Week 16: Medicinal Chemistry
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What structural class are most HSV antivirals?
acyclic nucleosides
What is the target of HSV nucleoside antivirals?
viral DNA polymerase (not host)
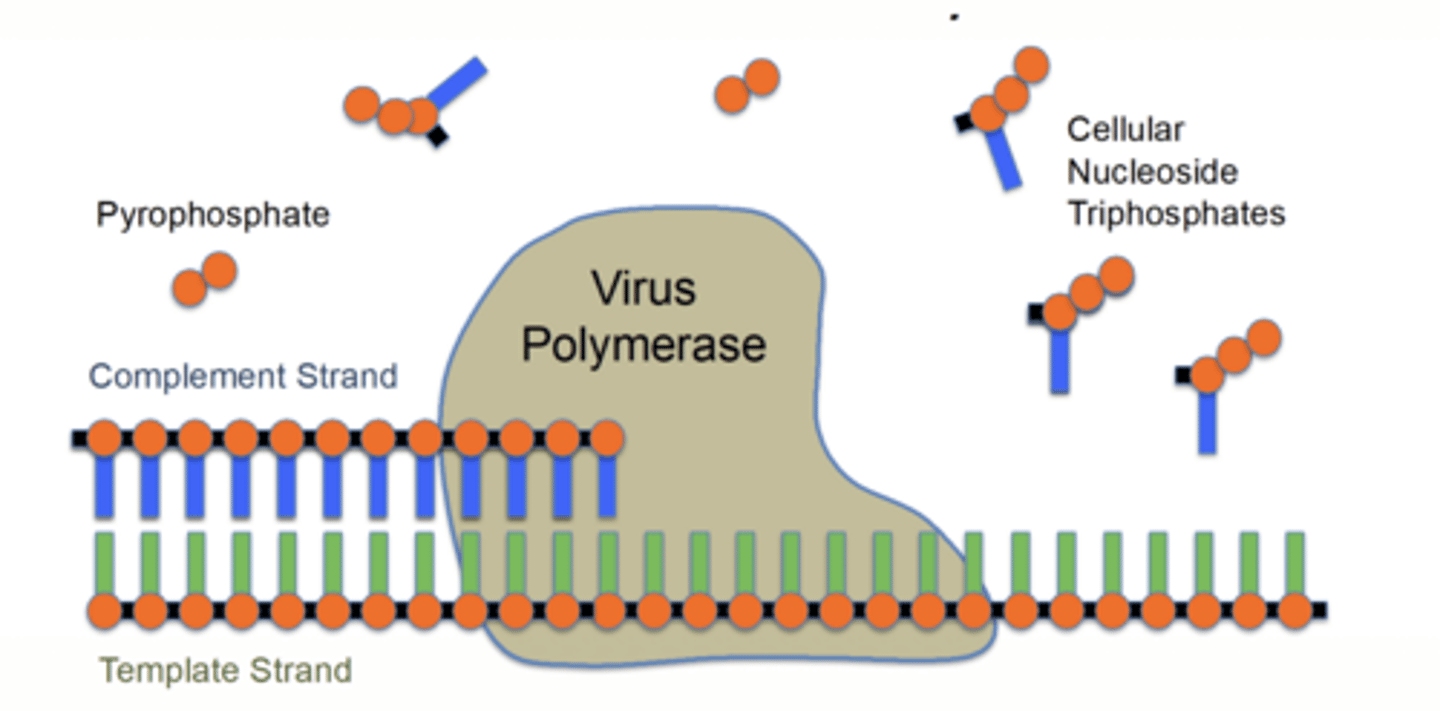
How are nucleoside antivirals activated?
phosphorylation → mono → di → triphosphate (active form)
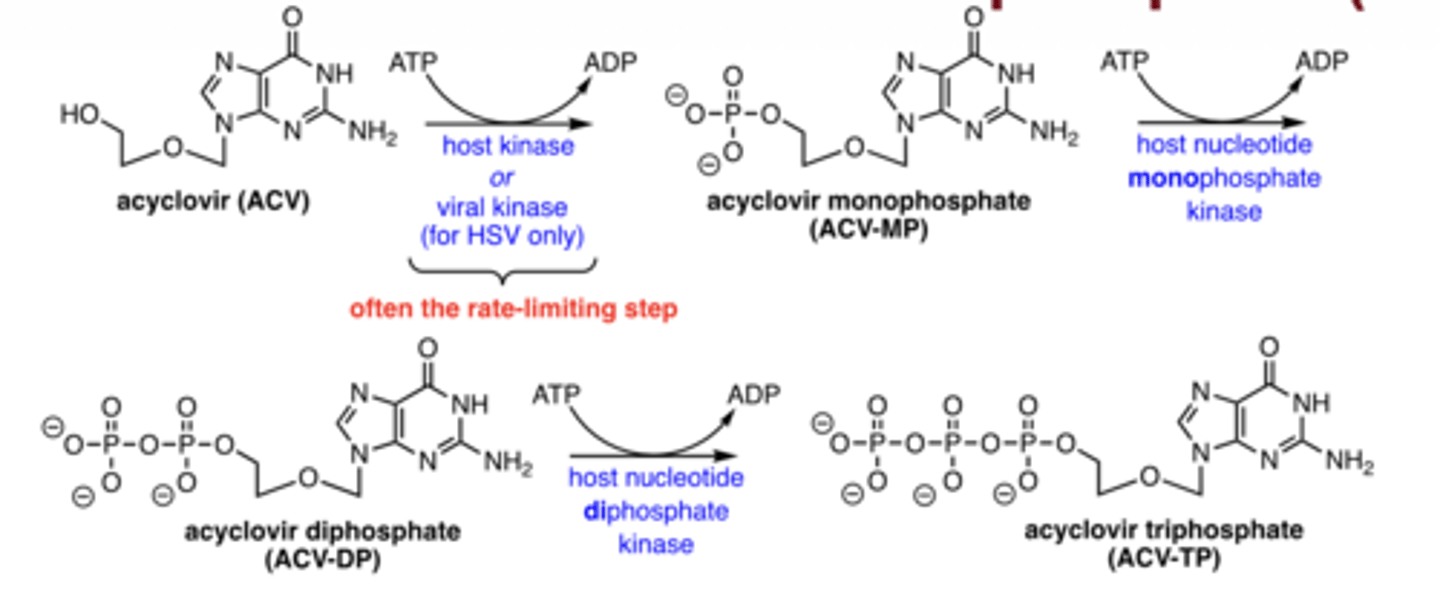
What enzyme is unique for HSV activation?
viral kinase performs initial phosphorylation (rate-limiting)
Why not directly use acyclovir triphosphate since it is the active drug?
highly charged → poor membrane permeability → poor oral absorption, distribution, and rapid clearance (cannot effectively enter cells)
Why is acyclovir a chain terminator?
lacks 3′-OH → cannot extend DNA chain
What is the major limitation of acyclovir?
poor oral bioavailability (10-20%)
How was acyclovir improved?
converted to prodrug (valacyclovir)
How does valacyclovir improve pharmacokinetics?
↑ bioavailability (~55%), ↑ Cmax
How is valacyclovir converted to acyclovir?
hydrolyzed by carboxypeptidase
How is acyclovir eliminated?
~90% excreted unchanged in urine
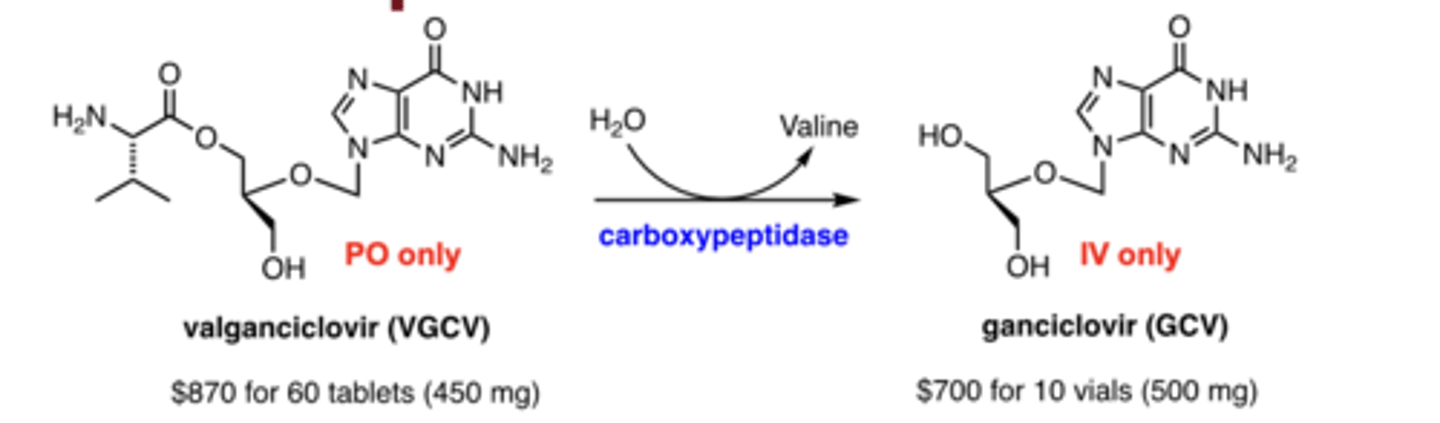
What is the difference between ganciclovir and valganciclovir?
Ganciclovir = IV only; Valganciclovir = oral prodrug
Why is ganciclovir poorly absorbed?
high polarity (multiple OH groups)
How is valganciclovir converted to ganciclovir?
via carboxypeptidase
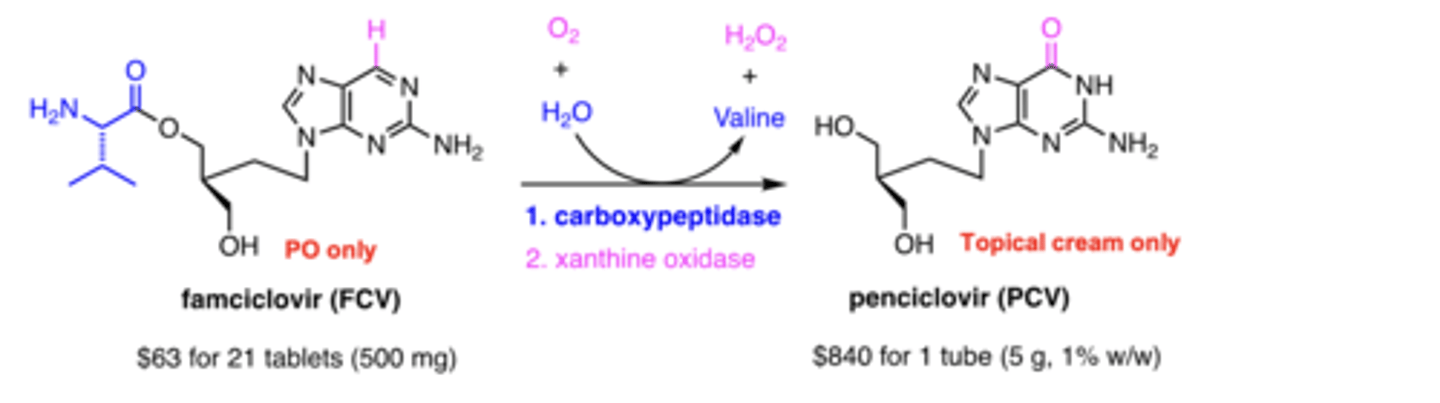
Relationship between famciclovir and penciclovir?
famciclovir = oral prodrug → penciclovir
How are they administered?
Famciclovir = PO, Penciclovir = topical
How is famciclovir converted to penciclovir?
step 1 - carboxypeptidase; step 2 - xanthine oxidase
Which drug covers HSV, CMV, and VZV?
ganciclovir
Which viruses do acyclovir-class drugs mainly treat?
HSV + VZV (limited CMV); only ganciclovir covers CMV

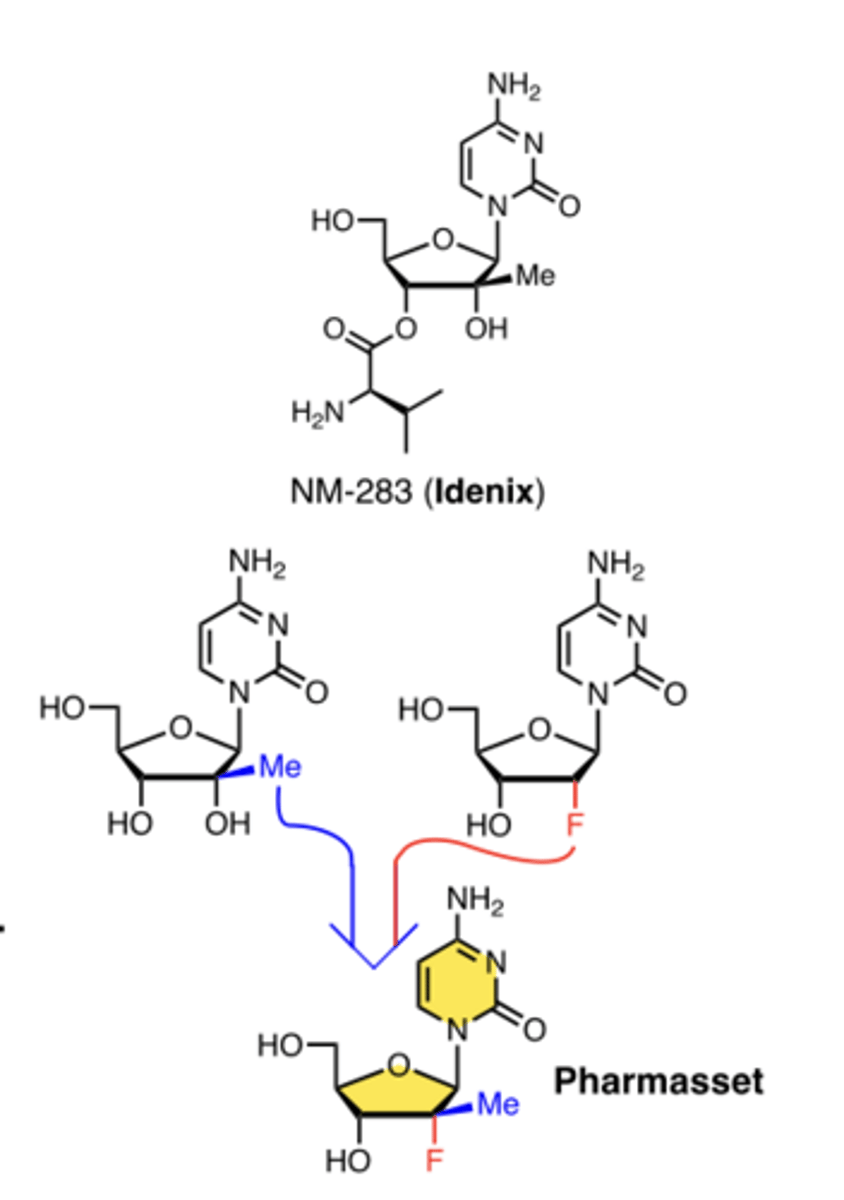
What major change improved HCV outcomes?
direct-acting antivirals (e.g., sofosbuvir) ↑ cure rates dramatically
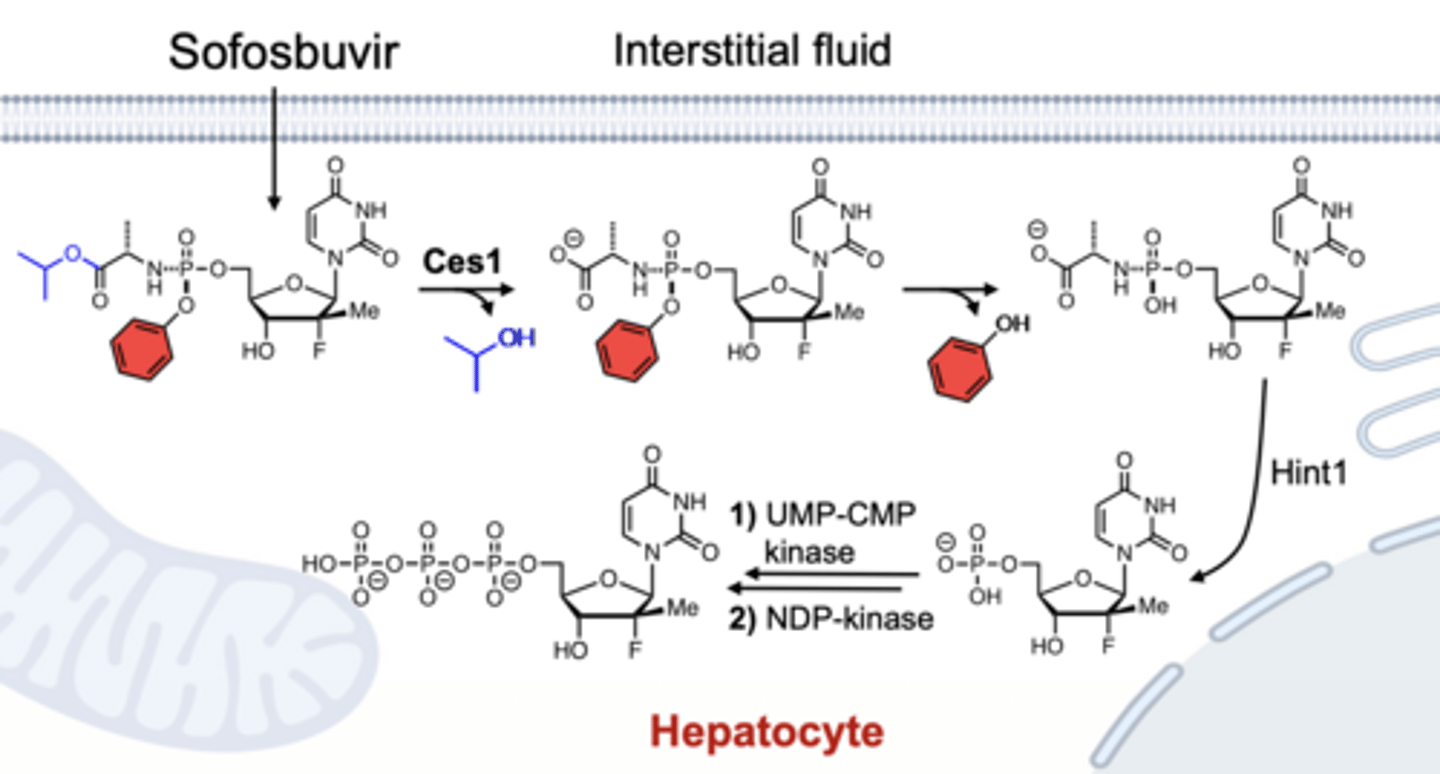
MOA of sofosbuvir?
RNA-dependent RNA polymerase inhibitor (NS5B), chain terminator
MOA of velpatasvir?
NS5A inhibitor → disrupts replication machinery; synergises with sofosbuvir
What key structural feature made sofosbuvir effective?
2′-methyl + 2′-fluoro modifications
What enzyme does sofosbuvir inhibit?
viral RNA-dependent RNA polymerase (RdRp)
How is sofosbuvir activated?
prodrug → monophosphate (via CES1/HINT1) → triphosphate (active)
Why does sofosbuvir inhibit RNA synthesis despite having 3′-OH?
steric hindrance (methyl/fluoro) disrupts elongation
How do physical properties affect drug design?
↑ polarity → ↓ bioavailability; prodrugs improve absorption
What determines antiviral toxicity?
selectivity for viral vs host polymerase