Gen Chem II Lab 17: Titration Curves of Polyprotic Acids
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
polyprotic acid
an acid that can lose more than one per molecule in an acid base reaction
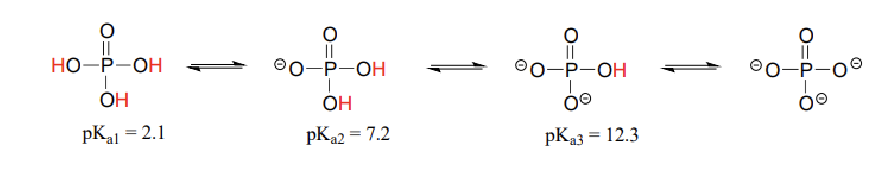
why is pKa2>pKa1?
the second proton disassociation involves removing a proton from a already negatively charged species. Thus, there is greater resistance faced in removing the second proton.
why is Ka1>Ka2 ?
The conjugate base and acid resulting from the first proton disassociation have more H+ ions (more acidic) than the conjugate base and conjugate acid in the proton disassociation
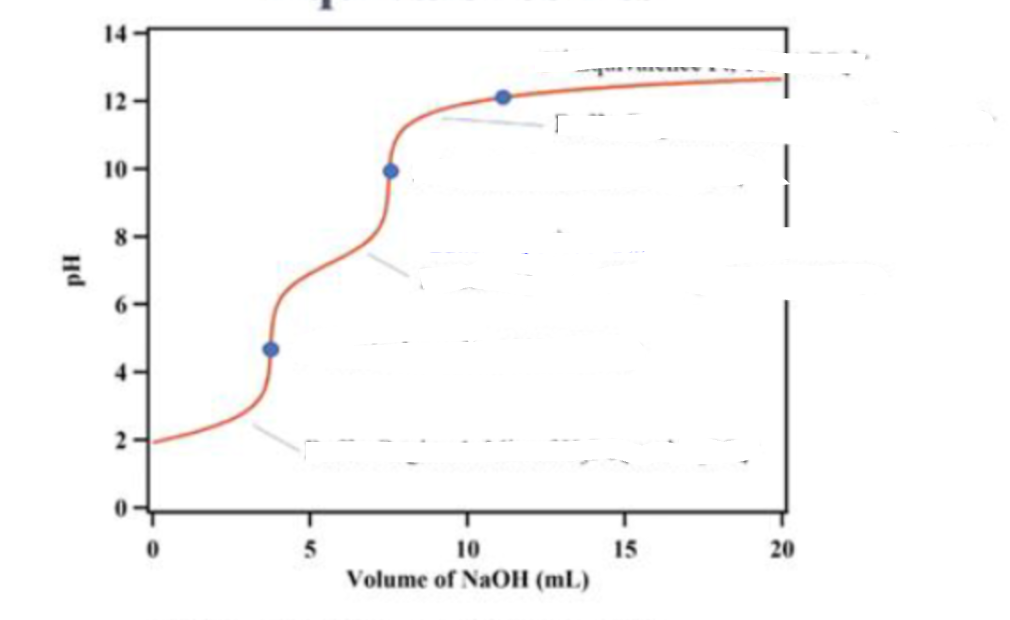
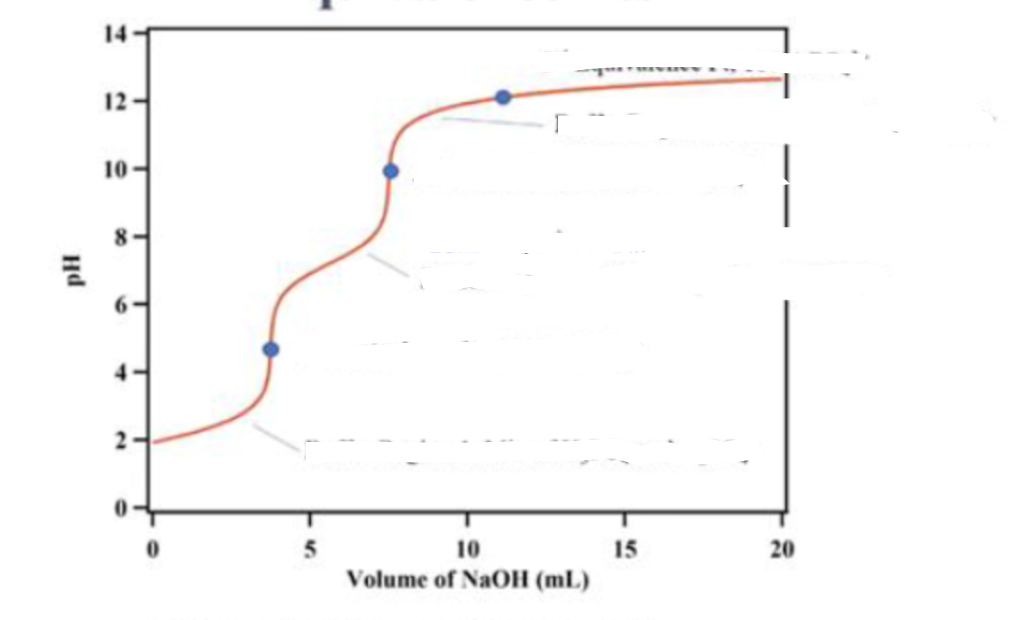
why don’t we see the third equivalence point ?
the acid strength of HPO42-(conjugate acid of the final ion PO43-) is close to that of water and as we add water the concentration of the acid is diluted
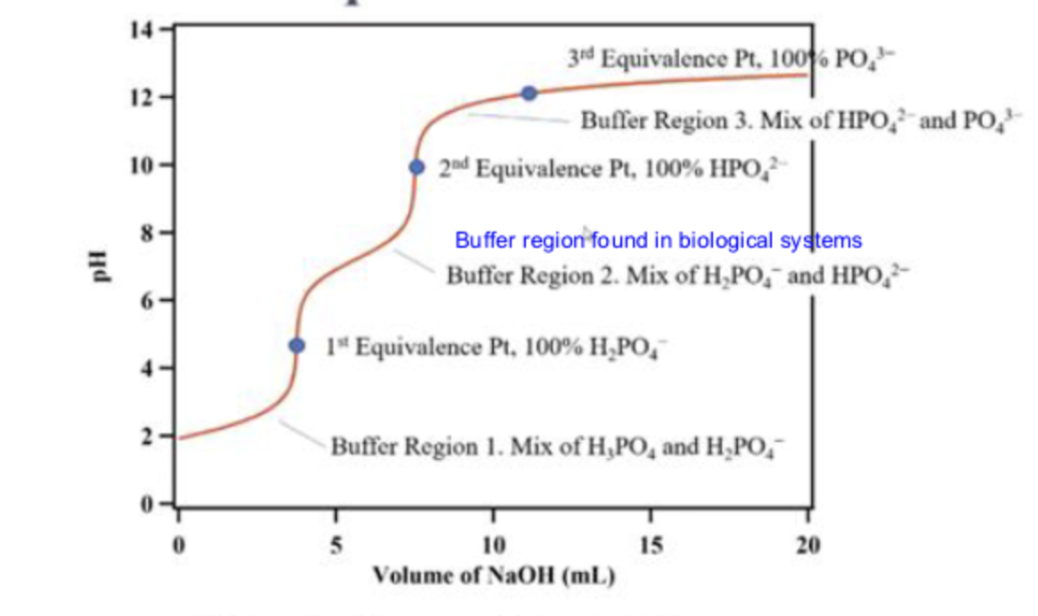
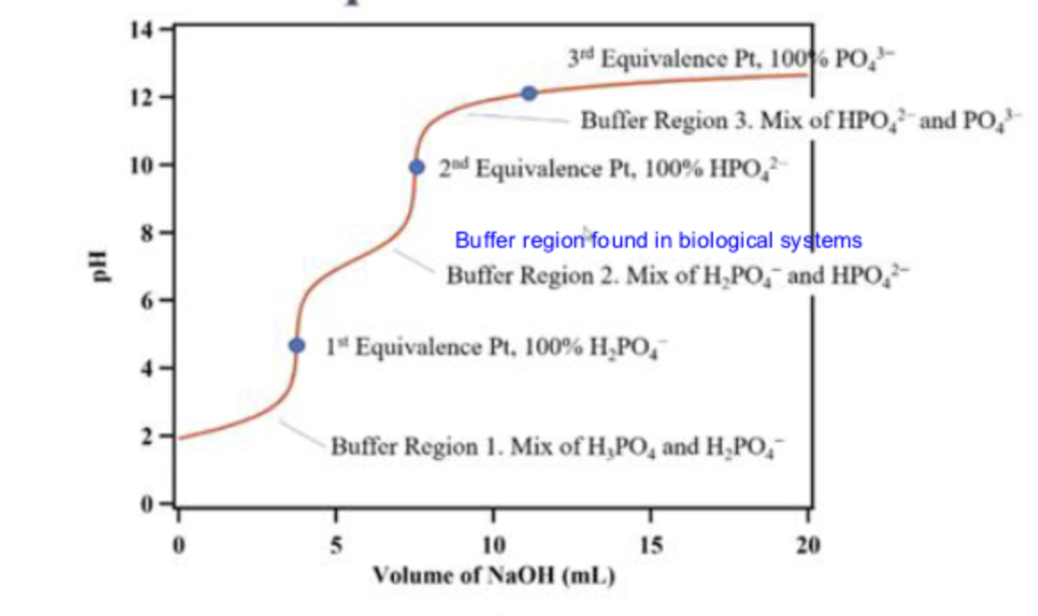
what is the first line pointing to
buffer region 1
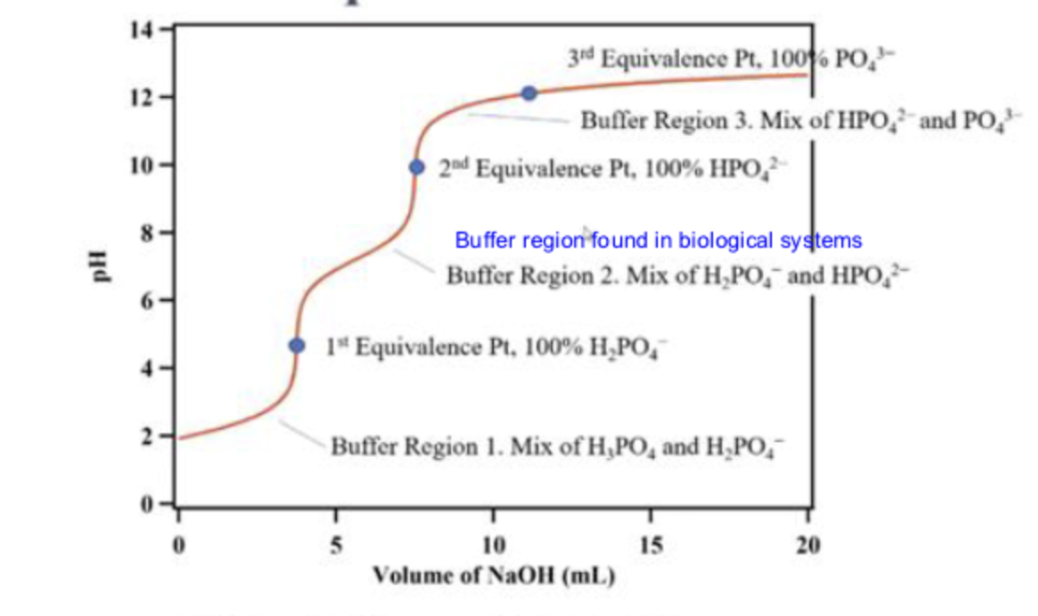
at buffer region 1, what is in the solution
a mix of H3PO4 and H2PO4-
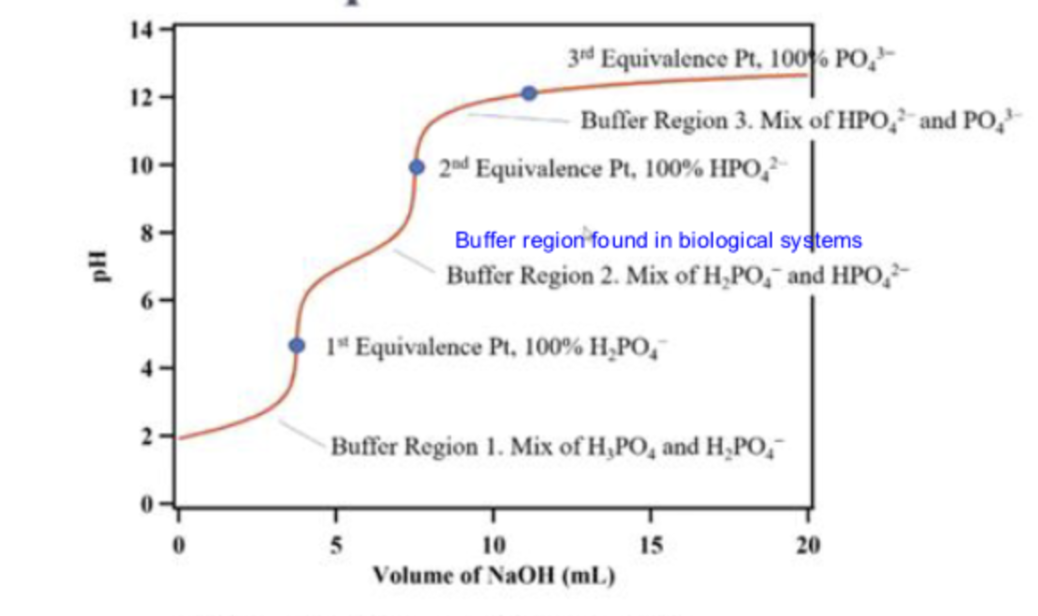
what is the first dot pointing to
the 1st equivalence point
molecular equation for reaction at 1st equivalence point
H3PO4(aq)+NaOH(aq)→NaH2PO4(aq)+H2O(l)
what is the second line pointing to
buffer region 2
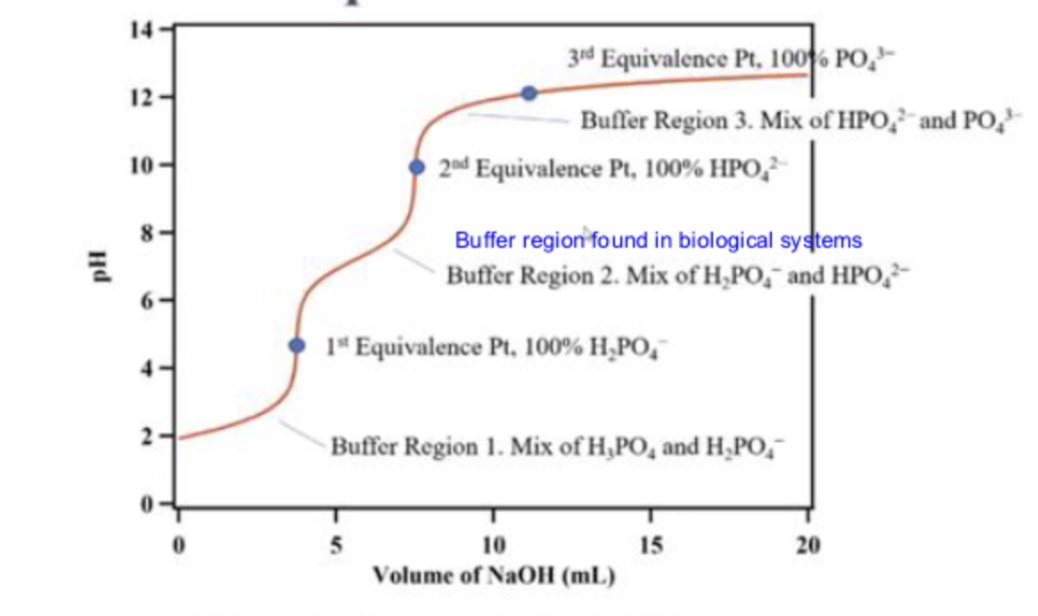
what is in the solution in buffer region 2?
a mix of H2PO4- and HPO2-
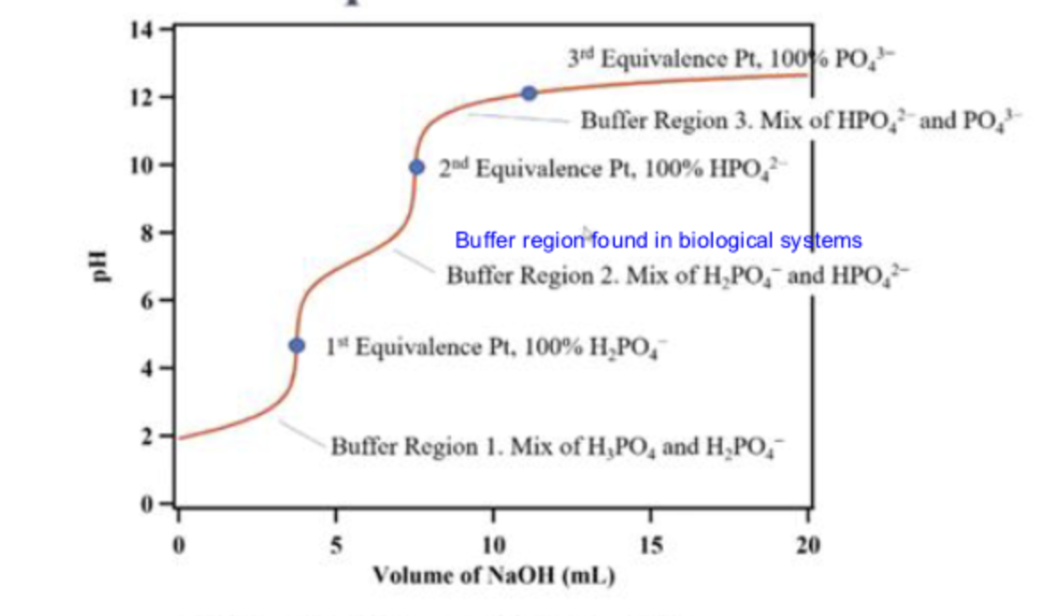
what is the second dot pointing to ?
the second equivalence point
molecular equation for reaction at second equivalence point
NaH2PO4(aq)+NaOH(aq)→Na2HPO(aq)+H2O(l)
what is the third line pointing to ?
buffer region 3
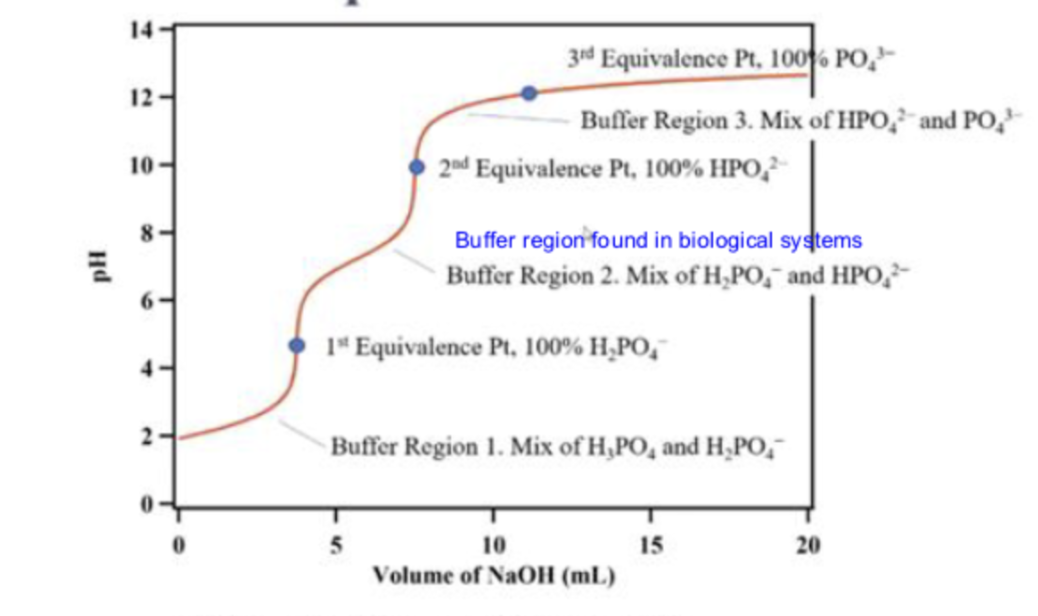
what is in the solution in buffer region 3?
a mix of HPO42-and PO43-
what is the third dot pointing to ?
equivalence point 3
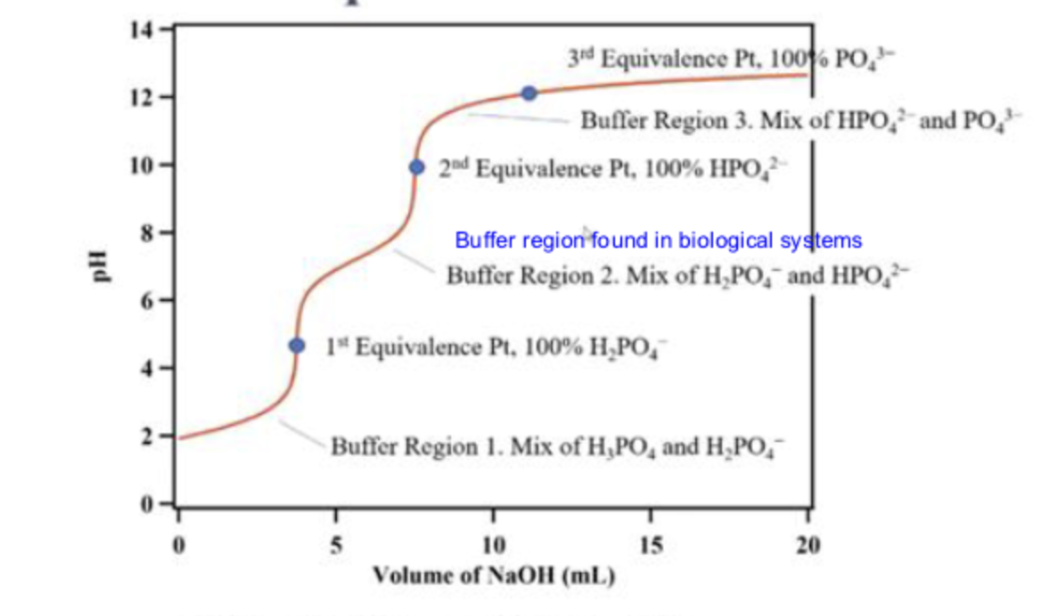
molecular equation at equivalence point 3
Na2HPO4(aq)+NaOH(aq)→Na3PO4(aq)+H2O(l)
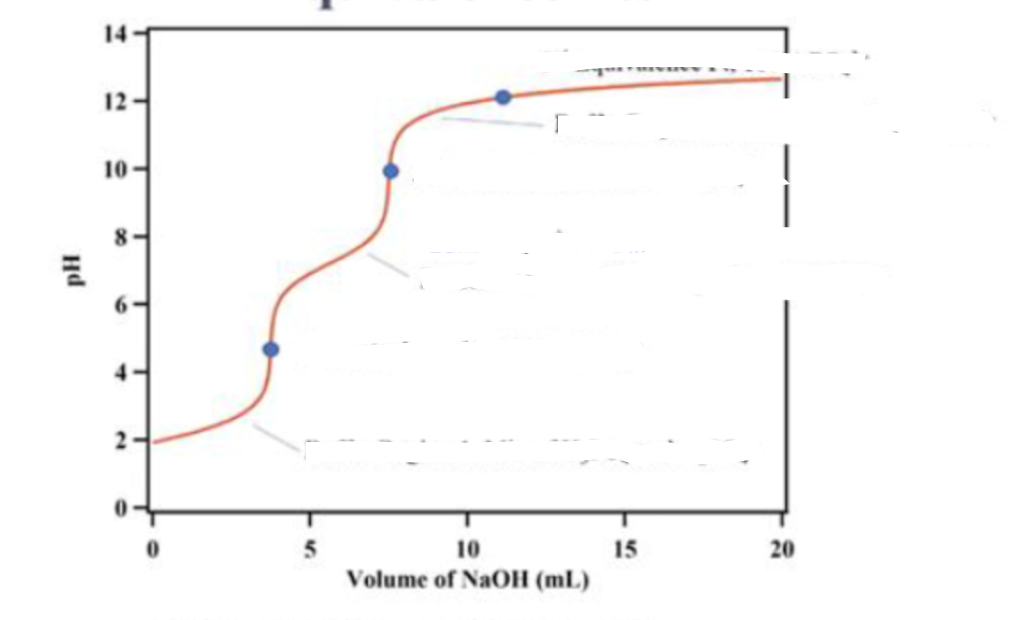
what is in the titration in the third dot ?
100% PO43-
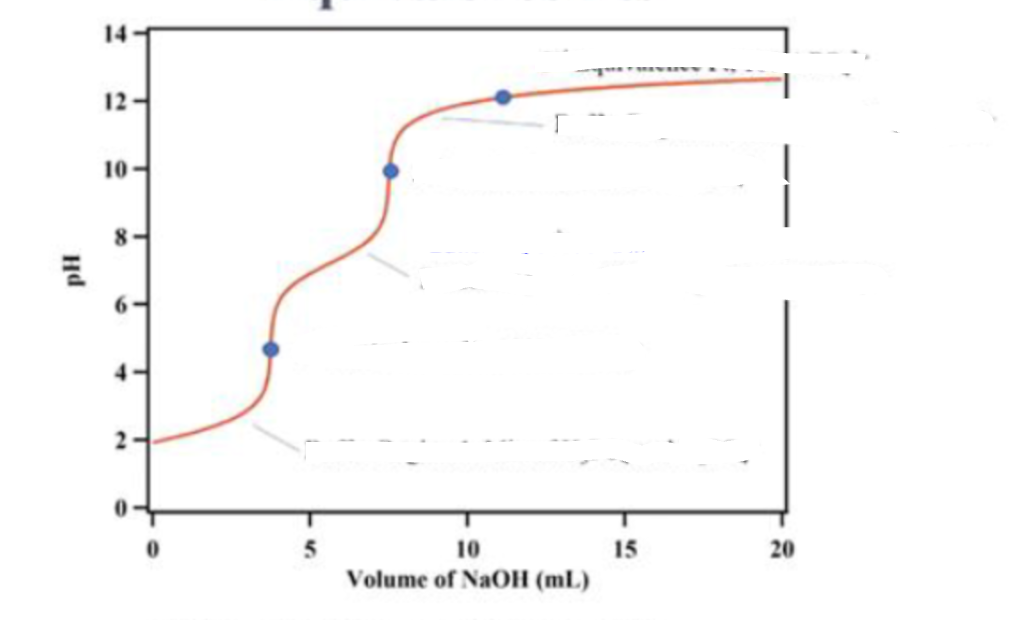
what is in the titration at the second dot
100% HPO42-
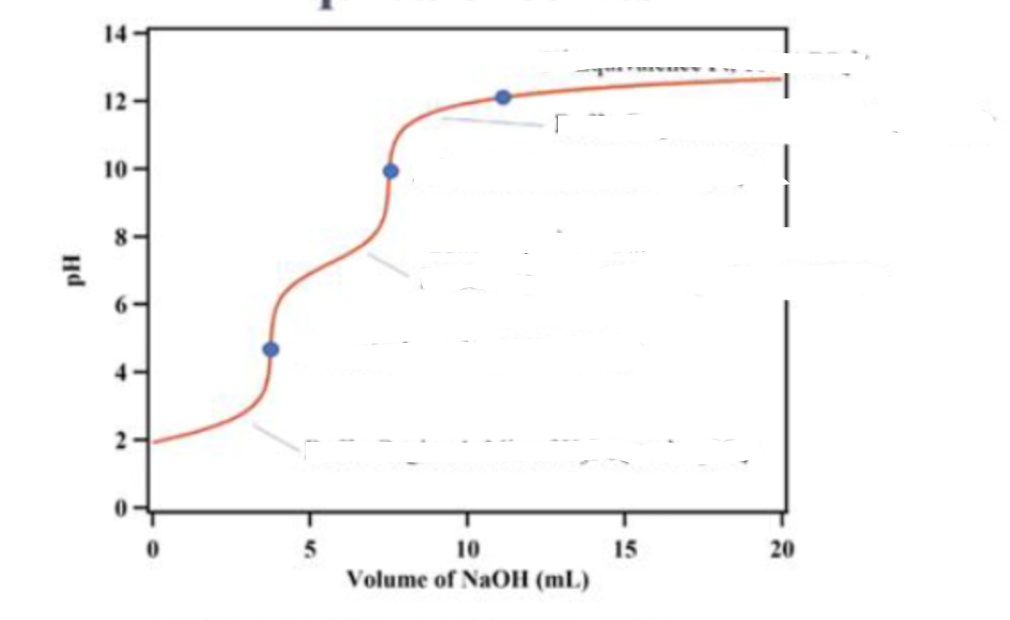
what is in the titration at the first dot
100% H2PO4-
pKa→Ka
Ka=10-pKa
how to find equivalence point from second derivative graph
take the point where the parabola crosses 0, that is the volume at ½ the equivalence point, double it to find vol at equivalence point
determining concentration of acid from equivalence pt
vol at equivalence point x [NaOH]= mol NaOH =mol acid, mol acid /vol in L= [acid]
Ka
the acid disassociation constant
the higher the Ka the greater
disassociation into products
Ka expression
= [A-][H3O+]/[HA]