Major SEE/NCE Flashcard
1/247
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
248 Terms
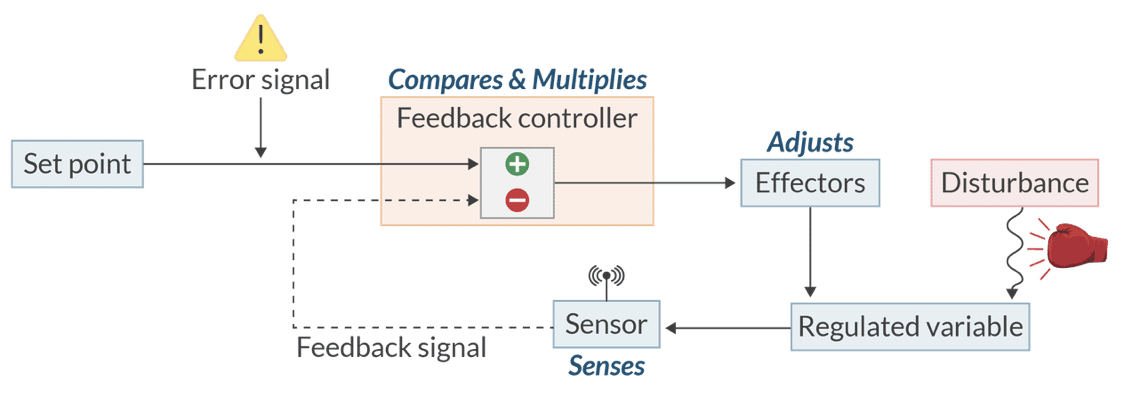
In a physiologic control system, which component is responsible for comparing the input signal with the internal reference value?
The feedback controller (or integrator).
(It assesses the difference between the actual input and the desired set point, adjusting outputs to maintain homeostasis.)
Explain the concept of autocrine signaling and provide a clinical example involving norepinephrine.
Autocrine signaling occurs when a cell responds to its own secreted molecules, such as norepinephrine, binding to presynaptic alpha2 receptors to inhibit further release.
Where are the sensors located that detect changes in H+ concentration within the cerebrospinal fluid for respiratory control?
Central chemoreceptors in the retrotrapezoid nucleus of the medulla.
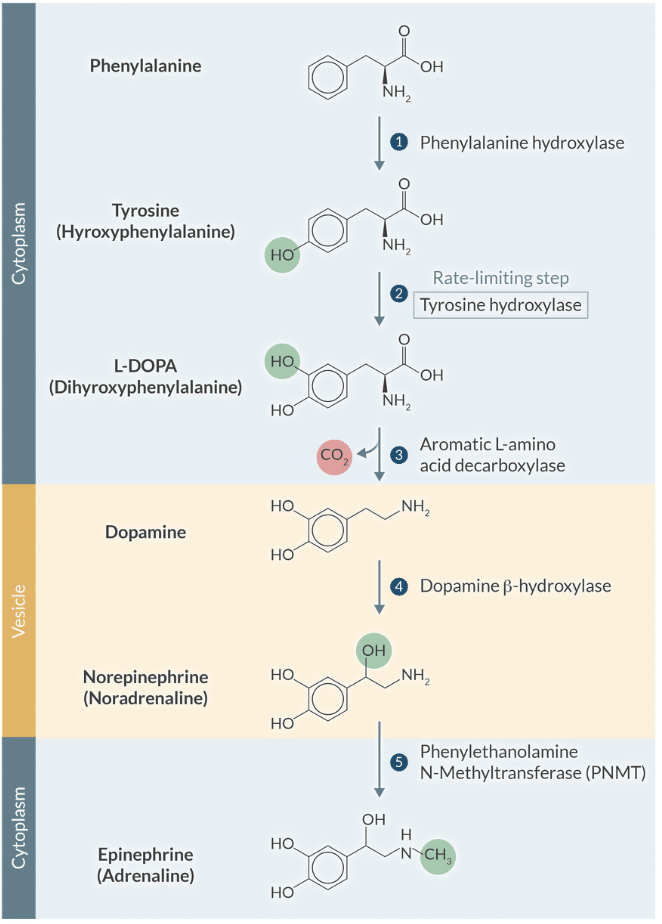
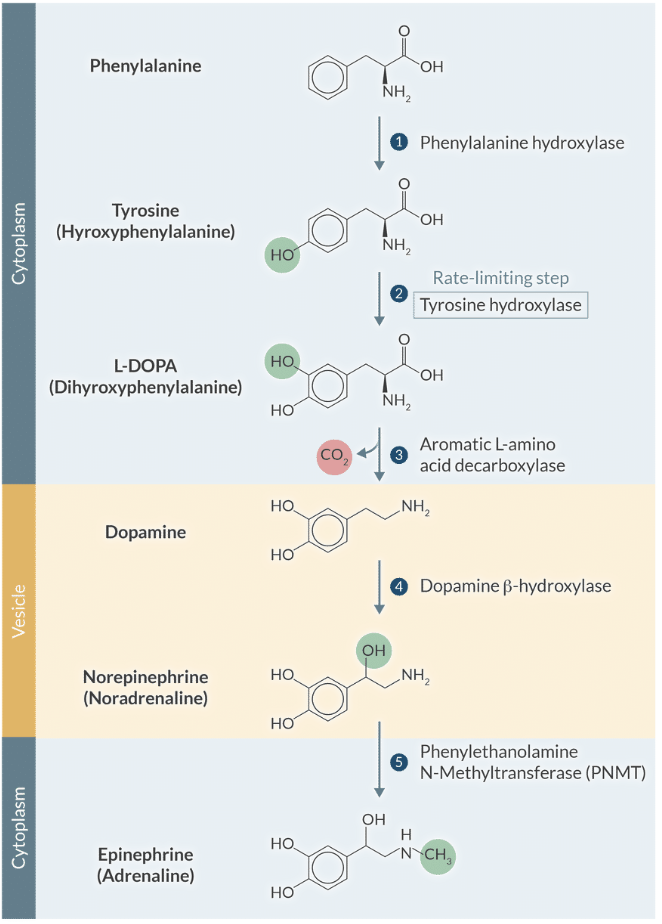
Explain the rate-limiting step in catecholamine synthesis and how it is physiologically regulated.
The hydroxylation of tyrosine by tyrosine hydroxylase is the rate-limiting step and is regulated by feedback inhibition from dopamine and norepinephrine.
Which type of feedback system is characterized by reversing a disturbance to maintain stability and homeostasis?
Negative feedback
During parturition, what serves as the 'sensor' in the positive feedback loop?
Stretch receptors in the cervix.
Identify three specific physiological examples of beneficial positive feedback mentioned in the text.
Childbirth (parturition)
The blood coagulation cascade
The generation/propagation of an action potential
Explain the G-protein coupling for alpha1, alpha2, and beta-adrenergic receptors.
Alpha1 receptors couple to Gq proteins, leading to phospholipase C activation and increased intracellular calcium.
Alpha2 receptors couple to Gi proteins, inhibiting adenylate cyclase and reducing cAMP levels.
Beta-adrenergic receptors couple to Gs proteins, stimulating adenylate cyclase and increasing cAMP levels.
Explain the cellular mechanism by which beta1 stimulation increases cardiac inotropy.
Activation of the Gs pathway increases cAMP, activating Protein Kinase A to phosphorylate L-type Ca2+ channels and increase Ca2+ influx.
Explain the mechanism of beta1-mediated lusitropy and why it is essential for cardiac hemodynamics.
Phosphorylation of phospholamban by PKA relieves inhibition of the SERCA2 pump, accelerating Ca2+ reuptake to promote faster myocardial relaxation.
Explain how beta1 stimulation increases chronotropy at the cellular level in SA nodal cells.
Phosphorylation of L-type Ca2+ channels and phospholamban leads to an increased rate of Phase 4 depolarization, resulting in a faster heart rate.
Explain the physiological significance of presynaptic alpha2 receptors for a CRNA managing sympathetic outflow.
Activation of these Gi-coupled receptors inhibits the release of norepinephrine, providing a negative feedback mechanism to reduce sympathetic tone.
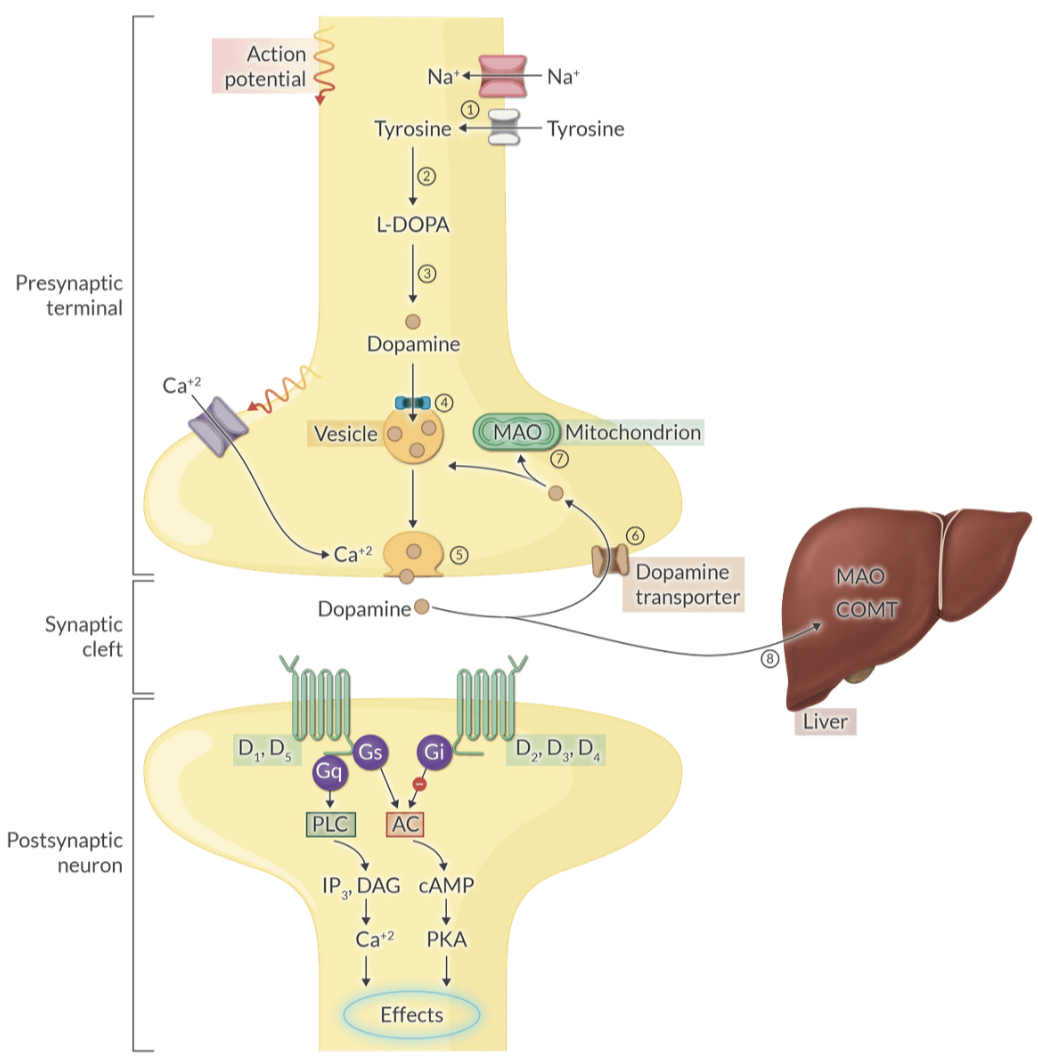
Explain the primary mechanism for the termination of catecholamine action in the synaptic cleft.
Active reuptake into the presynaptic terminal is the most important mechanism for removing catecholamines from the synaptic cleft.
Explain the metabolic pathway of dopamine and its primary end-product excreted in the urine.
Dopamine is metabolized by MAO and COMT into homovanillic acid (HVA), which is then eliminated by the kidneys.
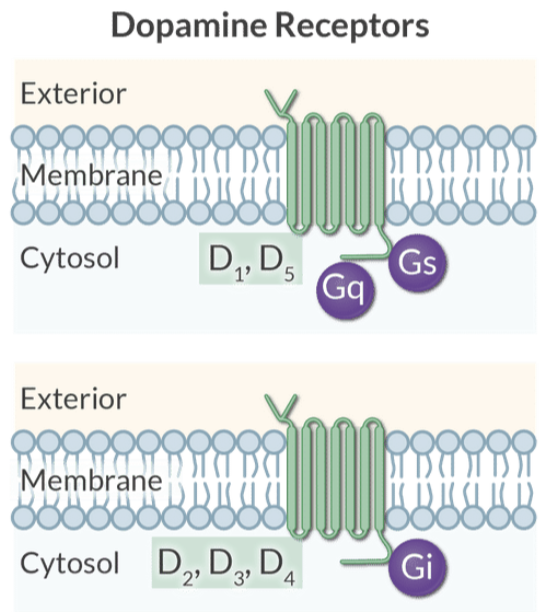
Explain the intracellular coupling of D1-like versus D2-like dopamine receptors.
D1-like receptors (D1, D5) activate adenylyl cyclase and PKA.
D2-like receptors (D2, D3, D4) inhibit adenylyl cyclase.
Explain the clinical consequences of overactive D2-like receptors and degenerating dopaminergic neurons in the substantia nigra.
Overactive D2 receptors are implicated in schizophrenia
Degeneration of neurons in the substantia nigra leads to Parkinson's disease
Explain the unique ionotropic property of the 5-HT3 receptor compared to other serotonin receptors.
The 5-HT3 receptor is a nonselective cation channel (inotropic) that causes rapid membrane depolarization, unlike most other serotonin receptors which are metabotropic.
Explain why 5-HT3 antagonists like ondansetron are effective for treating postoperative nausea and vomiting.
They block 5-HT3 receptors in the vagus nerve and area postrema, which are critical components of the body's vomiting reflex center.
Explain the physiological role of histamine in the CNS and the clinical side effect of first-generation antihistamines (Benadryl).
Histaminergic neurons regulate arousal and attention, so blocking these receptors frequently results in drowsiness.
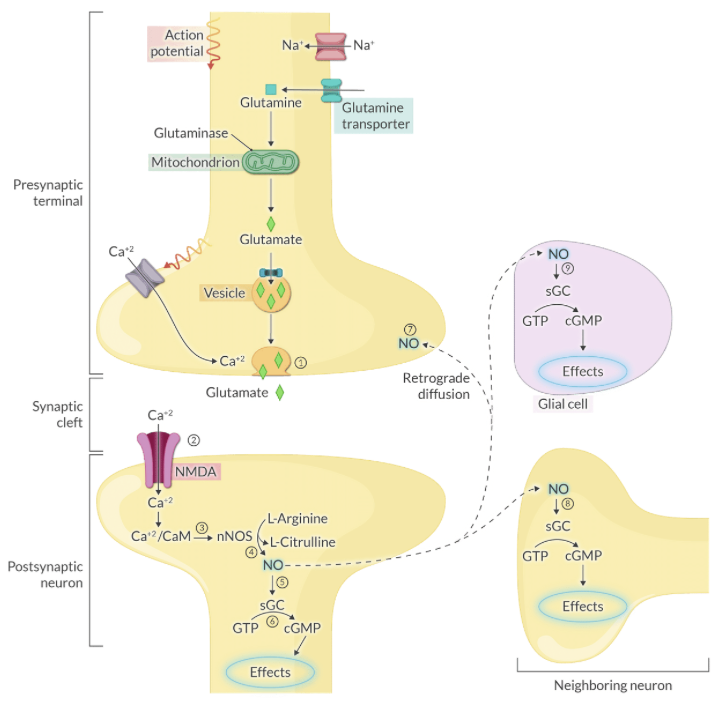
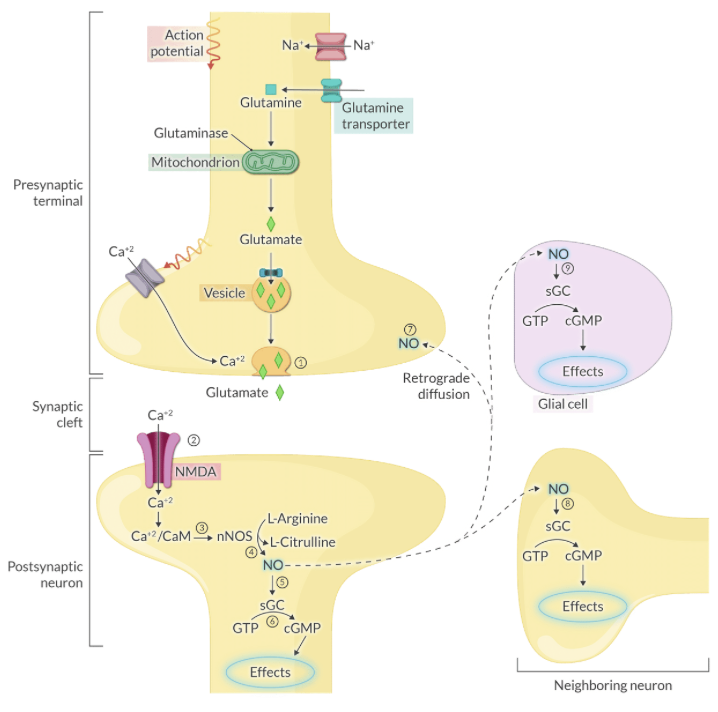
Explain the biochemical synthesis of Nitric Oxide (NO) and its immediate target in the postsynaptic neuron.
NO is synthesized from L-arginine by Nitric Oxide Synthase (NOS) and subsequently activates soluble guanylyl cyclase (sGC).
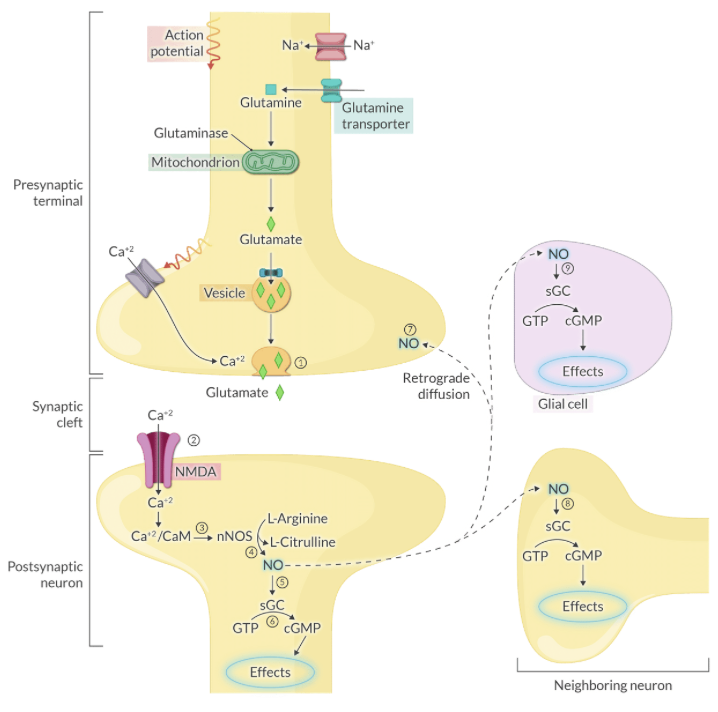
Explain the mechanism by which NO acts as a retrograde messenger to influence neurotransmitter release.
NO diffuses from the postsynaptic neuron back to the presynaptic terminal to enhance and prolong neurotransmitter release via positive feedback.
Explain the role of S-nitrosylation in protecting neurons from excitotoxicity.
At high concentrations, NO performs S-nitrosylation of the NMDA receptor to inactivate it, preventing dangerous hyperactivation.
Explain why glutamate is considered the primary excitatory neurotransmitter and its relationship to memory formation.
Glutamate mediates most excitatory signals in the brain and is essential for Long-Term Potentiation (LTP), the cellular basis for memory consolidation.
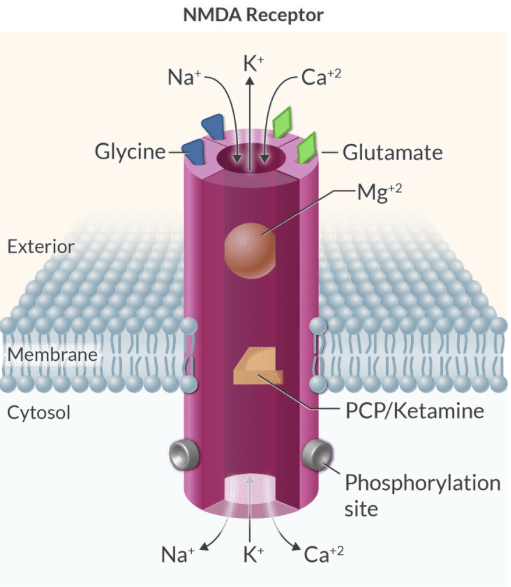
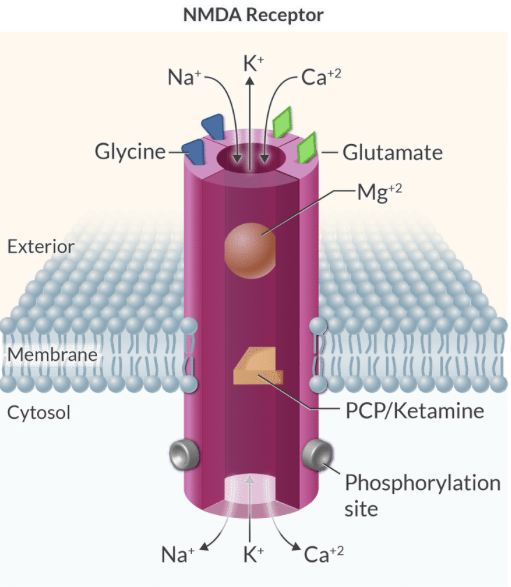
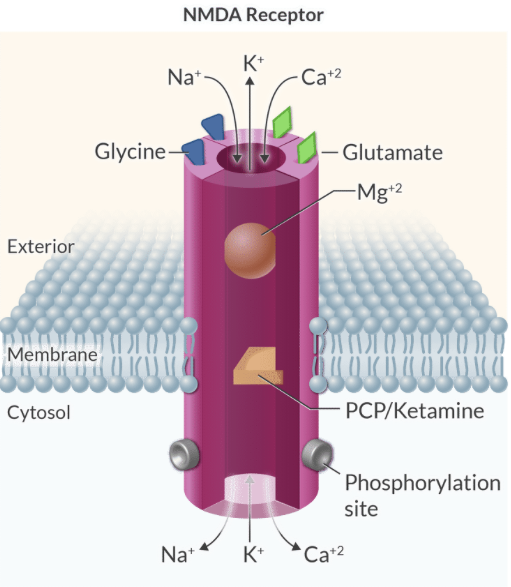
Explain the 'safety locks' of the NMDA receptor and their importance in preventing neuronal death.
NMDA activation requires voltage-dependent Mg2+ removal, two glutamate molecules, and two glycine molecules to prevent excessive Ca2+ influx and excitotoxicity.
Explain the mechanism of excitotoxicity and list three neurodegenerative diseases where it is implicated.
Excitotoxicity is neuronal injury caused by excessive glutamate receptor activity and Ca2+ overload, implicated in Huntington's, Alzheimer's, and ALS.
Explain the functional difference between AMPA and NMDA receptors during glutamatergic transmission.
AMPA receptors provide fast-gating, rapid excitatory transmission
NMDA receptors provide slower, delayed transmission that is Ca2+ permeable
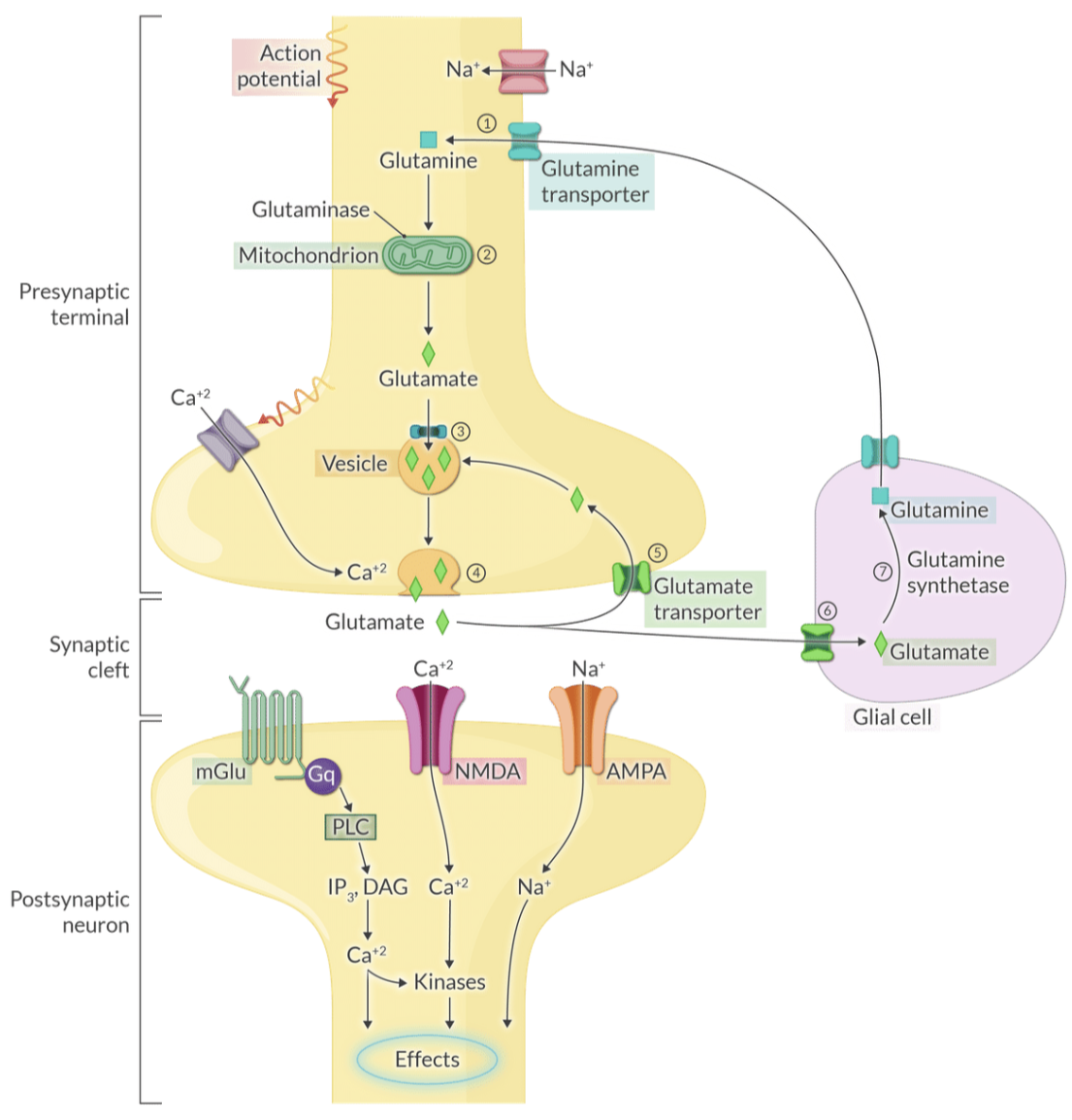
Explain the role of glial cells in glutamate metabolism and the 'glutamine-glutamate cycle'.
Glial cells take up glutamate from the synapse and convert it to glutamine via glutamine synthetase, which is then returned to neurons for reconversion to glutamate.
Explain the ionotropic mechanism of GABAA receptors and their clinical relevance to anesthesia.
GABAA receptors are ligand-gated Cl- channels that hyperpolarize neurons, serving as the primary target for propofol, barbiturates, and benzodiazepines.
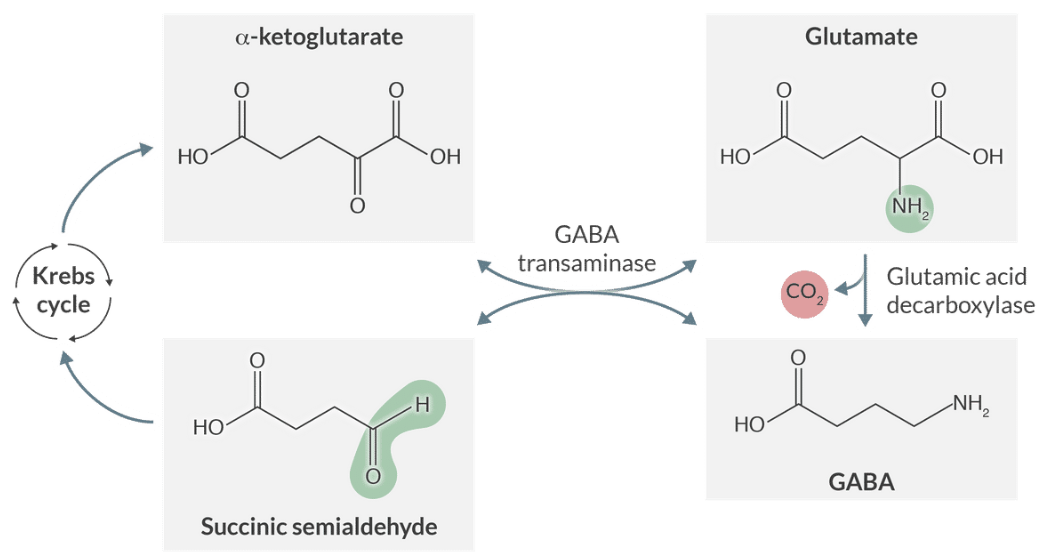
Explain the metabolic relationship between Glutamate and GABA via the Krebs cycle.
Glutamate and GABA are interconvertible through Krebs cycle intermediates like alpha-ketoglutarate and succinic semialdehyde.
Explain the coupling of GABAB receptors and their resulting inhibitory effects.
GABAB receptors are metabotropic Gi-coupled receptors that inhibit adenylyl cyclase and open K+ channels or close Ca2+ channels.
Explain the physiological significance of Glycine in the spinal cord and the condition known as hyperekplexia.
Glycine is the primary inhibitory neurotransmitter in the spinal cord, and mutations in its receptor cause hyperekplexia, this inhibition is impaired, leading to exaggerated startle responses.
Explain why glycine is considered a co-activator of the NMDA receptor.
Full activation of the NMDA receptor requires the binding of glycine molecules to a specific site on the receptor complex alongside glutamate.
Explain the structural composition of Nicotinic Acetylcholine Receptors (nAChR) and their classification as ionotropic receptors.
They are pentameric ligand-gated nonselective cation channels that require the binding of two acetylcholine molecules to open.
Explain where NM and NN nicotinic receptors are located and their distinct physiological roles.
NM receptors are found at the neuromuscular junction for muscle contraction.
NN receptors are found in autonomic ganglia for postganglionic neuron depolarization.
Explain the G-protein coupling for the five types of muscarinic ACh receptors (M1 through M5).
Odd-numbered receptors (M1, M3, M5) are coupled to Gq
Even-numbered receptors (M2, M4) are coupled to Gi
Explain the cardiac effect of M2 receptor stimulation by acetylcholine.
Activation of Gi inhibits adenylyl cyclase and opens K+ channels, leading to slowed spontaneous depolarization and decreased heart rate.
Explain how ionotropic receptors differ from metabotropic receptors in terms of response speed and mechanism.
Ionotropic receptors are direct ligand-gated ion channels that act within milliseconds
Metabotropic receptors use G-proteins and second messengers, taking seconds to minutes.
Explain the difference between direct and indirect effects of neurotransmitters at the postsynaptic membrane.
Direct effects involve ligand-activated ion channels altering conductance
Indirect effects are mediated by G-protein coupled receptors and second messengers.
Explain how volatile anesthetics inhibit excitatory neurotransmission regarding specific receptor targets.
Volatile anesthetics inhibit excitatory transmission via NMDA and nicotinic ACh receptors while enhancing inhibitory transmission via GABA and glycine receptors.
Explain the ionic currents that produce an Excitatory Postsynaptic Potential (EPSP).
Excitatory responses are typically produced by an inward current of Na+ or Ca2+ that depolarizes the membrane.
Explain the ionic currents that produce an Inhibitory Postsynaptic Potential (IPSP).
Inhibitory responses occur when an outward current of K+ or an inward current of Cl- hyperpolarizes the membrane.
Explain the synthesis of serotonin and the name of its primary precursor amino acid.
Serotonin (5-HT) is synthesized from the amino acid tryptophan via hydroxylation and subsequent decarboxylation.
Explain the enzymatic conversion of norepinephrine to epinephrine and where this primarily occurs.
Norepinephrine is methylated by Phenylethanolamine-N-methyltransferase (PNMT) to form epinephrine, occurring primarily in the adrenal medulla.
Explain the physiological significance of the 'locus coeruleus' in noradrenergic neurotransmission.
The locus coeruleus in the pons contains the major concentration of noradrenergic neurons in the CNS, projecting widely to regulate arousal and vigilance.
Explain the difference between 'autocoid' and 'paracrine hormone' in medical terminology.
Both terms refer to molecules that act locally, but 'autocoid' often suggests an autocrine function where the cell responds to its own signal.
Explain why Mg2+ is considered a voltage-dependent blocker of the NMDA receptor.
At normal resting membrane potentials, a Mg2+ ion clogs the channel pore, and it is only relieved when the membrane is sufficiently depolarized.
Explain the role of the G beta/gamma subunit in M2 receptor signaling in the heart.
The beta/gamma subunit directly opens GIRK (K+) channels to cause hyperpolarization and slow the heart rate.
Explain how PKA activation leads to increased myocardial Ca2+ availability during beta1 stimulation.
PKA phosphorylates L-type Ca2+ channels to increase trigger Ca2+ and phosphorylates phospholamban to increase Ca2+ stores in the sarcoplasmic reticulum.
Explain the biosynthetic origin of Histamine and the enzyme responsible for its production.
Histamine is synthesized from the amino acid L-histidine through a decarboxylation reaction catalyzed by histidine decarboxylase.
(Histidine decarboxylase is the enzyme that converts L-histidine into histamine.)
Explain the clinical utility of 5-HT3 antagonists in chemotherapy-induced nausea.
They exert their effect by blocking 5-HT3 receptors in the area postrema and vagus nerve, preventing the activation of the vomiting center by emetogenic drugs.
Explain how glutamate is removed from the synaptic cleft to terminate its signal.
Glutamate is removed via active reuptake into both presynaptic terminals and surrounding glial cells by specific transporters.
Explain the effect of Gq protein activation on intracellular Ca2+ levels.
Gq activates phospholipase C (PLC), which produces IP3 and DAG, ultimately leading to the release of Ca2+ from the endoplasmic reticulum.
Explain the primary physiological role of dopamine in the substantia nigra for a patient's movement.
Dopaminergic neurons in the substantia nigra are crucial for coordinating movement; their loss leads to the motor deficits seen in Parkinson's.
Explain the role of SERCA2 in cardiac lusitropy during sympathetic stimulation.
The SERCA2 pump actively moves Ca2+ from the cytosol into the sarcoplasmic reticulum; its activity is increased during beta1 stimulation to speed relaxation.
Explain why epinephrine is considered the primary hormone of the adrenal medulla.
The adrenal medulla contains the enzyme PNMT, which allows for the final step of catecholamine synthesis (conversion of NE to Epi) for release into the blood.
Explain the significance of 'Slower-gating' in NMDA receptors compared to AMPA receptors.
Slower-gating allows the NMDA receptor to mediate more prolonged excitatory postsynaptic currents, which is vital for synaptic plasticity and memory.
Explain the mechanism of Action of nitric oxide on soluble guanylyl cyclase (sGC).
Nitric oxide binds to the heme moiety of sGC, activating the enzyme to catalyze the conversion of GTP to cGMP.
Which intrinsic enzymatic activity of the G protein alpha subunit is responsible for terminating the signal?
GTPase activity (hydrolyzing GTP back to GDP).
(This intrinsic enzymatic activity allows the G protein to return to its inactive form, thereby terminating the signaling cascade.)
Explain the cellular response to Gi protein activation in terms of cAMP levels.
Gi inhibits adenylyl cyclase, decreasing cAMP production, and reducing PKA activity.
Explain the physiological basis for rapid arterial oxygen desaturation during induction in a term parturient.
A decreased Functional Residual Capacity (FRC), paired with a 20% increase in oxygen consumption (VO2), hastens the onset of hypoxemia during apnea.
Clinically, why should a CRNA avoid nasal airway adjuncts in the pregnant patient?
Increased progesterone, estrogen, and relaxin cause upper airway vascular engorgement and friability, making the tissue highly susceptible to trauma and bleeding.
What specific airway equipment modification is recommended for a parturient with large breasts to facilitate laryngoscopy?
The use of a short-handled laryngoscope (Datta handle) helps clear the chest wall/breasts during blade insertion.
What is the primary respiratory stimulant responsible for the 50% increase in minute ventilation during pregnancy?
Progesterone
How does the maternal PaCO2 of 30 mmHg affect the fetal-maternal gas exchange gradient?
A lower maternal PaCO2 of 30 mmHg enhances oxygen transfer to the fetus by increasing the gradient for carbon dioxide elimination and facilitating the delivery of oxygen.
In the context of the Oxyhemoglobin Dissociation Curve, what is the clinical significance of the maternal P50 shifting to the right?
A right shift (increased P50) facilitates the unloading and transfer of oxygen from maternal blood to the fetus.
Explain the 'dilutional anemia of pregnancy' in terms of plasma and erythrocyte volume changes.
Plasma volume increases by 45%, which outpaces the 20% increase in erythrocyte volume, leading to a lower relative hemoglobin concentration.
Why is the term parturient at a 6-fold higher risk for Deep Vein Thrombosis (DVT)?
Pregnancy creates a hypercoagulable state characterized by increased clotting factors (1, 7, 8, 9, 10, 12) and decreased anticoagulants (Protein S and Antithrombin).
Clinically, how does the CRNA manage the hemodynamic consequences of 'syndrome of supine hypotension'?
Implementation of Left Uterine Displacement (LUD) by elevating the mother's right torso 15 to 30 degrees to decompress the vena cava and aorta.
Explain why uterine blood flow is highly sensitive to maternal hypotension and Systemic Vascular Resistance (SVR) changes.
Uterine blood flow is not autoregulated; it is a low-resistance system entirely dependent on Mean Arterial Pressure (MAP) and Cardiac Output.
During which stage of labor and delivery does Cardiac Output (CO) reach its absolute peak?
The immediate postpartum period (Stage 3), where CO can increase by 80% due to autotransfusion from the contracting uterus and relief of caval compression.
What is the expected change in baseline Minimal Alveolar Concentration (MAC) for the parturient, and when does this change begin?
MAC decreases by 30 - 40% due to increased progesterone levels, a process that starts at 8 - 12 weeks gestation.
Why is every laboring mother considered a 'full stomach' regardless of NPO status?
While gastric emptying is unchanged before labor, the onset of labor pain and the administration of opioids significantly slow gastric emptying time.
How does pregnancy affect the dosing requirements for neuraxial local anesthetics?
Dose requirements are decreased because progesterone increases nerve sensitivity and engorged epidural veins reduce the volume of the subarachnoid/epidural spaces.
What are the four primary physiochemical characteristics of a drug that favor its transfer across the placenta?
Low molecular weight (< 500 Daltons)
high lipid solubility
non-ionized state
non-polar nature
Which four specific anesthetic-related drugs do NOT cross the placenta due to their chemical properties?
Neuromuscular blockers
Glycopyrrolate
Heparin
Insulin
As a CRNA, why would you prefer Phenylephrine over Ephedrine for treating post-spinal hypotension in a healthy parturient?
Recent evidence shows Phenylephrine is as efficacious as Ephedrine and results in higher (less acidic) fetal pH values.
Stage 1 labor pain is primarily visceral; identify the spinal segments involved and the specific anatomical structures generating the signals.
The pain originates from the lower uterine segment and cervix, traveling via posterior nerve roots at T10 - L1.
Stage 2 labor pain adds a somatic component; identify the additional spinal segments involved and the nerve pathway for perineal pain.
It adds signals from the vagina and perineum traveling via the Pudendal nerve to the S2 - S4 posterior nerve roots.
Explain the 'needle through the needle' technique used in a Combined Spinal Epidural (CSE).
The epidural space is identified first, then a long spinal needle is passed through the epidural needle to inject local anesthetic into the subarachnoid space.
Why is 0.75% Bupivacaine specifically contraindicated for epidural use in obstetrics?
There is a high risk of fatal cardiotoxicity if the drug is accidentally injected into the intravascular space.
Which local anesthetic is known to reduce the efficacy of epidural morphine, and what is the clinical implication?
2-Chloroprocaine; it can interfere with the mu-opioid receptors, leading to poor postoperative analgesia.
A patient experiences respiratory arrest 15 minutes after epidural dosing. What is the most likely etiology and its pathophysiology?
Subdural injection; a small volume of local anesthetic in the potential subdural space causes an excessive, delayed cephalad spread and high block.
What is the primary mechanism of action for Oxytocin (Pitocin) when used for uterine atony?
It stimulates uterine contractions by binding to specific receptors in the myometrium, combatting hypotonia and hemorrhage.
Identify the critical side effect of rapid IV bolus administration of Oxytocin.
Rapid IV administration can cause profound hypotension and cardiovascular collapse.
Why is Methergine (Methylergonovine) strictly administered via the IM route only?
IV administration can cause severe, life-threatening vasoconstriction, hypertension, and cerebral hemorrhage.
In which patient population is Prostaglandin F2 (Hemabate) relatively contraindicated due to its side effect profile?
Patients with asthma, as it can cause significant bronchospasm.
When is the most ideal time to perform nonobstetric surgery during pregnancy to minimize fetal and maternal risks?
The second trimester is the best time, as organogenesis is complete and the risk of preterm labor is lower than in the third trimester.
According to current consensus, at what gestational age should a pregnant patient be treated as a 'full stomach' for RSI?
Between 18 and 20 weeks gestation.
How does maternal hyperventilation (resulting in PaCO2 < 20 mmHg) during surgery negatively impact the fetus?
It reduces placental blood flow, increasing the risk of fetal asphyxia and acidosis.
What is the hemodynamic goal when managing a patient with Preeclampsia, and what is the treatment for a BP > 160/110?
The goal is to prevent cerebrovascular accidents and placental abruption; first-line treatments include Labetalol (20 mg IV) or Hydralazine (5 mg IV).
Describe the imbalance of vasoactive substances that characterizes the pathophysiology of Preeclampsia.
There is an increase in Thromboxane (vasoconstrictor/platelet aggregator) and a decrease in Prostacyclin (vasodilator/inhibitor of aggregation).
Why must the CRNA check a platelet count before placing a neuraxial block in a preeclamptic patient?
Preeclampsia causes endothelial damage and platelet consumption; a count < 50,000/\mu L poses a high risk for spinal-epidural hematoma.
What is the definitive treatment for Preeclampsia, Eclampsia, and HELLP syndrome?
Delivery of the fetus and the placenta.
How does Magnesium Sulfate affect the CRNA's use of neuromuscular blocking agents (NMBAs)?
Magnesium increases sensitivity to both depolarizing and non-depolarizing NMBAs, potentially prolonging their effects.
Identify the mnemonic 'VEAL CHOP' for interpreting fetal heart rate decelerations.
Variable = Cord compression
Early = Head compression
Accelerations = OK
Late = Placental insufficiency
What does a complete lack of Fetal Heart Rate (FHR) variability suggest regarding the fetus's status?
It is a worrisome finding indicating potential fetal distress, hypoxemia, acidosis, or the effects of CNS depressant drugs.
Differentiate between Placenta Accreta, Increta, and Percreta.
Accreta attaches to the myometrium surface
Increta invades the myometrium
Percreta penetrates through the myometrium and may involve other organs
Compare the clinical presentation of Placenta Previa versus Placental Abruption.
Placenta Previa presents with painless vaginal bleeding
Placental Abruption presents with painful vaginal bleeding and fetal hypoxia
What is the most important clinical indicator of adequate ventilation during neonatal resuscitation?
The resolution of bradycardia (heart rate rising above 100 bpm).
In neonatal resuscitation, at what heart rate threshold should chest compressions be initiated?
When the heart rate remains < 60 bpm despite 30 seconds of effective Positive Pressure Ventilation (PPV).