orgo 1 exam 3
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
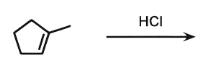
can be HBr, HI
markovnikov
add Cl to most sub side
remove double bond
check if chiral
if chiral = stereo
CARBOCATION CAN REARRANGE = RING EXPANSION
the H goes to the less sub side, the carbocation goes to the most sub side, CHECK if rearrangement needed, then the Cl Attacks it.
wherever there is a chiral center, related or not, ALWAYS STEROCHEM
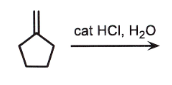
can be cat H2SO4 H2O
markovnikov
add OH to most sub side
remove double bond
check if chiral
if chiral = stereo
CARBOCATION CAN REARRANGE
wherever there is a chiral center, related or not, ALWAYS STEROCHEM
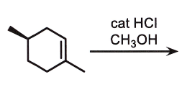
markovnikov
add OCH3 to most sub side
remove double bond
check if chiral
if chiral = stereo
CARBOCATION CAN REARRANGE
wherever there is a chiral center, related or not, ALWAYS STEROCHEM
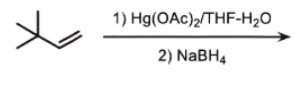
can be iPrOH or EtOH
markovnikov
add OH to most sub side
remove double bond
check if chiral
if chiral = stereo
NO CARBOCATION
wherever there is a chiral center, related or not, ALWAYS STEROCHEM
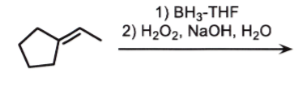
ANTI markovnikov
add OH to LEAST sub side
remove double bond
check if chiral
if chiral = stereo
NO CARBOCATION
wherever there is a chiral center, related or not, ALWAYS STEROCHEM
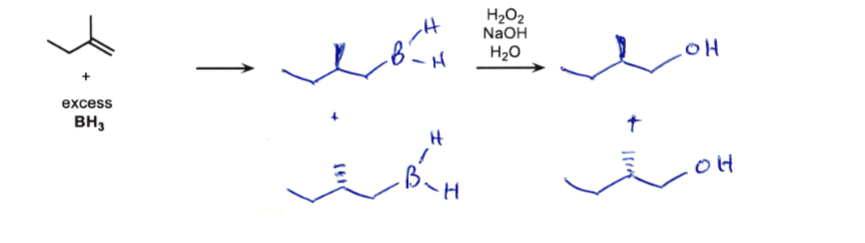
double bond → add H and B
then → change B into OH
DONT FORGET STEREOCHEM
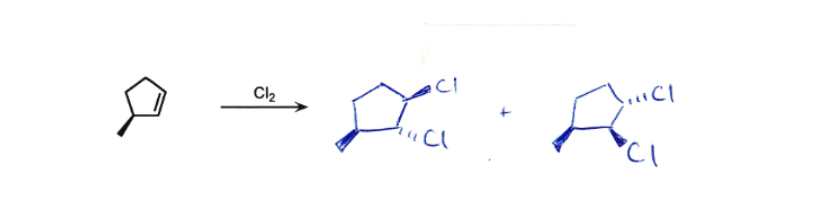
Anti on double bond
stereochem!!!!! ONLY on chiral
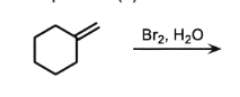
Add across the double bond
OH goes to MORE substituted carbon (Markovnikov)
Br goes to LESS substituted carbon
ANTI addition (opposite sides)
NO CARBOCATION
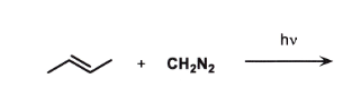
hv (light) → makes :CH₂ (carbene)
carbene adds across double bond
👉 You take the double bond and turn it into a triangle
This is SYN addition
So:
both new bonds form from the same side
Because carbene can come from:
front
back
👉 gives 2 stereoisomers
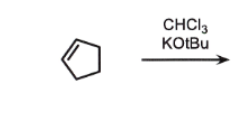
Or CHCl3 NaOH
You have a ring with a double bond
A tiny triangle piece (:CCl₂) comes in
It “snaps” onto the double bond
Now you get a 3-membered ring attached
forms :CCl₂ (dichlorocarbene)
👉 adds across double bond → makes:
cyclopropane
with 2 Cl on the SAME carbon
forms :CCl₂ (dichlorocarbene)
👉 adds across double bond → makes:
cyclopropane
with 2 Cl on the SAME carbon
🔥 Key idea:
👉 both Cl stay on the same carbon
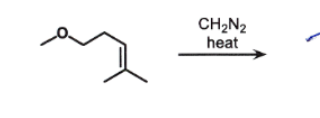
Or CH₂I₂ + Zn(Cu)
makes :CH₂ carbene
What to do:
find the double bond
draw a triangle across it
Stereochemistry:
SYN addition again
so:
front attack → one product
back attack → another
STEREOCHEM ON CHIRAL
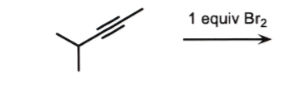
or Cl2
alkyne to an alkene
add 1 Br to each alkene carbon
trans
no wedge or dash
1 equiv Br2 or Cl2 starting w/ alkene
alkene to alkane
add 1 Br or Cl to each alkane carbon
wedge /dash if chiral
still anti addition
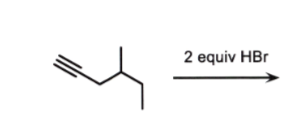
w/ no peroxides (no ROOR)
markov
alkyne to alkane
add two Brs to same carbon
no cis/trans
Is it HBr?
Are peroxides (ROOR) present?
How many equivalents? w/ Alkyne
How many equivalents? w/ Alkene
Is it HBr?
Yes → continue
No → ignore peroxide idea
Are peroxides (ROOR) present?
Yes → ANTI-Markovnikov
No → Markovnikov
How many equivalents? w/ Alkyne
1 → alkene NO wedges/dashes, add only 1 Br
2 → alkane NO wedges/dashes, add two Br same carbon
How many equivalents? w/ Alkene
1 → alkane Possible wedges/dashes if chiral, add 1 Br
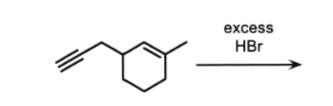
alkyne to alkane - mark, ADD 2 Brs!
alkene to alkane - mark, chiral? wedges dashes
Type of H | Structure | pKa (approx) |
|---|---|---|
strong acid (protonated O) | O+-H | ~ -2 to 0 |
carboxylic acid | C=O-OH | ~ 4–5 |
ammonium (R₃NH⁺) | ~ 9–11 | |
phenol | benzene ring–O-H | ~ 10 |
alcohol | R–OH | ~ 16 |
water | H₂O | 15.7 |
terminal alkyne | RC≡CH | ~ 25 |
amine (R–NH₂) | ~ 35–40 | |
alkane | ~ 50 |