CH. 20 | Antimicrobial Drugs
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
20 - 1 Identify the contributions of Paul Ehrlich and Alexander Fleming to chemotherapy.
Paul Ehrlich
Coined the term chemotherapy
Developed the concept of selective toxicity (magic bullet; target and destroy pathogens without harming the host)
Laid the foundation for all antimicrobial drug development
Alexandar Fleming
In 1928, he observed that the mold P. chrysogenum was inhibiting the growth of S. aureus on a contaminated petri plate
Led to the isolation and identification of penicillin (the first true antibiotic)
Observations led to the concept of antibiosis (the inhibitory effect that microorganisms can have on one another)
Antibiosis / Antibiotic
An antimicrobial agent produced by microorganisms that inhibit the growth of, or kill bacteria
→ Penicillin is a classic example of antibiosis since the antibiotic was produced by a fungus mold, none other than the Penicillium / Penicillium chrysogenum, which acts as an antibiotic on other microorganisms
20 - 2 Name the microbes that produce most antibiotics.
Streptomyces bacteria
Provides a broad spectrum of antibiotics
Found in soil
Gram +
20 - 3 Describe the problems of chemotherapy for viral, fungal, protozoan, and helminthic infections.
Main Idea: These pathogens are either eukaryotic (like human cells) or viruses (hide within human cells), making selective toxicity very difficult
Chemotherapy: The use of chemical agents (antibiotics, antivirals, antifungals, etc) to treat infectious diseases by inhibiting the growth of, or killing microorganisms without harming the host (selective toxicity)
Viral Infections
Viruses are obligate intracellular pathogens — meaning that they must be inside a host cell to replicate
Viral genetic material hijacks the host cell to produce more viruses — meaning you’d have to disrupt normal human cell processes to stop it
Few antiviral drugs compared to antibiotics — the selective toxicity problem is much harder to solve when the pathogen is essentially "hiding inside you.”
Antibiotics do not affect viruses, by the way
Fungal, Protozoan & Helminthic Infections
All three are eukaryotic, so they closely resemble human cells at the cellular level
A drug that kills the pathogen will most likely damage host tissue as well
Arsenal against these is far more limited than antibacterial drugs
Superinfection Risk (Compounding problem)
Using broad-spectrum drugs to fight these can wipe out normal microbiota
This allows resistant or insensitive organisms (Candida albicans) to flourish as opportunistic pathogens
20 - 4 Define the following terms: spectrum of activity, broad-spectrum antibiotic, superinfection.
Spectrum of Microbial Activity
The range of different microbial types that a drug is effective against
Targets few types
Penicillin G has a narrow spectrum — it affects gram + bacteria, but very few gram - bacteria
Broad-Spectrum Antibiotic
An antibiotic that is effective against a wide range of both gram + and gram - pathogenic bacteria
Advantage: Useful when the pathogen identity isn’t immediately known, since you can just use the antibiotic right away (saves time)
Disadvantage: Destroys normal microbiota, which can lead to a superinfection
Superinfection
The growth of a pathogen that has developed resistance to an antimicrobial drug being used; the growth of an opportunistic pathogen
Loss of microbiota can lead to an overgrowth of other microbes
Occurs in 2 ways:
→ When broad-spectrum drugs wipe out normal microbiota, allowing insensitive organisms to overgrow as opportunistic pathogens
→ When a target pathogen develops resistance to the antibiotic, allowing the resistant strain to replace the original sensitive strain and continue the infection
Overview for next L.O
Antimicrobial drugs are either bactericidal (they kill bacteria directly), or bacteriostatic (inhibit/prevent the growth of bacteria)
Mode of Action
How a drug works — specifically, what it targets and how it kills or inhibits the pathogen
Understanding the mode of action helps explain selective toxicity — if a drug targets something unique to the pathogen (like a bacterial cell wall that human cells don't have), it can kill the pathogen without harming the host
20 - 5 Identify 5 modes of action of antimicrobial drugs.
1) Inhibiting Cell Wall Synthesis
Penicillin can prevent the synthesis of peptidoglycan (found in bacteria only), resulting in the cell wall being weakened and undergoing lysis
Only actively growing cells are affected
Primarily disrupts and targets the synthesis of the bacterial cell wall by binding to penicillin-binding proteins (PBP) to inhibit peptidoglycan cross-linking
Little toxicity for host cells since humans don’t have peptidoglycan cell walls
2) Inhibiting Protein Synthesis
Eukaryotic cells — 80S Ribosomes
Prokaryotic cells — 70S Ribosomes
→ The difference in ribosomal structure accounts for the selective toxicity of antibiotics that affect protein synthesis
However, the problem here is that mitochondria also contain 70S ribosomes. So, antibiotics targeting 70S ribosomes can accidentally affect mitochondria, causing side effects in the host
A few examples of antibiotics that use this is chloramphenicol, erythromycin, streptomycin, and tetracyclines
Bacteriostatic
3) Injuring the Plasma Membrane
Bacteria:
Polypeptide antibiotics change the permeability of the plasma membrane, → causes the cell to lose metabolites it needs to survive
Some can disrupt both the inner and outer membranes of gram-negative bacteria
Ionophores (a special type):
Allow uncontrolled movement of cations (charged ions) across the membrane — disrupting normal cell function
However, they are not used in human medicine
Instead, they are used in cattle feed to alter gut microbiota, improving digestion and promoting growth
Fungi:
Antifungal drugs target sterols in the fungal plasma membrane
By binding to these sterols, the drug disrupts the membrane
4) Inhibiting Nucleic Acid Synthesis
Antibiotics interfere with the DNA replication and transcription of microorganisms
Blocks bacterial topoisomerase or RNA polymerase
5) Inhibiting the Synthesis of Essential Metabolites
Competitive Inhibition
→ Antimetabolite closely resembles the normal substrate for the enzyme
→ Enzyme gets “fooled”, and binds to the drug instead of the real substrate
→ Blocks the enzyme from doing its job
![<p><span style="color: yellow;">1) Inhibiting Cell Wall Synthesis</span></p><ul><li><p>Penicillin can prevent the synthesis of peptidoglycan (found in bacteria only), resulting in the cell wall being weakened and undergoing lysis</p><ul><li><p>Only actively growing cells are affected</p></li></ul></li><li><p><mark data-color="#a7a992" style="background-color: rgb(167, 169, 146); color: inherit;">Primarily disrupts and targets the synthesis of the bacterial cell wall</mark> by binding to penicillin-binding proteins (PBP) to inhibit peptidoglycan cross-linking</p></li><li><p>Little toxicity for host cells since humans don’t have peptidoglycan cell walls<br><br></p></li></ul><p><span style="color: yellow;">2) Inhibiting Protein Synthesis</span></p><ul><li><p>Eukaryotic cells — 80S Ribosomes</p></li><li><p>Prokaryotic cells — 70S Ribosomes<br>→ The difference in ribosomal structure accounts for the selective toxicity of antibiotics that affect protein synthesis</p></li></ul><p></p><ul><li><p>However, the problem here is that mitochondria also contain 70S ribosomes. So, antibiotics targeting 70S ribosomes can accidentally affect mitochondria, causing side effects in the host</p></li><li><p>A few examples of antibiotics that use this is chloramphenicol, erythromycin, streptomycin, and tetracyclines</p></li><li><p>Bacteriostatic<br><br></p></li></ul><p><span style="color: yellow;">3) Injuring the Plasma Membrane</span></p><p><strong>Bacteria:</strong></p><ul><li><p>Polypeptide antibiotics change the <em>permeability</em> of the plasma membrane, → causes the cell to lose metabolites it needs to survive</p></li><li><p>Some can disrupt <strong>both</strong> the inner and outer membranes of gram-negative bacteria</p></li></ul><div data-type="horizontalRule"><hr></div><p class="font-claude-response-body break-words whitespace-normal leading-[1.7]"><strong>Ionophores</strong> (a special type):</p><ul><li><p>Allow <strong>uncontrolled movement of cations</strong> (charged ions) across the membrane — disrupting normal cell function</p></li><li><p>However, they are not used in human medicine</p></li><li><p>Instead, they are used in cattle feed to alter gut microbiota, improving digestion and promoting growth</p></li></ul><div data-type="horizontalRule"><hr></div><p class="font-claude-response-body break-words whitespace-normal leading-[1.7]"><strong>Fungi:</strong></p><ul><li><p>Antifungal drugs target <strong>sterols</strong> in the fungal plasma membrane</p></li><li><p>By binding to these sterols, the drug disrupts the membrane<br><br></p></li></ul><p><span style="color: yellow;">4) Inhibiting Nucleic Acid Synthesis</span></p><ul><li><p>Antibiotics interfere with the DNA replication and transcription of microorganisms</p></li><li><p>Blocks bacterial topoisomerase or RNA polymerase<br><br></p></li></ul><p><span style="color: yellow;">5) Inhibiting the Synthesis of Essential Metabolites</span></p><ul><li><p>Competitive Inhibition<br>→ Antimetabolite closely resembles the normal substrate for the enzyme<br>→ Enzyme gets “fooled”, and binds to the drug instead of the real substrate<br>→ Blocks the enzyme from doing its job</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/58cbaec3-9109-4a7f-aa76-439356ef86a1.png)
20 - 6 Explain why the antibacterial drugs described in this section are bacteria-specific.
Antibacterial drugs are not harmful to human cells, and are bacteria-specific because of selective toxicity. In other words, they exploit on the structural and metabolic differences unique to bacteria, that are generally absent or lacking in eukaryotic human cells
20 - 7 List the advantages of each of the following over penicillin: semisynthetic penicillins, cephalosporins, and vancomycin.
Penicillin
A group of antibiotics produced by Penicillium
Common structure containing B- lactam ring
Natural Penicillins Disadvantage:
Narrow spectrum of activity
Susceptibility to penicillinases
→ Penicillinases are enzymes produced by bacteria (Staphylococcus species, to be exact) that split and destroy the B- lactam ring of the penicillin molecule
Semisynthetic Penicillins
Penicillinase-resistant (e.g., oxacillin): Natural penicillins are vulnerable to penicillinase enzymes that bacteria produce to destroy them — semisynthetic versions were engineered to resist this destruction
Broad-spectrum (e.g., ampicillin, amoxicillin): Natural penicillin G has a narrow spectrum — these semisynthetic versions work against both gram-positive AND gram-negative bacteria, making them more versatile
Cephalosporins
Inhibits cell wall synthesis in the same way as penicillin does
Differ slightly in their B- lactam ring structure, which makes them more widely used than any other B- lactum antibiotic
Vancomycin
Works by inhibiting cell wall synthesis through a different mechanism than penicillin, bypassing existing resistance
Considered as a “last resort” for antibiotic defense for the treatment of S. aureus infections that are resistant to other antibiotics
However, its widespread use to treat MRSA has led to the appearance of vancomycin-resistant enterococci (VRE)
→ Gram + pathogens
20 - 12 Describe how sulfa drugs inhibit microbial growth.
Sulfa Drugs
Antimicrobial bacteriostatic agent that targets pathogens
Folic Acid Production
Bacteria need folic acid as a coenzyme to synthesize proteins, DNA, and RNA — it's an essential nutrient needed for their survival
Bacteria must make their own folic acid using PABA as a starting material
How Sulfa Drugs Work:
Sulfa drugs are structurally similar to Para-Aminobenzoic Acid (PABA)
Since they look so alike, the sulfa drugs competitively bind to the enzyme that normally processes PABA
As a result, folic acid production gets blocked, bacteria wouldn’t be able to make proteins, DNA, or RNA, and growth is inhibited
20 - 16 Explain 2 tests for microbial susceptibility to chemotherapeutic agents.
1) Disk-Diffusion Method (Kirby-Bauer Test)
An agar-diffusion test to determine microbial susceptibility to chemotherapeutic agents
Measures the diameter of the zone of inhibition surrounding the antimicrobial-soaked disk to determine the susceptibility of bacteria to drugs
How it works:A filter paper disk is soaked with the chemical agent, which is then placed on an agar plate that has been inoculated with the test organism. Afterwards, the plate gets incubated, and the results are up for discussion
To interpret results:
→ Clear zone of inhibition around disk = Chemical is effective, meaning that bacteria cannot grow in the zone
→ No clear zone = Chemical is ineffective, meaning that bacteria can grow
2) Broth Dilution Test
Useful in determining broth, the minimal inhibitory concentration (MIC), and the minimal bactericidal concentration (MBC) of the antimicrobial drug
How it works:Decreasing concentrations of the drug are prepared in a series of broth wells
The test bacteria are added to all wells simultaneously
After incubation, wells are checked for visible growth
This determines the MIC (Minimal Inhibitory Concentration) — the lowest concentration that prevents visible growth
Wells showing no growth are then subcultured into drug-free broth to determine the MBC (Minimal Bactericidal Concentration) — lowest concentration of an antibiotic that kills a certain percentage of bacteria (typically 99.9%)
20 - 17 Describe the mechanisms of drug resistance.
Vertical Transmission: A resistant bacterium reproduces and passes the mutation to all its "offspring."
Horizontal Transmission: Bacteria "swap" resistance genes (often carried on plasmids) with each other through processes like conjugation or transduction.
Mechanism of Antibiotic Resistance
Blocking the drug’s entry into the cell
→ The bacteria change their outer “shell” or entry points to keep the drug from getting inside the cell
→ Gram-negative bacteria are naturally resistant to this because their cell wall restricts absorption
→ Mechanism: Bacteria can modify porins (the openings in their cell walls) so the antibiotic cannot enter the periplasmic spaceInactivation of the drug by enzymes
→ Bacteria produce enzymes that physically break down or neutralize the antibiotic before it can do its job
→ Some bacteria produce B-lactamase, an enzyme that destroys the structure of penicillin and cephalosporins
→ Because of this, it led to the development of resistant strains like MRSA (Methicillin-Resistant Staphylococcus aureus)Alteration of the drug’s target site
→ Antibiotics usually work by binding to a specific part of the bacteria (i.e., protein, ribosome). Bacteria can change the shape of that “target” so the drug no longer fits
→ A minor modification to a ribosome can neutralize an antibiotic like tetracycline without stopping the cell from making proteins
→ MRSA Strategy: It changed its Penicillin-Binding Protein (PBP) so that methicillin could no longer bind to itEfflux of the drug from the cell
→ Bacteria use specialized “pumps” in their membranes to kick the antibiotic out of the cell as soon as it enters
→ This prevents the drug from reaching a high enough concentration to kill the bacteria
→ Many bacteria use these pumps to eliminate various toxic substances, including major classes of antibiotics
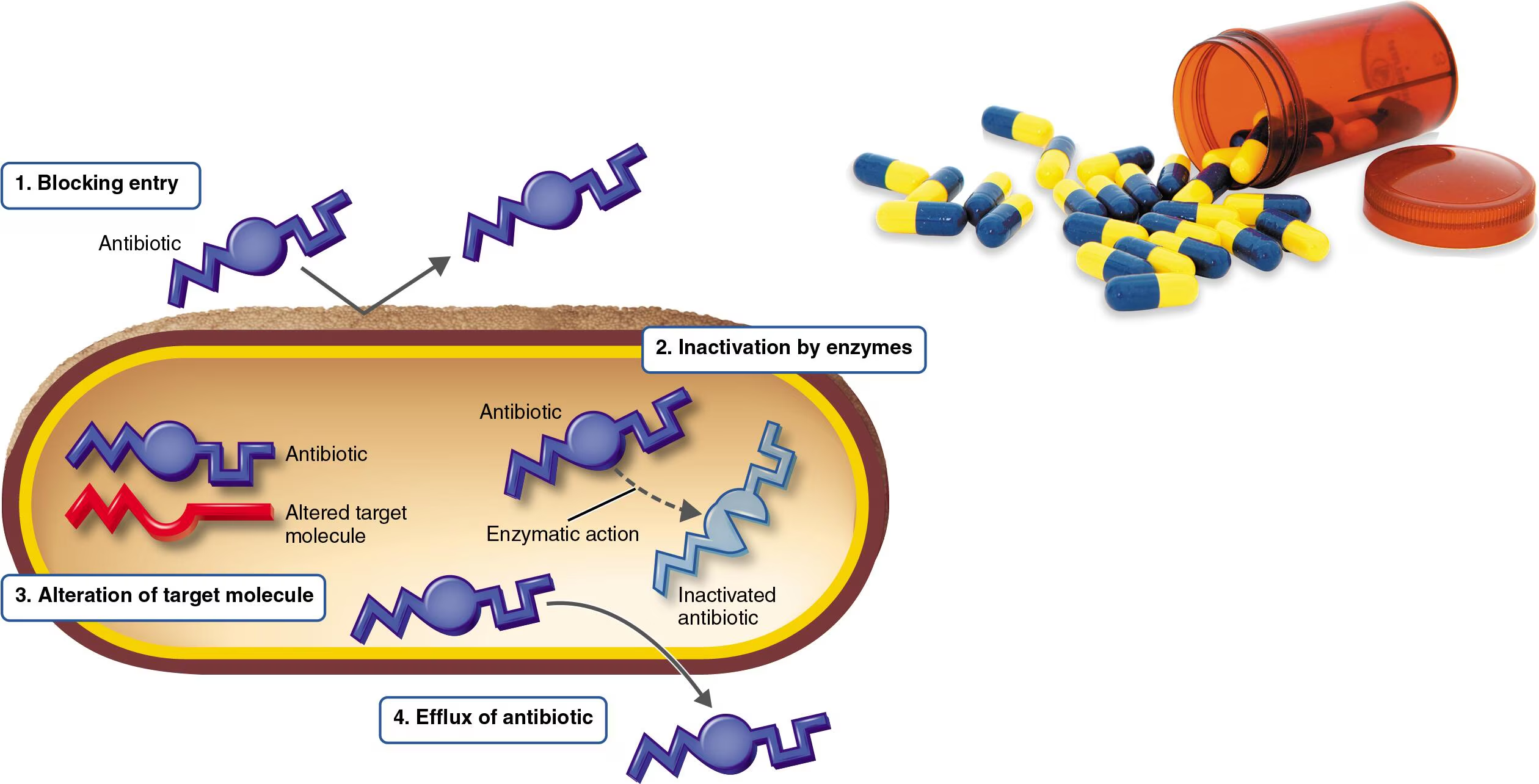
20 - 18 Compare and contrast synergism and antagonism.
Synergism
The effectiveness of 2 drugs used simultaneously is greater than that of either drug used alone
The drugs essentially enhance each other's effectiveness
Also beneficial when we are unaware with the organism that is causing the infection
Can also prevent antibiotic resistance
Reduce toxicity by using lower doses of each drug
→ Penicillin + Streptomycin for bacterial endocarditisPenicillin damages the cell wall, making it easier for streptomycin to enter the bacteria
Together, they work better than separately
Antagonism
When 2 drugs are less effective than either one alone
Competition among microbes
One drug actually interferes with the other's ability to work
→ Penicillin + TetracyclineTetracycline is bacteriostatic — it stops bacterial growth
Penicillin needs bacteria to be actively growing to work
So tetracycline accidentally undermines penicillin's effectiveness
20 - 19 Name 3 areas of research on new chemotherapeutic agents.
1) Targeting Virulence Factors
Instead of targeting the microbe itself, new research focuses on targeting what makes it dangerous
Examples: neutralizing bacterial toxins (like cholera toxin) or sequestering iron that pathogens need to grow
This is a fresh approach since existing antibiotics have relied on a limited range of targets and keep running into resistance problems
2) Antimicrobial Peptides
Many organisms naturally produce antimicrobial peptides — birds, amphibians, plants, and mammals all make them as part of their defense systems
Magainins (from amphibian skin) attack bacterial membranes and have existed for an indefinite time without significant resistance developing — making them very promising
Bacteriocins (produced by bacteria themselves) show broad or narrow spectrum activity and have shown promising early results against C. difficile
3) Phage Therapy
Uses bacteriophages — viruses that naturally kill bacteria — as an alternative to antibiotics
Particularly promising for treating antibiotic-resistant infections since phages are highly specific to their host bacteria
Already used for over 50 years in Russia, Georgia, and Poland
Naturally abundant — bacteriophages in soil kill roughly half of Earth's bacteria every few days