Dual Nature of Light
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
List the following
Ancient Greek had differing thoughts on the nature and behavoir of light. Some thought that light is
wave or disturbance that travels through spaces in the air
a substance carrying particles that flow with measurable velocity from a light source.
a stream of rays that comes that great vel. from eyes of an observer.
The reuslt of rays that leave the eyes reflect off an object, an interact with sunlight to becomes visible.
Newton’s Prism Experiment
In the 1600s, Sir Isaac Newtons performed an experiment that seperated white light into colors
He thought of light as a stream of tiny particles discharged by luminous objects that travel in straight paths.
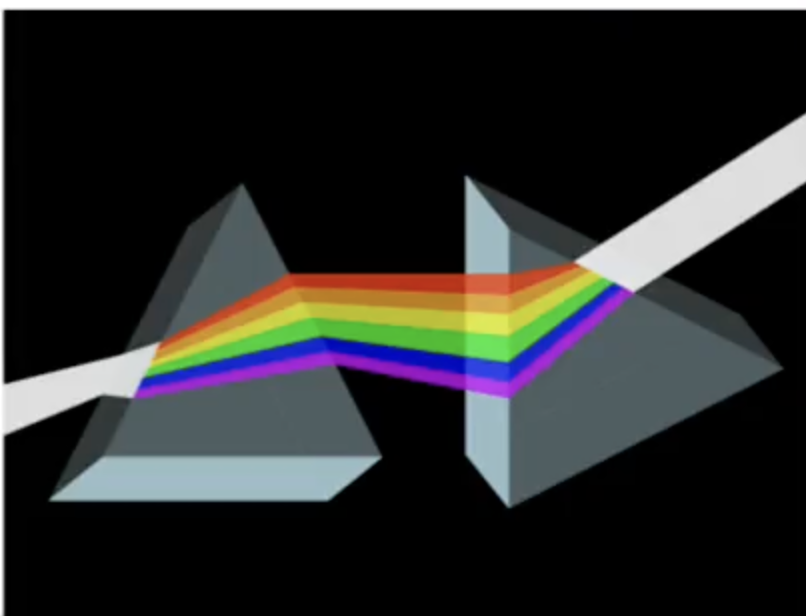
Which ancient idea of the nature of light did Isaac Newton build upon?
Light is a substance through which particles flow from a light source.
What theory explains relfection and refraction
theory of light.
Reflections
Particles bounce off a surface much like an elastic ball bounces off a wall.
Refractions define it
Particles refract as forces cause light pass through one medium to another.
In 1678, Christiaan Huygens theorized what
theorized that light is made of waves that can bend and spread out.

diffraction
diffracitons can explain by
wave theory.
Interference can be explain by
wave theory.
relfection can be explain by
particle and wave theories.
refraction
both particle and wave theories.
Young’s Double Slit Experiment
In 1803, Thomas Young performed the double slit experiment
Experimental results supported the wave theroy of light
Waves spread through slits, in accordance with Huygen’s princples.
Wave interfered resulting in pattern called the diffraction pattern.
Maxwell’s Electromagnetic Theory
In 1873, James Clerk Maxwell built on the work of other scientists to mathematically explain how electric and magnetic field can induce each other.
He concluded that light is produced by the interaction bertween electric and magnetic fields.
Hertz’s Experiment
In the 1880, Heinrich hertz observed that radio waves have the same properties as light so he conluded that light is made of waves.
Heinrich Hertz
discovered the photoelectric effect, the emissions of electrons from a metal when it is struck by light of certain freq.
Each metal has it own freq. threshold which is the min, freq required to ejected electrons.
The Wave Theory of Light and Photoelectric effect
The results of experiment with the photoelectric effect were different from scientsits predictions which were based on the classical physics explaination of wave behavoir.
these reuslts weakened the wave theroy and stregthened the particle theory.
Expected reuslts; Light of any freq. (color) will cause the ejections of electrons
Freq. of light lower than the freq. threshold of the metal did not eject electrons
Expected Results: There will be a time gap between when the light strikes the metal and when electrons are ejected.
As soon as light struk the metal, electrons were ejected. NO TIME GAP.
Expected results: increasing the intensity of light will cause the kinetic energy of the ejected electrons to increase.
The kinetic energy of the ejected electrons depended only on the freq. of light.
Which result would support the particle theory of light?
Frequencies of light that fall below the frequency threshold of the metal do not eject electrons.
Einstien Phontons
Explained the reuslt of the ______________
photoelectric experiments
Einstin Photon
by suggesting that a _________ of ________ energy with _______caused the ______ of an electrons
quantum, electromagnetic, zero mass
Photons used the Explainations of Results.
Experimental reuslt: Regqueincnes of light were lowers than the freeq. threshold od the metl did no eject electrons
Explainations of reuslt: the energy in eahc photons is determined by its freq. Electrons are ejected only if the nenergy of the photons is at least equal to the threshold freq.
Experimental reuslts: As soon as light struck the metal electrons were rejected
Explainations od reuslts” Atoms absorb phtons as soon as light strikes the metal.
Experimental Results: Kinetic energy of the ejected electrons depends only on the freq. of light
Increasing the intensity increases the number of photons hitting the metal, not energy of eahc phtons. Increasing the intensity reuslts only in the ejections of more electrons.
Which concept explains why frequencies of light that fell below the frequency threshold of the metal did not eject electrons?
Photons of different kinds of light carry different amounts of energy.
The photoelectric effect in everday life.
What uses the phtotelectric effect
digital cameras.
image sensors in cameras absorb photons and release electrons
acculmularted eletrons are responsible for the pixels in an image.
The uncertainty Principle.
In the 1927, the German physicist.
Werners Heisenberg formulated the uncerity principle.
The principle states that the speed or locaitons fo quantum particles cannot be measured simultaneously.
This principle applies
Electrons
Light
Superpositions and Quantum Compeuting
what is the superpositions principle? (in detial).
states that a wave or particle canexist in the same postion at the same time
Quantim computing takes the principles of supoerions and the uncertainty principle to create a new way that info or daya can be handled by computers.
traditional computers input data in 0 and 1
In quantum computing, 0 and 1 can exist superpostions and thus 0 and 1 can exist at the same time.
This allows a computer to calculate or process multiple data all at once.
Which statement best describes how today’s scientific community views light?
Light behaves like both waves and particles.
Planck’s constant
6.6 × 10^-34
what is the units for freq.
hertz (Hz).
energ ypf photons is ________-
directly proportional to the freq.