CHEM130 Midterm 2 (Chapters 4.1 - 7.4)
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Ionic Bonds
electrostatic forces of attraction between oppositely charged cations and anions
Binary Ionic Compounds
compounds composed of just two elements: a metal (which forms the cations) and a nonmetal (which forms the anions)
Covalent Compounds (non-metals)
result when atoms share, rather than transfer, electrons (formed between atoms which have similar tendencies to attract electrons to themselves)
Pure Covalent Bond
if the atoms that form a covalent bond are identical (homonuclear) then the electrons in the bond must be shared equally
Polar Covalent Bond
when the atoms linked by a covalent bond are different, bonding electrons are more attracted to one atom than the other, causing a shift of electron density
Representing a Polar Covalent Bond
using an arrow that points from the less electronegative atom to the more electronegative atom (positive to negative)
use δ+ and δ- to denote which atom attracts more electrons
Pauling Electronegativity
the tendency of an atom to attract electrons towards itself
Notes on Pauling Electronegativity
the more strongly an atom attracts the electrons in its bonds, the larger its electronegativity
electrons in a polar covalent bond are shifted toward the more electronegative atom
the more electronegative atom is the one with the partial negative charge
the greater the difference in electronegativity, the more polarized the electron distribution is
bond with the “least ionic character” is the one with the smallest electronegativity difference
Electronegativity Difference for a Pure Covalent Bond
< 0.4
Electronegativity Difference for a Polar Covalent Bond
between 0.4 and 1.8
Electronegativity Difference for an Ionic Bond
> 1.8
Polyatomic Ions
groups of atoms covalently bonded together that act as a single unit with an overall net charge
(form ionic compounds by combining with ions of opposite charge)
Naming Ionic Compounds (monatomic ions)
name of the cation + anion + ide
Naming Ionic Compounds (polyatomic ions)
cation + anion + ide/ate
(anion ends in –ate if it’s polyatomic, –ide if it’s not)
Naming Ionic Compounds (transition metal ions)
cation + charge of the metal ion in roman numerals + anion + ide
Ionic Hydrates
ionic compounds that contain water molecules as integral components of their crystals (use a black dot that denotes that it’s not a bond, just a loose association)
Naming Covalent Compounds
prefix + more metallic element + prefix + more nonmetallic element + –ide
(prefixes: di-, tri-, tetra-, penta-, hexa-, hepta-)
Binary Acid
compounds composed of hydrogen and one other nonmetallic element
Naming Binary Acids
hydro + nonmetallic element name + –ic + acid
Oxyacids
compounds that contain hydrogen, oxygen, and at least one other element
Naming Oxyacids
anion + –ate/ic/ite/ous + acid
(you omit “hydrogen” in the name for oxyacids)
Limiting Reactant
the substance in a chemical reaction that runs out first and determines the maximum amount of product that can be formed
Excess Reactant
a substance that is not completely consumed during a chemical reaction
Find Volume or Mass of an Aqueous Solution
need to use this equation: molarity (M) = mol/liter
(molarity is similar to concentration)
How do you find the amount of a reactant/product based off another reactant/product?
divide the mass of the reactant by its formula mass
multiply the number of moles by the conversion factor
Mass Percent/Percent Composition
the mass of an element expressed as a percentage of the total mass of the compound it is part of
(molar mass of element/molar mass of compound)
Finding the Empirical Formula
convert mass percent to grams (if only mass percent is given, assume you have 100 g of the compound)
convert to moles by dividing the mass by its formula/molar mass
divide each element’s molar amount by the smallest molar amount
multiplying all coefficients by an integer, if necessary, to ensure that the smallest whole-number ratio of subscripts is obtained
Finding the Molecular Formula
calculate the empirical formula mass
divide the compound’s given molar mass by the empirical formula mass
multiply all subscripts by this value to find the molecular formula
Lewis Symbol
consists of an elemental symbol surrounded by one dot for each of its valence electrons
Lewis Structures
drawings that describe the bonding in molecules and polyatomic ions, using a dash/line to indicate a shared pair of electrons
Lone Pairs
electrons that are not used in bonding
Octet Rule
the tendency of main group atoms to form enough bonds to obtain eight valence electrons
Steps to Draw a Lewis Structures
determine the total number of valence electrons
remember to check if it has a charge
draw a skeleton structure of the molecule, arranging the atoms around a central atom
the least electronegative element should be placed in the center, if electronegativities are the same, it’s the atom which will make more bonds
connect each atom to the central atom with a single bond
distribute the remaining electrons as lone pairs
place all remaining electrons on the central atom
rearrange the electrons of the outer atoms to make multiple bonds with the central atom
aim to obtain octets and ensure the electrons used equals the total number of valence electrons
Exceptions to the Octet Rule
odd-electron molecules have an odd number of valence electrons, and therefore have an unpaired electron (called free radicals)
electron-deficient molecules have a central atom without a full outer shell (generally, these molecules have central atoms from groups 2/13)
hypervalent molecules have a central atom that has more than 8 valence electrons (they can share more than four pairs of electrons with other atoms because they have d orbitals)
Formal Charge
formal charge = valence electrons (that it had before bonding) – (# of bonds + non bonding electrons)
formal charge should add up to the ionic charge
Using Formal Charge to Predict Molecular Structure
a molecular structure with formal charges of zero is more likely than one in which some formal charges are not zero
the arrangement with the smallest non-zero formal charges is better
Lewis structures are preferable when adjacent formal charges are zero or of the opposite sign
the structure with negative formal charges on the more electronegative atom is better
Resonance Arrow
a double-headed arrow between Lewis structures indicates that they are resonance forms
Length of Bonds
single bonds are longer than double bonds, and double bonds are longer than triple bonds
Bond Angle
the angle between two bonds that include a common atom, measured in degrees
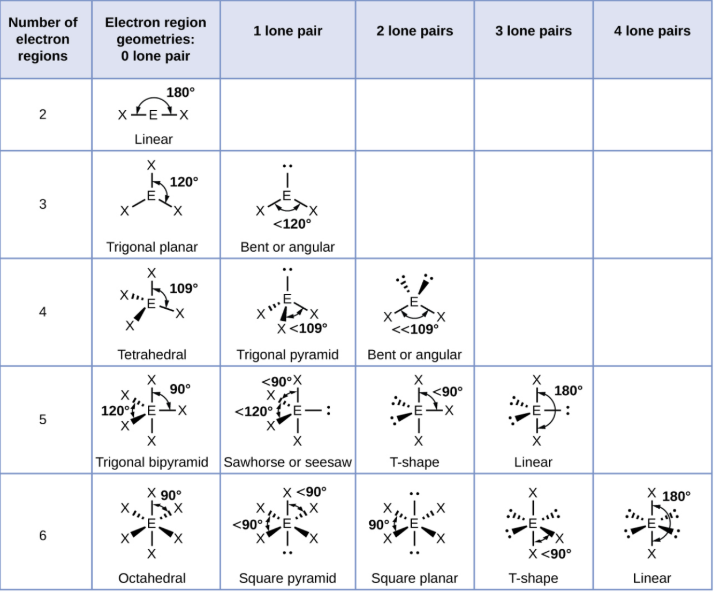
Valence Shell Electron-Pair Repulsion Theory (VSEPR)
enables us to predict a molecular structure from the number of bonds and lone electron pairs in s Lewis structure
electrons in the valence shell of a central atom form either bonding pairs or lone pairs
assumes that electron pairs in the valence shell of a central atom will maximize the distance between electron pairs
Electron-Pair Geometry (if 2 things are connected)
linear (180 degree bond angles)
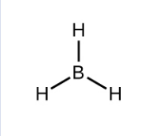
Electron-Pair Geometry (if 3 things are connected)
trigonal planar (120 degree bond angles)
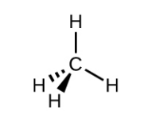
Electron-Pair Geometry (if 4 things are connected)
tetrahedral (109.5 degree bond angle)
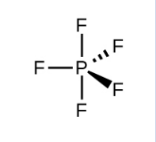
Electron-Pair Geometry (if 5 things are connected)
trigonal pyramidal (90 and 120 degree bond angles)
Electron-Pair Geometry (if 6 things are connected)
octahedral (90 and 180 degree bond angles)
How to Draw a Linear Bond Geometry
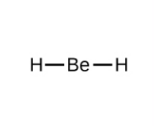
How to Draw a Trigonal Planar Bond Geometry
How to Draw a Tetrahedral Bond Geometry
How to Draw a Trigonal Pyramidal Bond Geometry
How to Draw a Octahedral Bond Geometry
Electron-Pair Geometry vs Molecular Structure
electron-pair geometries describe all regions where electrons are located, bonds AND lone pairs
molecular structures only include the placement of the atoms in the molecule, not the electrons (molecular is more specific)
Order of Electron-Pair Repulsions
lone pair – lone pair > lone pair – bonding pair > bonding pair – bonding pair
AND lone pair > triple bond > double bond > single bond
Ideal Molecular Structures
MEMORIZE THIS
Axial and Equatorial Positions (for trigonal bipyramidal geometry)
the axial positions are the two bonds pointing straight up and down, while the equatorial positions are the three bonds arranged in a flat triangle
Steps to Predict Electron Pair Geometry and Molecular Structure
draw the Lewis structure of the molecule
count the number of regions of electron density (lone pairs and bonds)
a single, double, or triple bond counts as one region of electron density
identify the electron-pair geometry based on the number of regions of electron density
use the number of lone pairs to determine the molecular structure
minimize repulsions
in trigonal bipyramidal arrangements, repulsion is minimized when every lone pair is in an equatorial position
in an octahedral arrangement with two lone pairs, repulsion is minimized when the lone pairs are on opposite sides of the central atom
Bond Polarity
when the the separation of charge in polar covalent bonds gives rise to a bond dipole moment
Bond Polarity Geometry
when a molecule contains more than one bond, the geometry must be taken into account
if the bonds in a molecule are arranged such that their bond moments cancel, then the molecule is non-polar
if the molecular structure is bent and the dipole moments won’t cancel, then the molecule is polar
Properties of Polar Molecules
they align with the positive end of the molecule oriented toward the negative plate when placed in an electric field (attracted to electrically charged objects as well)
Valence Bond Theory
describes a covalent bond as the overlap of half-filled atomic orbitals
the mutual attraction between this negatively charged electron pair and the two atoms’ positively charged nuclei serves to physically link the two atoms
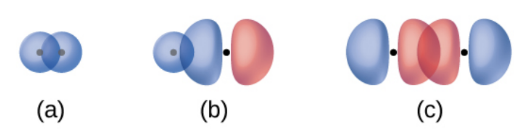
Sigma Bonds
the overlap of two s orbitals, an s orbital and a p orbital, and the end-to-end overlap of two p orbitals (usually px orbitals)
electron density is concentrated in the region along the internuclear axis (a line between the nuclei would pass through the center of the overlap region)
single bonds in Lewis structures are described as σ bonds in valence bond theory
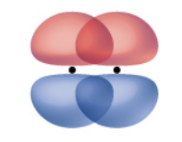
Pi Bonds
results from the side-by-side overlap of two p orbitals
egions of orbital overlap lie on opposite sides of the internuclear axis
along the internuclear axis is a node
multiple bonds consist of both σ and π bonds (first bond is a σ, second and third are a π)
Hybridization
taking atomic orbitals and turning them into molecular orbitals to make covalent bonds, the new orbitals that result are called hybrid orbitals
Hybridization Rules
HYBRIDIZATION HAPPENS BY COMBINING TWO ORBITALS FROM THE SAME ELEMENT!
hybrid orbitals have different shapes and orientations from those of the atomic orbitals in isolated atoms
a set of hybrid orbitals is generated by combining atomic orbitals, the number of hybrid orbitals in a set is equal to the number of atomic orbitals that were combined
all orbitals in a set of hybrid orbitals are equivalent in shape and energy
the type of hybrid orbitals formed in a bonded atom depends on its electron-pair geometry as predicted by the VSEPR theory
hybrid orbitals overlap to form σ bonds, unhybridized orbitals overlap to form π bonds
What is the hybridization if a central atom is connected to…
2 things: sp hybridized
3 things: sp2 hybridized
4 things: sp3 hybridized
5 things: sp3d hybridized
6 things: sp3d2 hybridized
Valence Bond Theory vs Molecular Orbital Theory
Molecular Orbital Theory
describes electrons in a molecule as occupying new molecular orbitals, which are formed by the combination of atomic orbitals and can extend over multiple atoms
orbitals are either bonding or non-bonding
linear combination of atomic orbitals: the mathematical process of combining atomic orbitals to generate molecular orbitals
S Orbitals
created by the overlap of two atomic s orbitals on adjacent atoms
when in-phase waves combine, constructive interference occurs, producing the σs molecular orbital
when out-of-phase waves combine, destructive interference occurs, producing the σs* molecular orbital
P Orbitals (overlapping end to end)
in normal p orbitals, the two lobes have opposite phases
when lobes of the same phase overlap, they form a bonding (σ) orbital
when lobes of the opposite phase overlap, they create an anti-bonding (σ*) orbital
when two px orbitals on different atoms overlap end to end along the x-axis, they specifically form the σpx and σpx* orbitals
P Orbitals (overlapping side by side)
when they overlap side by side they form a π bonding orbital with electron density above and below the internuclear axis
they also form a π* anti-bonding orbital with two nodal planes
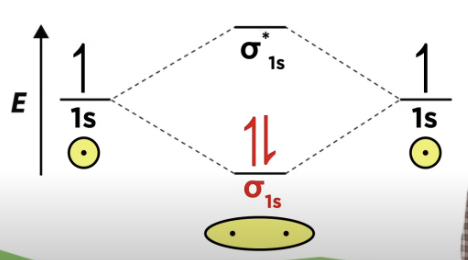
Orbital Diagrams
each horizontal line represents one orbital that can hold two electrons
the molecular orbitals formed by the combination of the atomic orbitals are in the center
dashed lines show which of the atomic orbitals combine to form the molecular orbitals
the bonding orbitals have lower energy, and the anti bonding orbitals have higher energy
Bond Order
bond order = ((number of bonding electrons) - (number of anti-bonding electrons)) / 2
Paramagnetism
a weak form of magnetism in which materials are weakly attracted to an external magnetic field due to the presence of unpaired electrons
Diamagnetic
materials in which all of the electrons are paired and are thus not magnetic