Exam 1 Material
1/150
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
151 Terms
Molecule
A group of two or more atoms chemically bonded together that act as a single unit.
Molecular Weight
The total mass of a molecule calculated by adding the atomic masses of all atoms in the molecule.
Element
A pure substance made of only one type of atom.
Atom
The smallest unit of an element that retains the chemical properties of that element.
Covalent Bond
A chemical bond formed when two atoms share one or more pairs of electrons.
Ionic Bond
A chemical bond formed when one atom transfers electrons to another, creating oppositely charged ions that attract each other.
Hydrogen Bond
A hydrogen bond is a weak electrostatic interaction between a partially positive hydrogen atom (attached to an electronegative atom) and a nearby electronegative atom.
Van der Waals Force/Interactions
Weak attractions between molecules caused by temporary shifts in electron distribution.
Electronegativity
The ability of an atom to attract electrons toward itself in a chemical bond.
Electropositive Atom
an atom that tends to lose electrons and therefore often develops a partial positive charge (δ⁺) or a full positive charge (cation).
Electronegative Atom
an atom that strongly attracts electrons, often developing a partial negative charge (δ⁻).
Polar Covalent Bond
A covalent bond where electrons are shared unequally because of differences in electronegativity.
Nonpolar Covalent Bond
A covalent bond where electrons are shared equally between atoms.
Hydrophilic Interactions
Interactions between water and substances that are attracted to water, usually polar or charged molecules.
Hydrophobic Interactions
Interactions where nonpolar substances avoid water and tend to group together.
Macromolecule
A large, complex molecule made up of smaller subunits; includes proteins, nucleic acids, and carbohydrates.
Polymer
A large molecule composed of repeating structural units (monomers) connected by covalent bonds.
Monomer / Subunit
The basic building block of a polymer; a small molecule that can join with others to form a polymer.
Polysaccharide
A polymer made of many monosaccharide (sugar) units linked by glycosidic bonds.
Membrane lipids
Lipids that form the structural components of cell membranes, primarily phospholipids and cholesterol.
Proteins
Polymers of amino acids that perform various functions including catalysis, structure, and signaling.
Nucleic acids
Polymers of nucleotides that store and transmit genetic information (DNA and RNA).
Monosaccharide
A single sugar molecule, the simplest form of carbohydrate (e.g., glucose).
Amino Acid
The monomer of proteins; consists of a central carbon bonded to an amino group, carboxyl group, hydrogen, and R group.
Nucleotide
The monomer of nucleic acids; composed of a sugar, phosphate group, and nitrogenous base.
Condensation reaction
Two molecules join together, endergonic (energetically unfavorable), non-spontaneous, consumes energy, water produced
Hydrolysis reaction
Molecule is broken up, exergonic (energetically favorable), releases energy, spontaneous, requires input of water
Energetically favorable
A reaction that releases free energy and can occur spontaneously.
Energetically unfavorable
A reaction that requires an input of energy to proceed.
Directional / Structural Polarity
Molecules like nucleic acids and proteins have a direction (e.g., 5' to 3' or N- to C-terminus) due to the way subunits are added.
Amphipathic
A molecule that has both hydrophilic (water-loving) and hydrophobic (water-fearing) regions.
Glycosidic bond
A covalent bond that joins two sugar molecules.
Peptide bond
A covalent bond formed between the amino group of one amino acid and the carboxyl group of another.
Phosphodiester bond
A covalent bond that links the phosphate group of one nucleotide to the sugar of another in nucleic acids.
Reaction coupling
The use of an energetically favorable reaction to drive an energetically unfavorable one.
Activated carrier
A molecule that stores energy or chemical groups in a form that can be donated to other reactions (e.g., ATP).
Metabolism
All chemical reactions that occur in a cell, including catabolic and anabolic pathways.
Free energy
The portion of a system’s energy that is available to do work (Gibbs free energy).
Anabolic
Complex molecules being made from simple molecules. Endergonic, consumes free energy (positive G), condensation reactions
Catabolic
Complex molecules breaking down into simple molecules. Exergonic, releases free energy (negative G), hydrolysis
Endergonic
A reaction that absorbs free energy from its surroundings (non-spontaneous).
Exergonic
A reaction that releases free energy (spontaneous).
Activation Energy
The energy required to start a chemical reaction.
Enzyme
A protein that acts as a catalyst (without being consumed!) by lowering activation energy of a reaction
Active Site
The region of an enzyme where substrate molecules bind and undergo a chemical reaction.
Substrate
The specific reactant that an enzyme acts upon.
Activated carrier
A molecule that stores energy or chemical groups in a form that can be donated to other reactions (e.g., ATP).
Intramolecular
Interactions that occur within a single molecule.
Intermolecular
Interactions that occur between different molecules.
Disulfide bond
A covalent bond formed between two cysteine residues, stabilizing protein structure.
Primary level of protein structure
The linear sequence of amino acids in a polypeptide chain.
Secondary level of protein structure
Local folding patterns such as alpha helices and beta-pleated sheets, stabilized by hydrogen bonds in the peptide backbone.
Tertiary level of protein structure
The overall 3D shape of a polypeptide, formed largely by interactions between R groups.
Quaternary level of protein structure
The arrangement of multiple polypeptide subunits in a protein supported largely by interactions between R groups.
Alpha helix
A coiled region stabilized by hydrogen bonds between every fourth amino acid.
Beta-pleated sheet
A sheet-like structure formed by hydrogen bonding between segments of a polypeptide chain.
Protein domain
An independently folding and distinct functional and structural unit within a protein.
Protein subunit
A single polypeptide chain that assembles with others to form a functional protein complex.
Chaperone
Proteins that assist in the proper folding of other proteins.
Proteasome
A protein complex that degrades misfolded or damaged proteins tagged with ubiquitin.
Protein conformation
The specific three-dimensional shape of a protein that determines its function.
Monomer: Sugars
Polymers: Polysaccharides, glycogen, and starch (in plants)
Monomer: Fatty acids
Polymers: Fats and membrane lipids
Monomer: Amino Acids
Polymer: Proteins
Monomer: Nucleotides
Polymer: Nucleic acids
Carbohydrates
Formula: (C1H2O1)n. n= 3,4,5,6
Source of chemical energy for cells through covalent bonds. Can also be structural
Ring structure in aqueous environment
Polymerization of Sugars
Monosaccharides combine through condensation. The bond is called a glycosidic bond. Reaction is reversible (with hydrolysis)
Polypeptides
Subunit: amino acids. Consist of central carbon, amino group, carboxyl group, single H, and R group (functional group)
Protein function
Enzymes, structural, transportation, etc.
Polymerization of proteins
Amino acids bond through condensation reactions. Resulting bond is called “peptide bond”, connecting O=C-N.
Polypeptides are read in this direction:
N (amino) terminus → C (carboxyl) terminus
What allows a protein to fold?
The covalent bonds surrounding the central carbon. They’re flexible and allow the polypeptide chain to twist.
Components of nucleic acids
Subunits: nucleotides
5 carbon sugar, phosphate group, N-containing base
Functions of nucleic acids
Carry genetic information through DNA and RNA
also in rRNA, siRNA, etc.
Polymerization of nucleic acids
Bond through condensation reactions, forming phosphodiester bonds (O-P-O). Phosphate group bonds with the O on Carbon 3’.
Directionality of nucleic acid
From C5’-PO4 to C3’-OH
DNA
Nitrogenous base: C, G, A, T
Sugar: deoxyribose (C2’ has H only)
RNA
Nitrogenous base: C, G, A, U
Sugar: Ribose (C’2 has OH)
Fatty Acids/Lipids
Doesn’t have traditional “subunits,” doesn’t form traditional polymers through condensation
Two regions: Hydrophobic tail (pure hydrocarbons), Hydrophilic head (carboxylic)
Fatty acids function
Phospholipids make up membranes bc they’re amphipathic. They have two fatty acid tails that point inwards, away from water. Glycerol is on the head, points outwards bc its hydrophilic. Creates a two-layer membrane
Saturated fats
Do not possess a double bond
Unsaturated fats
Possess a double bond (kink)
Directional polarity in proteins
Proteins are read and synthesized from their N-terminus to their C-terminus.
How are anabolic and catabolic reactions linked?
Energy released in catabolic reactions drive anabolic reactions
Energy in endergonic reactions
Reactants have less energy than products, not spontaneous, and energy is absorbed, NEEDS outside energy to push it to completion
Energy in exergonic reactions
Reactants have more energy than products, spontaneous, and energy is released, this is the reaction that fuels other, not spontaneous reactions with its excess energy
How does an enzyme affect the transition state?
It stabilizes its structure
What determines an enzyme’s function?
The shape and structure. Substrate must be able to fit its active site.
What enzymes break down carbohydrates?
Sucrase, lactase, maltase
What enzymes break down proteins?
trypsin, pepsin, proteases
What enzyme breaks down lipids?
lipases
What enzyme breaks down nucleotides?
nucleases
What is often used as an energy source for reaction coupling?
Adenosine triphosphate (ATP)
How does ATP carry energy?
It possesses 3 negative phosphate groups that CONSTANTLY repel each other (building energy in their bonds). Once that bond is broken (P-O), you get an explosion of energy released.
How do cells use energy released from ATP?
They use it to drive endergonic reactions.
How do cells use exergonic reactions in regards to ATP?
They use to form ATP from ADP + P (“recharging” the battery)
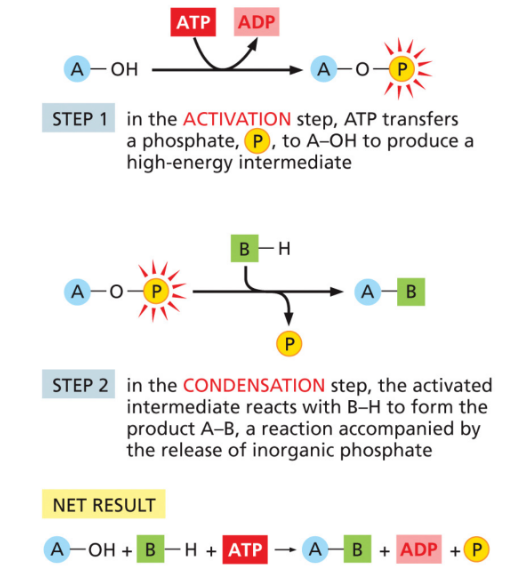
Reaction coupling with ATP
Since condensation reactions are energetically unfavorable, we must couple them with ATP hydrolysis.
Activation. ATP exchanges a phosphate for the H of an -OH, creating a high energy intermediate A-O-P and leaving ADP
Condensation. Intermediate reacts with B-H to form A-B. Releases the phosphate and a molecule of water. 1 H comes from A-OH and one from B-H, and O from A-O-P
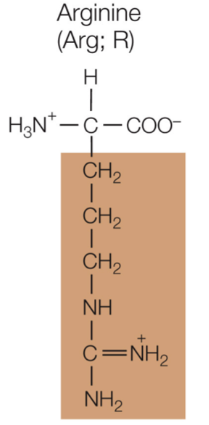
Arginine
Positively charged, hydrophilic
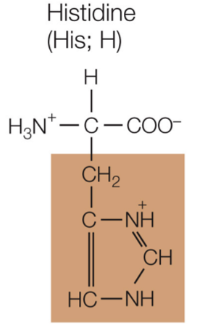
Histidine
Positively charged, hydrophilic
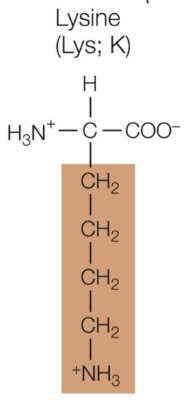
Lysine
Positively charged, hydrophilic