BME 520 Exam 3: Tissue Development and Regeneration
1/145
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
146 Terms
composition of ectoderm
epiblast cells that do not ingress through the primitive streak
how is the ectoderm induced
active repression of differentiation via the inhibition of TGF-B
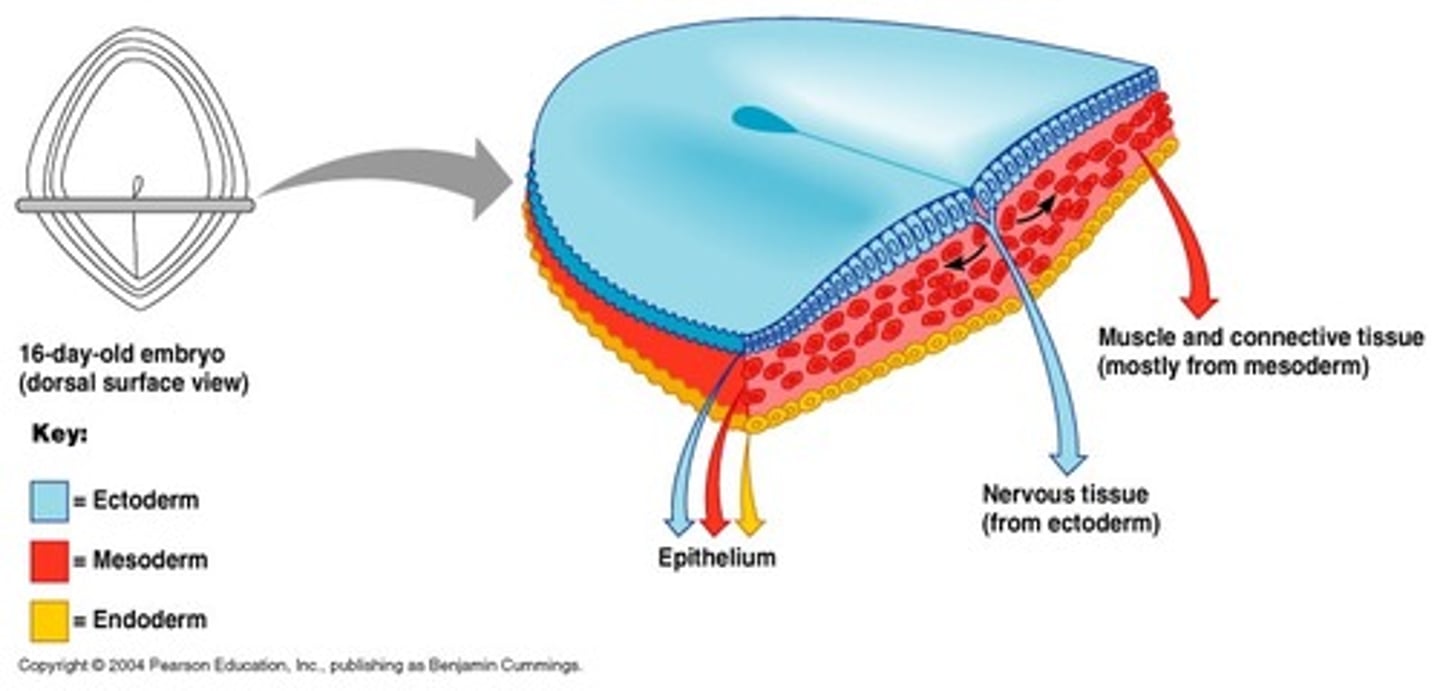
main derivatives of the ectoderm and their sub-parts
surface ectoderm-> skin, corneal lens
neural crest cells-> peripheral nervous system and support cells
neural tube -> CNS and retina of eye
limbs
steps of formation of the primitive neural tube
-primitive streak regresses, leaving behind the a divot on top of the underlying notochord. walls of the divot fold up
-the portion of the ectoderm closest to the underlying notochord (neural plate) becomes fated to the neural lineage and neurulation is initiated to form the primitive neural tube
neuroepithelium
cells that form the neural tube
what growth factors does the neuroepithelium posses
Pax6, Sox2, and CDH2 (N-Cadherin)
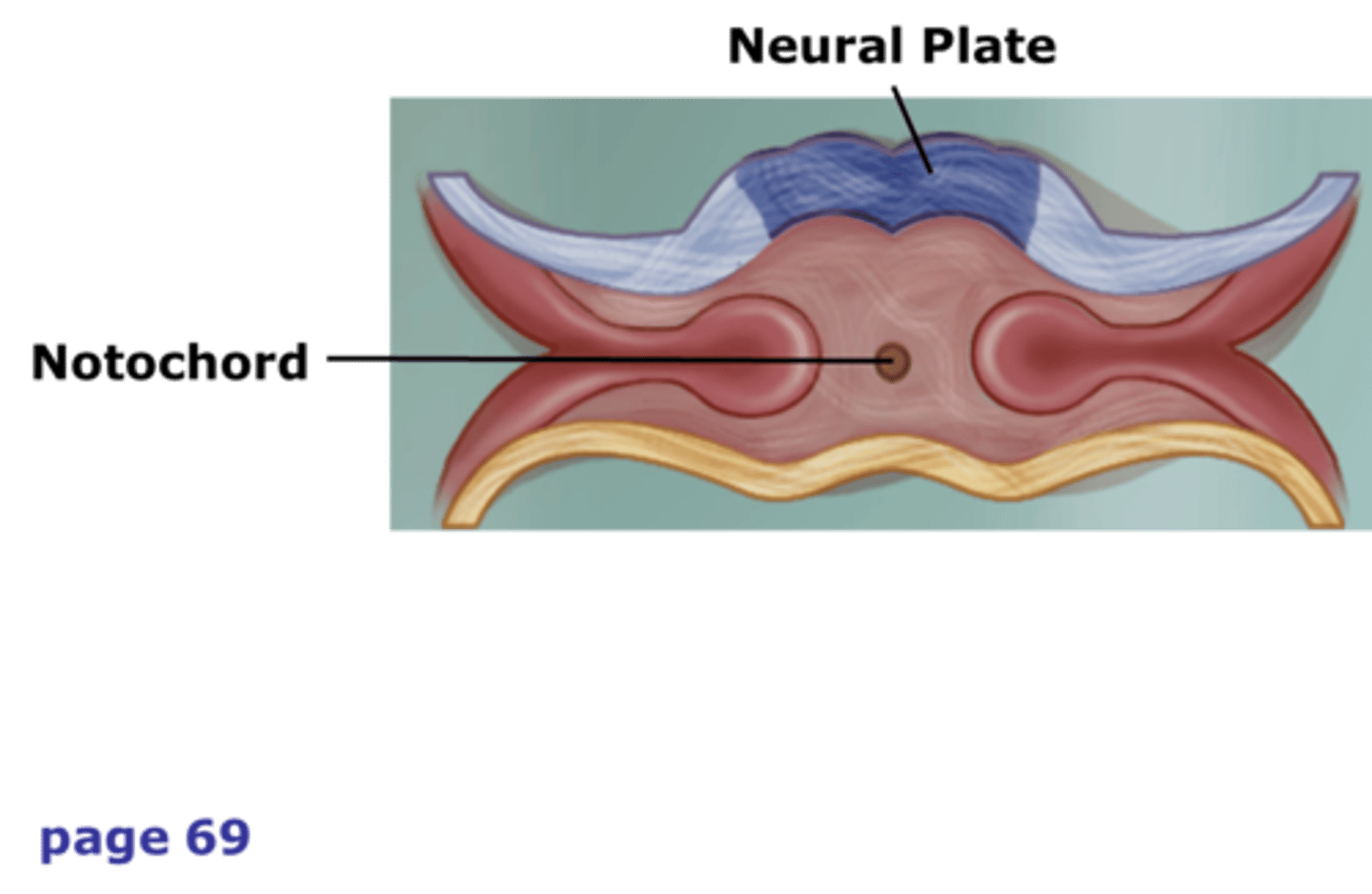
notochord
A flexible rod that supports a chordate's back
neuroectoderm
portion of the ectoderm that becomes the nervous system
neural plate
thickened region of the ectodermal layer, proximal to underlying notochord, that gives rise to the neural tube
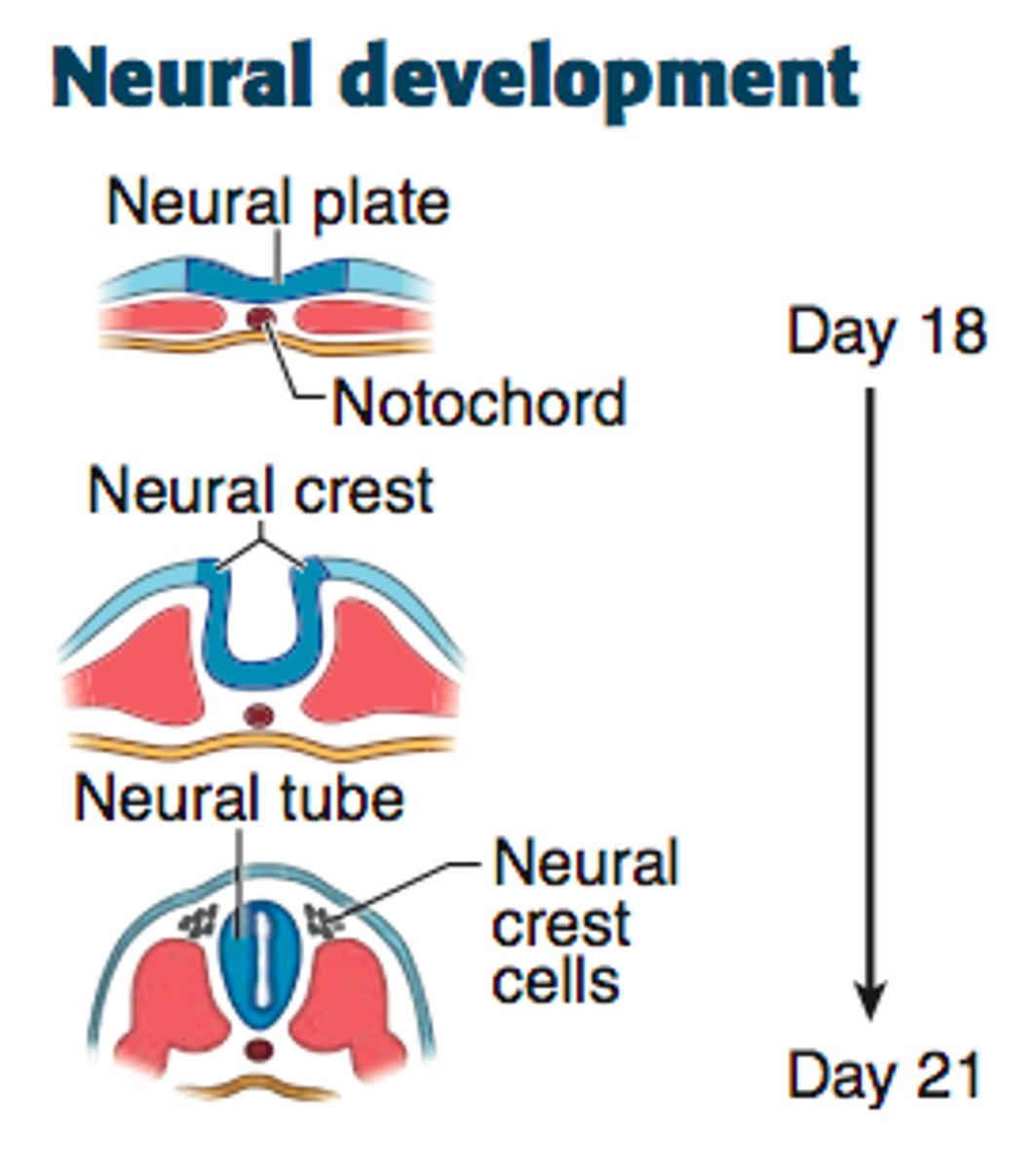
primary neurulation
the neural plate creases inward until the edges come in contact and fuse
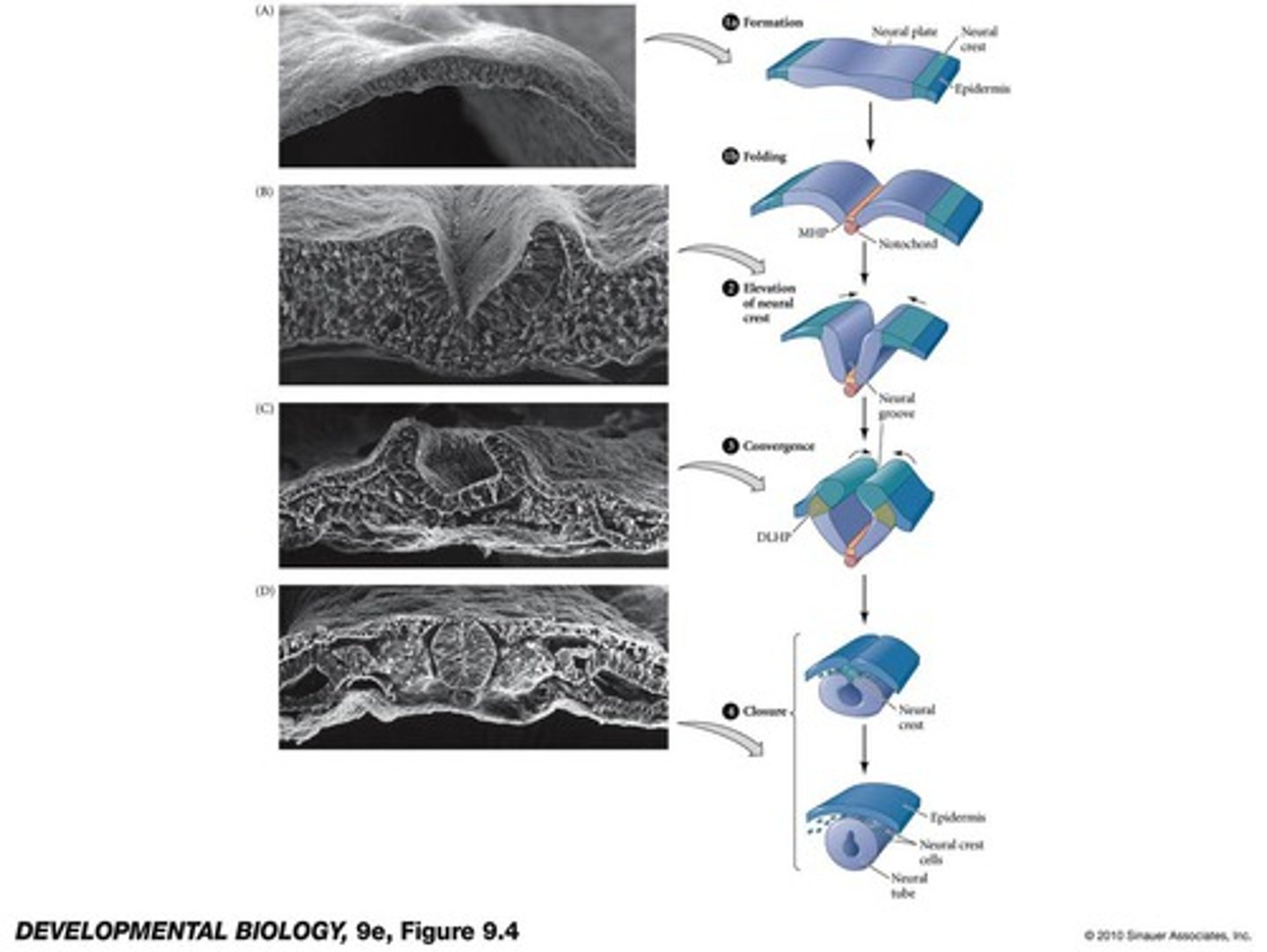
four phases of primary neurulation
1. formation
2. elevation
3. convergence
4. closure
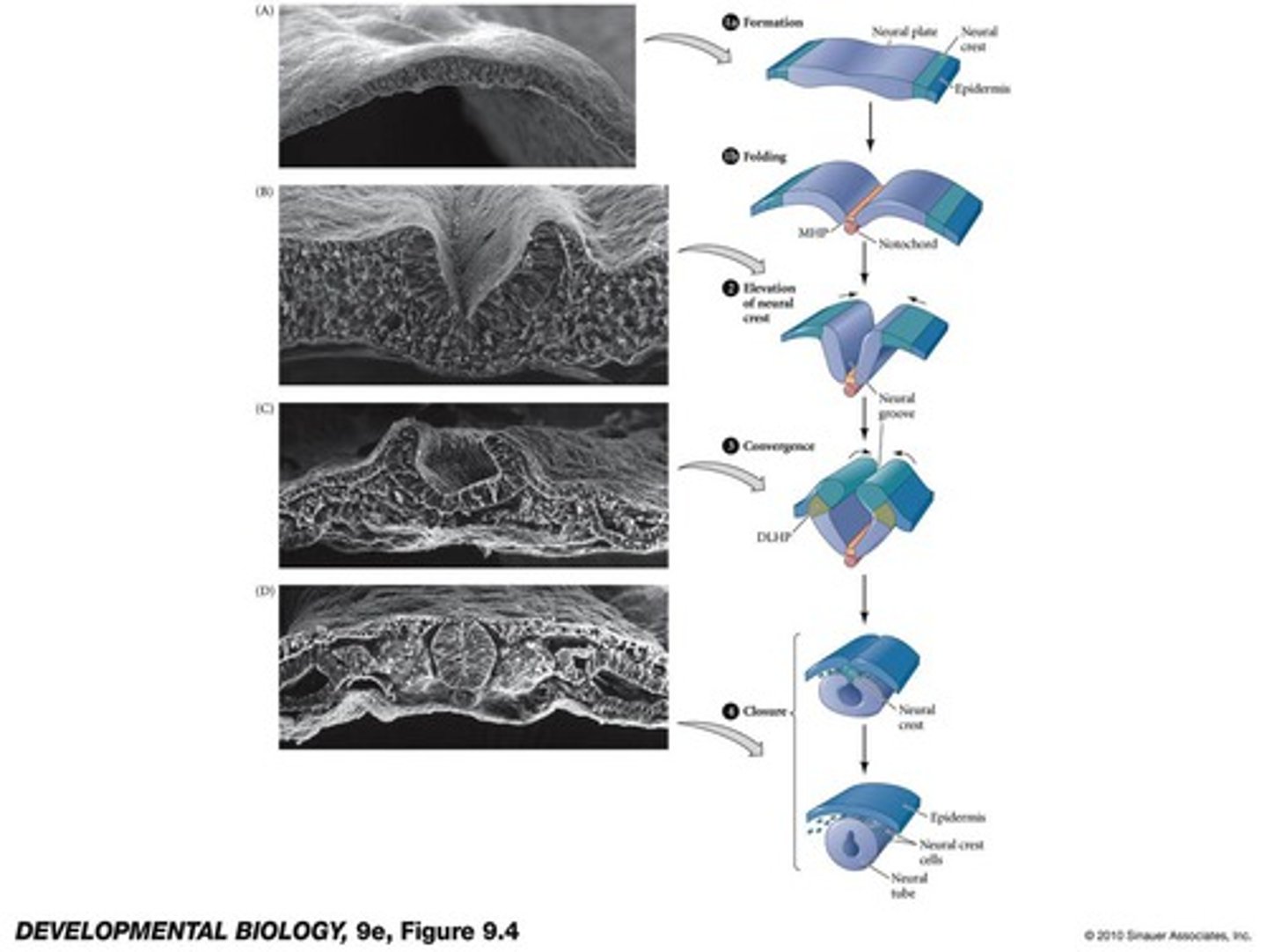
in primary neurulation, the original ectoderm divides into three groups of cells..
-neural tube: brain and spinal chord
-surface ectoderm: epidermis of the skin
-neural crest
role of cadherins in primary neurulation
aid in sorting heterogeneous cell populations into homogeneous groups, there is differential expression in CDH2 (N-cadherin) and CHD1 (E-cadherin) between the ectodermal cells that will become the neural tube vs the epidermis
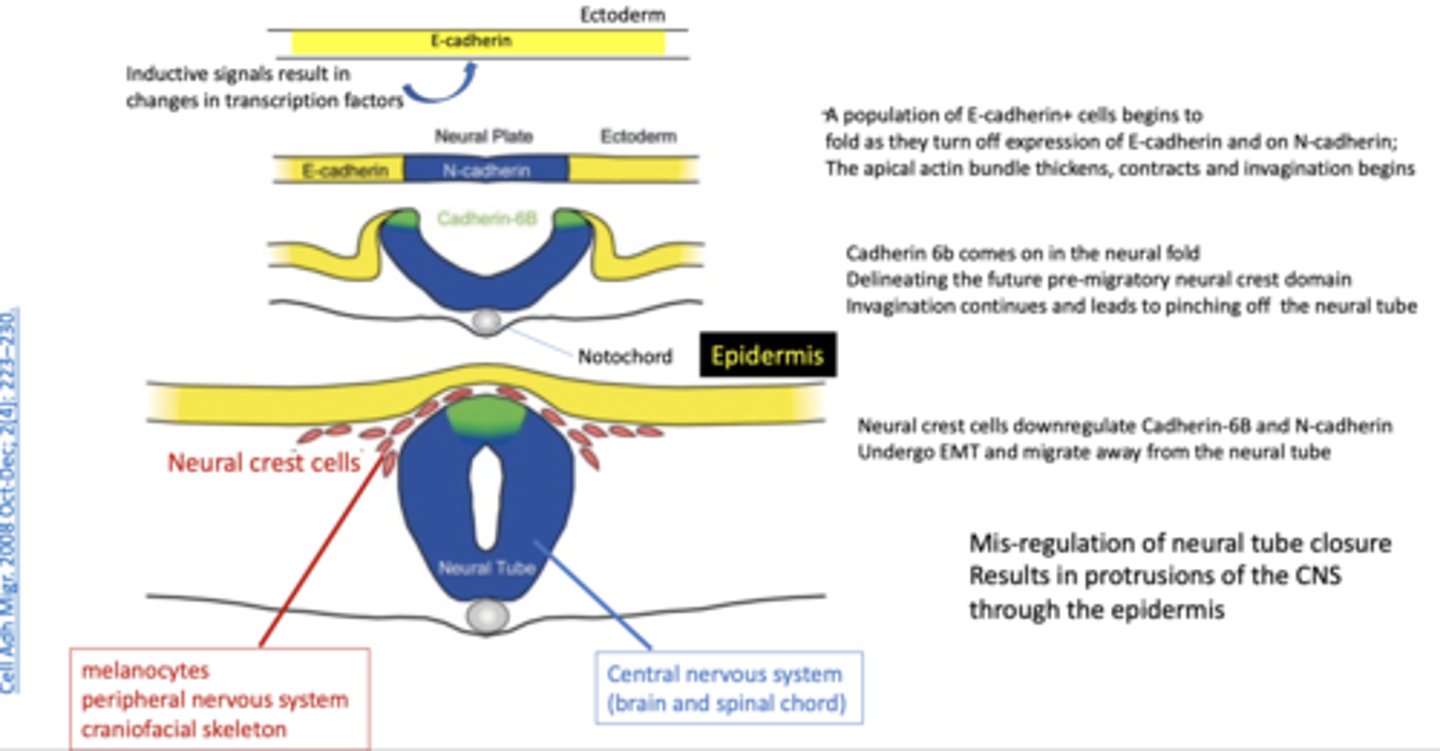
how to tell if theres good neurulation in vitro
columnar neuroepithelial cells, look like rosettes in vitro
rostrocaudal
head-to-tail
dorsoventral
back to stomach
in which direction is the neural tube pattered
both rostocaudal and dorsoventral
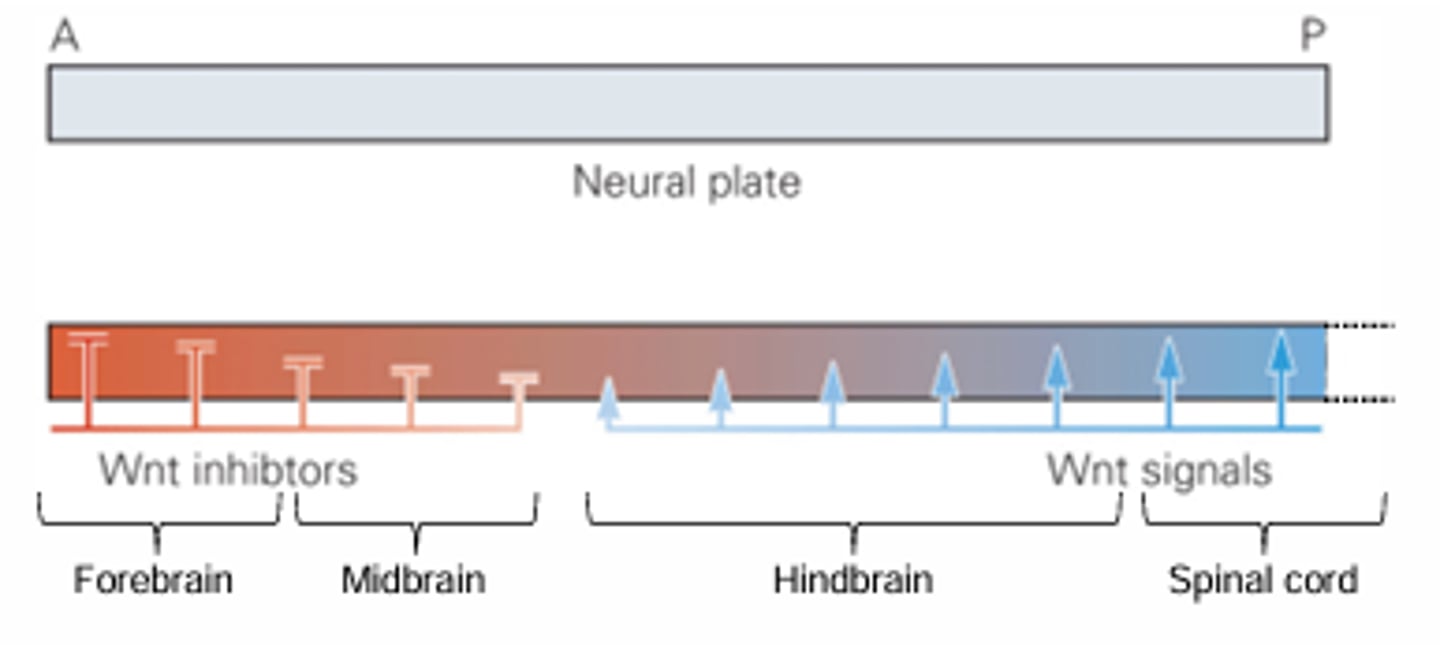
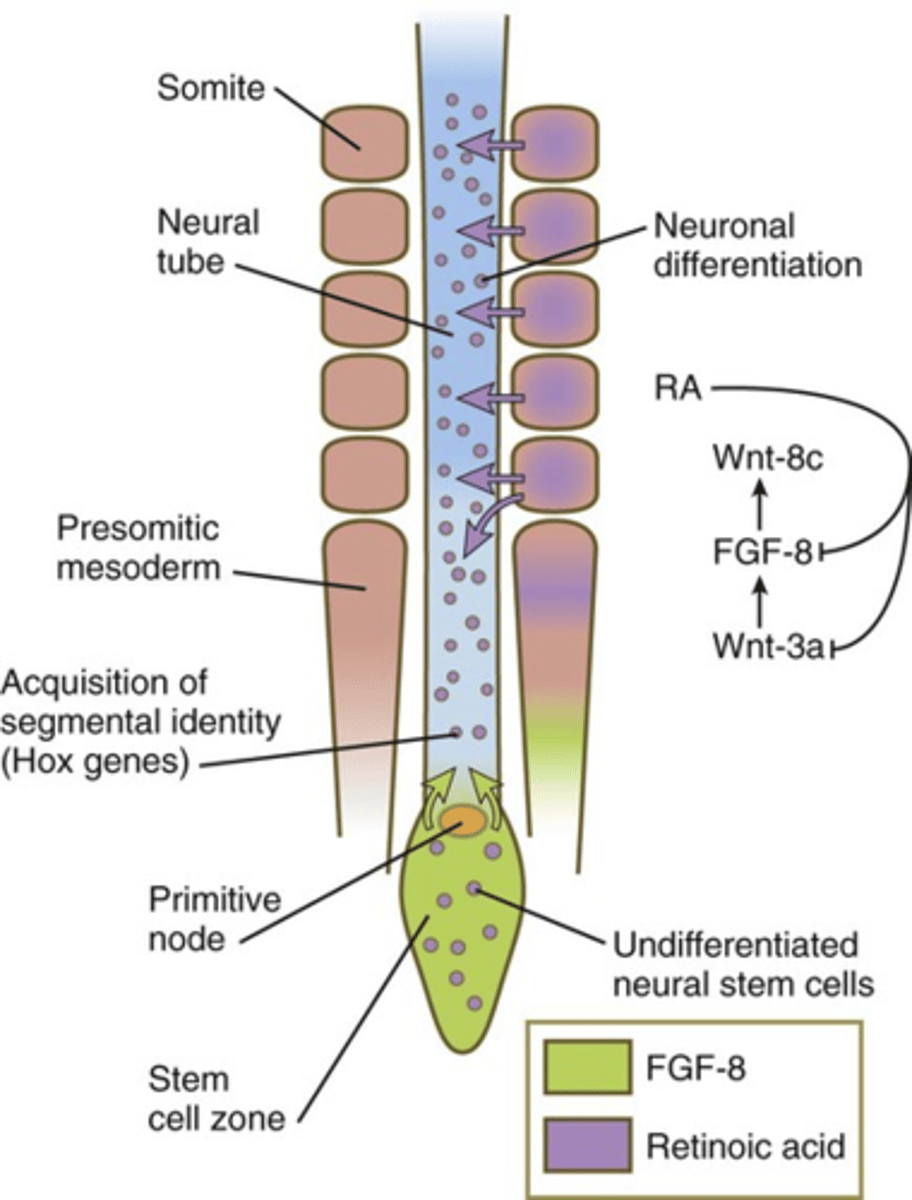
rostrocaudal patterning in the neural tube is most obvious in..
the development of vesicles of the brain (forebrain, midbrain, hindbrain) and spinal chord
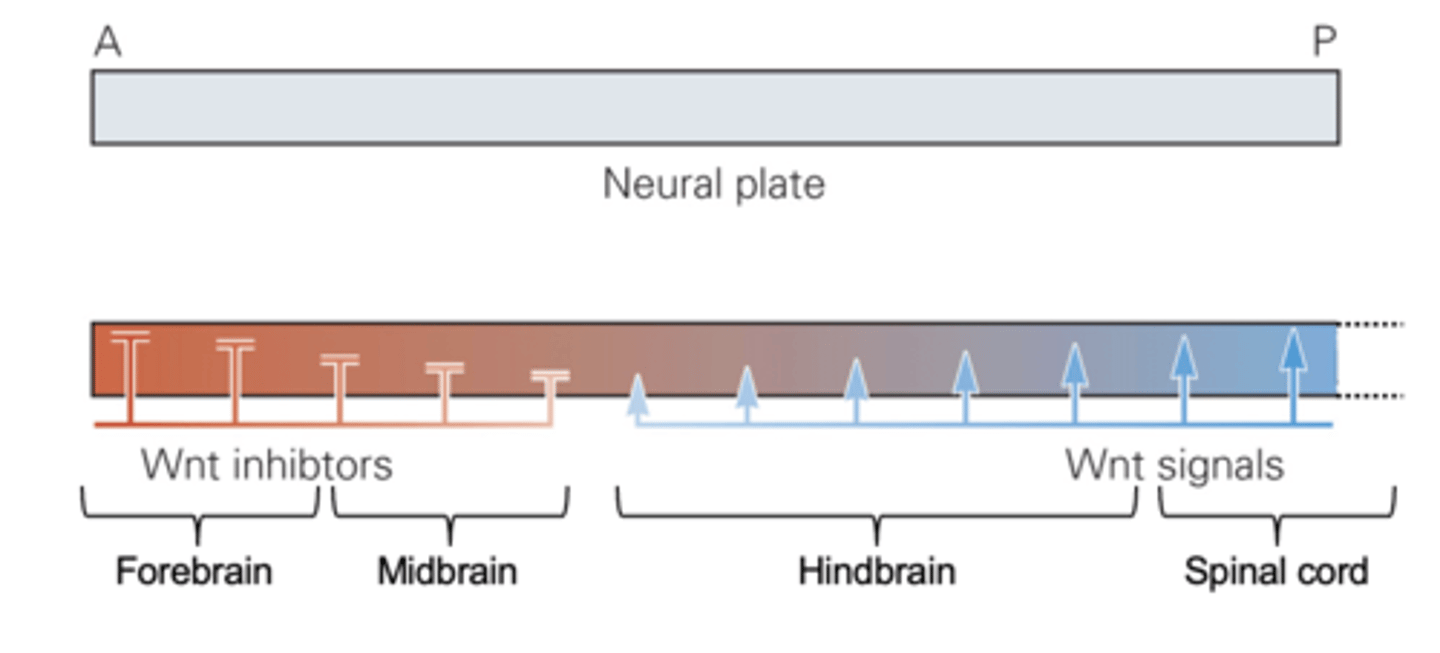
the graded presence of which four molecules are responsible for rostrocaudal patterning in the neural tube
BMP, FGF, Wnt, and retinoic acid
hox code
Unique combinations of Hox proteins along the body axis specify positional information. four clusters: HOX A, B, C and D
where is HOX code all found in development
rostrocaudal axis in neural tube (as discussed)
neural crest
paraxial mesoderm
surface ectroderm
Hensen's node
the front of the primitive streak
stem zone
a region in the brain that contains bipotent neuromesodermal progenitors. created by regression of hensens node
dorsoventral patterning in the neural tube
first, induced by sonic hedgehog (shh) secreted by notochord on ventral side/ floor plate
then, upon neural tube closure, the overlying ectoderm forms a BMP signaling center on the dorsal side/ roof plate
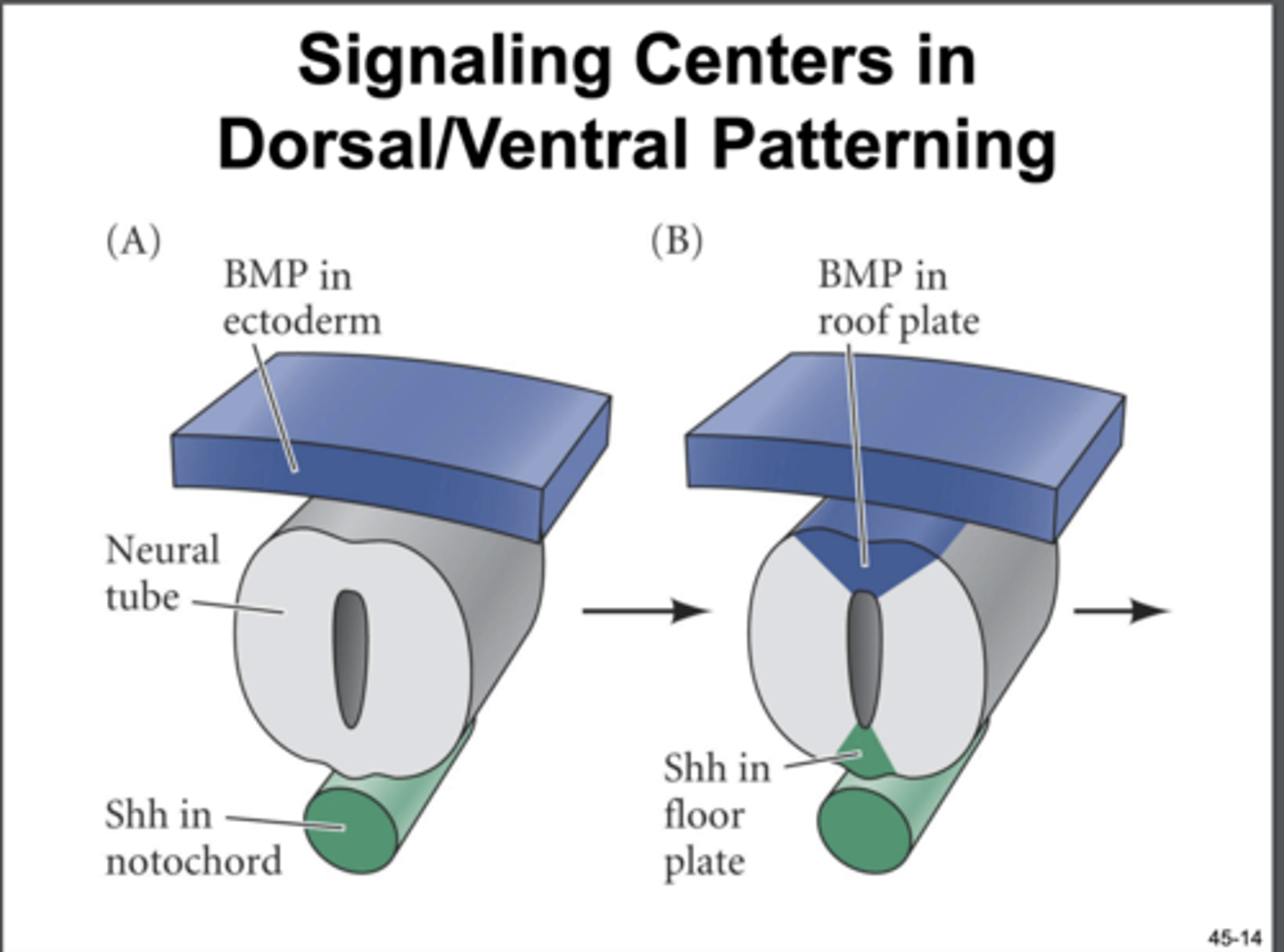
the graded presence of which molecules are responsible for dorsoventral patterning in the neural tube
TGF-B/ BMP, Shh. transcription factors induced by opposing gradients co-repress each other to generate a defined transcription factor code for 12 different progenitor domains
interkinetic nuclear migration
the nuclei of the neuroepithelial cells in the neural tube undergo a constant up-and-down migration through the stages of the cell cycle to divide. mitosis only takes place on the ventricular surface (moving outwards)
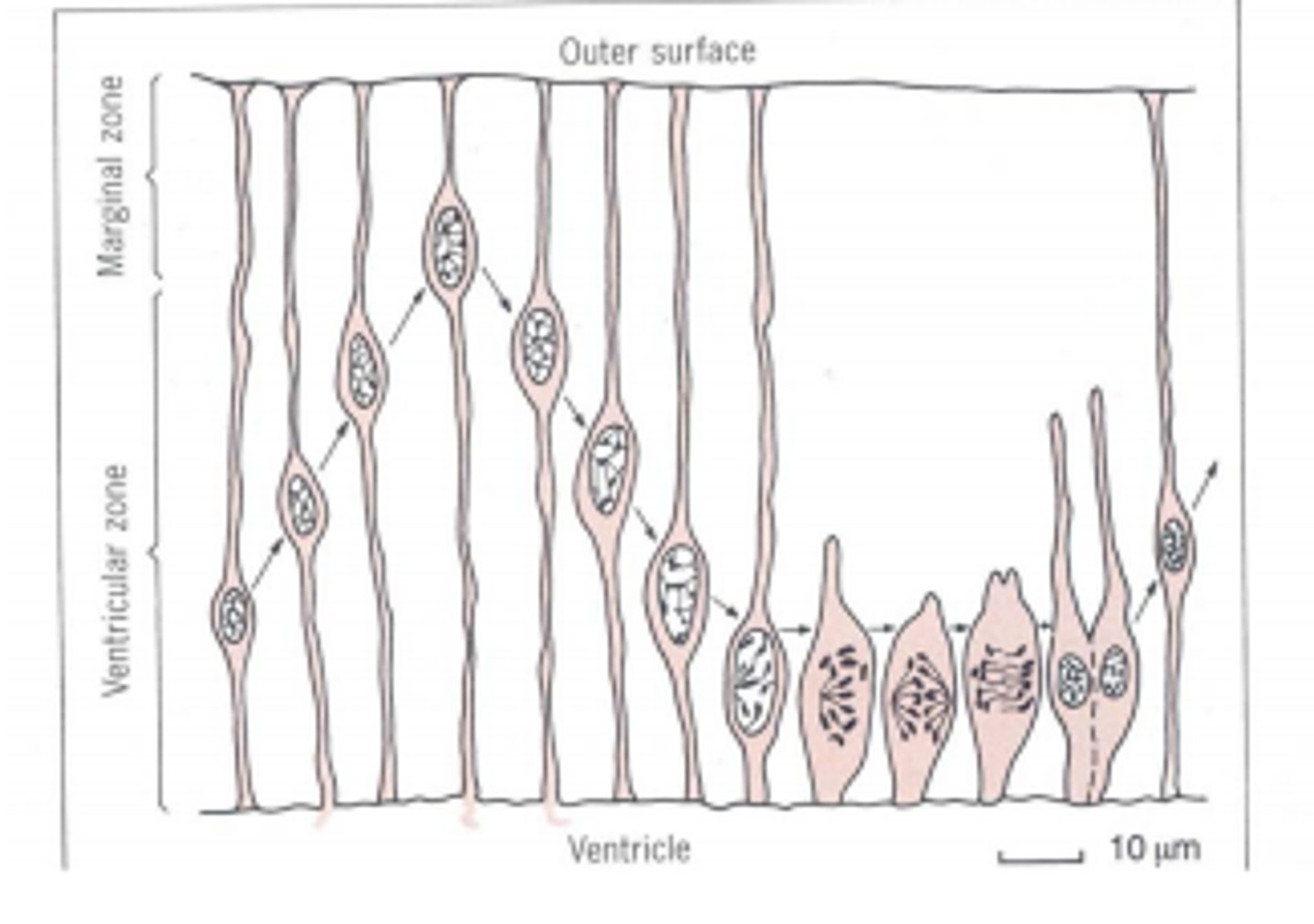
expansion of neural tissue
new born cells become post-mitotic and migrate along the axonal processes to form various CNS tissue
can hPSC's mimic neurulation
hPSC-derived neural stem cells keep some aspects of interkinetic nuclear migration and radial stratification spontaneously in vitro =neural organoids/ 'brain in a dish'
3 steps after neurulation
neurogenesis, astrogenesis, gliogenesis
steps of the very beginning of eye formation
neuroepithelium in the diencephalic vesicle (just a specific chunk of the neural tube) proliferates and extends until it comes in contact with the surface head of the ectoderm. This makes head ectoderm thicken and form the lens placode
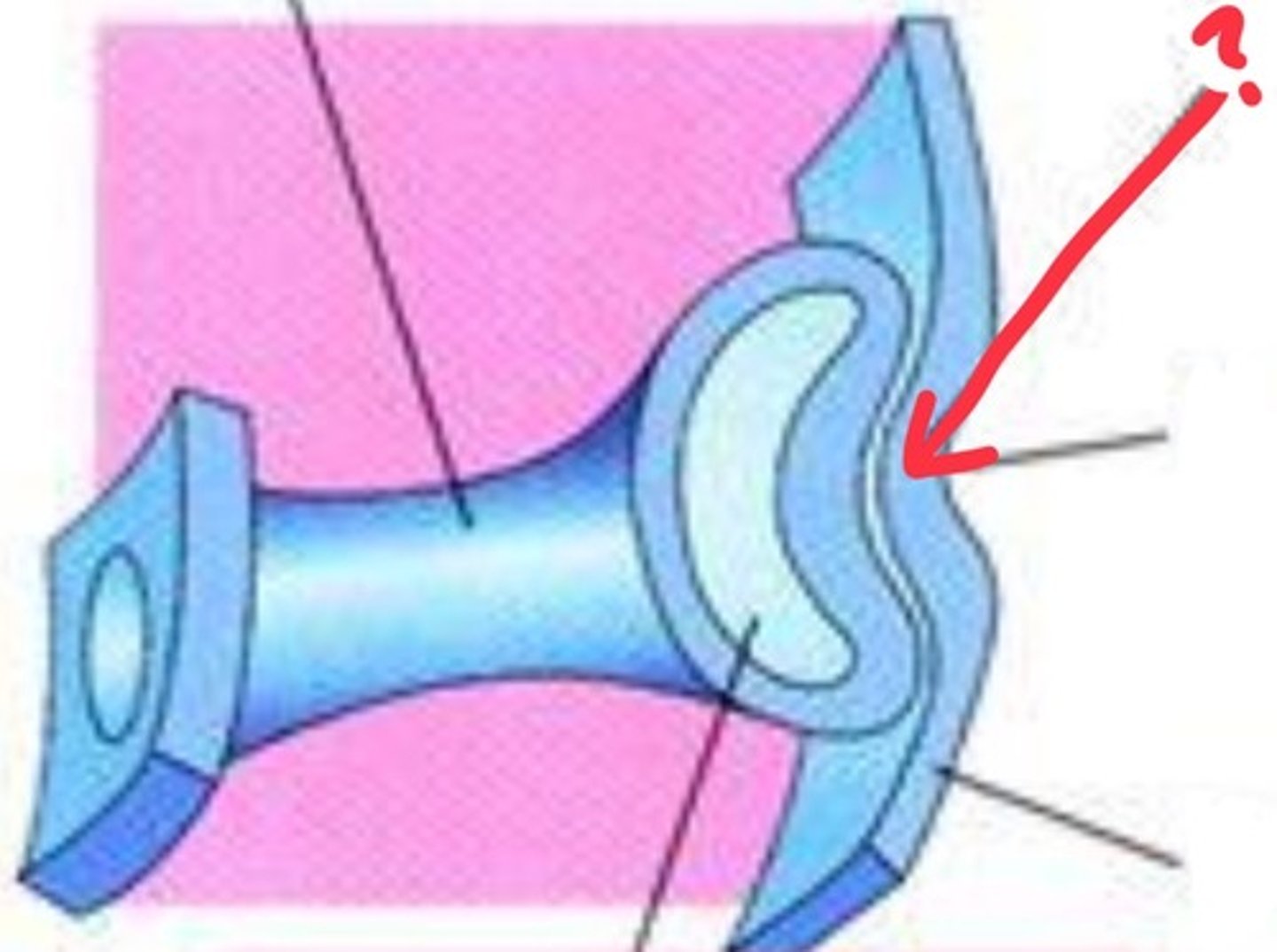
lens placode
thickening of surface ectoderm that invaginates to form the lens vesicle
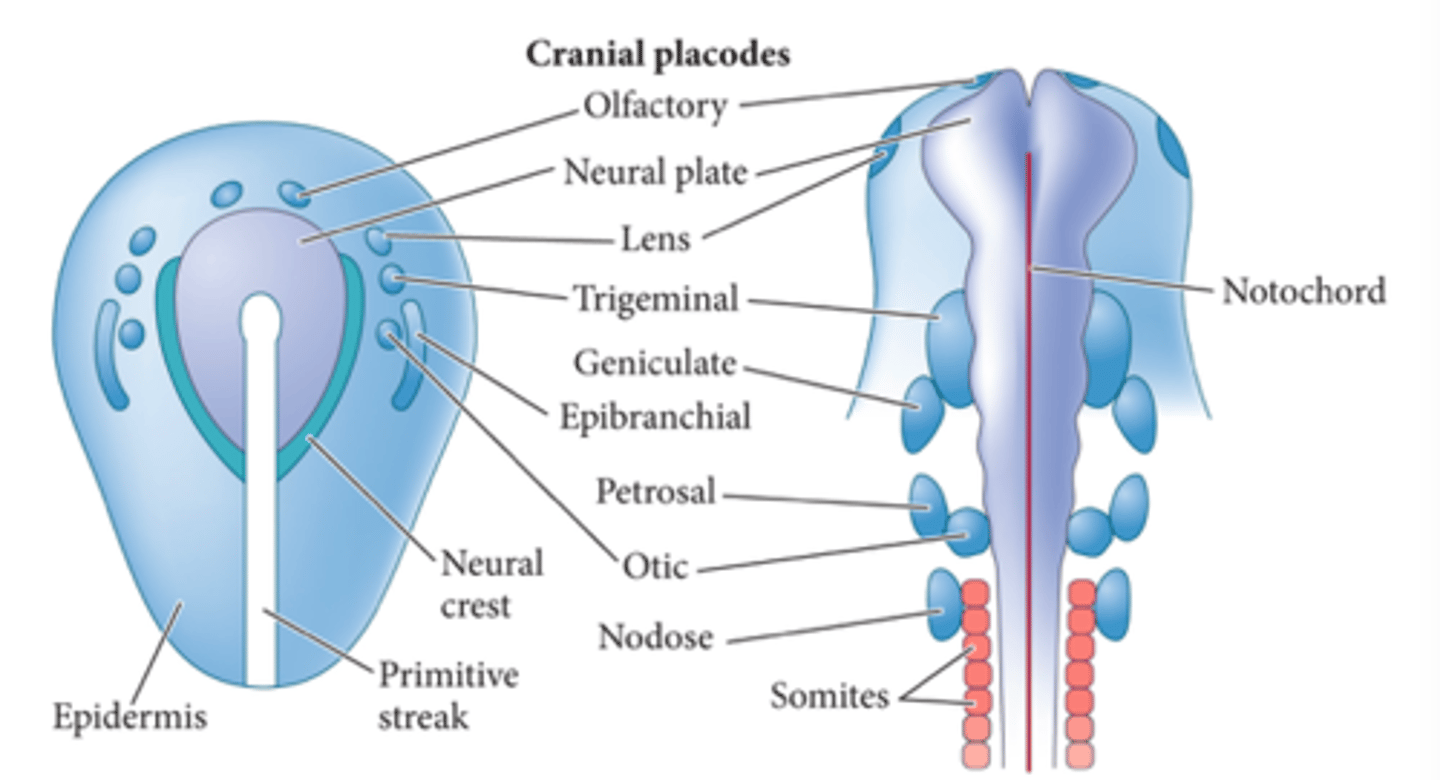
sensory placodes
form specific parts of special sensory organs. occur throughout the surface ectoderm and have some neurogenic potential
how does the lens placode further form the eye
lens placode induces optic vesicle to differentiate into pigmented versus neural retina and also signals to overlying ectoderm inducing corneal differentiation
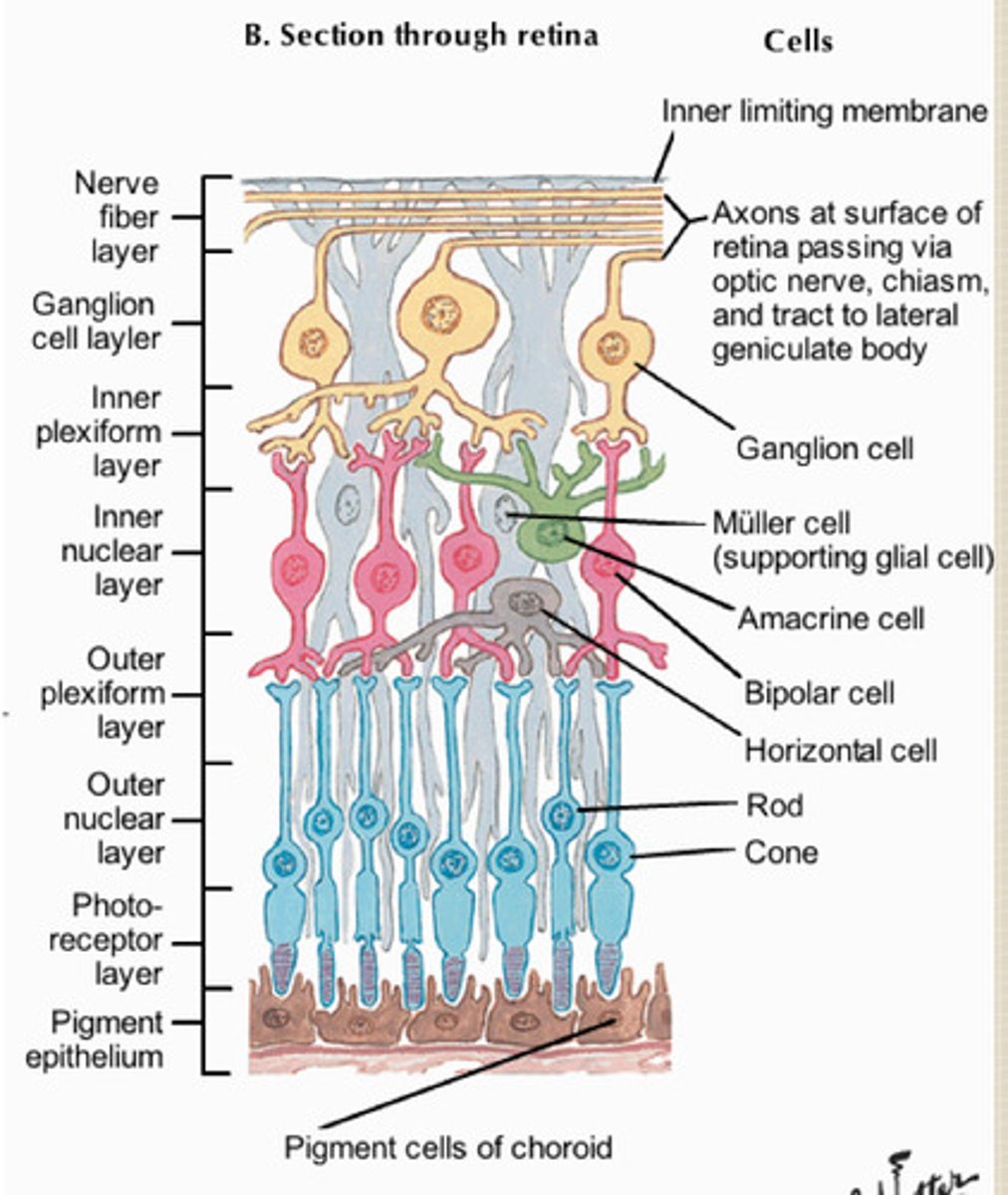
neural retina cells
act as neural stem cells and proliferate to form stratified layers with various glia, ganglion cells, interneurons, and photreceptors
what complex signaling interactions orchestrate this process
regulation of gene expression controls when and where each cell type of the retina and cortex arises
how did the research paper example begin to engineer the optic cup
mESC embryoid body is induced to a retinal cell using Pax6 and Rx GF's and activin and matrigel treatment. by day 10, epithelial vesicles invaginate without surface ectoderm to form optic cup-like structures
how is the gene expression profiles different between the distal and proximal layers of the cup?
distal/neural retina: Pax 6/ Rx/Chx10
Pigmented epithelium: Pax 6/ Rx/ Mitf
how close is the in vitro experiment to a real eye
only appears to be missing the lens from the surface ectoderm, the neural retinal cells were making structures similar to those found in post-natal
spatio-temporal dynamics of the retinal layer formation
1. progenitor cells differentiate into retinal neurons
2. plexiform layers emerge, becoming stratified between nuclei layers
3.photoreceptor development, differentiation of glial cells
"fourth germ layer"
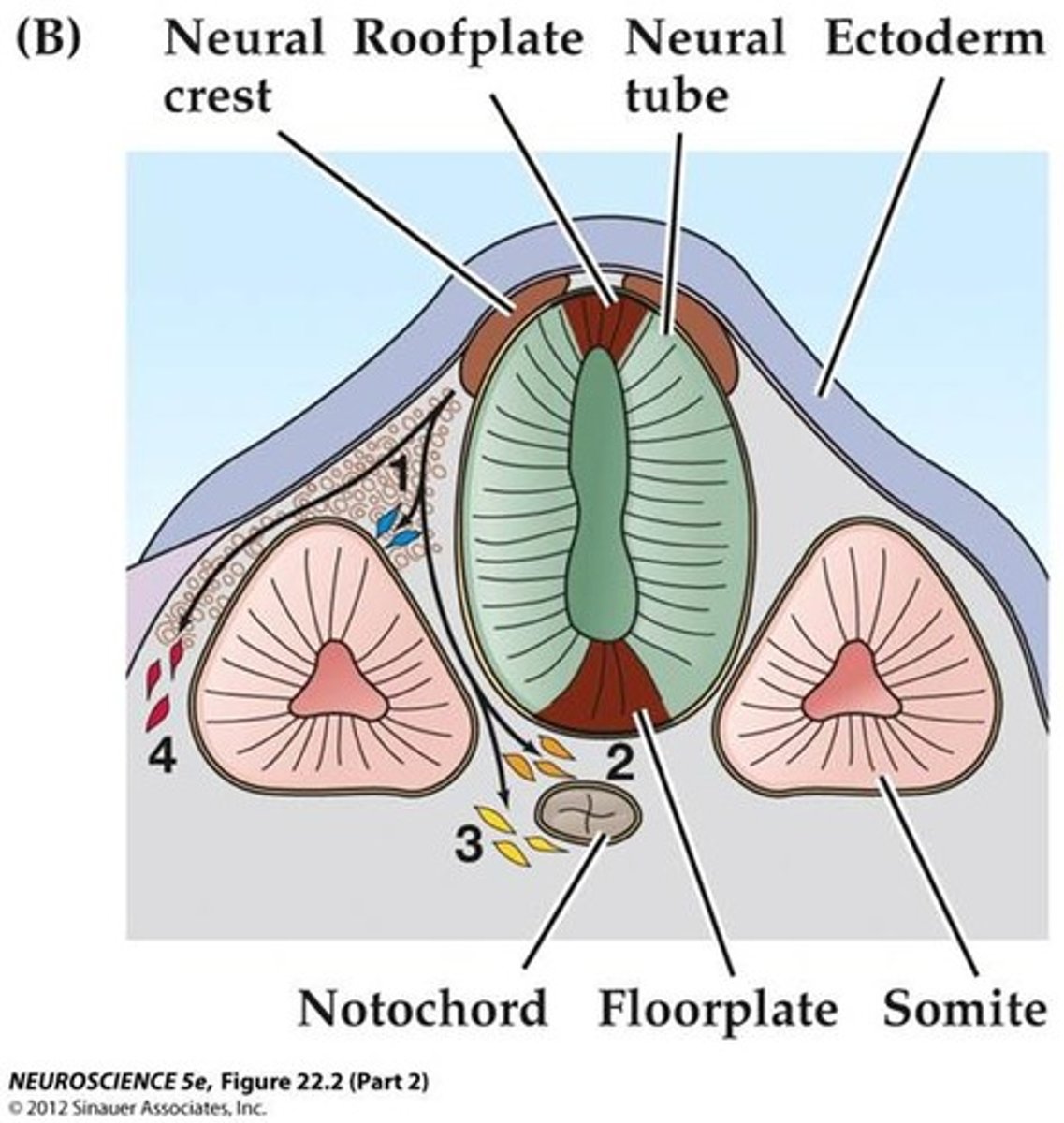
neural crest cells: they migrate extensively and generate a ton of differentiated cell types. fate determined by where they end up
some differentiated cell types of neural crest cells
-neurons and glial of sympathetic and parasympathetic nervous system
-epinephrine cells of adrenal gland
-skeletal and connective tissue of head
neural crest transcription factors
Pax7, Snail2, Sox9, FoxD3, induced again by BMPs and Wnts, same as neural tube
where are neural crest cells generated?
during closure of the neural tube, all along the rostrocaudal axis
do we fully understand what guides neural crest cells migration and differentiation
nah
main categories of neural crest cells
cranial
cardiac
trunk
vagal and sacral
how do the axons of neurons know how to connect properly to target organs?
growth cones in axons can sense and respond to signals. different transcription factors expressed by neurons in different HOX regions of the spinal cord impart the cells with different cell surface receptors
limb bud
lateral motor neurons of the spinal cord project to limb muscles, so technically from ectoderm
retinotectal patterning
the process by which the visual information from the retina is organized and projected onto the tectum (midbrain) in a precise, topographic manner, forming a "retinotopic map" that allows for accurate visual processing and behavior
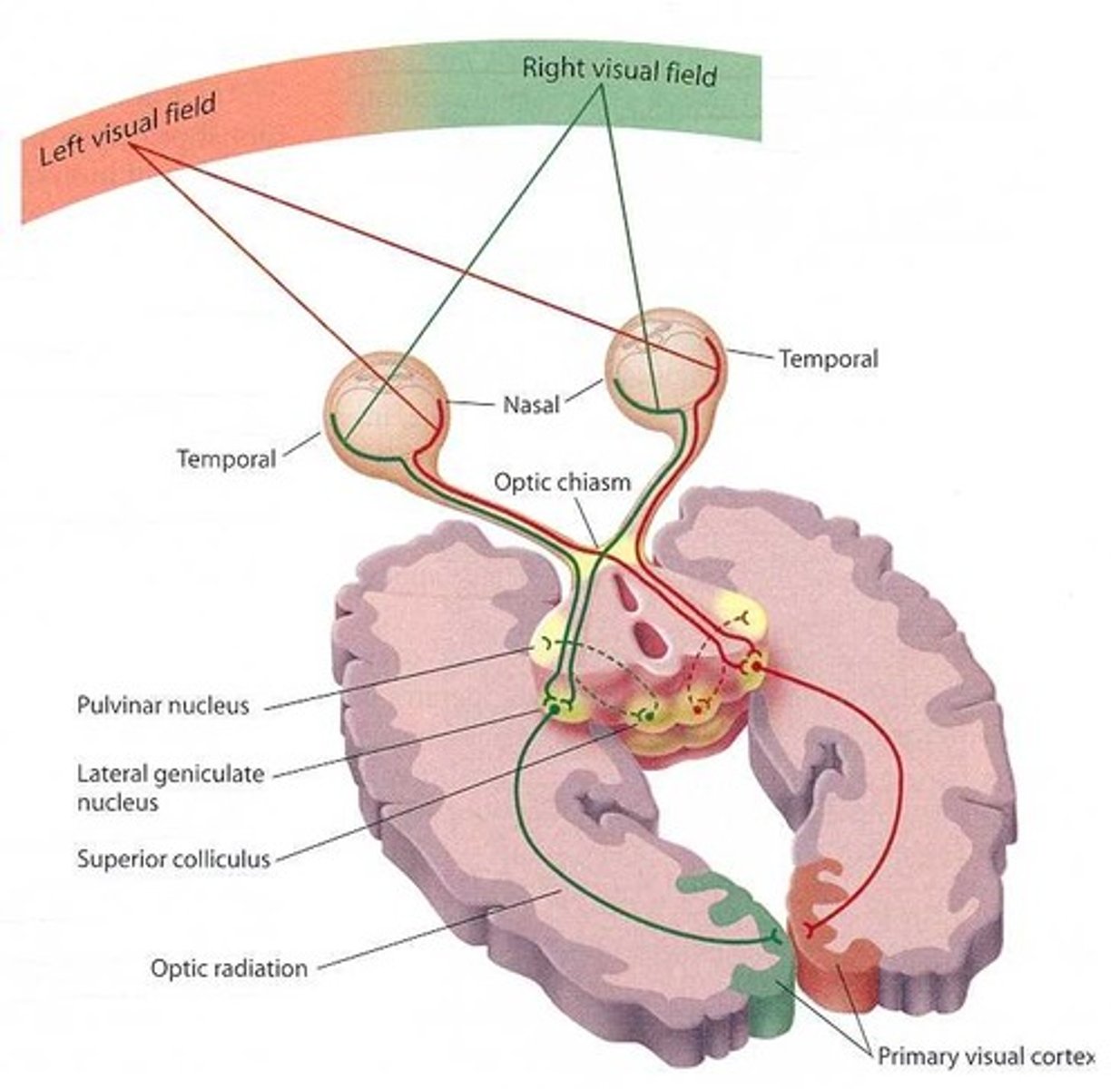
why is retinotectal patterning a great example of axonal guidance?
multiple cues are used to guide nerve axons from the retina to various parts of the tectum in the midbrain
how does this patterning work?
RGC axons go from the inner margin of the retina towards the optic disc. When the signal travels along this axon and reaches the optic tectum, the gradient expression of Ephrin ligands by tectum and differential expression of eph receptors by the axons tell the axons where to terminate
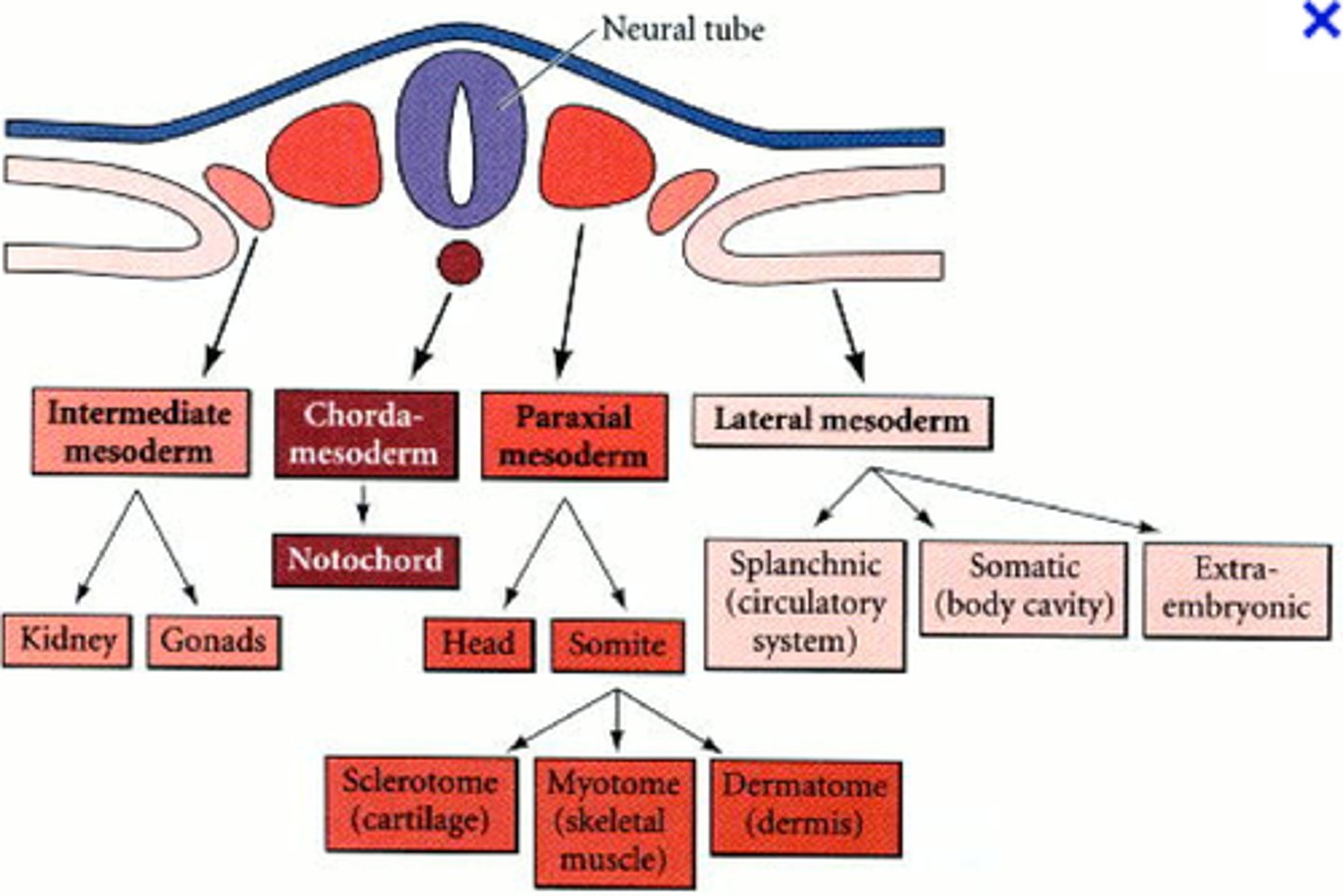
tissues of the mesoderm
-lining of digestive and respiratory tracts
-muscles
-skeletal system
-cardiovascular system
-gonads, kidneys
through what transcription factors is the mesoderm formed?
Brachyury (T) and Tbx6, induced by HOX code/ wnt, BMP, FGF and Ra gradients
how is the mesoderm formed
The mesoderm is formed by cells migrating between the ectoderm and endoderm during gastrulation
Chordamesoderm
notochord: only present during early development
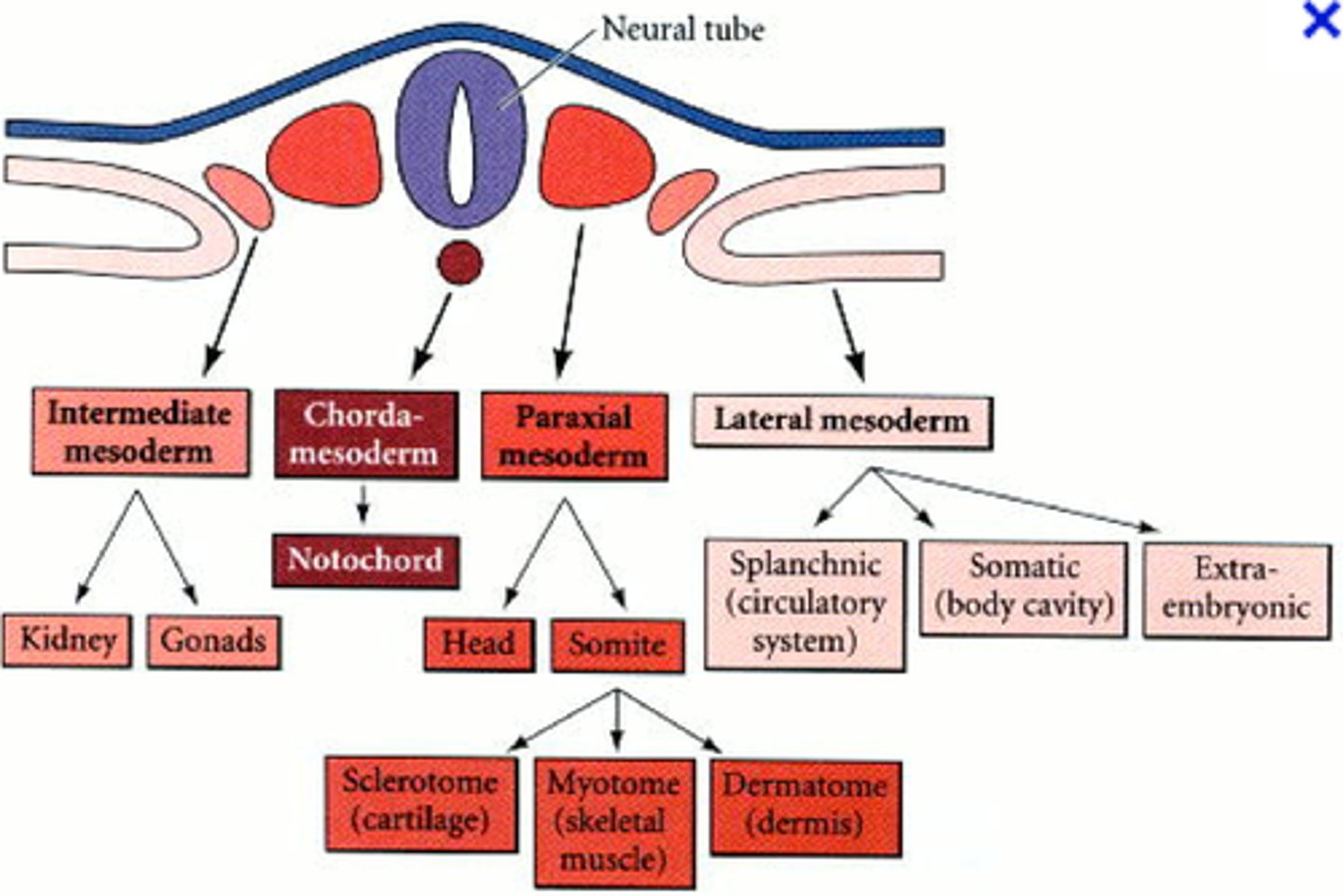
paraxial mesoderm
closest to neural tube: forms somites that produce connective tissues of the back (muscle, vertebrae, ribs, intervertebral vasculature, etc)
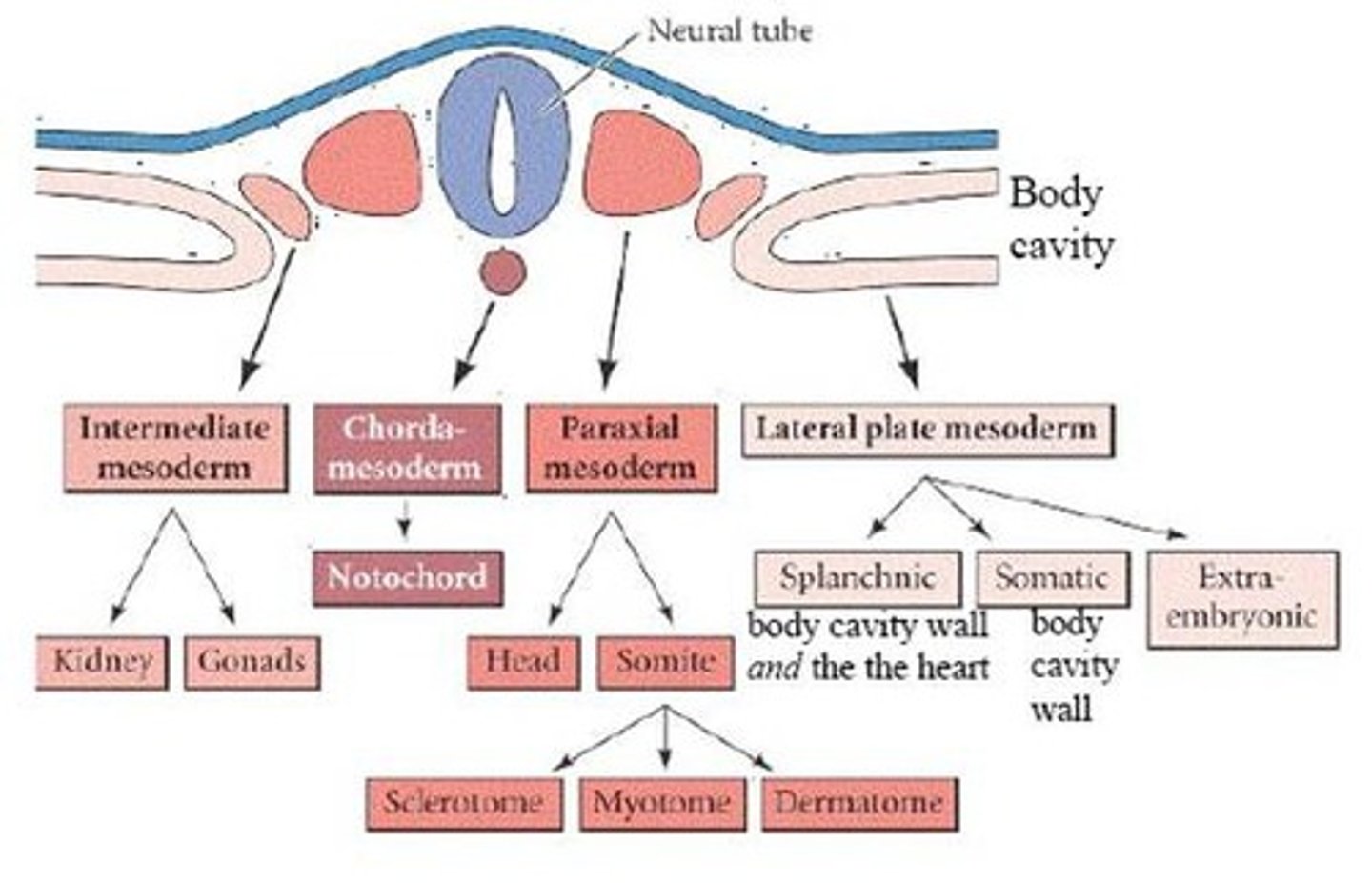
intermediate mesoderm
second from the neural tube: forms urogenital system (kidneys and gonads)
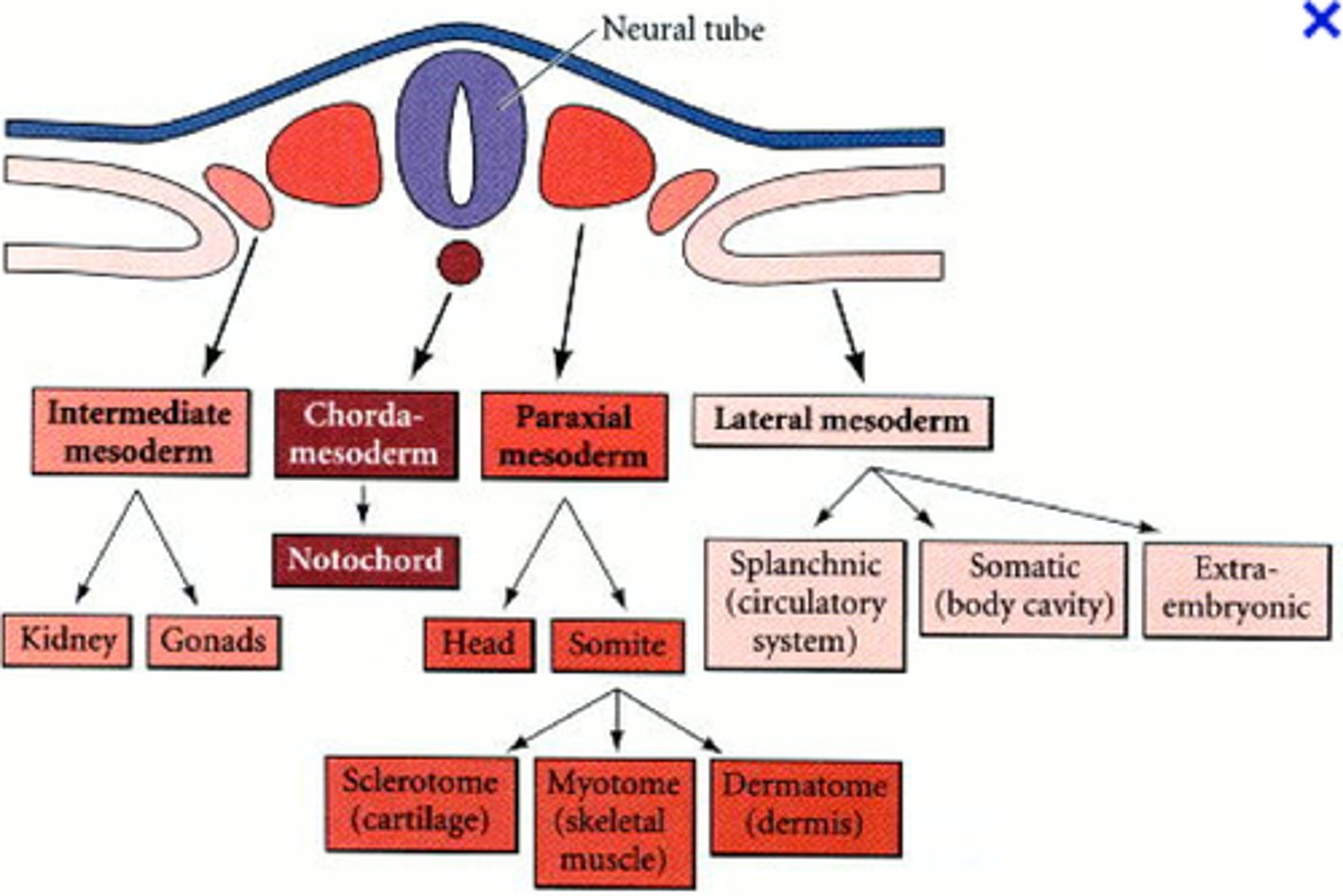
lateral plate mesoderm
furthest from the neural tube: forms the organs of the circulatory system, body cavity lining, pelvic and limb skeleton (*limb musculature is from paraxial mesoderm)
head mesoderm
skull, muscles and connective tissue of head, dentine. unsegmented anterior paraxial mesoderm
somites
segmental structures in the mesoderm
Somitogenesis steps
salt and pepper expression of MESP2 in somite segment -> somite compartmentalizes into anterior and posterior identity
mediolateral mesodermal patterning to form the 4 mesodermal sections (paraxial, intermediate, lateral plate)
four mesodermal subdivisions specified along this axis by increasing amounts of BMP and Fox. BMP4 is expressed at high levels in the lateral mesoderm and you can change specification by altering BMP expression, but true mechanism of is unknown
when does mesoderm form in relation to ectodermal tissues
synchronously
when and where does somite formation occur
paraxial mesoderm forms somites when the primitive streak regresses first and BMP signals are repressed. Timing is important
what direction does somitogenesis occur in
rostral-caudal direction
five important components of somitogenesis
1. periodicity
2. fissure formation
3. epithelialization
4. specification
5. differentiation
1. periodicity
clock and wave mechanism: timing of somites is regulated by negative feedback loops with Notch signaling. Its cynical because there are time delays for transcription and translation of the Notch
-The "clock" = oscillating Notch genes
-The "wavefront" = the point where cells become competent (determined) due to the low FGF/Wnt and high retinoic acid.
how long does it take to develop a somite and how many somites form
new somite every 90 mins, appearing on both sides of the neural tube in the same number. total # determined by species
Notch signaling pathway
-FGF8/Wnt signals are high in the posterior and suppress Delta, a Notch ligand
-in the anterior pre-somatic tissue, retinoic acid replaces FGF/Wnt allows cells to start responding to Notch signaling and form somites
marker of finished somite segmentation
Mesp2
2. fissure formation
Mesp2 induces EphA4 in the postreior border cells, which induces EphB2 in the cells across the border, and the repulsion of these two creates a fissure-> somites segregate from each other
3. epithelialization
ectodermal signals act on GTPases in border cells to initiate formation of epithelial cells: epithelial cells on border and mesenchymal cells in the middle
4. specification
-along rostal-caudal axis
-governed by HOX code
can a somite be re-specified like iPSCs?
NO, hox code is present prior to formation of somites and cannot be respecified
5. differentiation
paracrine factors from the neural tube, notochord and epidermis and intermediate mesoderm influence adjacent somite regions
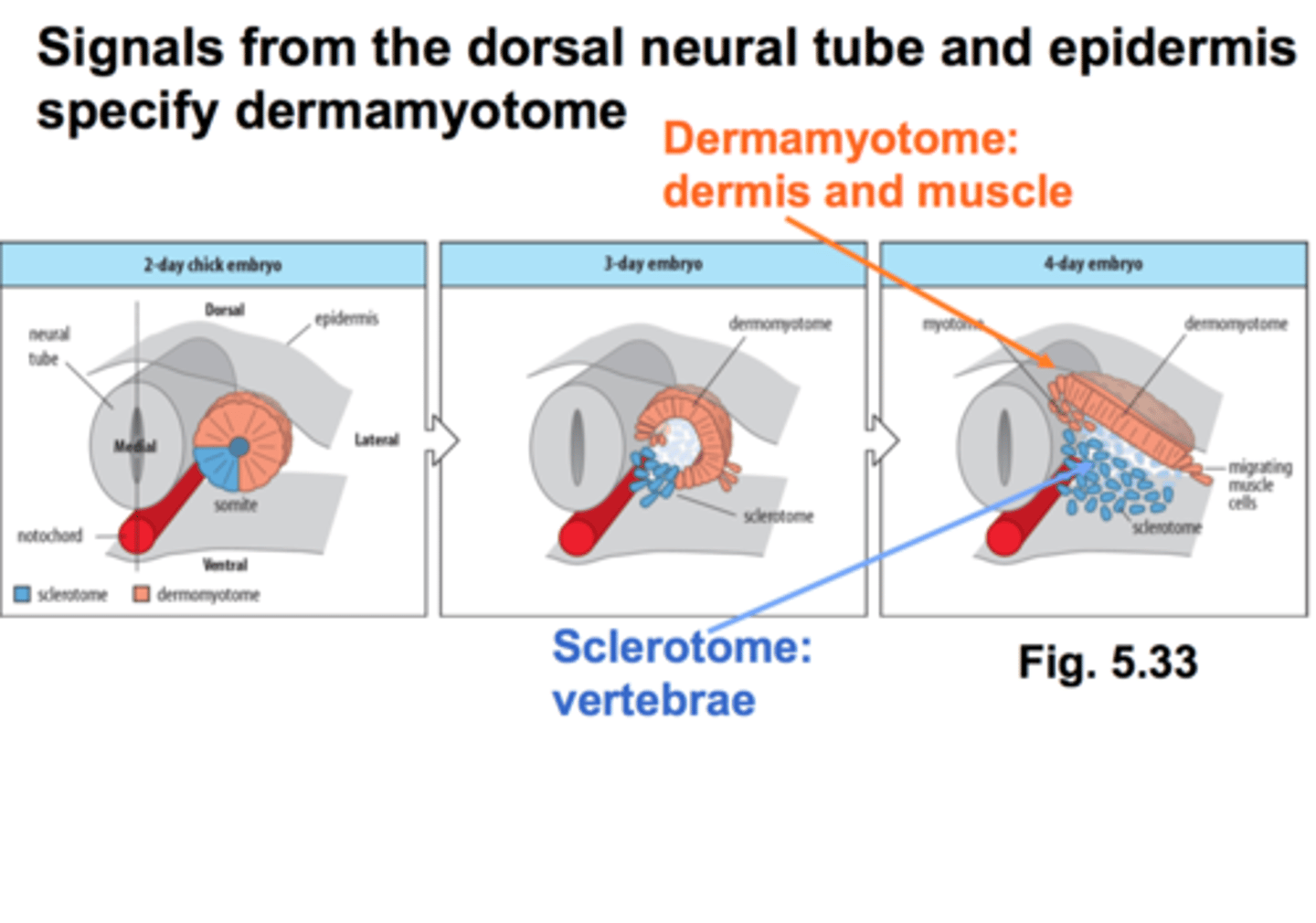
the paraxial mesoderm forms somites and then what do the somites differentiate into
sclerotome and dermomyotome
sclerotome
somites closest to notochord: forms vertebrae and ribs
dermamyotome
becomes the dermis and skeletal muscle
satellite cells
stem cells found in adult muscles
Where do myoblasts origninate from?
cells in the central dermatome break off from the epithelial plate
where does all skeletal musculature in the vertebrate body come from
dermamyotome (except for head muscles)
Basic helix-loop-helix (bHLH)
family of transcription factors like MyoD and Myf5
------ signaling to the primaxial and abaxial dermamyotome induce transcription of-----
Wnt and Shh, MyoD and Myf5
what does MyoD do once activated?
positively regulates its own gene expression
what factors do satellite cells express
Pax3 and Pax7, which combined, repressed MyoD expression, thus preventing muscle differentiation and keeping it in a pluripotent state
steps of myogenesis
1. MyoD expressing myoblasts align and fuse to form multi-nucleated myotubes
2. they secrete fibronectin into the ECM and grab on with integrins
3. they align (using caherins) and fuse
4. recruit other myotubes to fuse
myoblasts must become ---before they can fuse
post-mitotic (no FGF or Shh signaling) but must express myogenin
where do satellite cells sit in adults
The basal lamina surrounding the myotube
which parts of the mesoderm form which bones?
somites-> vertebrae and ribs
lateral plate mesoderm-> limb skeleton
Neural crest (ectoderm)-> pharyngeal arches and craniofacial bones and cartilage
endochondral ossification
Process of transforming cartilage into bone.
step one of osteogenesis
mesenchymal cells commit to cartilage lineage by expressing Pax1 in response to Shh
step two of osteogenesis
Pax 1 committed mesenchyme condenses into compact nodules and differentiate into chondrocytes, depends on BMP and FGF
role of BMPs in step two of osteogenesis
BMP's induce cadherins and Sox9 expressions, which induces expression of collagen 2 and aggrecan
step 3 of osteogenesis
proliferation and secretion of cartilage-specific ECM, triggered by Wnt expression
step 4 of osteogenesis
become post-mitotic and hypertrophic via Runx2 expression...adding collagen 10 and fibronectin to matrix to enable mineralization and VEGF for vascularization
step 5 of osteogenesis
hypertrophic chondrocytes die and are replaced with osteoblasts brought in with invading vasculature
precursors of osteoblasts
sclerotomal precursors
Osterix
Required for final commitment of progenitors to preosteoblasts
what do osteoblasts become
osteocytes or bone lining cells
Osteoclast
bone cell that absorbs and removes unwanted bony tissue, derived from blood cell lineage of the lateral mesoderm