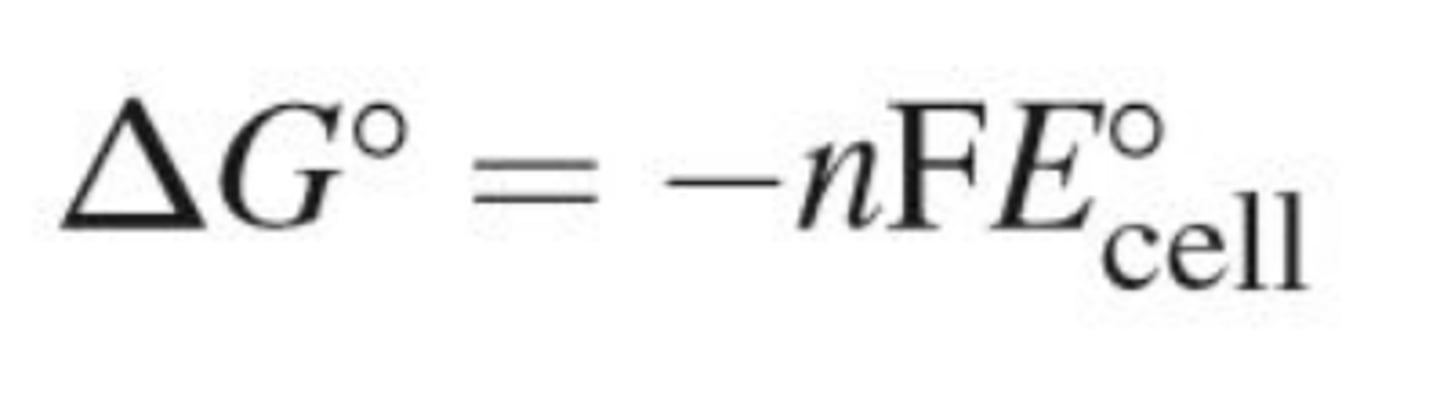AP Chemistry Full Review
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms
Dalton's Law
at constant volume and temperature, the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of the component gases
ionic solid properties
-electrostatic attraction
-high melting point
-hard, brittle
-conductivity: only in aq solution
metallic solid properties
-electrostatic attraction between cations and sea of electrons
-different melting points
-malleable
-conductivity: high
covalent network solid properties
-properties vary depending on the nonmetals involved
-strong bonds
-high melting points
-good conductors of heat
-no conductors of electricity (except graphite)
-insoluble in water
molecular solid properties
-intermolecular forces
-low melting point
-soft
-conductivity: none
strong acids
HCl, HBr, HI, HNO3, H2SO4, HClO4
strong bases
LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
Pauli Exclusion Principle
An atomic orbital may describe at most two electrons, each with opposite spin direction
ionization energy
The amount of energy required to remove an electron from an atom (increases left to right and decreases from up to down)
electron affinity
the energy needed to remove an electron from a negative ion to form a neutral atom or molecule (increases left to right and decreases from up to down)
Electronegativity
the ability of an atom to attract electrons when the atom is in a compound (increases left to right and decreases from up to down)
atomic radius trends
increases from up to down groups. decreases left to right
formal charge formula
[# of valence electrons on atom] - [non-bonded electrons + number of bonds] (ex: formal charge of oxygen in H2O is 6-(4+2)=0)
how do we know which resonance structure is best?
- the one where most atoms have full octet
- the one where there are the fewest formal charges (meaning the more ones with formal charge of 0)
- negative charge on more electronegative atom and/or most positive charge on least electronegative atom
photoelectric effect
The emission of electrons from a metal when light shines on the metal
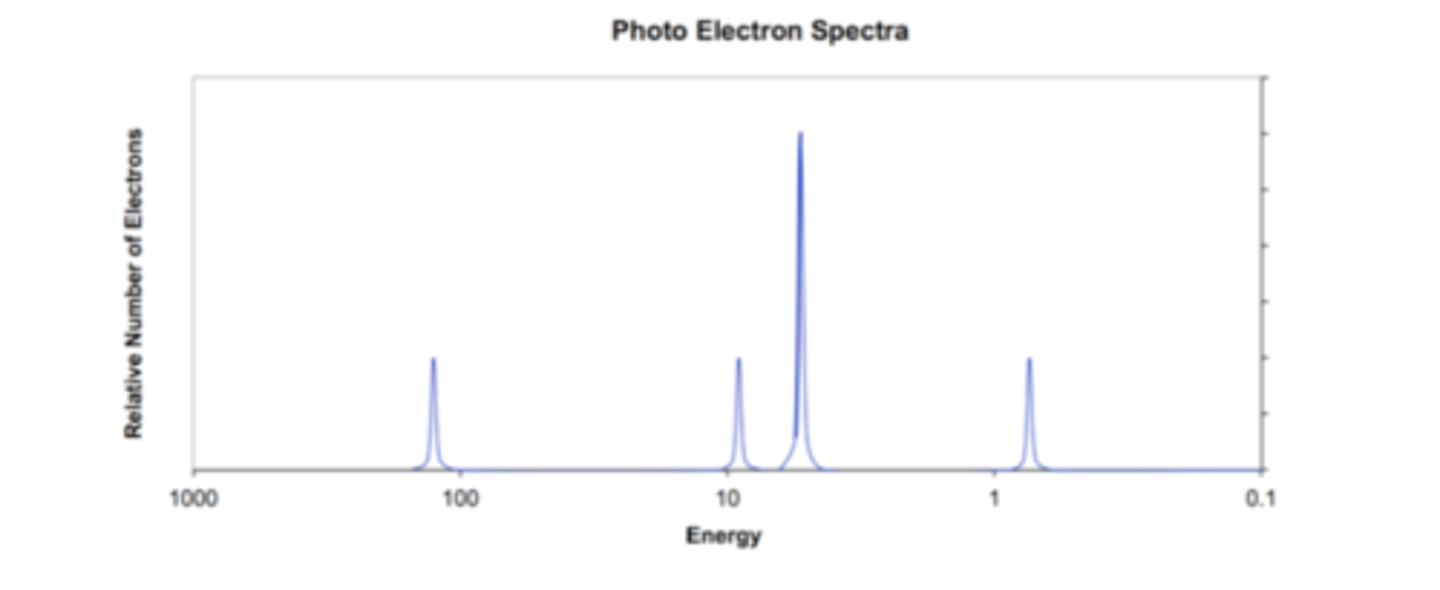
photoelectron spectroscopy (and how it works)
shines light on an electron, snatches electron off atoms. The ones that are easier to snatch move faster than the harder ones. These speeds show us the ionization energy of the electrons and thus show us the electron configuration (s,p,d,f)
Ion and Atom radius/size relationship
The negative ion is much larger than the atom from which it was formed, and positive is smaller
Coulumb's Law
F=kq1q2/r^2: bigger charges and smaller radius means bigger force between atoms (bigger lattice energy)
lattice energy
the energy released when one mole of an ionic crystalline compound is formed from gaseous ions; the greater the lattice energy, the higher the boiling point and melting points
covalent bond
A chemical bond that involves sharing a pair of electrons between atoms in a molecule (ex: O=O)
nonpolar covalent bond
a covalent bond in which the electrons are shared equally by the two atoms; equally electronegative (<0.4)
ionic bond
Formed when one or more electrons are transferred from one atom to another; high electronegativity difference (>1.4)
How to Draw a Lewis Structure
-1.) add the valence electrons for all the atoms in the molecule
-2.) use a pair of electrons to form a bond between atoms
-3.) arrange the remaining electrons to satisfy an octet/duet for each atom (completing outside atoms first)
-4.) molecules try to be symmetrical
-5.) the element that needs the most electrons will be in the center
exceptions to the octet rule
Hydrogen (2), Boron (3-6), Aluminum(sometimes 6, but 3), Phosphorus(10), Sulfur(up to 12).
formal charge
# of valence electrons - ( # dots + # lines)
Resonance structures
structures that occur when it is possible to draw two or more valid electron dot structures that have the same number of electron pairs for a molecule or ion
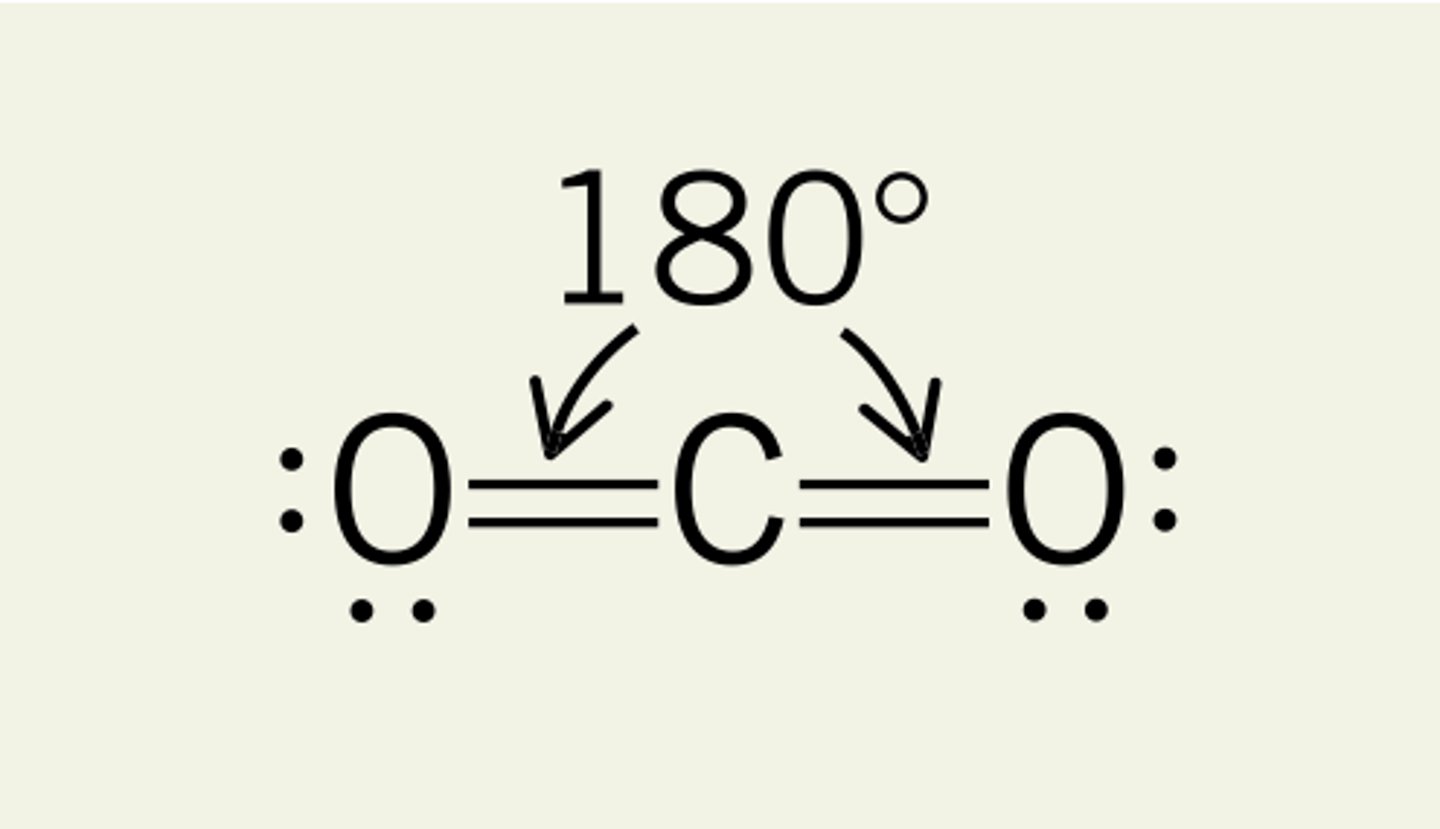
Linear VSEPR
straight line - 2 atoms bonded together, or 2 atoms bonded to the central atom with NO lone pairs, sp
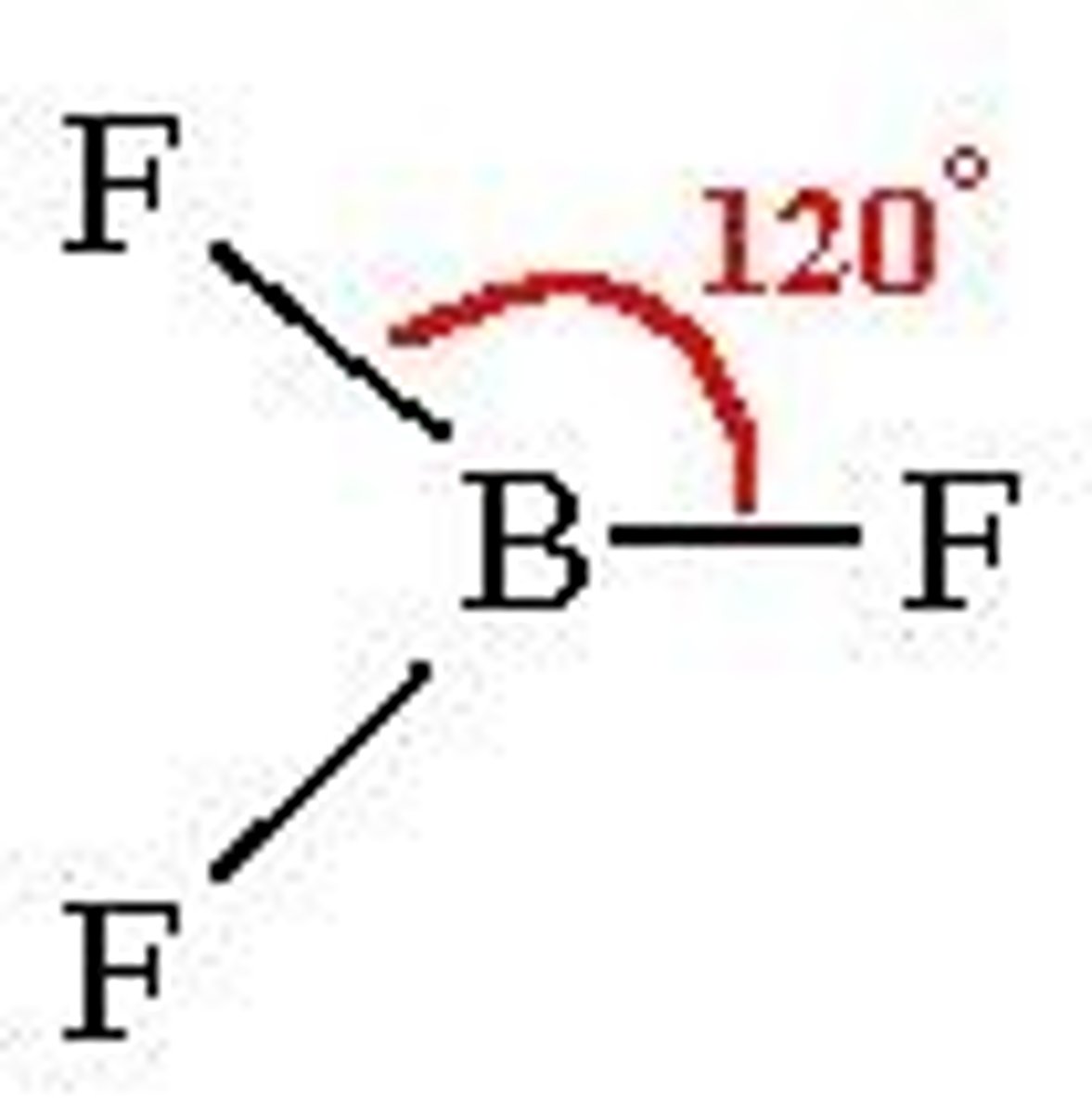
trigonal planar
3 bonds, 0 lone pairs, 120 degrees, sp2
Tetrahedral
4 bonds, 0 lone pairs, 109.5 degrees, sp3
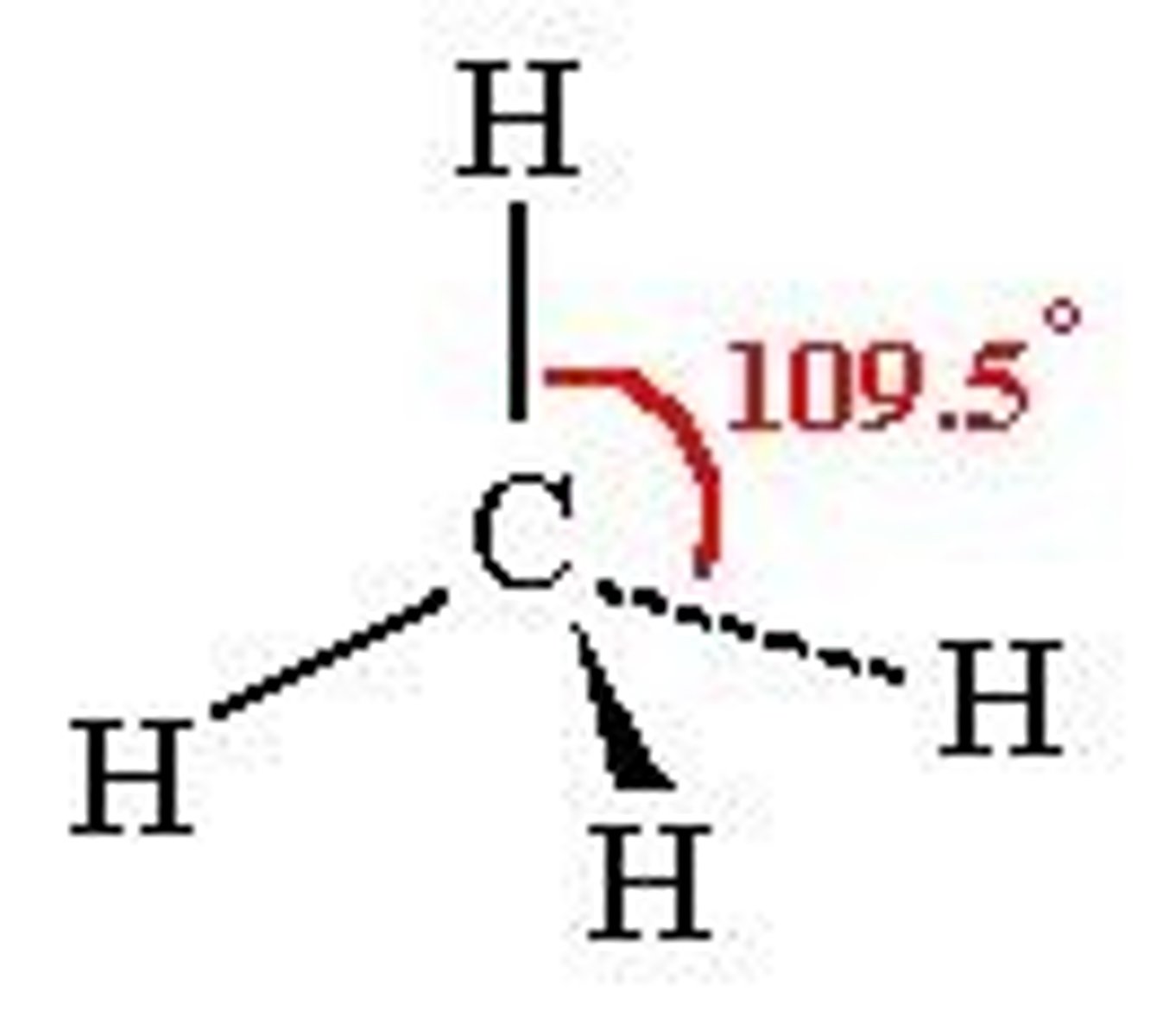
trigonal bipyramidal
5 bonds, 0 lone pairs, 90-120 degrees, sp3d
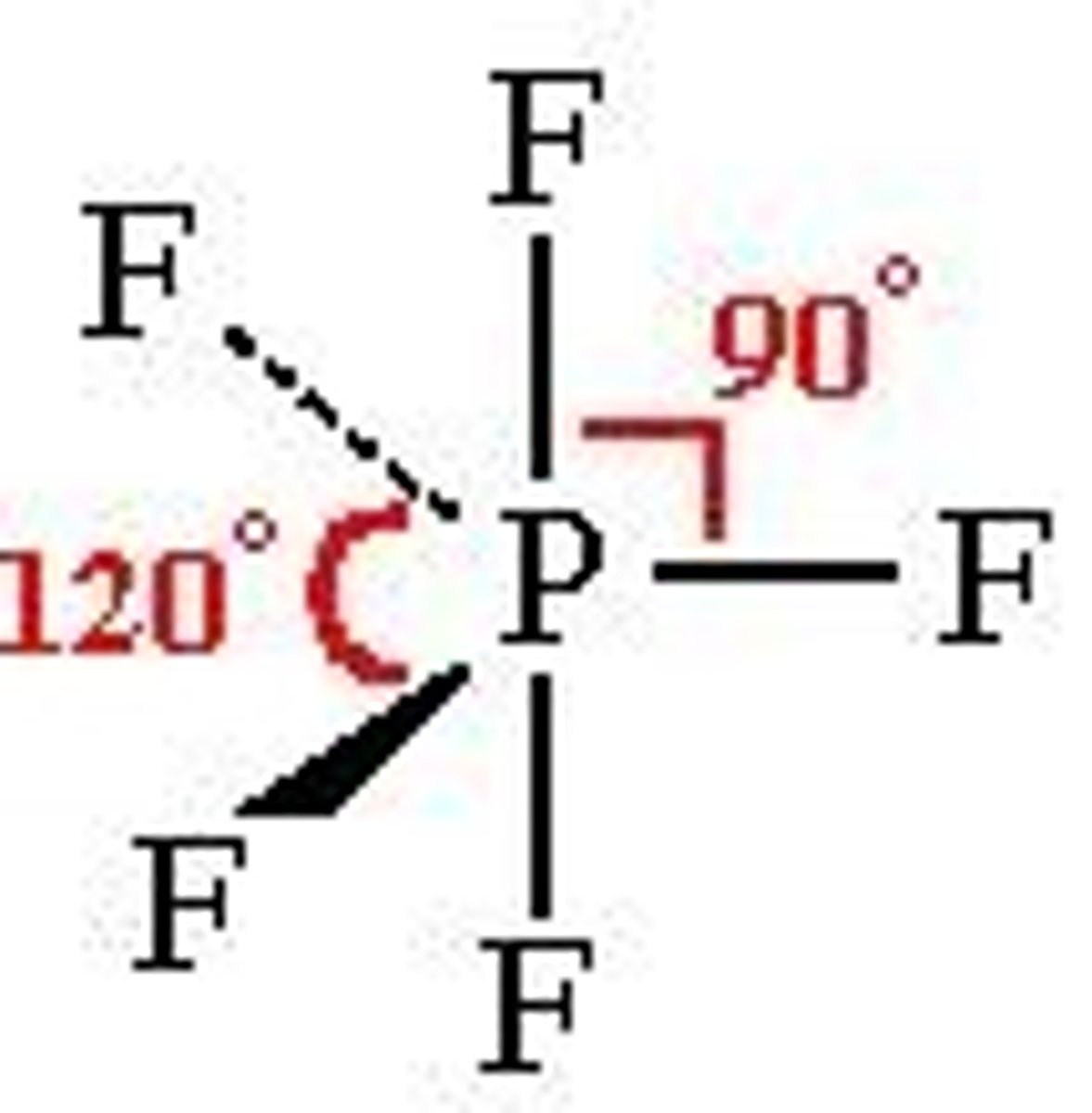
seesaw
4 bonds, 1 lone pair, 90-120, sp3d
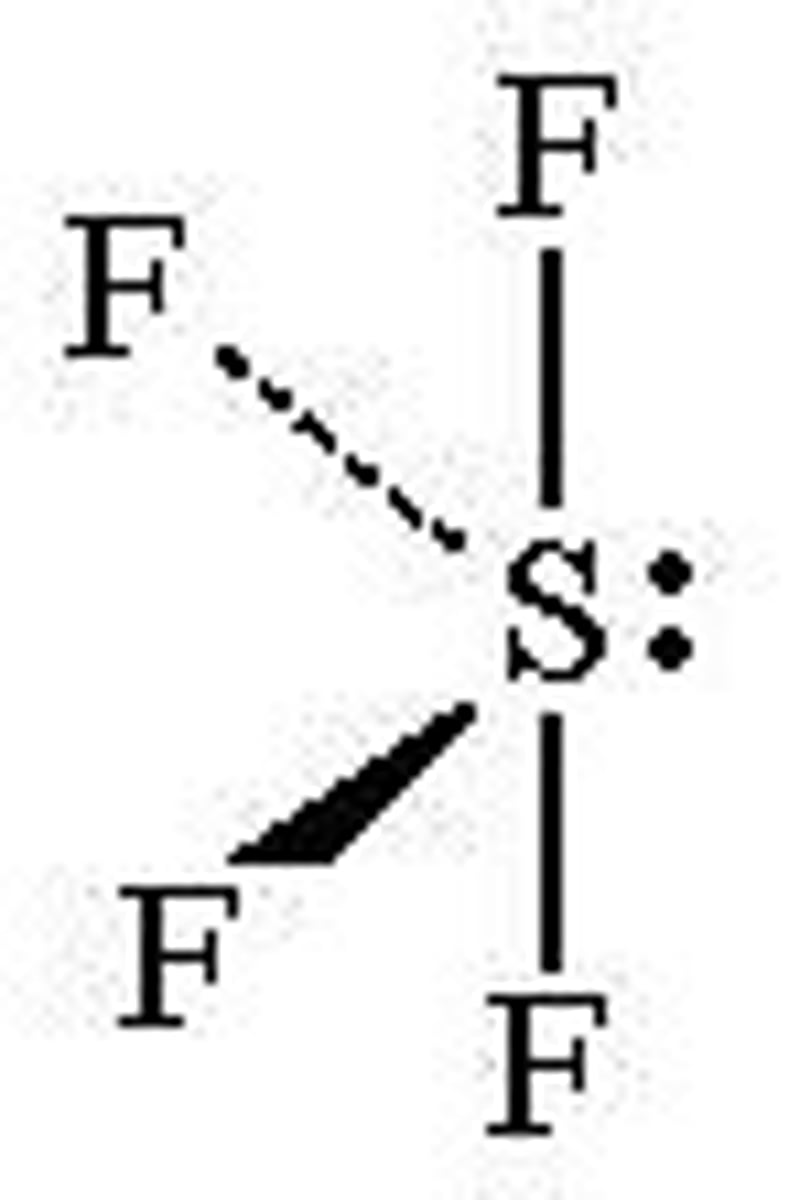
T-shaped
3 bonds, 2 lone pairs, 90 degrees, sp3d
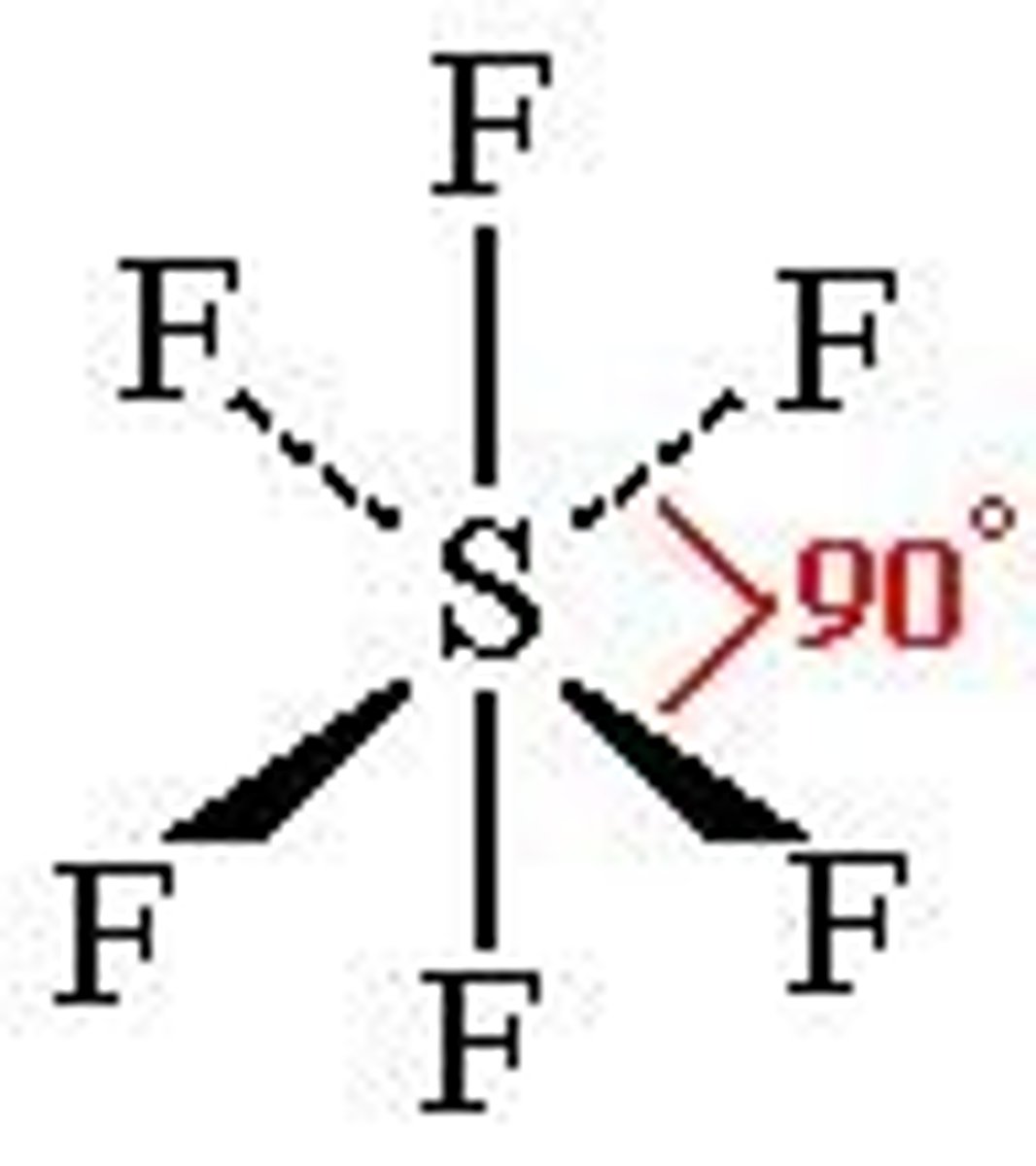
Octahedral
6 bonds, 0 lone pairs, 90 bond angle, sp3d2
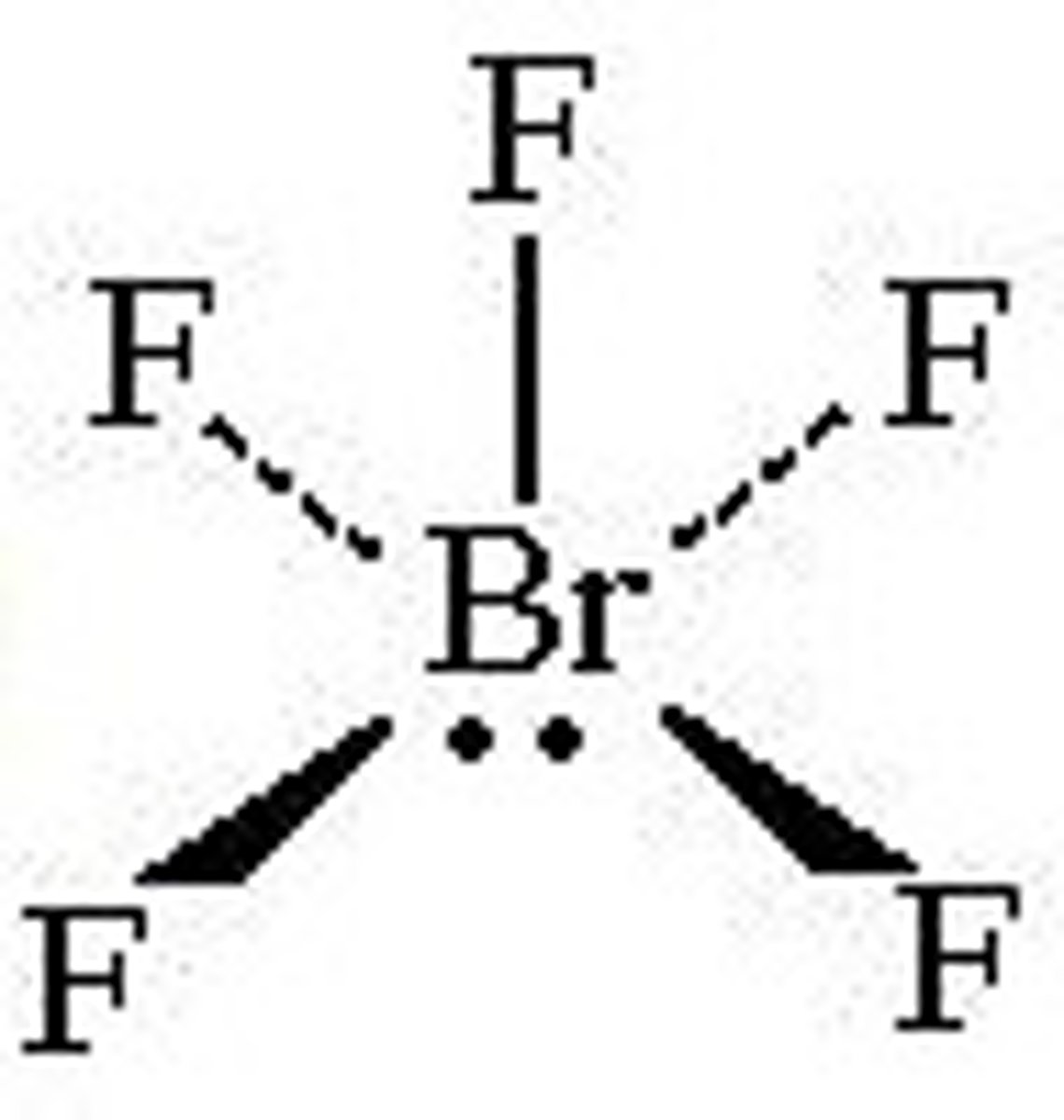
square pyramidal
5 bonds, 1 lone pair, 90 degrees, sp3d2
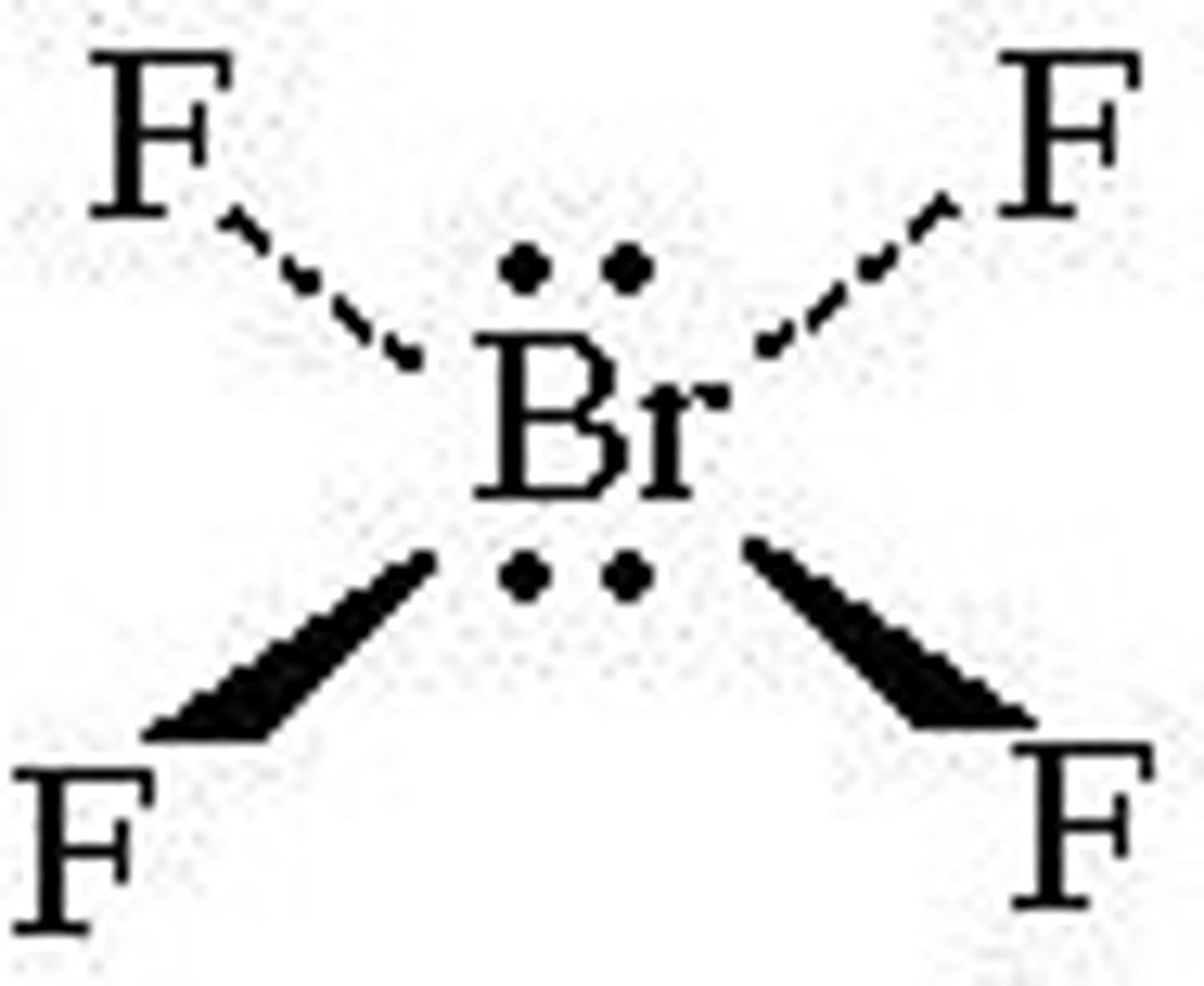
square planar
4 bonds, 2 lone pairs, 90 degrees, sp3d2
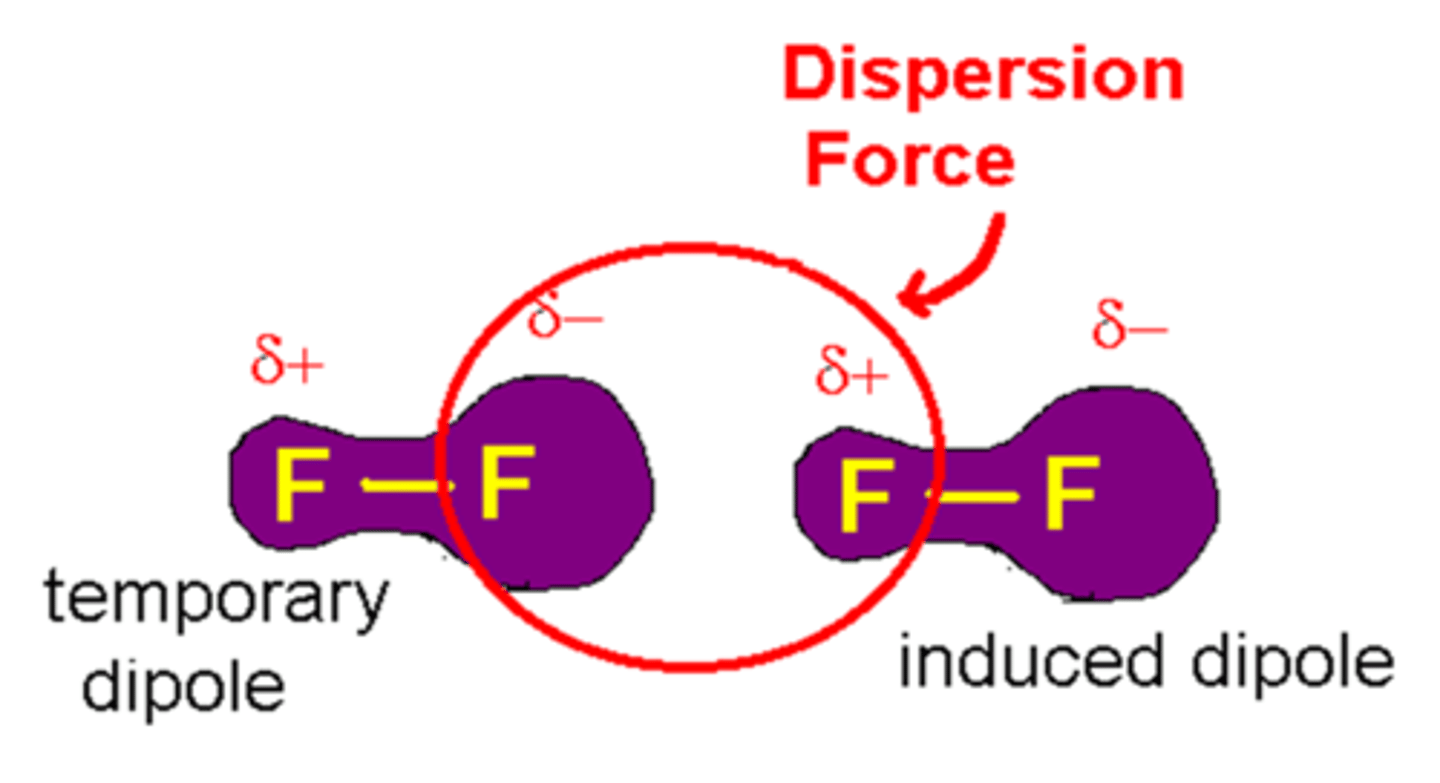
London dispersion forces
The intermolecular attractions resulting from the constant motion of electrons and the creation of instantaneous dipoles, the only force between nonpolars
dipole-dipole forces
attractions between oppositely charged regions of polar molecules
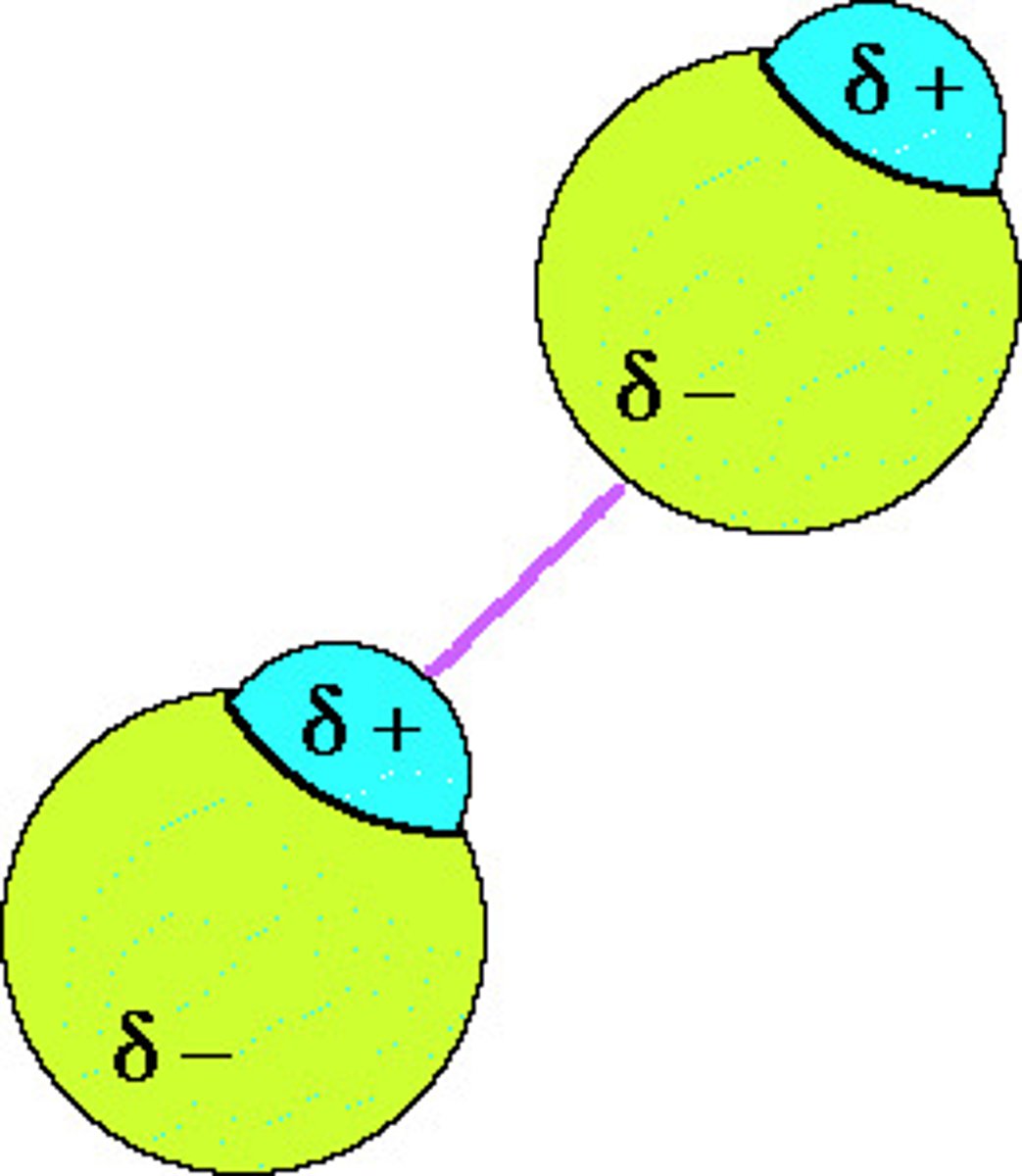
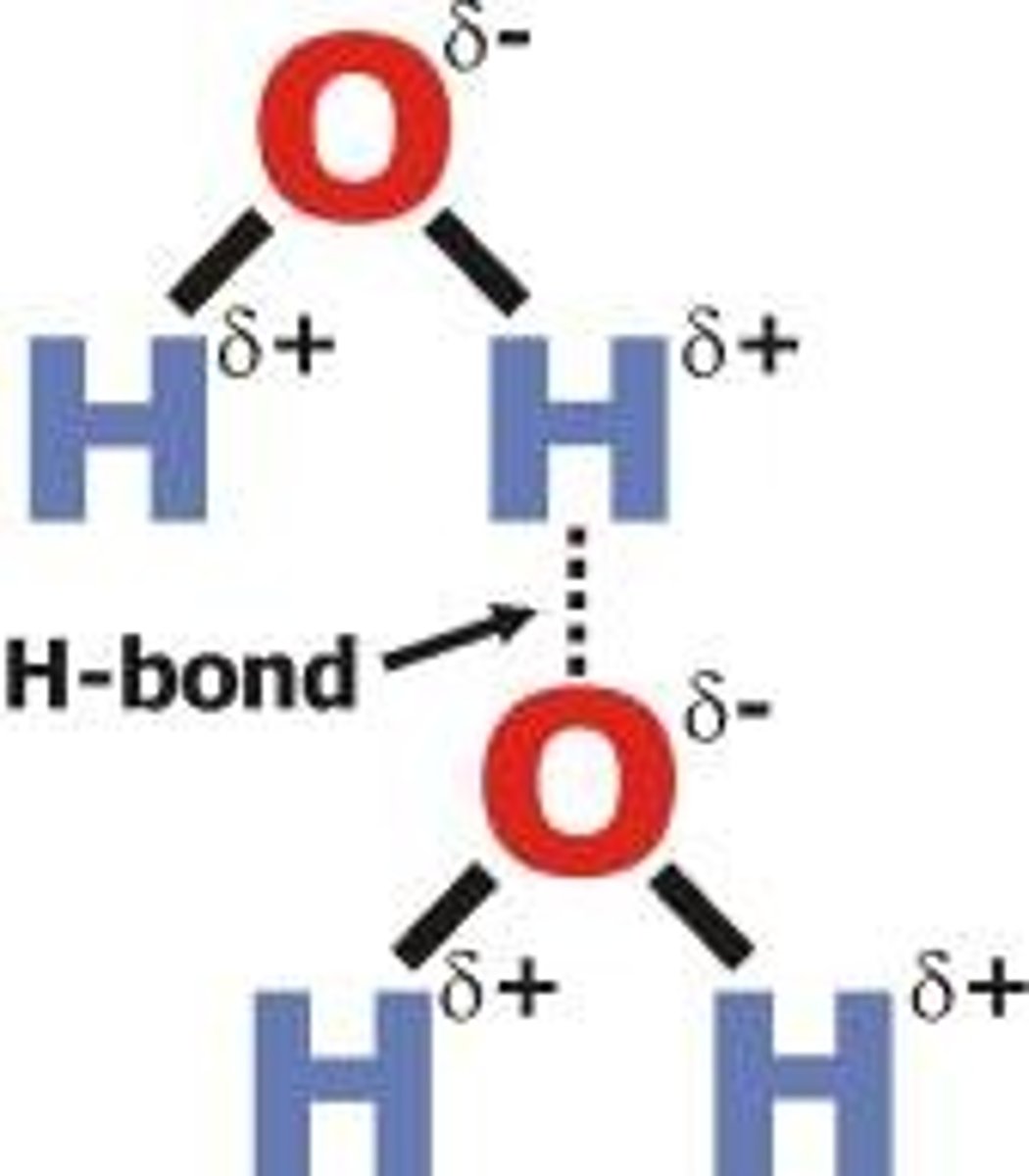
Hydrogen bonds
hydrogen bonding to F,O,N: very strong, polar only
heat
mcAt or mHf/mHv
Ideal Gas Law
PV=nRT; all collisions are elastic, no IMFs, individual gas particles don't take up volume. (be able to explain it using kinetic molecular theory)

Kinetic Molecular Theory
the theory that all matter is composed of particles (atoms and molecules) moving constantly in random directions
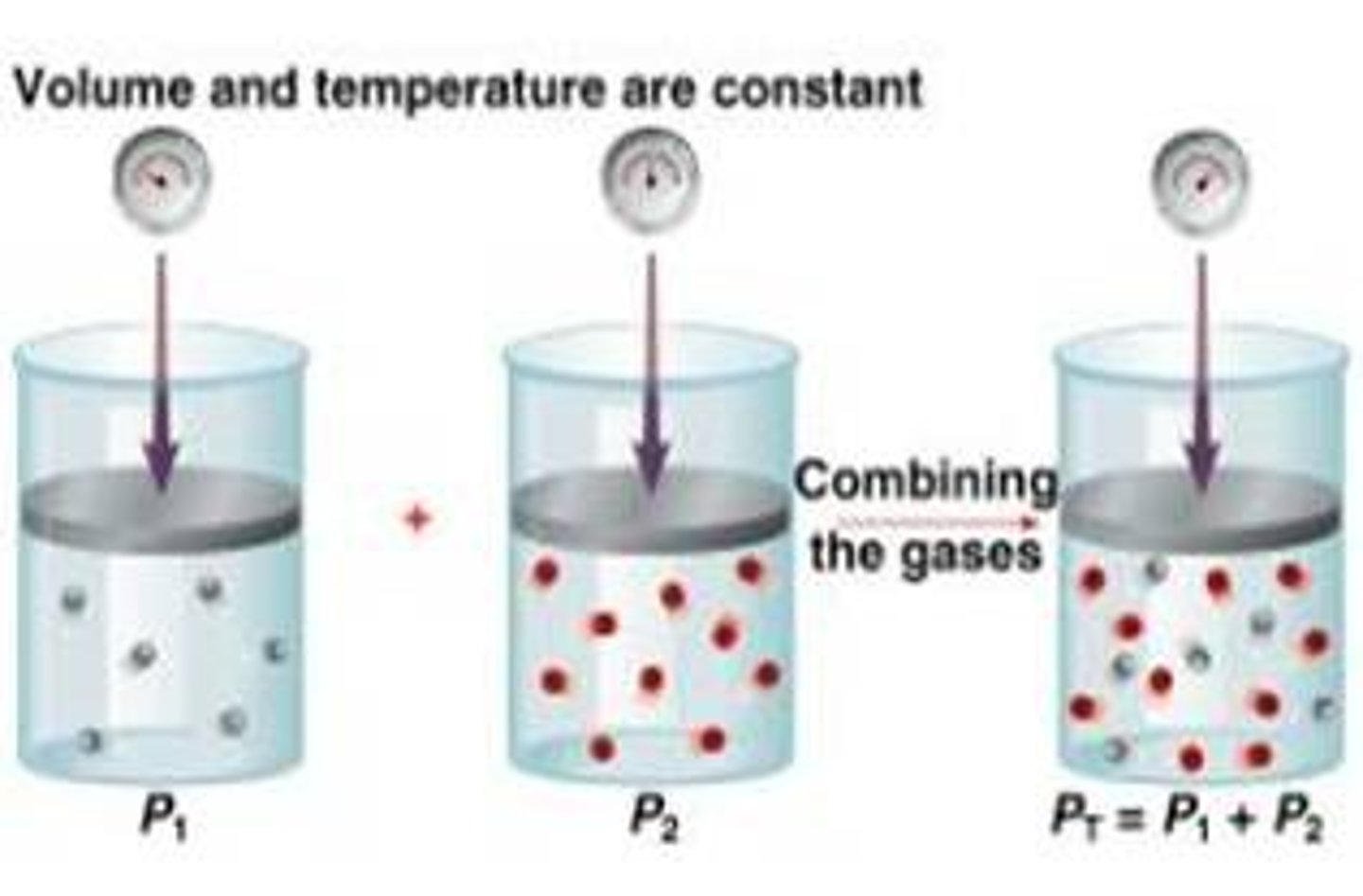
partial pressure
the pressure of each gas in a mixture; Ptotal = P1+P2+... (applies for constant temperature and volume)
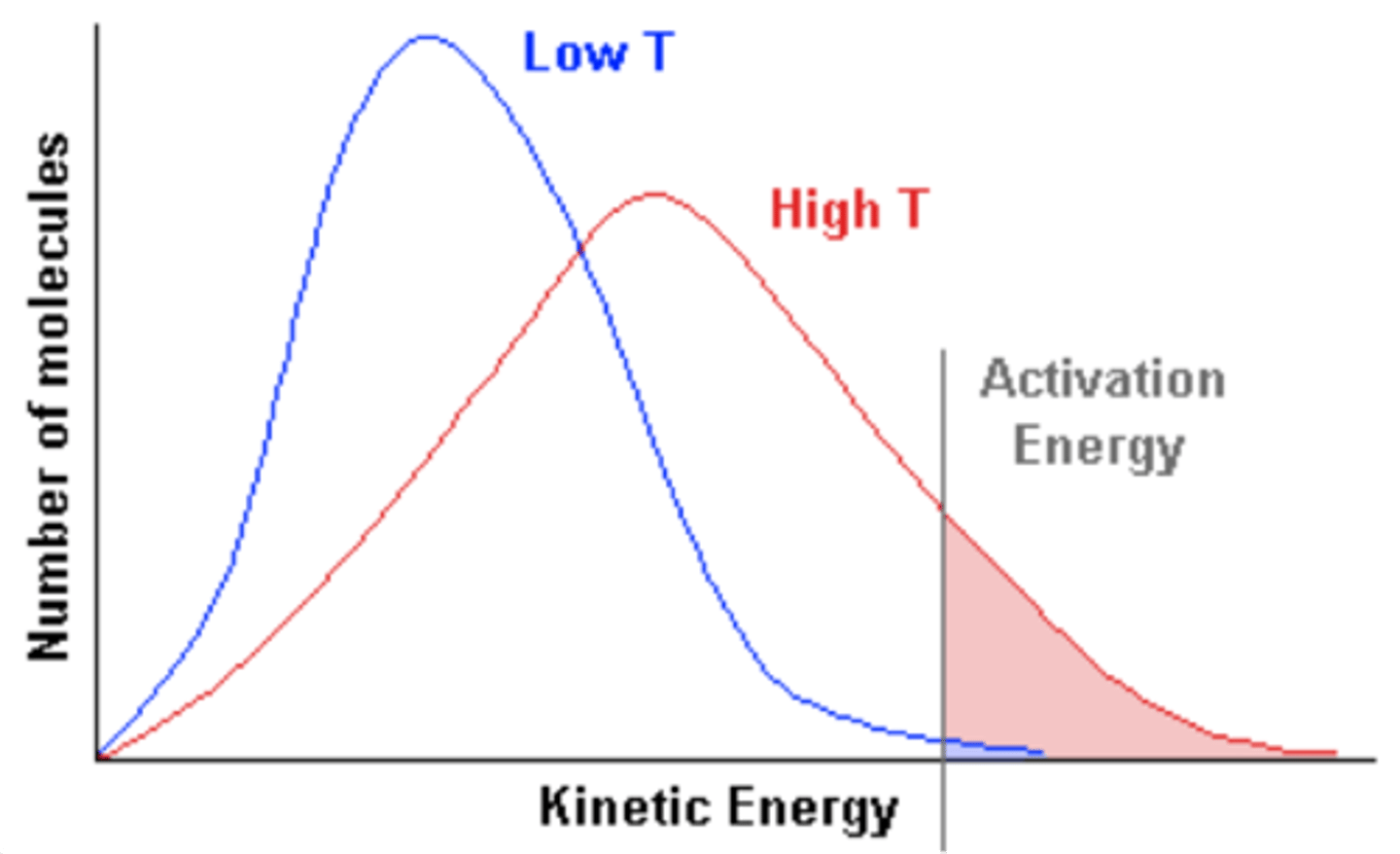
Maxwell-Boltzmann distribution
the higher the temperature, the lower the curve and wider the range. The smaller the mass of the element, the lower the curve and wider the range.
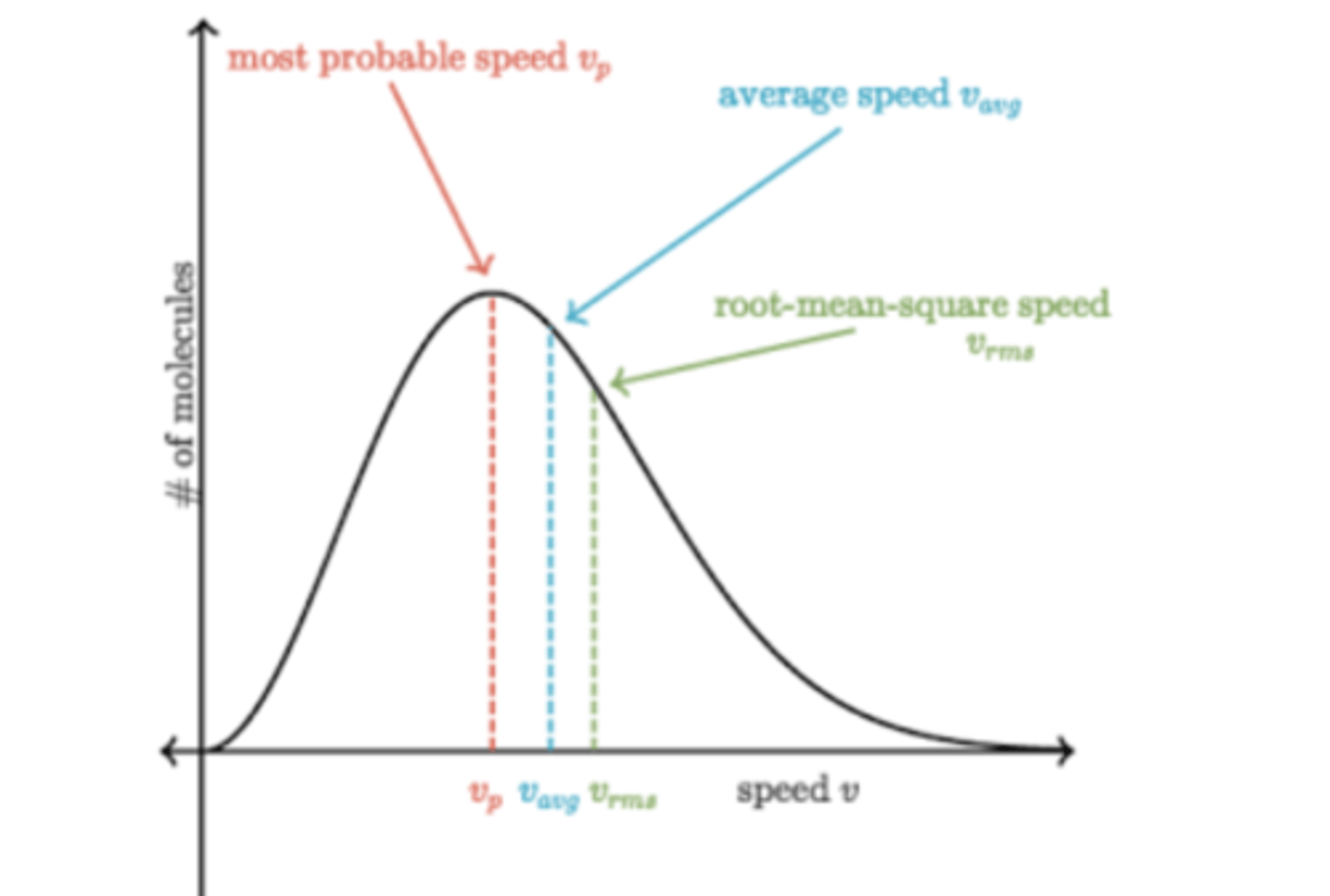
most probable maxwell-boltzmann diagram
peak is most probable speed, average speed found a bit after, root-mean-squared speed a bit after that
Decomposition
A chemical reaction that breaks down compounds into simpler products.
Synthesis
combining parts into a whole
double replacement
AB + CD = AD + CB
Single Replacement
A + BC --> B + AC
rate of reaction
measure of how fast a chemical reaction occurs; when rate of reaction equals reverse, equation is at equilibrium
Zeroth order rate law
Rate = k (change in concentration has no effect on the rate)
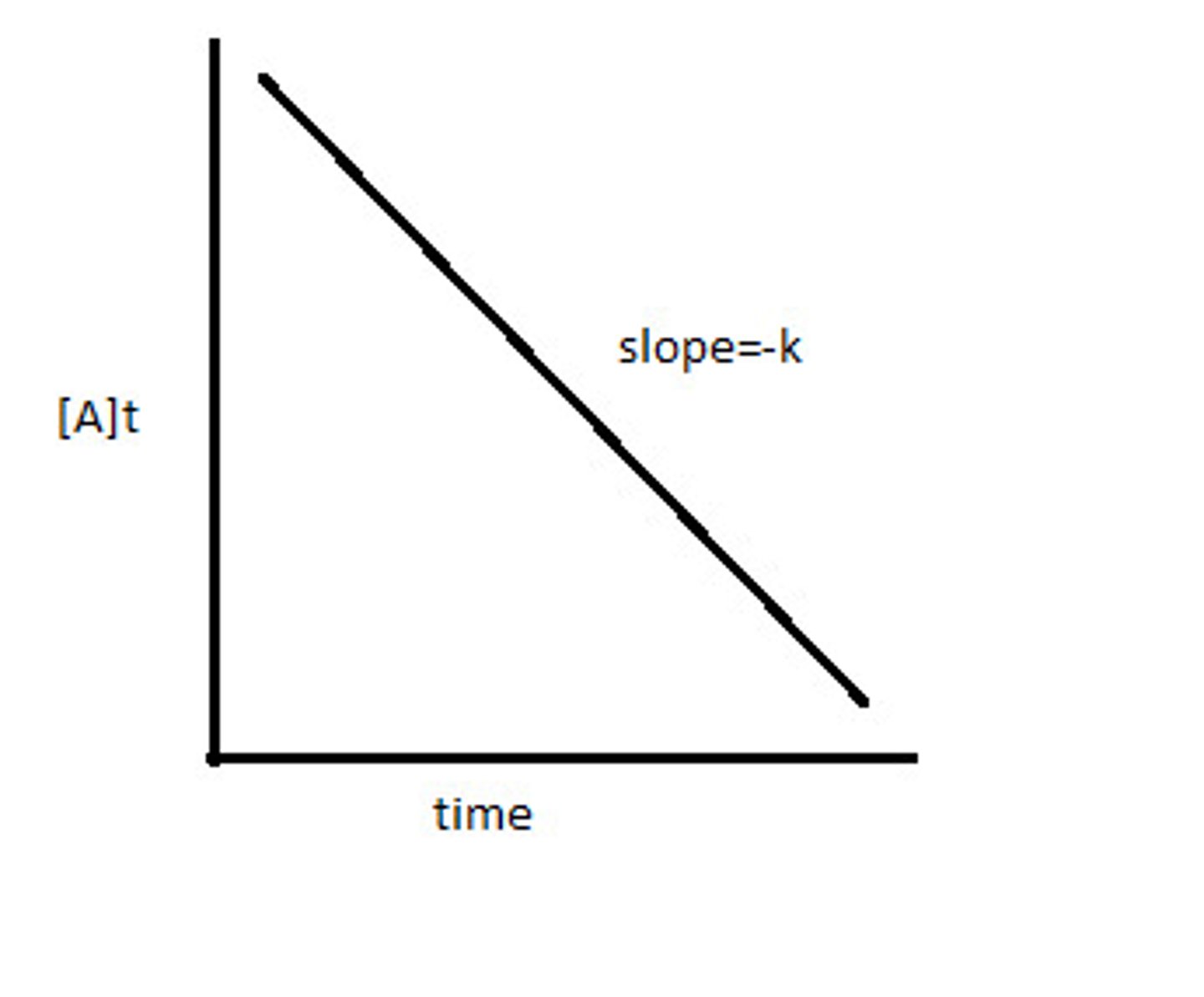
first order rate law
ln[A]t = -kt + ln[A]0
![<p>ln[A]t = -kt + ln[A]0</p>](https://knowt-user-attachments.s3.amazonaws.com/1d731b5b-bceb-48b8-a16e-b730d7ed86f3.png)
second order rate law
1/[A]t = kt + 1/[A]0
![<p>1/[A]t = kt + 1/[A]0</p>](https://knowt-user-attachments.s3.amazonaws.com/3e0c568b-9bdd-4889-b38d-ae5502c252ac.png)
Zeroth order half life
t1/2 = [A]0 / 2k
![<p>t1/2 = [A]0 / 2k</p>](https://knowt-user-attachments.s3.amazonaws.com/daf1d84d-c5d0-48ed-9bf6-46fee183bb8a.png)
first order half life
t1/2 = 0.693/k (doesn't change over time)
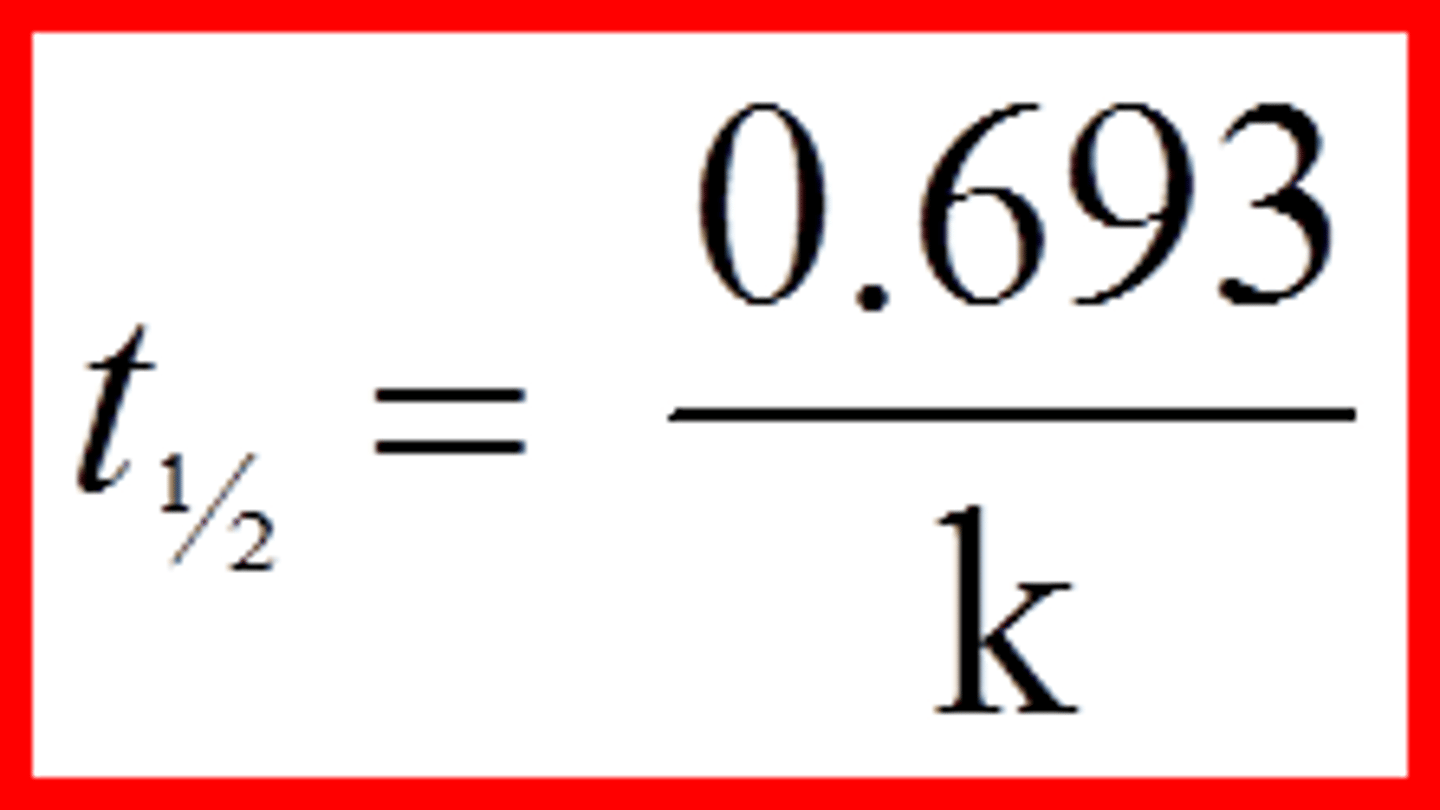
second order half life
t1/2 = 1 / k[A]0
![<p>t1/2 = 1 / k[A]0</p>](https://knowt-user-attachments.s3.amazonaws.com/f105fc26-d08d-4960-8e4f-45bd2f5c837d.jpg)
collision theory
states that atoms, ions, and molecules must collide with enough energy and right angle in order to react
Arrhenius equation
k=Ae^(-Ea/RT)
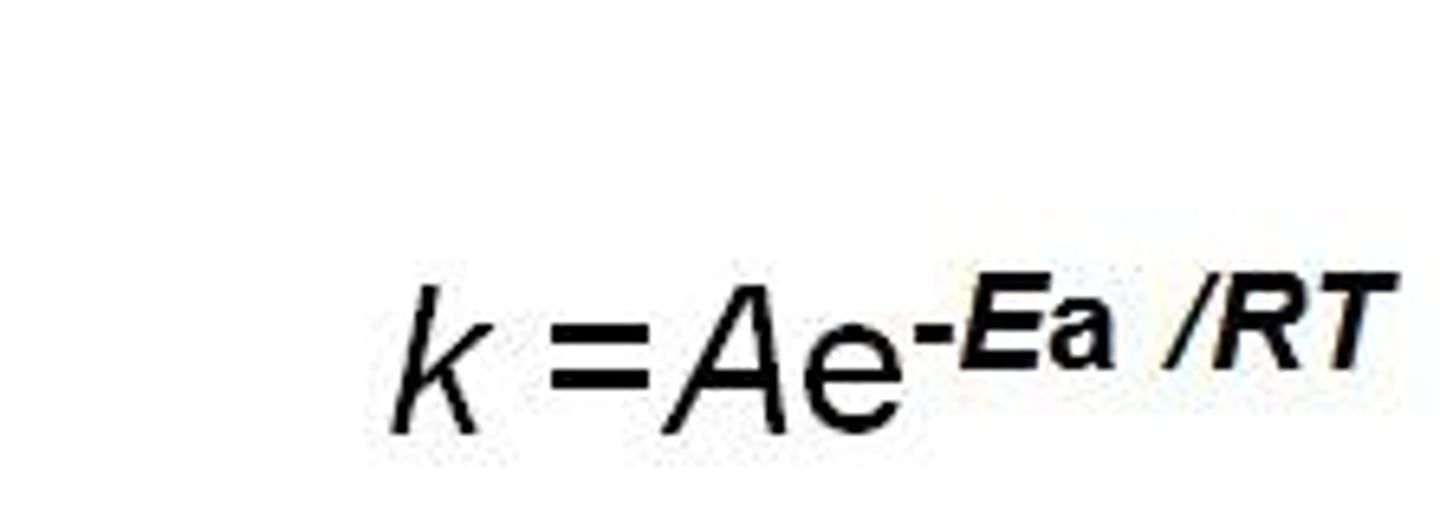
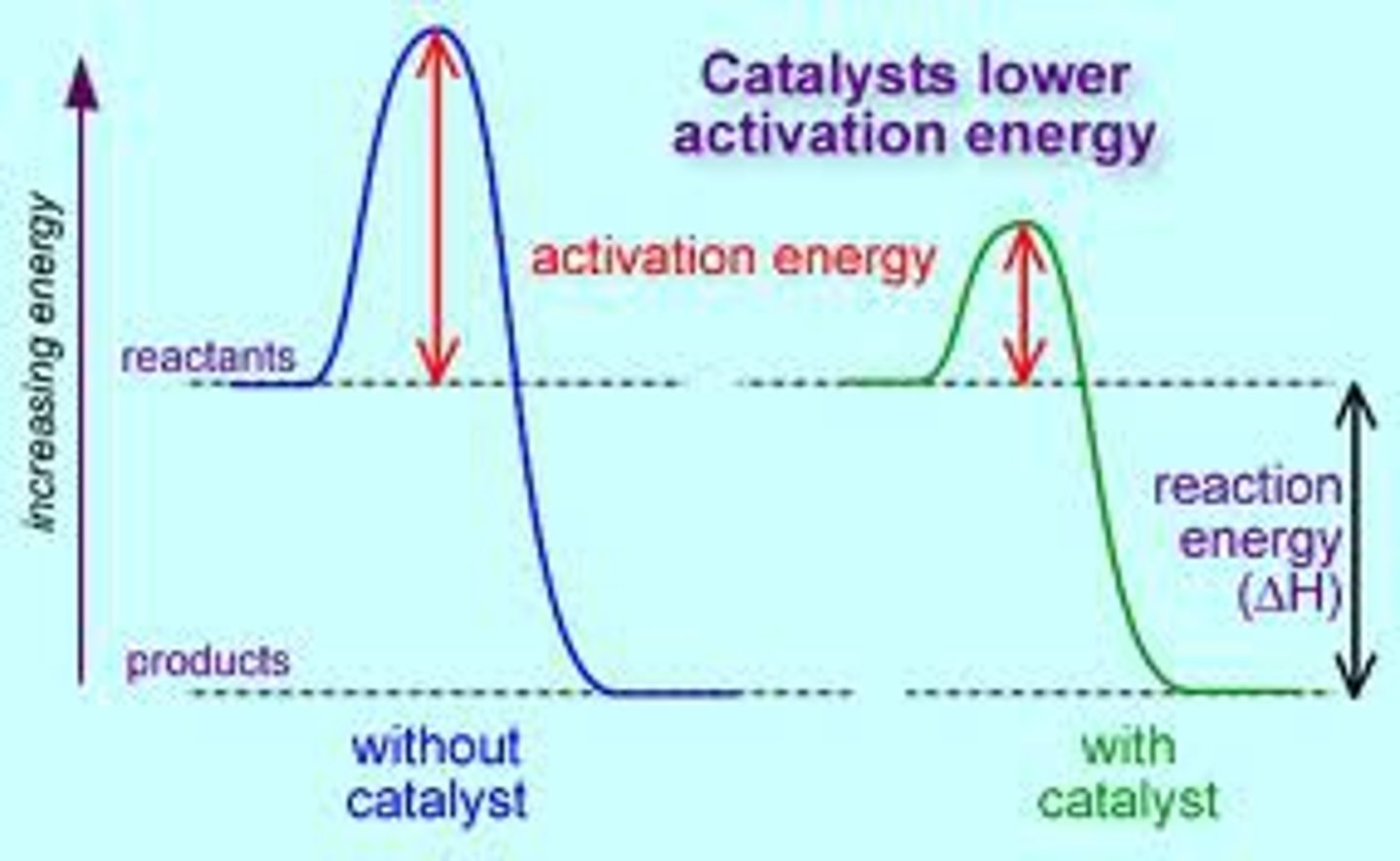
catalyst
substance that speeds up the rate of a chemical reaction
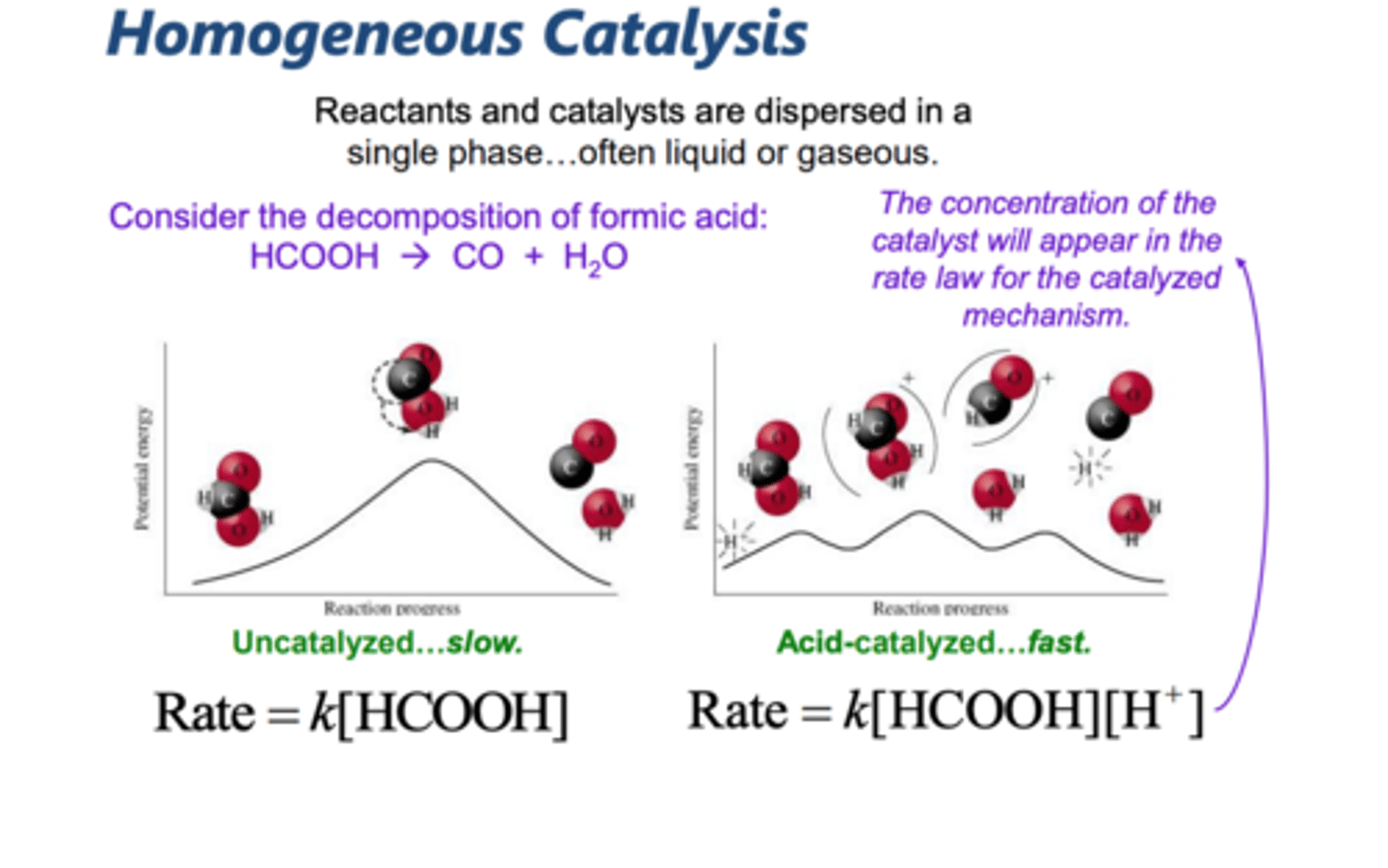
homogenous catalyst
a catalyst that is in the same phase as all the reactants and products in a reaction system
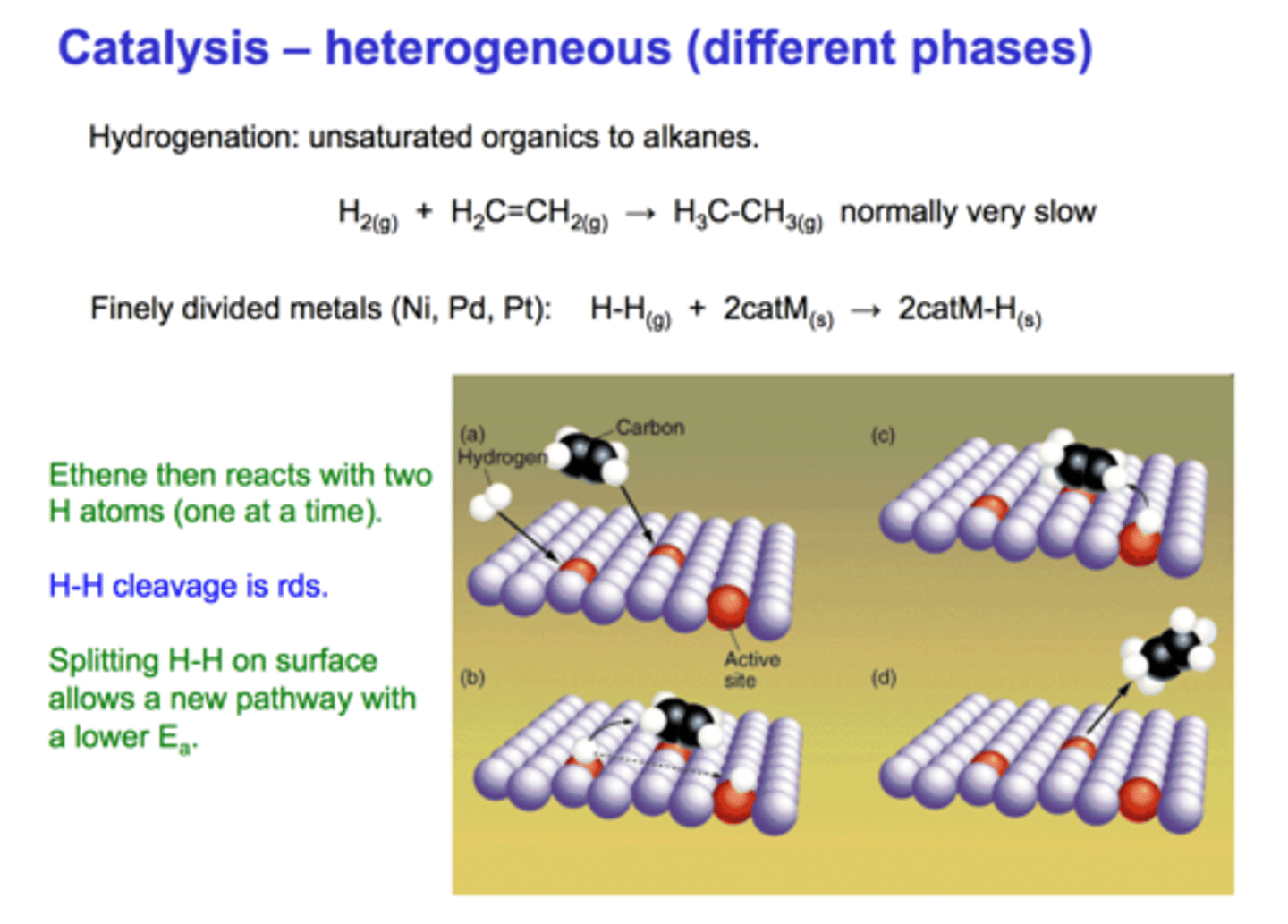
heterogenous catalyst
a catalyst whose phase is different from the reactants
Beer's Law
A=ebc

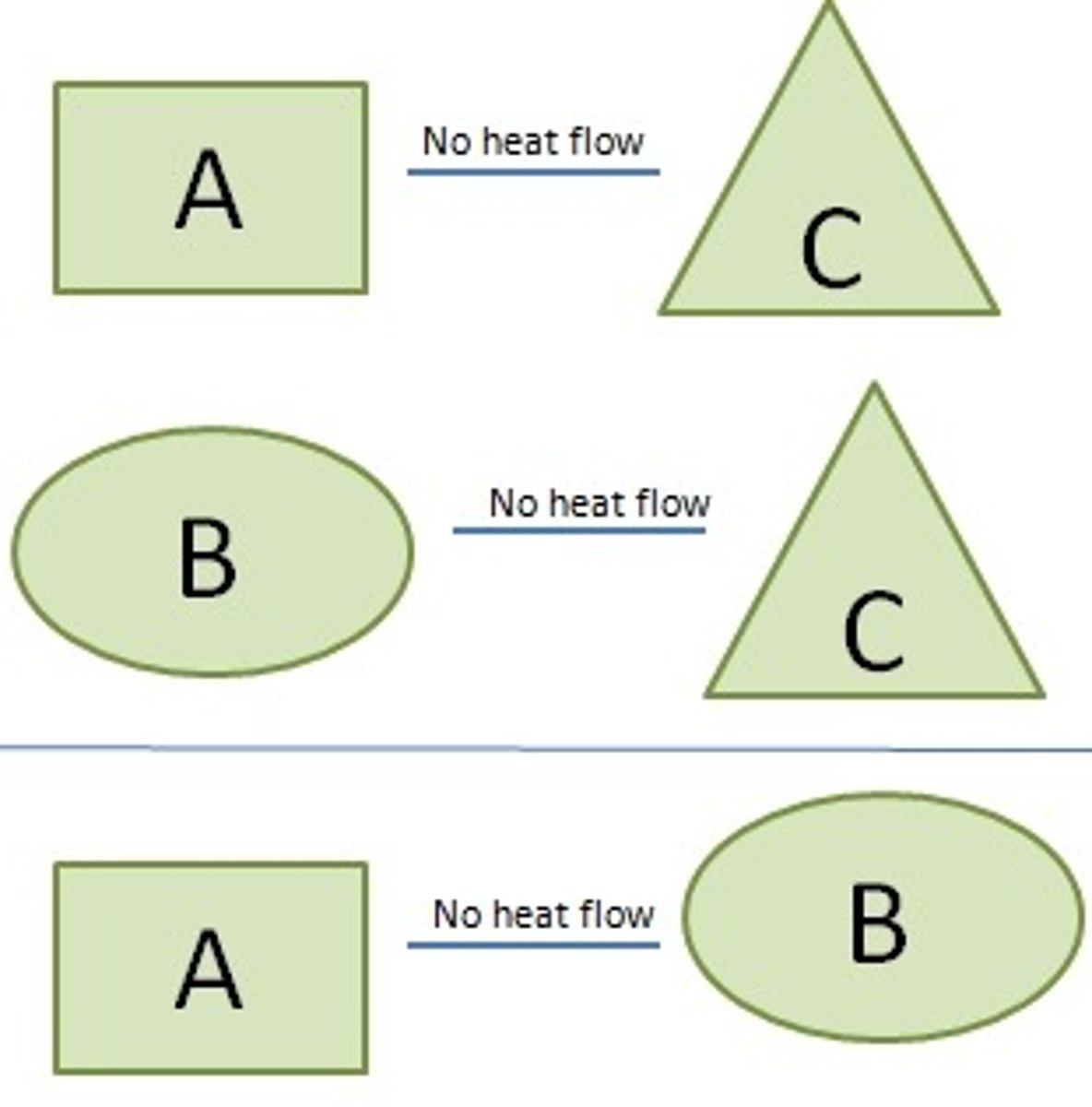
Zeroth Law of Thermodynamics
A law that if two systems are separately found to be in thermal equilibrium with a third system, the first two systems are in thermal equilibrium with each other; that is, all three systems are at the same temperature. Also known as thermodynamic equilibrium.
first law of thermodynamics
Energy can be transferred and transformed, but it cannot be created or destroyed; U=Q+W
second law of thermodynamics
Every energy transfer or transformation increases the entropy of the universe.
relationship between specific heat capacity and change in temperature
the lower the specific heat capacity, the greater the change in temperature
Hess's Law
the overall enthalpy change in a reaction is equal to the sum of enthalpy changes for the individual steps in the process
enthalpy of formation
the enthalpy change that accompanies the formation of a substance from the most stable forms of its component elements

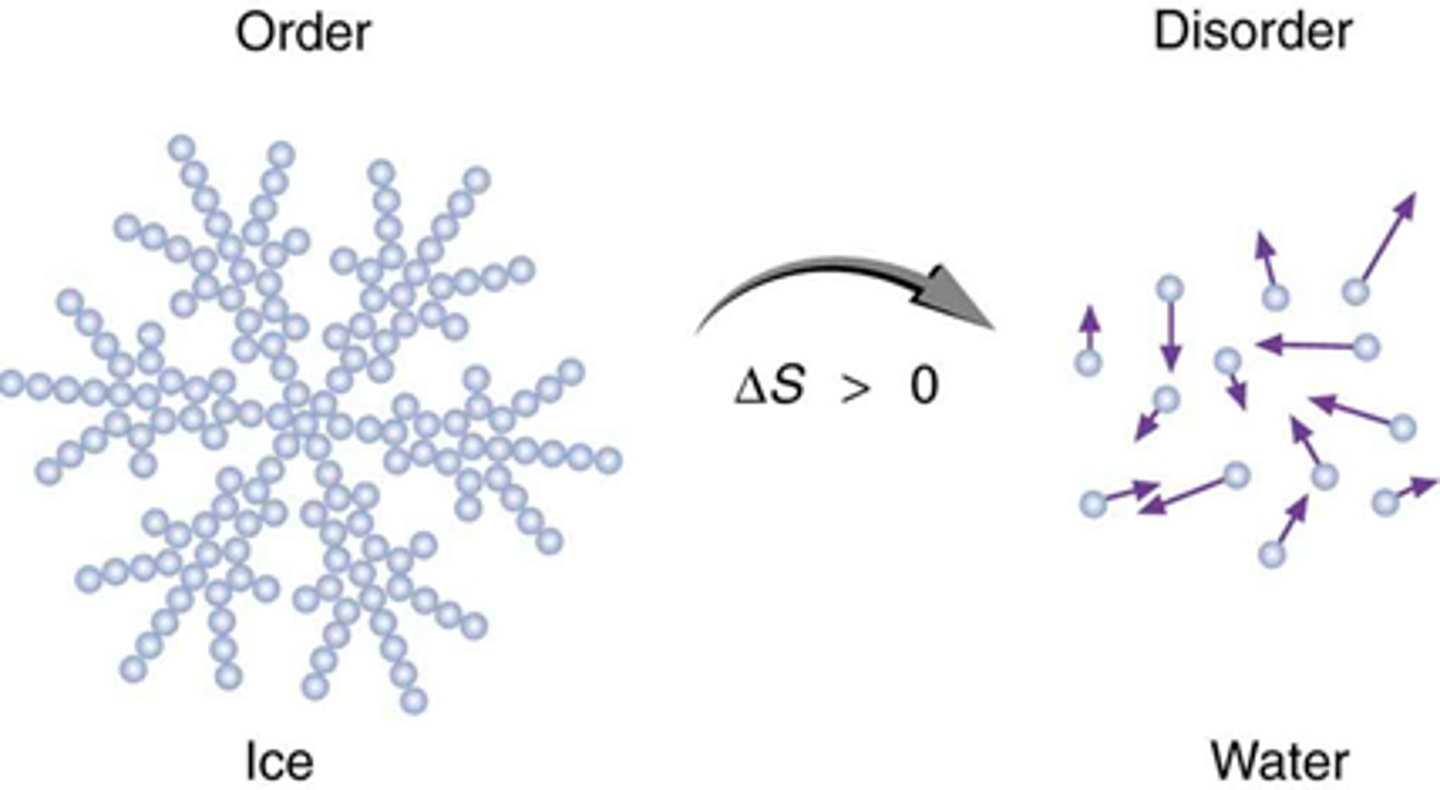
Entropy
A measure of disorder or randomness.
Things that mean entropy increased
- increased moles of same phase (ex: 1 mol gas --> 2 moles gas)
- more entropy phases (ex: solid --> gas)
Gibbs free energy formula
∆G = ∆H - T∆S

Delta G = Delta Go + RTlnQ
If Q is bigger than Keq than rxn shifts to the left or if Q is smaller than Keq then rxn shifts to the right.
Value of Delta G at equilibrium
0
Ksp
solubility product constant
Ksp=[A+]^m[B-]^n
do not include concentration of pure solid/liquids
![<p>solubility product constant</p><p>Ksp=[A+]^m[B-]^n</p><p>do not include concentration of pure solid/liquids</p>](https://knowt-user-attachments.s3.amazonaws.com/1e479247-5159-4d4a-9103-87ca257ea48f.jpg)
Le Chatelier's Principle
States that if a stress is applied to a system at equilibrium, the system shifts in the direction that relieves the stress.
- Increase reactants moves right
- Increase products moves left
- Increase temperature: if delta H is + go right, if negative go left
- increase pressure go to side with less moles
When asked to find rate law of overall reaction of an equation, which step do you use?
slow step: DON'T USE THE OVERALL EQUATION
pH formula
pH=-log[H+]
pOH formula
-log[OH-] and 14-pH
Strength of Acids
bigger atom and less hoggy means better acid
Kw
[H+][OH-] = 1.0 x 10^-14
If a salt comes from a strong acid and a weak base what pH will you get when you dissolve it in water
basic (pH>7)
If a salt comes from a strong base and a weak acid what pH will you get when you dissolve it in water
acidic (pH<7)
Buffers
weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH
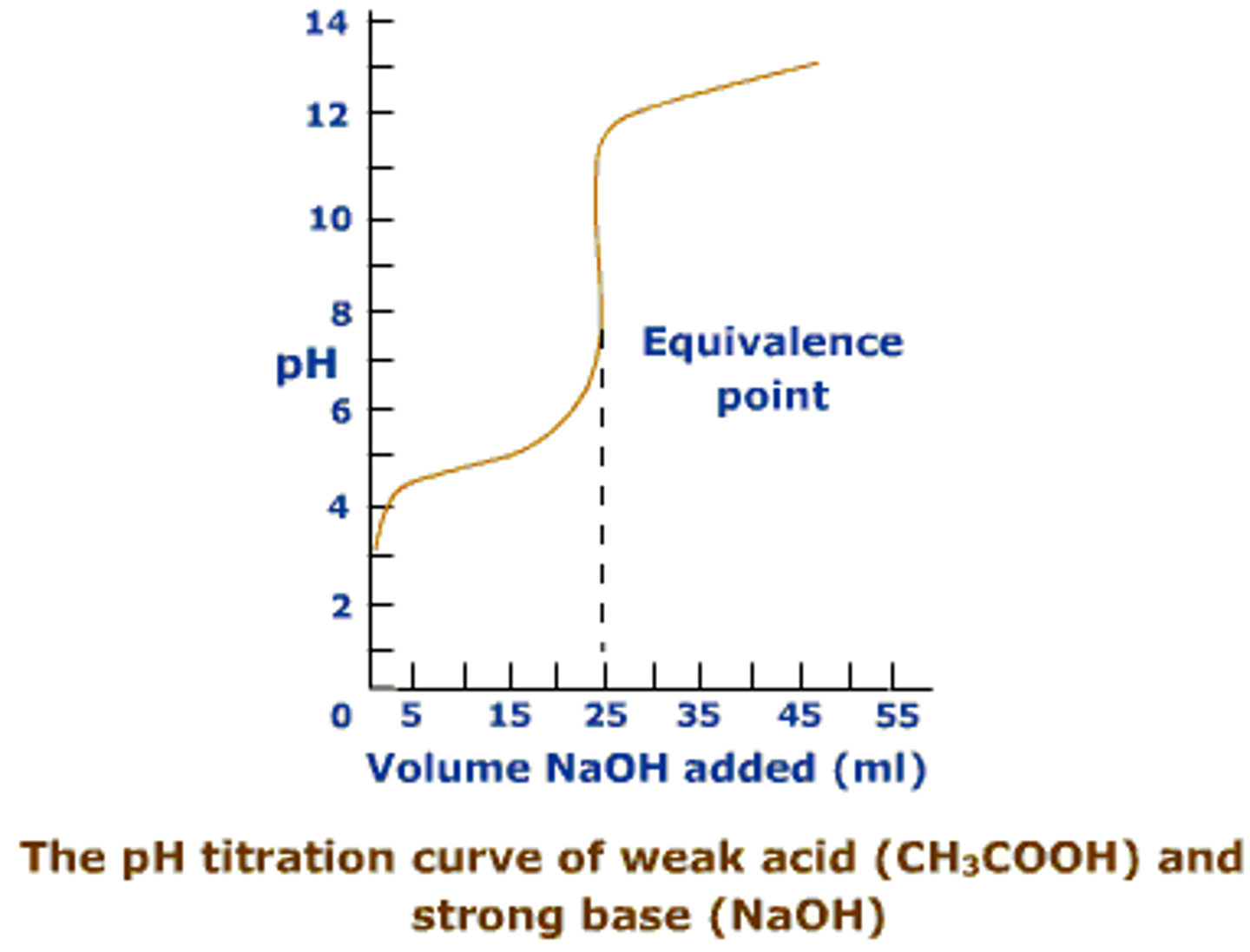
half equivalence point
pH=pKa
Henderson-Hasselbach
pH = pKa + log([A-]/[HA])
![<p>pH = pKa + log([A-]/[HA])</p>](https://knowt-user-attachments.s3.amazonaws.com/56ac3dfc-1e0e-4911-adbd-82ad1660cb0a.png)
When titrating weak acid and strong base...
remember to convert some of the weak acid to its conjugate first (depending on mols of strong base) and then applying Henderson Hasselbach
Oxidation state exceptions
- H2O2; Oxygen is -1
- F2O; F is going to be -1 and O is going to be +2
Galvanic Cells (voltaic)
-spontaneous
- anode=negative
-cathode=positive
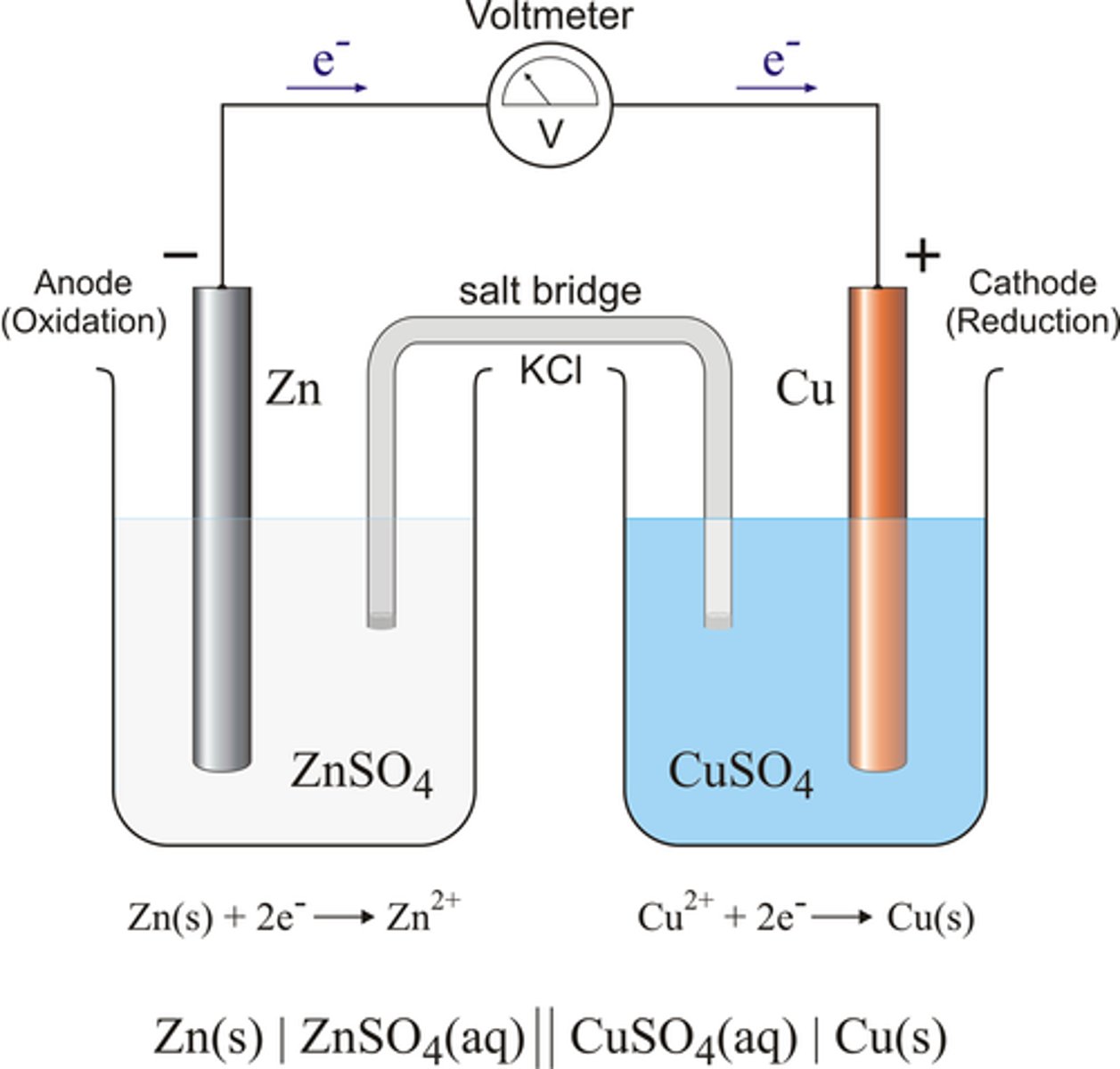
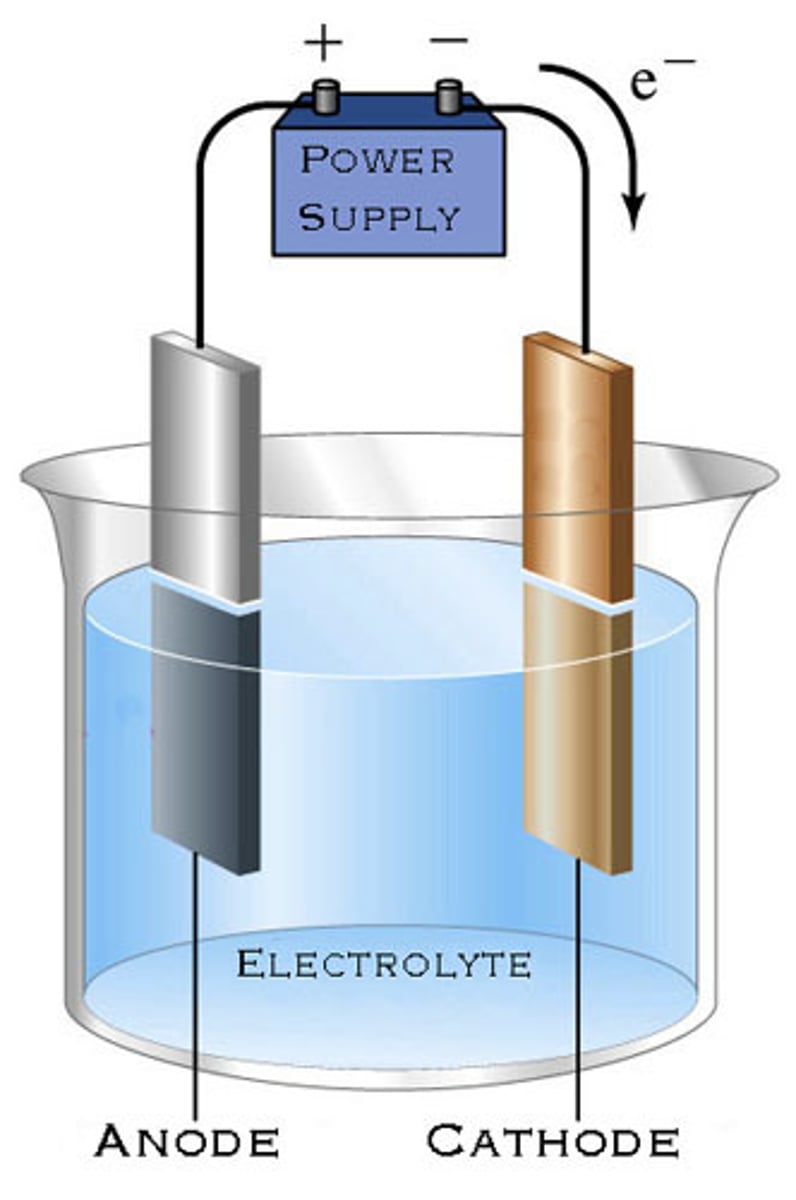
electrolytic cell
an electrochemical cell used to cause a chemical change through the application of electrical energy
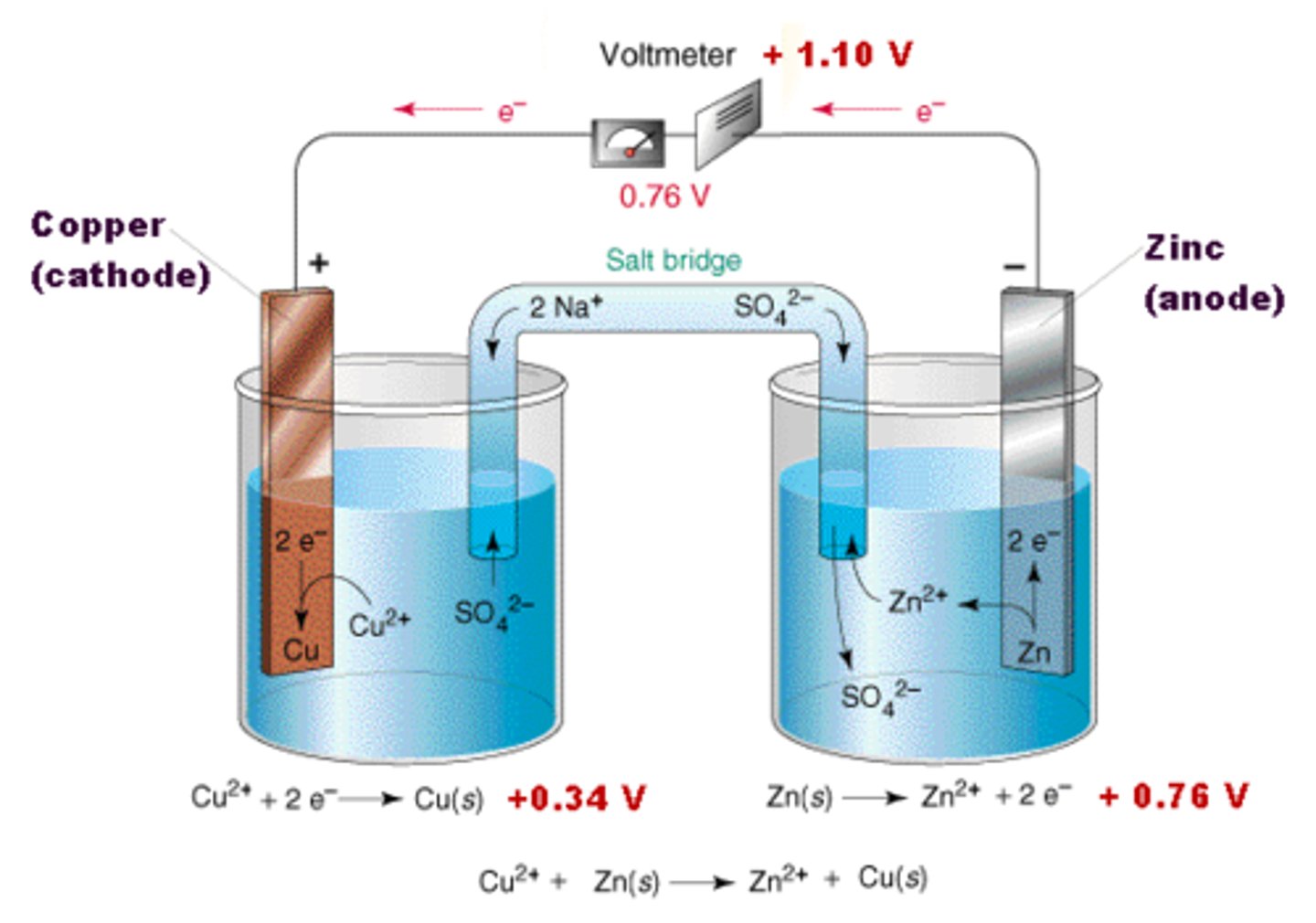
salt bridge
A tube that allows the slow transfer of ions and maintains the neutrality of the electrolyte solutions.
Nernst equation
Ecell= E°cell - (RT/nF) (lnQ)
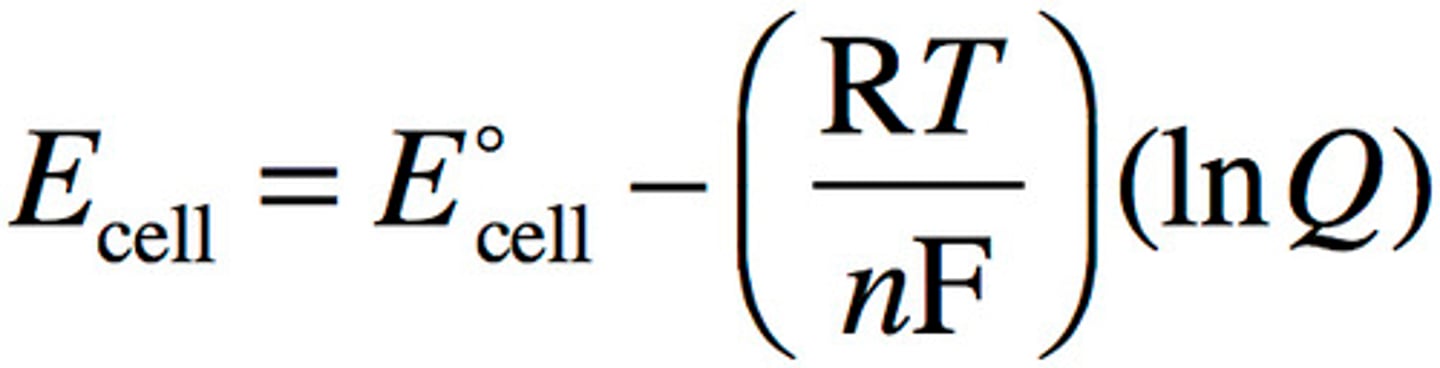
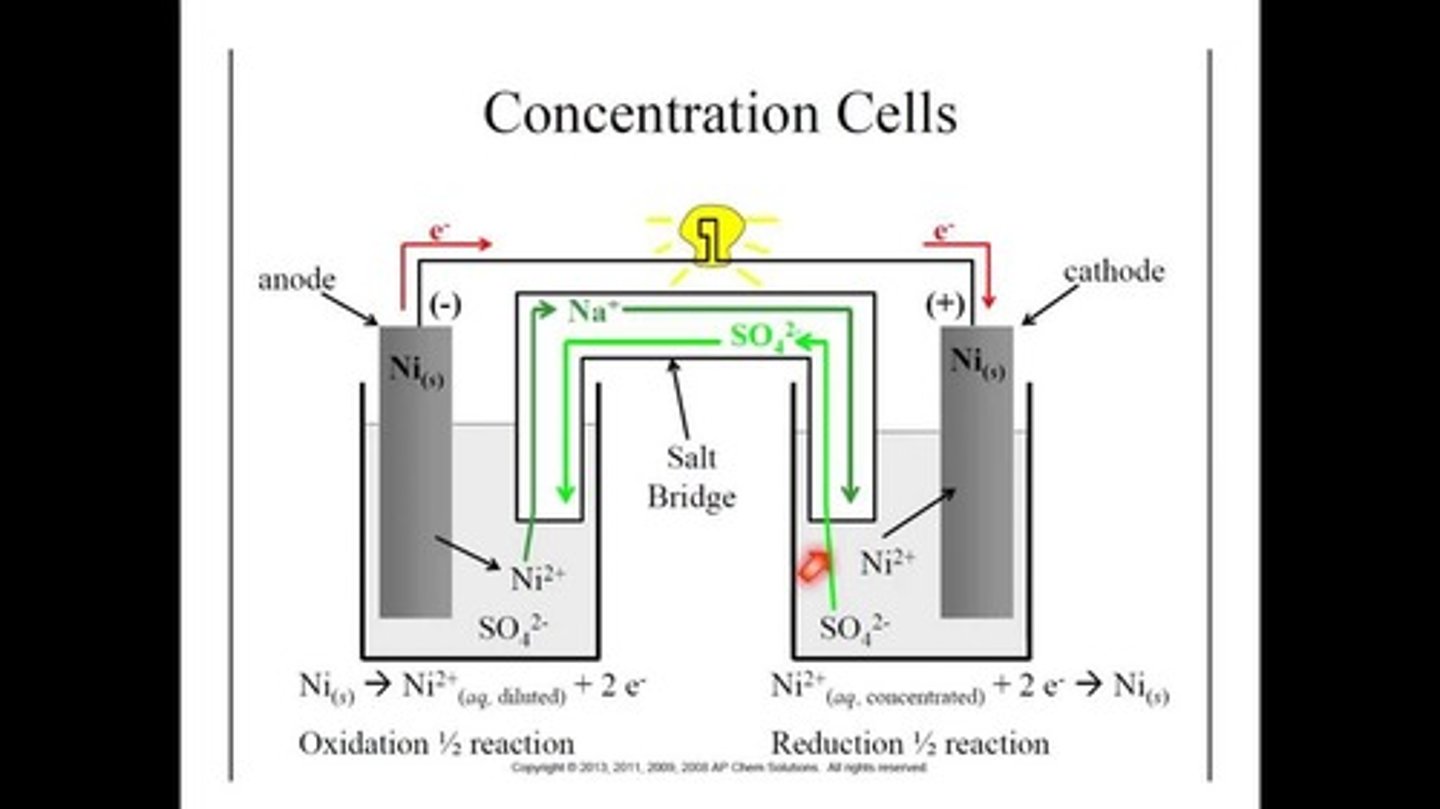
Concentration cells
- Current is generated as a function of a concentration gradient established between the two solutions surrounding the electrodes.
- Voltage is zero if the concentration is equal.
- electrons travel from less molarity to higher molarity
Delta g equation in redox
delta G = -nFE