unit 2 transition metals
1/66
Earn XP
Description and Tags
oxidation
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
Radii
generally decreases and then increases across a row
cations smaller
anions bigger

elctron config and ox states
-1/2 filled always wanna be d5 of d10
-catiosn loose s e-s before D e-s
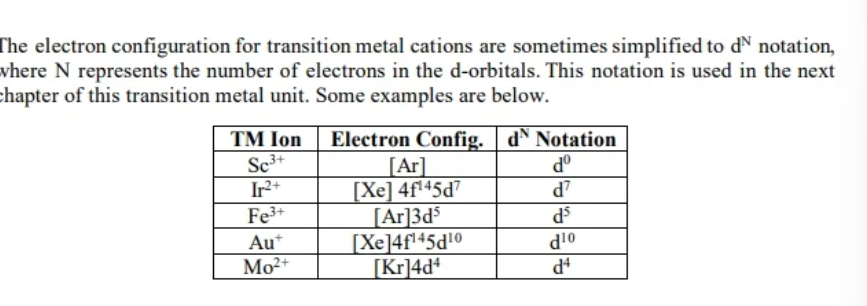
electron config transition metals D^n
*e- config of transition metal cations are sometimes simplified to d^N notation - N = number of e-s in the D orbittal
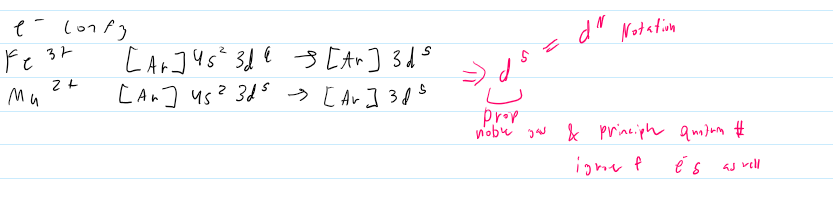
ionic and covalent
*Transition metals have both ionic and covalent charater.
covalent bonds : usually coordinate covalent bonds - both e-s are donated by one group (ligand)
color
any metal with partially filled d-subshells has color
e- transition occurs upon absorption of light energy (electronic transitiosn between d - orbs)
filled and unfilled d subshells colorless

Combination reaction
Decompisition reaction

single replacement reaction

Double replacemnt reaction
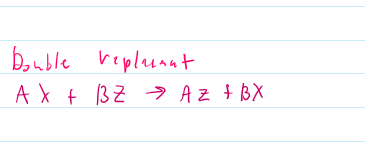
Most transition metals R paramagnetic ( 1 unpaired e-)
soem ferromagnetic - permanet domains of magnetism
catalysis
multiple ox states and coordination numbers are possible
ligands can easily bond and multiple bonding site s are avaliable
transition metals are generall coordinatively unsaturated
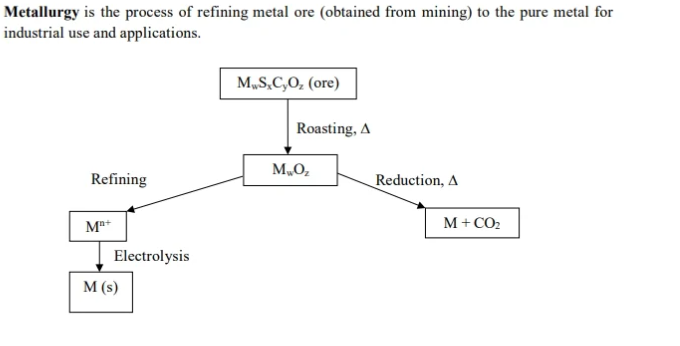
Metallurgy
*ore → metal
*alloy - moxing two mentals together
Scandium
d2
Sc+3
Common to Al, Y, La bc nonne got de-s
colorless and dia
high intensity lamps or alloys
Titanium
Ti+4
hight strenght/weight ratio
low corrosion
C,Si
Colorless
Vanadium
+2 … +5
V2O5 - catalyst
tough and strong steel
Chromium
+2, +3, +6 common ox
corrosion resistant
color red
Tungsten
W - woldfam
lightbulb
Hard substance used in drills
Maganese
+2 … +7
- MnO2
in a dry cell battery ()
Techntium
smallest element w/ no stable isotopes
very radioactive
Iron
most important
+2, +3
steel, alloys, heme
4.7% of earths crust
M-CO
Cobalt
+2, +3
cathode, Lion battery cell
color difficutl to isolate (blue)
in vitamin B12
Nickel
Corrosion resistant
+2 ox state
M-CO
Doubly magic - extreamly stable (#protons and # neutrons)
*NiCad cath
catalysit fo hydrogenation
Platinum
+2, +4
Not very reactive
copper
coinage metals cuz corrosion resistant
bronse Cu Sn
brass CU + Zn
very conductive
s e-s dominate its chemistry
Gold
dont oxidize in air or water
consistant conainge metal
malleable
Zinc
+2, d^10
colorless, Dia
galvanizing
dry cell anode
corrosion resistant
Cadium
+2
cathode NiCad
Tabacco
Mercury
+1, +2
toxic
liquid at STP
Lanthahides
inner transition metals
+3
highly reactive w/ halogens and chalcogens
F shell
e-s lost in S shell before f e-s
vesper

coordinate compounds
transition metal ions, in combo with ligands form these compunds and counter ions
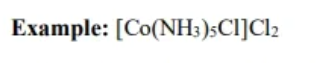
complex ion
Transition metal iosn in combo with ligand from - transition metal species has nonzero charge
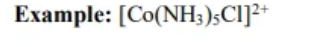
counter ions
aniosn or catiosn needed to produce a compund with no net charge
added to make coordination compund neutral

ligand
group surrounding transition metal ion

coordination number
number of nearest neighors to transition metal ion
neighbors, usually ligands
How to find: count number of ligands aroudn transition metal ion,
can vary from 2-12 , 6 most common, 2 or 4
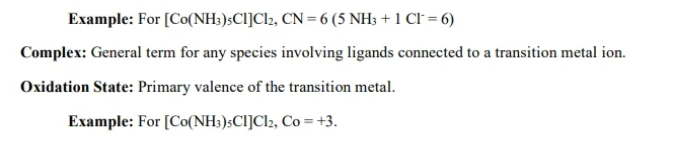
Ligand will have a
lone pair of e-s ready to form a coordinate covalent bond
neutral or negative charge

vesper summary
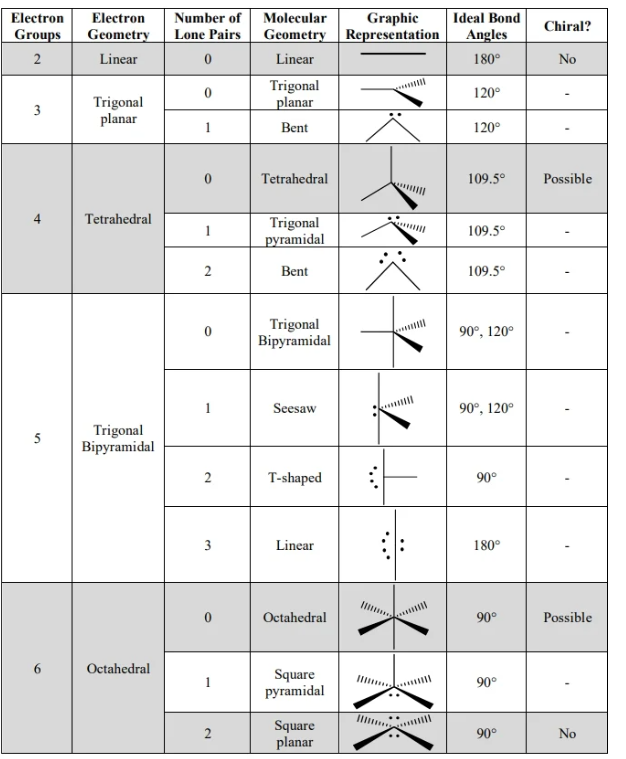
removing D e-s

d^n notation
As oxidation increases….
it becomes more acidic bc the ox state of metal increases so it enhances the metal oxide’s ability to accept e- pairs or donate protons.
3 types of magnetism…

As you move down the table.. (transition metals)
The stability of oxidation states increases - bc the V e-s are further form the nuc - affects removal E.
Stability…
influnced by # of e-s
increases as more e-s in d orb
most stable: Cu, Fe, Ni, Zn
Max e-s held in each orb?
p - 6
f - 14
d - 10
s - 2
Hybridization
s, p, d orbs together
counter ion
ion present to balance charge of the complex ion but is not directly bonded to the central metal by donating e-s
how many bonds can a single EDTA forms
6
Monodentate ligand
ligand with 1 pair of e-s to attatch
polydent
2 or more pair of e-s
Chelating
Bidentate ligand
2
tridentate
3
tetradentate
4
pentadente
5
Hexadentate ligand
6 donor sites
Why does the chelate effect lead to increased stability?
The replacement of multiple monodentate ligands by a single polydentate ligand results in increased entropy
monodenate ligands
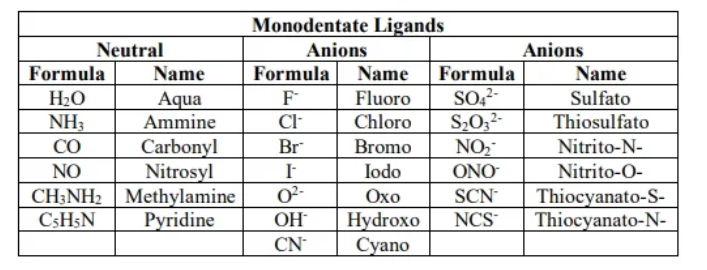
polydente ligand
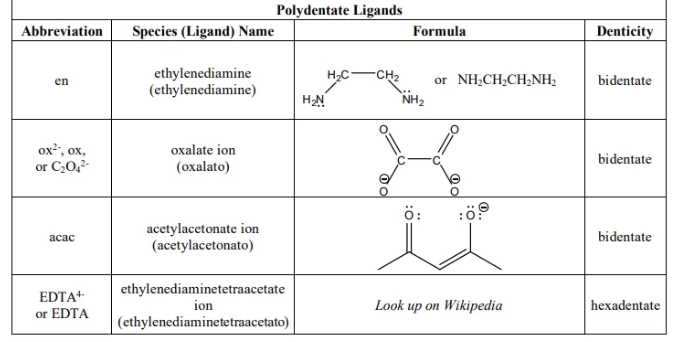
nomeclature new
1.cations named 1st, then anions
2. ligands named using:
-ide → -o
-ite → -ito
- ate → -ato
3. number of ligands is named by prefix (mono, bi, tri, tetra, penta, hexa)
Composite ligands - contain prefixes in its name (complicated ligands) but not all polyd ligands r composite,. YOU WILL HAVE TO USE composted PREFIXES EN,OX, ACAC AND EDTA
4. When naming complex ion, ligands are named alphabetically in order 1st (not including prefix) AND THEN the metal with ox state is named last
A. if complex ion is an anion, metal nust be named in latin (if possible) with the suffix - ate. latin stem in table below
(Iron - ferrate, Copper - cuprate, Tin - stannate, Silver Argentate, lead - plumbate, Gold - Aurate)
b. if complex ion is cation, metal is named english with no additional suffix
5. When writing a formula: list ligands in abc order
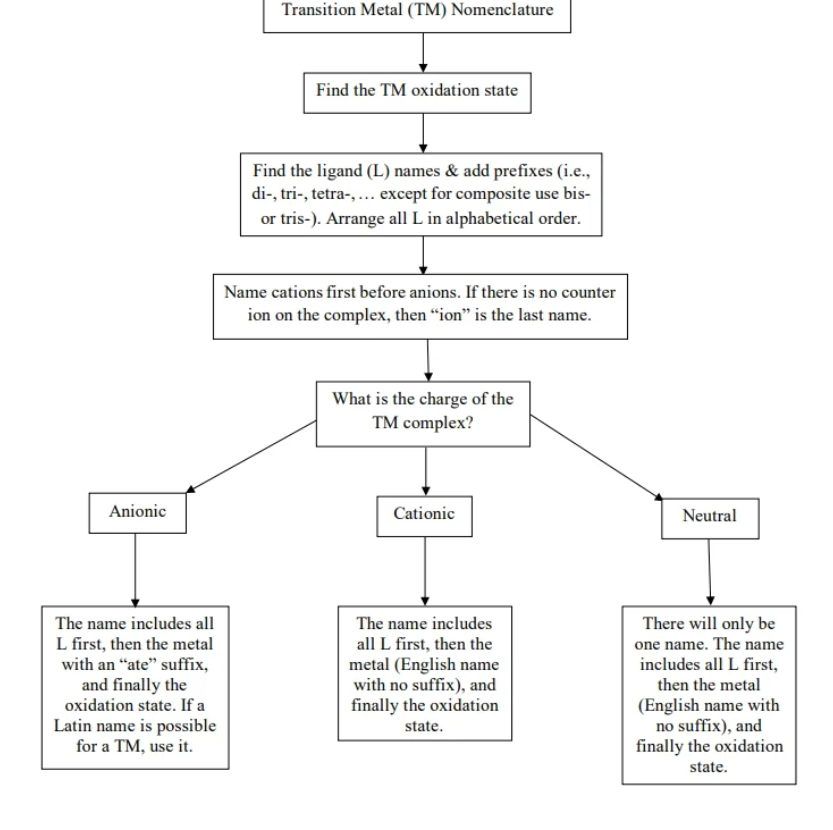
nomen new summary
1. identift coordination sphehre - central metal aion and ligands
2. ligand 1st - in abc order
A. neutral -
ammine - NH3
aqua - H2O
corbonyl - CO
nitrosyl - NO
B. Anionic - end with O
Cl- - chloro
OH- → hydroxo
CN- > cyano
O²- → oxo
C. prefixes
mono
di
tri
tetra
penta
hexa
D polydente ligands like EDTA
Bis - 2
Tris - 3
Tetrakis - 4
3. metal name
a.is neutral or cationic : use normal metal name
b. Complex is anionic - at ate to the end
4 dtermien and add metal od state
use form : ox state of metal - overall charge - sum of ligand charges
5. combo everything
If there are counter ions : name the cation 1st then anion - leave a space in between co
nomeclature old 1. Binary ionic compounds
Metals give e-s - catiosn
metals take e-s - anions
state cation then anion
1. A name = of element
2. B Name = elemnt name end s with - ide
complete charge shoudl = 0
Nomen old 2 - metals w/ more than 1 OX state
metals with more than 1 oxidation state
write name and then in () roman numeral of ox state given
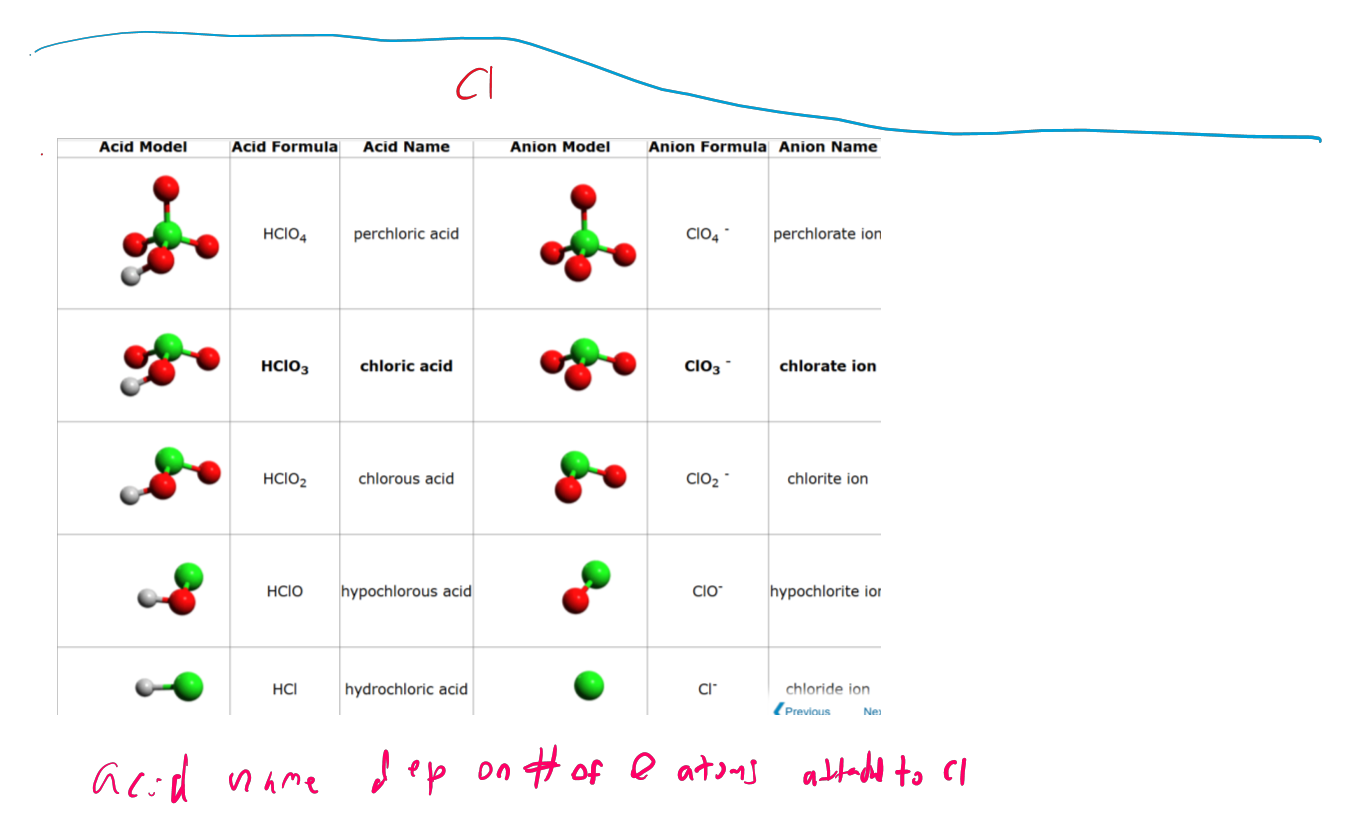
Nonmen 2 - acids and anions - Cl
acid name dep on # of atoms attached to Cl
parent - chloric acid
1 or more - perchloric acid
1 less is chlorous acid
2 less - hypo chloric acid
no O - hydrochloric
End depends of ending of acid
anions ate → ic acid
anions ite → ous acid
acid from only 2 e- (NO O) - anion
Nomen 3 oxianions
ite → ate by removing 1 O
Per - 1 more O than ate
hypo 1 less O than ite
polyatomic ions
ate - more O
Ite - less o
Per - ate is most O
hypo -ite is least O
nomen binary covalent
Nonmetals can bond with other nonmetals
*binary covalent comps always end with -ide
mono - neber used for 1st e-
di - 2
tri - 3
tetra - 4
penta - 5
hexa - 6
hepta - 7
octa - 8
nona 9
deca 10