Amino acids, proteins, and DNA
1/55
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Draw and label the general displayed formula of an amino acid.
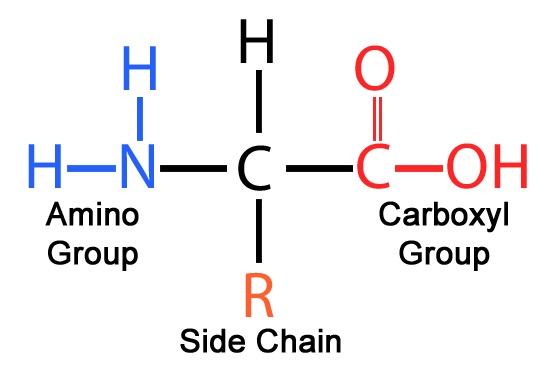
True or false? Amino acids are amphoteric.
True.
Why do amino acids show optical isomerism?
They contain a chiral carbon (the central carbon is the chiral carbon).
Talk me through how to name an amino acid using the IUPAC system.
1) Name the longest carbon chain as a carboxylic acid.
2) Number the carbons always starting from the carboxyl group carbon as number one.
3) Number which carbon the amino group is on, this will be the prefix of the name.
4) Name any other groups in the molecule.
What is a zwitterion?
A molecule that contains both positive and negative ions.
True or false? Amino acids always exist as zwitterions.
False. ZWITTERIONS ONLY EXIST AT THE ISOELECTRIC POINT OF THE AMINO ACID.
What is the isoelectric point of an amino acid?
The specific pH at which the amino acid has a net charge of zero.
Draw the general displayed formula of an amino acid zwitterion. How does the zwitterion form?
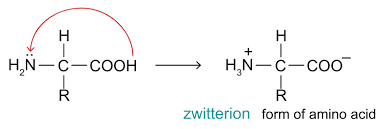
The hydrogen in the carboxyl group is donated to the nitrogen in the amino group as it has a lone pair of electrons.
Draw the general displayed formula of what happens to an amino acid zwitterion when the pH is lower (more acidic) than the isoelectric point. State what has happened to it.
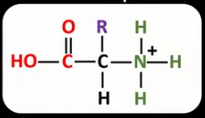
The COO- has accepted a proton (acted as a base).
Draw the general displayed formula of what happens to a zwitterion of an amino acid when the pH is greater than the isoelectric point. State what has happened to it.
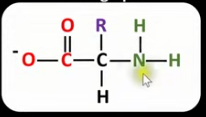
The NH3+ group has donated a proton (acted as an acid).
True or false? Different amino acids have different solubilities.
True.
What technique can you use to separate amino acids?
Thin layer chromatography (TLC).
True or false? Amino acids are colourful compounds.
False. AMINO ACIDS ARE COLOURLESS COMPOUNDS.
What is the monomer that makes up protein?
Amino acids.
What type of polymer is protein?
Condensation polymer.
What type of bond joins amino acids in protein?
Peptide bonds (amide links).
How can you break a polypeptide?
Hydrolysis (addition of water).
What molecule is formed when two amino acids are bonded together?
Dipeptide.
Draw and label the displayed formula of the reaction between two amino acids to form a dipeptide.
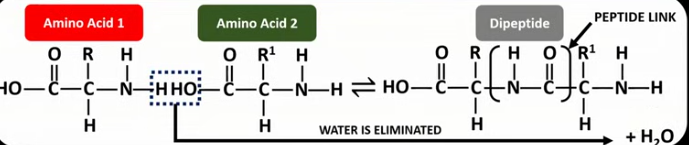
Explain the advantage of amino acids being difunctional.
It allows the amino acids at the ends of a polypeptide to undergo further reactions to make up a polymer chain and not be limited to dipeptides.
What conditions are needed to hydrolyse a protein?
6 mol dm-3 hydrochloric acid, 110 degrees C, reflux for 24 hours (very severe conditions).
What is the primary structure of a protein?
The sequence of amino acids that make up the protein.
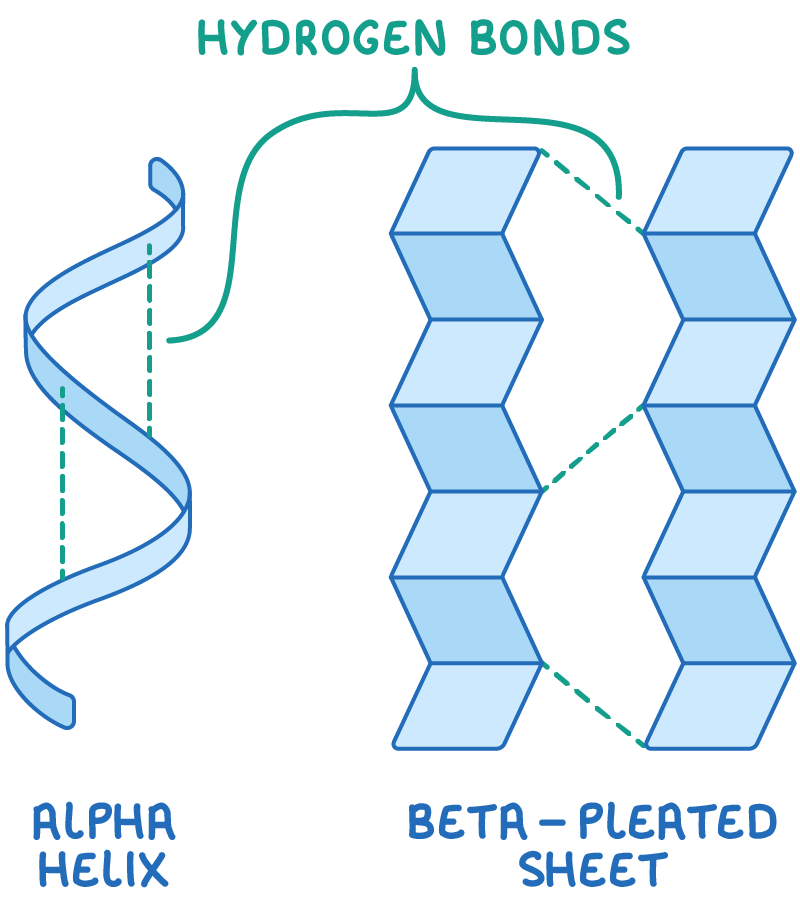
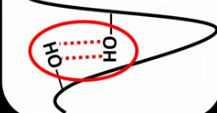
What is the secondary structure of a protein?
When the polypeptide has a coiled/pleated structure due to hydrogen bonding.
Describe the structure of an alpha helix and a beta pleated sheet.
What is the tertiary structure of a protein?
The unique shape of a protein caused by hydrogen bonding, ionic bonding, and disulfide bonds (disulfide bridges).
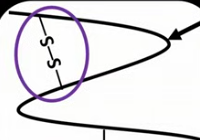
How do disulfide bonds form in a polypeptide?
The R group of cysteine contains -SH, this -SH can lose a hydrogen and form S-S bonds (disulfide bonds) with opposite R groups on cysteine.
Draw what disulfide bonds look like in a polypeptide.
Draw what hydrogen bonds look like in a polypeptide.
How do temperature and pH affect the shape of proteins?
They interfere with with the hydrogen bonds and disulfide bonds in the tertiary structure which changes the shape of the protein.
What is the function of enzymes?
They catalyse metabolic reactions.
Talk me through how enzymes work in catalysing reactions.
Enzymes have a specific tertiary structure that involves an active site that is complementary to a specific substrate. The substrate binds with the active site forming an enzyme-substrate complex. This puts strain on the bonds in the substrate making them easier to break.
What does it mean that the active sites of enzymes are stereospecifics?
Enzymes have enantiomers as they are made up of chiral amino acids. Only one enantiomer on an enzyme will fit into the substrate.
Give one way on which the rate of an enzyme-controlled reaction can be slowed down.
Add an inhibitor for the enzyme to the reaction mixture.
Talk me through how a competitive inhibitor works in slowing down the rate of an enzyme-controlled reaction.
1) The competitive inhibitor has a similar shape to the substrate so binds to the active site of the enzyme.
2) The already occupied active site prevents an enzyme-substrate complex from forming.
3) This means it takes longer for the substrate to be broken down meaning the rate of reaction decreases.
True or false? Different inhibitors bind to the active sites with different strengths.
True.
How does using an inhibitor with an increases strength of binding to the active site of the enzyme change the decreases in the rate of reaction?
Greater decrease in the rate of reaction.
How do some drugs work with enzyme inhibition? Use antibiotics as your example.
Some drugs are enzyme inhibitors.
1) The antibiotic blocks the active site of the enzyme that is responsible for making the cell wall of the bacteria.
2) The cell wall of the bacteria cannot be made causing the bacteria to burst and die.
True or false? It’s impossible for drugs to show optical isomerism.
False. CERTAIN DRUGS SHOW OPTICAL ISOMERISM.
What is the issue with a drug showing optical isomerism?
Only one of the enantiomers of the drug will be complementary to the stereospecific active site of the enzyme meaning the other enantiomer will be completely ineffective/have harmful side effects.
How can computers be used in drug modelling?
Computers can be used to design drugs that may bind to the active sites of enzymes without having to actually make the drug and test it.
What are the benefits of using computers in drug modelling?
Cheaper.
Quicker.
What is the monomer that makes up DNA?
(DNA) nucleotides.
Draw and label the general structure of a DNA nucleotide.
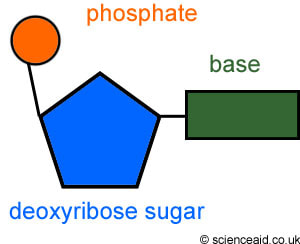
Name the four nitrogenous bases that can be found on DNA nucleotides.
Guanine, cytosine, adenine, thymine.
How do two DNA nucleotides bond together?
The phosphate group of one nucleotide forms a phosphodiester bond (a type of covalent bond) with the deoxyribose of the other nucleotide, this is a condensation reaction.
Describe the structure of DNA.
Two polynucleotide strands that are held together by hydrogen bonds between complementary base pairs, twisted into a double helix.
Which bases are complementary to each other?
Adenine is complementary to thymine, cytosine is complementary to guanine.
Using the data sheet, draw how hydrogen bonds form between adenine and thymine.
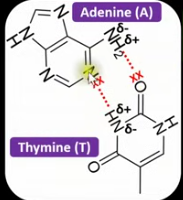
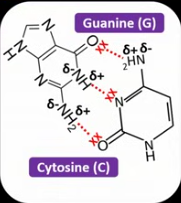
Using the data sheet, draw how hydrogen bonds form between cytosine and guanine.
How many hydrogen bonds form between A and T?
2
How many hydrogen bonds form between G and C?
3
Why does not further hydrogen bonding take place between complementary base pairs despite there being more partially charged atoms?
The partially charged atoms would be too close to each other and repel.
OR
The partially charged atoms would be too far apart to form hydrogen bonds.
Why is DNA a twisted double helix?
It allows the base pairs to perfectly align to form hydrogen bonds.
What sort of drug is cis-platin?
An anti-cancer drug.
Describe the structure of cis-platin.
Square planar ion complex consisting of a platinum ion surrounded by two ammonia ligands and two chloride ion ligands.
Draw the structure of cis-platin.