has a stereochem flip

The carbonyl becomes a negatively charged oxygen as a hydrogen leaves from a carbon next to it, creating a double bond. The Br leaves the chain, and the double bond created by the old carbonyl reaches out to that charged carbon, and then oxygen goes back to becoming a carbonyl.
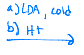
Becomes a negatively charged oxygen, and then LDA reaches out to a hydrogen that is on the carbon next to it, and a double bond can be created.
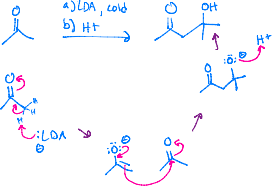
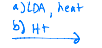
Becomes a negatively charged oxygen, and then LDA reaches out to a hydrogen that is on the carbon next to it, and a double bond can be created. LDA then reaches out to another hydrogen, on the carbon between your oxygens, and another double bond forms as the hydroxyl created leaves.
Adds the carbon chain to a side of the carbonyl, oxygen in part B is removed, and double bond remains

Adds the carbon chain to a side of the carbonyl, but the oxygen turns into a hydroxyl, and there is no double bond
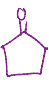
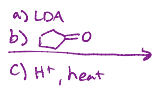
LDA, heat
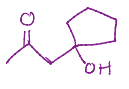
Reacts with itself one of the oxygens is removed and there is a double bond
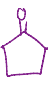
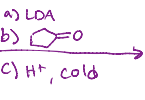
LDA, cold
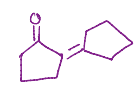
Reacts with itself, both oxygens remain, but one becomes a hydroxyl, no double bond
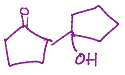
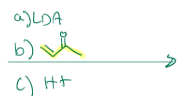
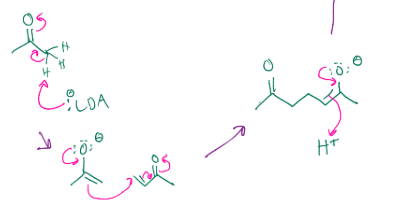
Gets rid of that double bond and the carbonyl shifts the electrons up so the carbon is charged, and then creates a double bond, which reaches out there
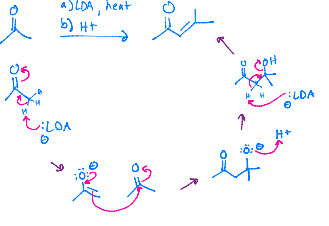
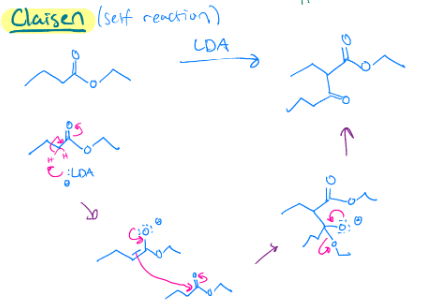
LDA (Claisen self-reaction)
LDA makes that double bond by the carbonyl. Then since it is a self reaction, next reactant is going to do the oxygen shift so that carbon is charged, and the double bond can reach out to it. Then the double bond doesn’t want to be charged anymore, so the electrons go back down to make a double bond, and that oxygen with the chain leaves
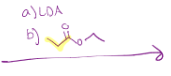
Crossed Claisen, much like Claisen, but not self reacting

Acetals as protecting groups
Convert a ketone or aldehyde into an acetal so that it doesn’t react with certain reagents to get a specific product, then can revert it
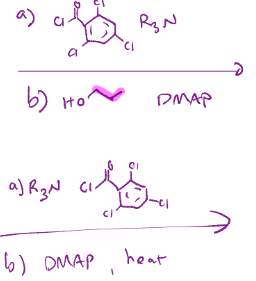
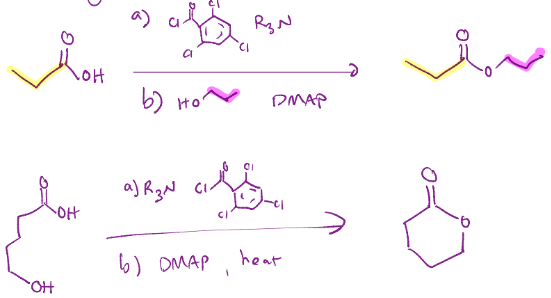
Yamaguchi macrolactonization, just a fancy esterification.
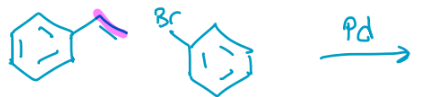
Heck
Makes a carbon carbon double bond between the two. Br gets removed