chem231 exam 2 practice
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms

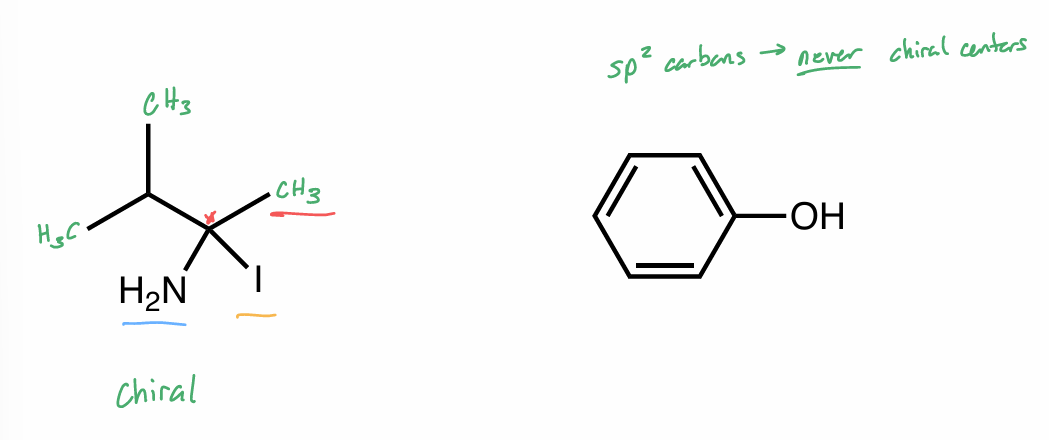
Is this chiral or achiral?
If chiral, label chiral center with *


Is this chiral or achiral?
If chiral, label chiral center with *
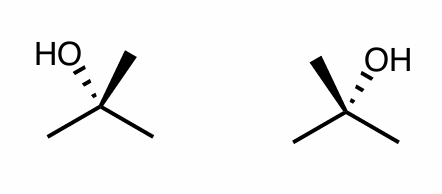
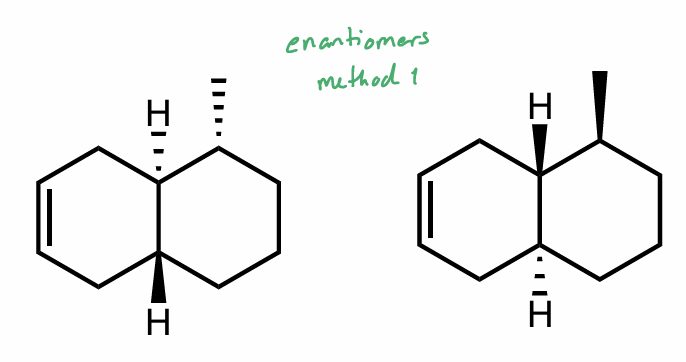
Are these molecules chiral?
What is their relationship?
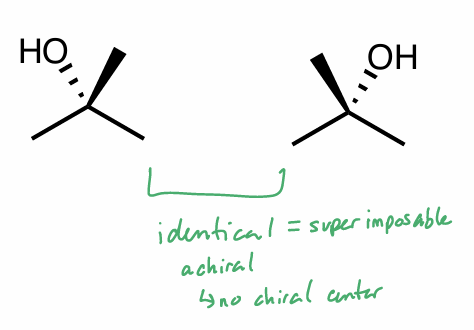

Are these molecules chiral?
What is their relationship?
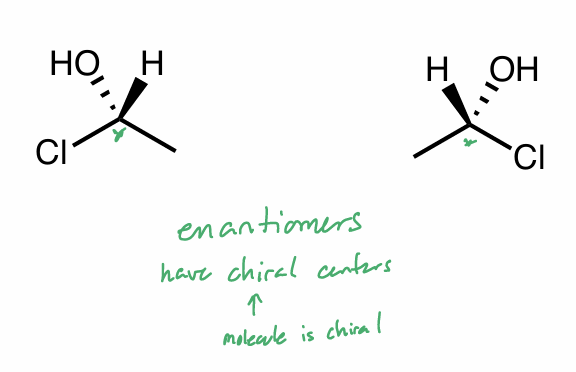
What is the relationship between this pair?
How do you know?
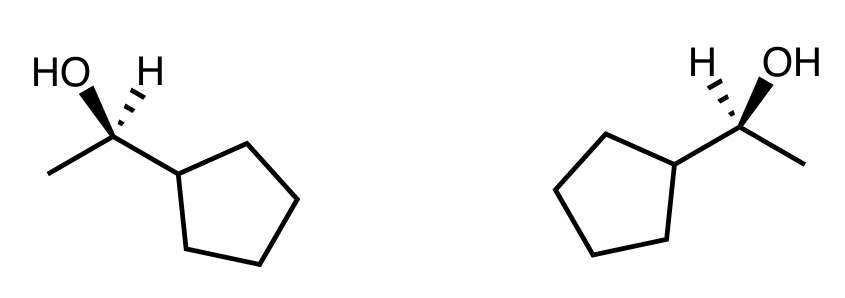
What is the relationship between this pair?
How do you know?
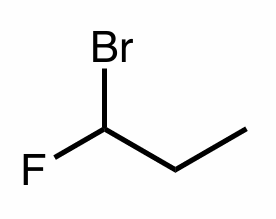
Nomenclate the following molecule
Add the R/S configuration in the right location
1-bromo-1-fluropropane

Nomenclate the following molecules
Add the R/S configuration in the right locations
Are they the same molecule?
No, they have different R/S configurations
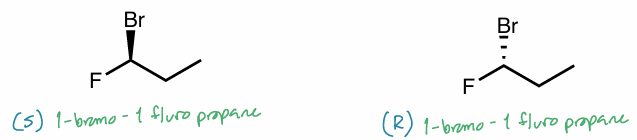
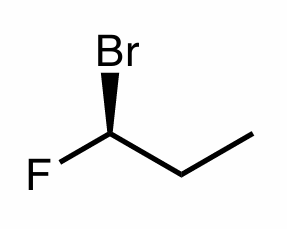
Assign the R/S configuration to the chiral center
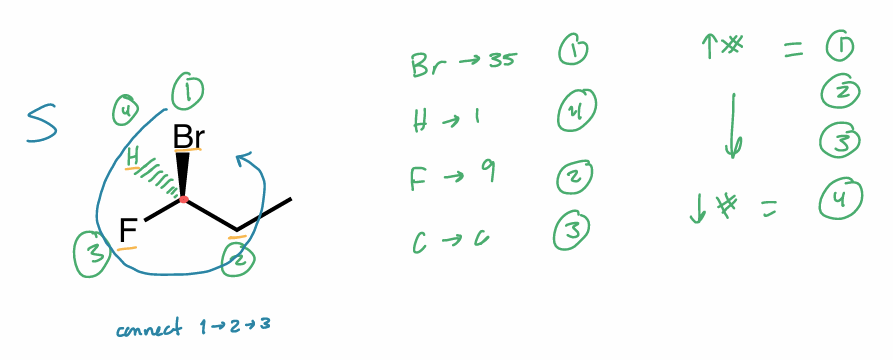
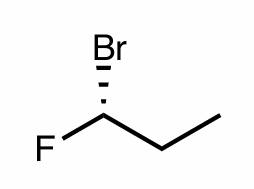
Assign the R/S configuration to the chiral center
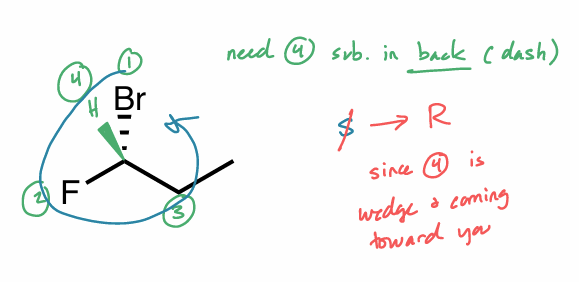
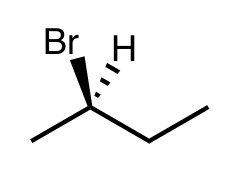
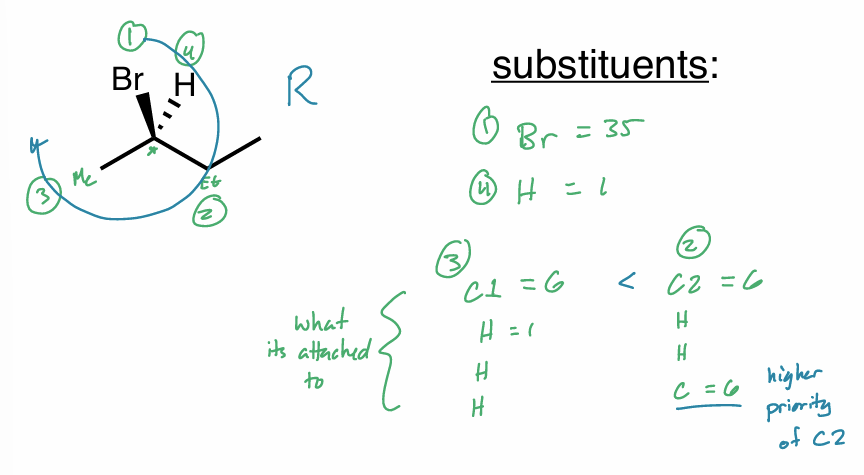
Assign the R/S configuration to the chiral center
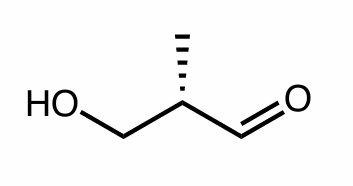
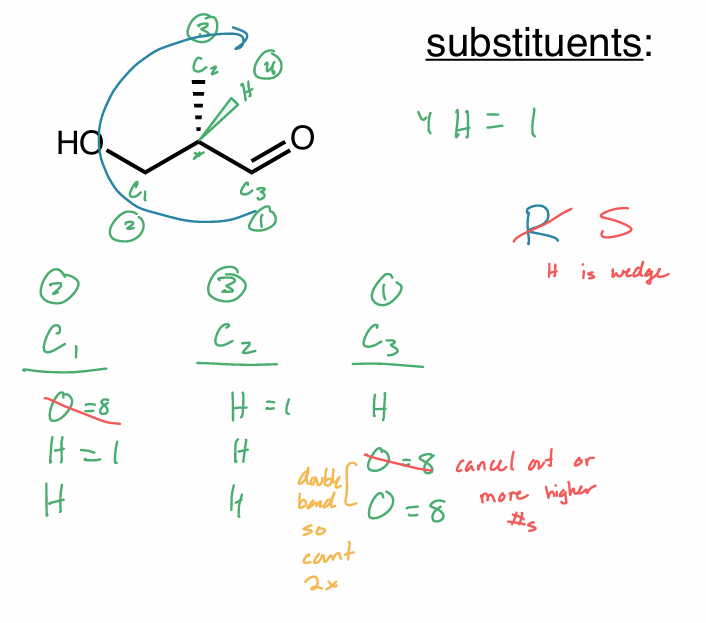
Assign the R/S configuration to the chiral center
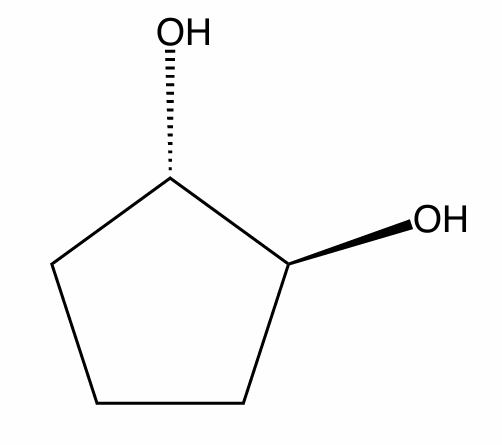
Assign the R/S configuration to the chiral center
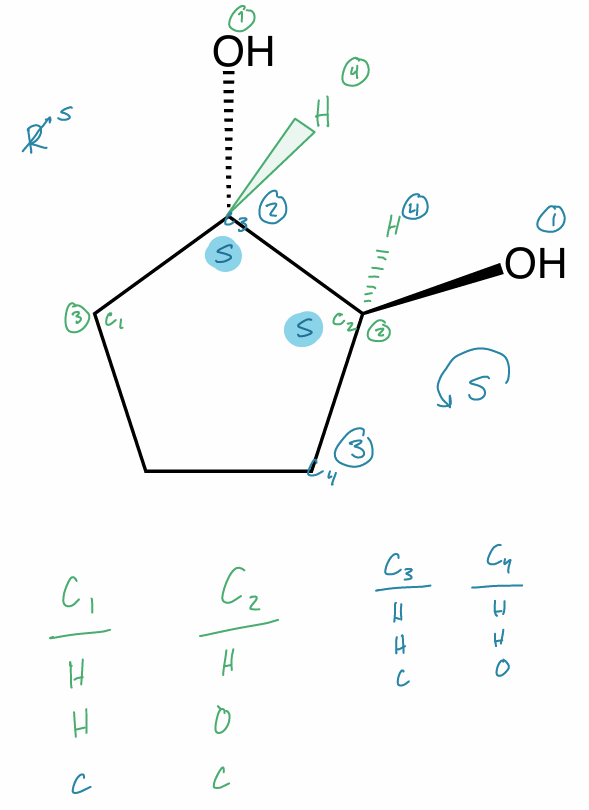
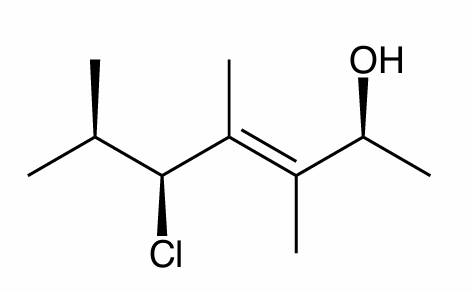
Assign the R/S configuration to the chiral center
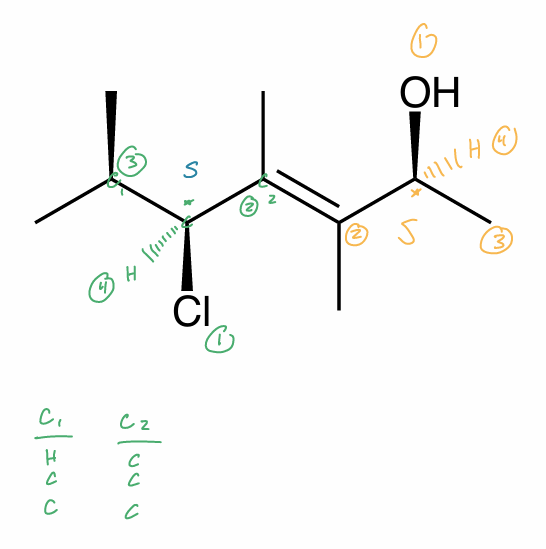
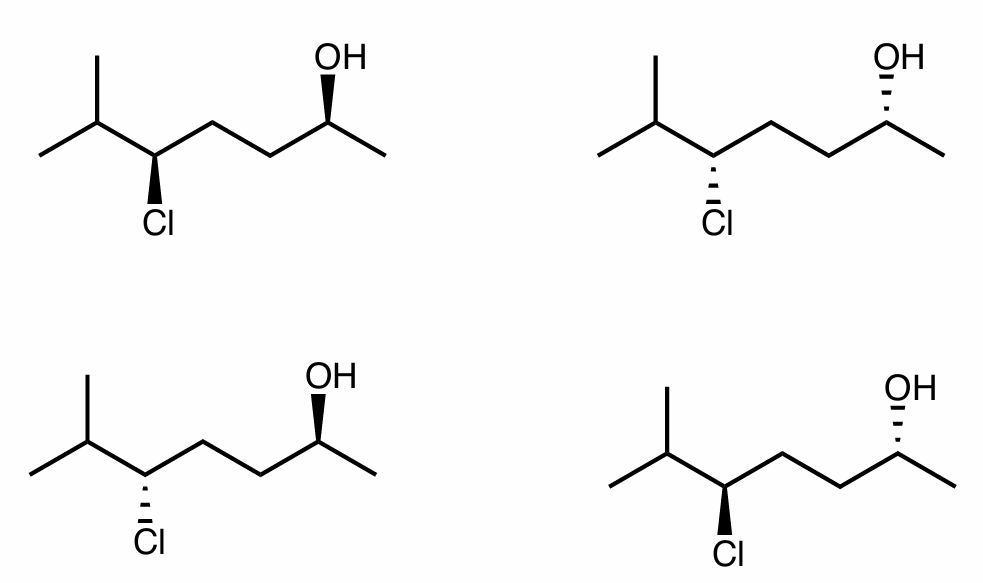
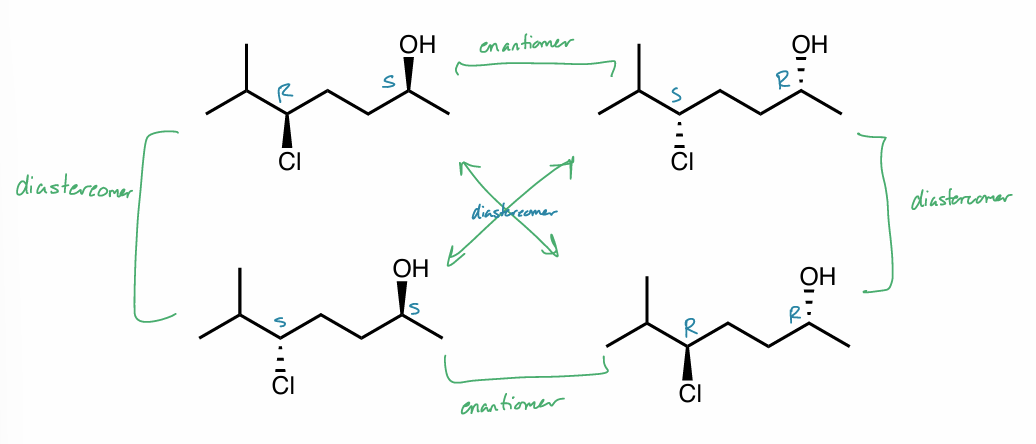
Find the stereochemical relationship between each of these molecules (hint: assign R/S)
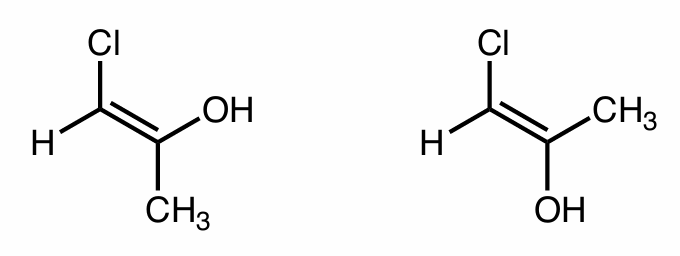
Find the stereochemical relationship between these molecules (hint: alkenes)
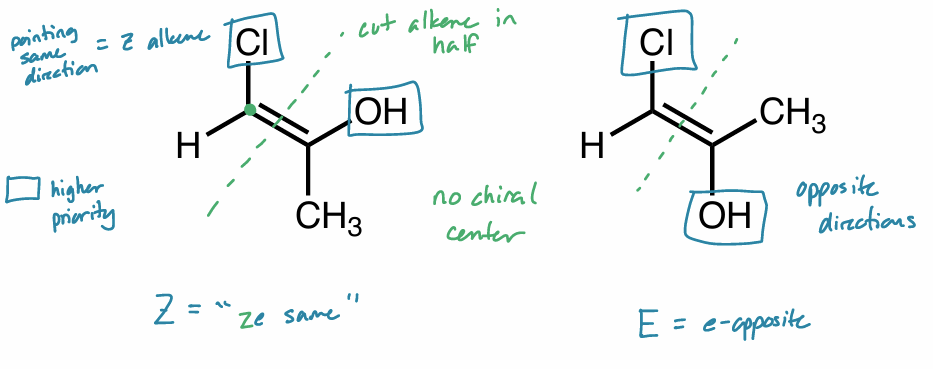
Find the stereochemical relationship between these molecules (hint: alkenes)
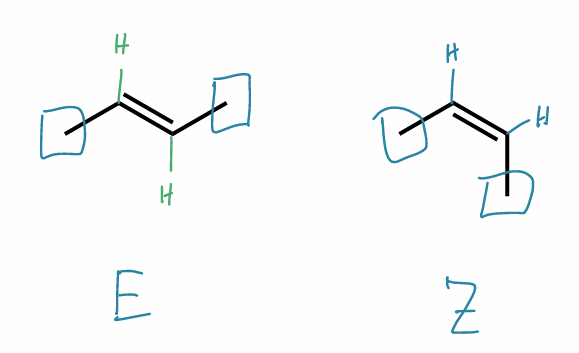
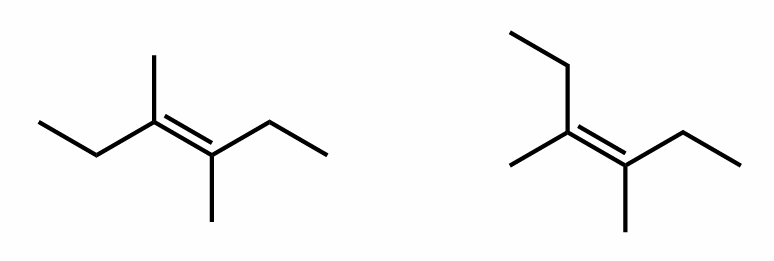
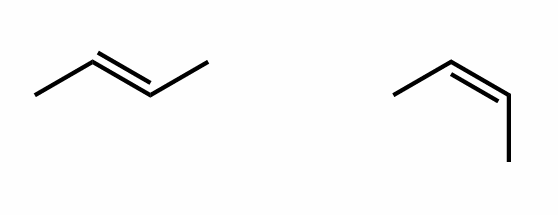
Find the stereochemical relationship between these molecules (hint: alkenes)
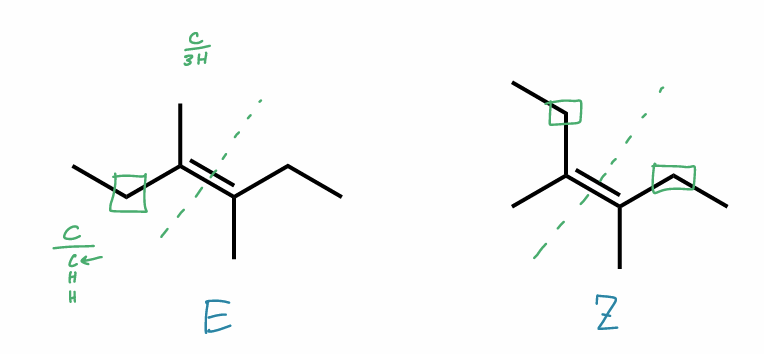
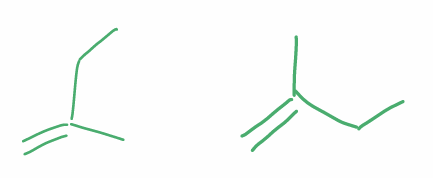
Find the stereochemical relationship between these molecules (hint: alkenes)
identical → neither E or Z
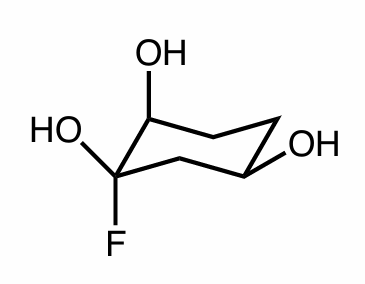
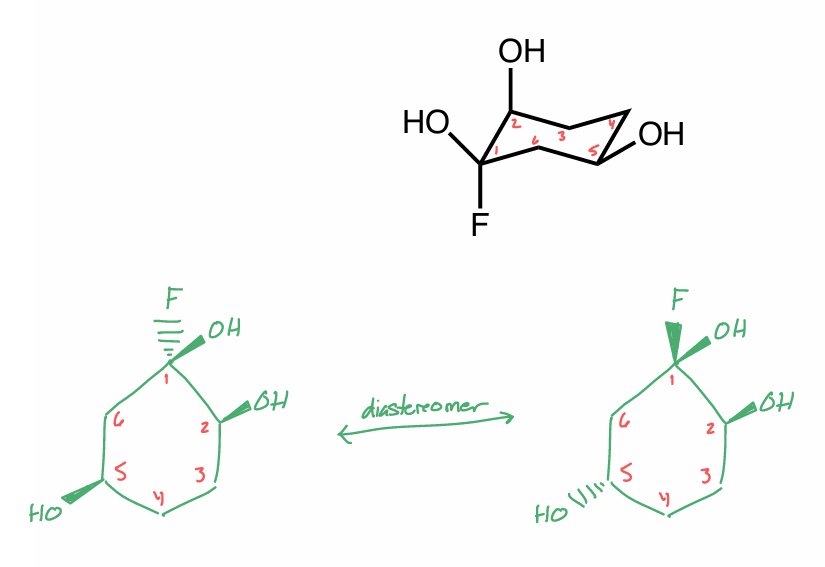
Draw one diastereomer of the molecule
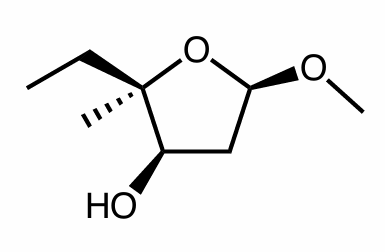
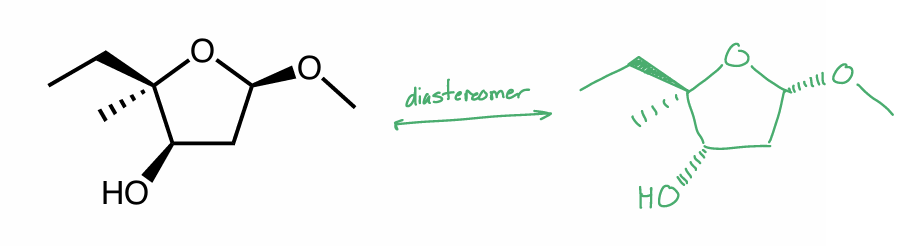
Draw one diastereomer of the molecule
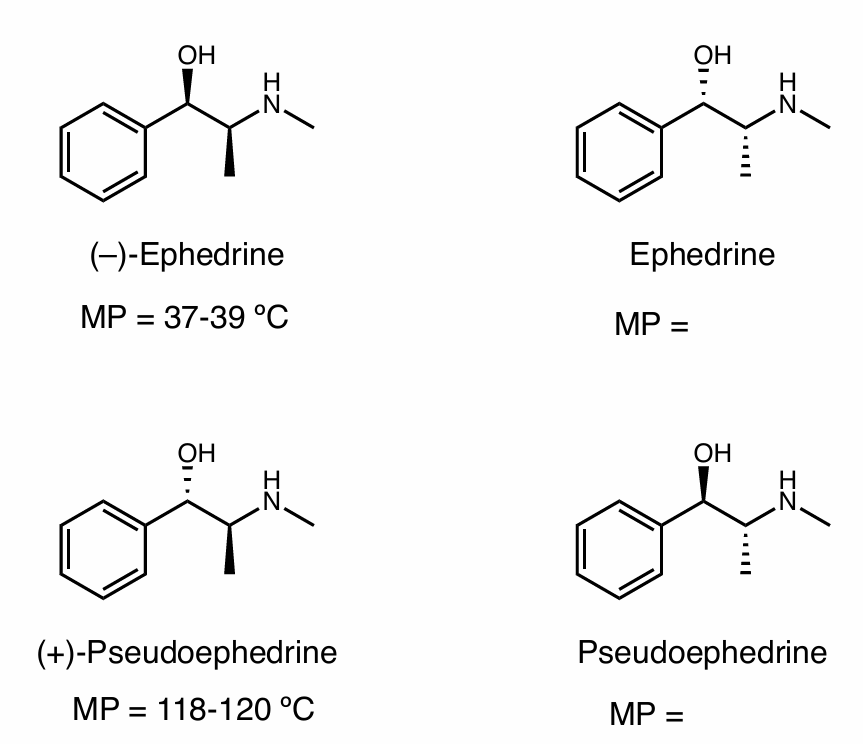
Find the stereochemical relationship between each of these molecules
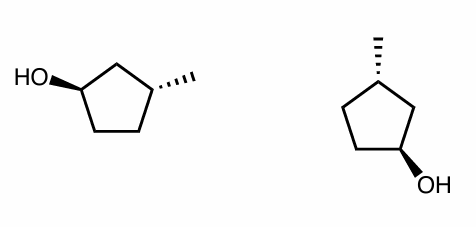
Are the following compounds identical, constitutional isomers, enantiomers, or diastereomers? Use whatever method works for you.
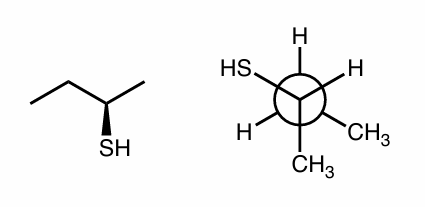
Are the following compounds identical, constitutional isomers, enantiomers, or diastereomers? Use whatever method works for you.
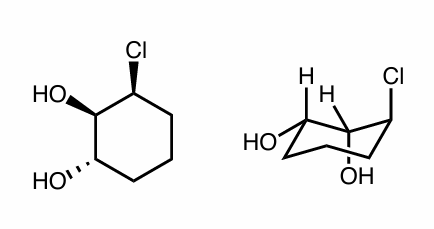
Are the following compounds identical, constitutional isomers, enantiomers, or diastereomers? Use whatever method works for you.
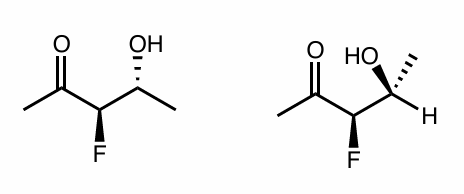
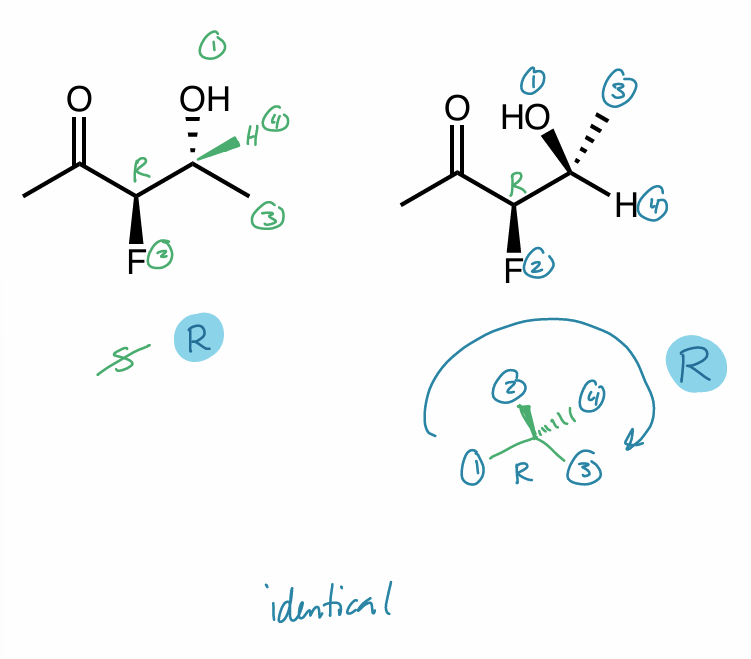
Are the following compounds identical, constitutional isomers, enantiomers, or diastereomers? Use whatever method works for you.
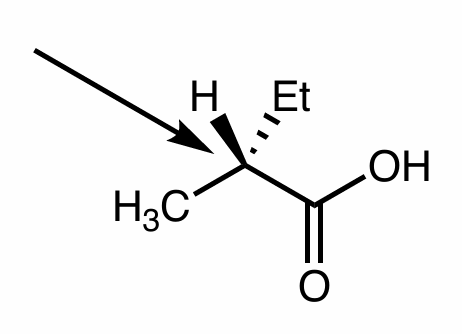
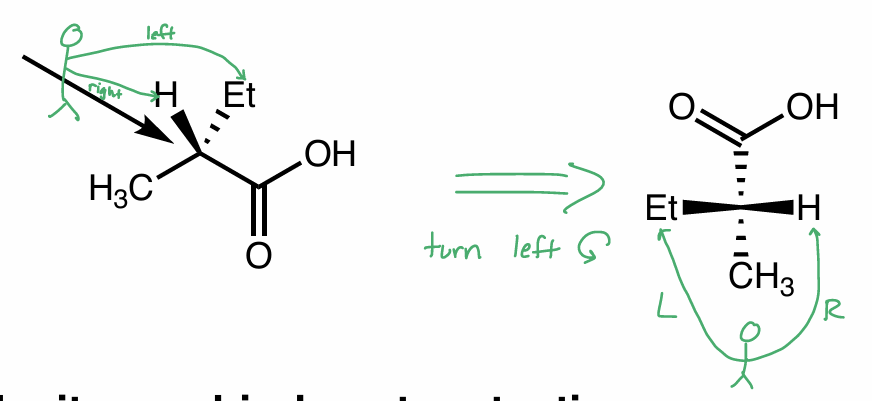
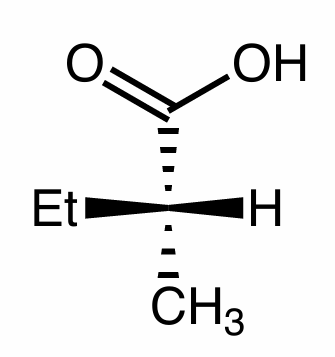
Convert this molecule to a Fischer Projection
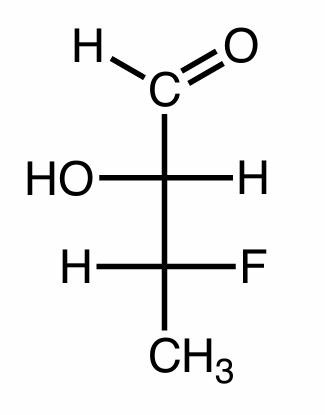
Convert this Fischer Projection into a basic structure

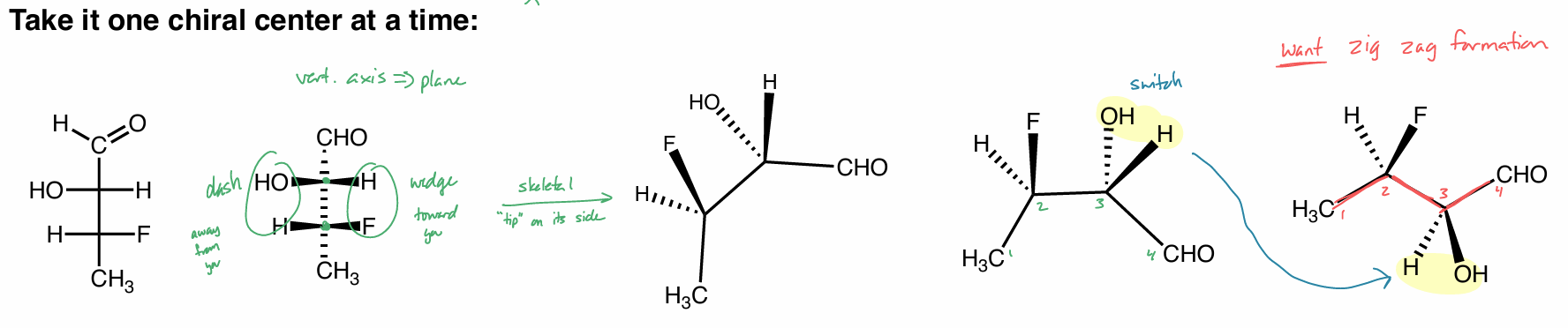
Convert this molecule to a Fischer Projection (FP)
Conver the FP into a skeletal structure (hint: rotate skeletal)
to skeletal
vertical axis → plane (solid line)
1 side is dashed, the other is wedged
“tip” on its side
want carbons in zig zag formation
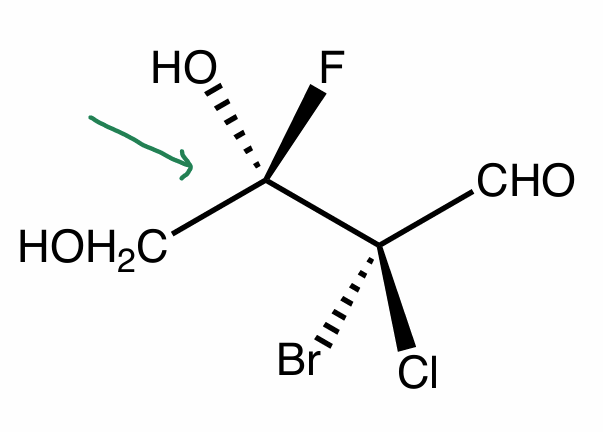
Convert skeletal to NP to FP
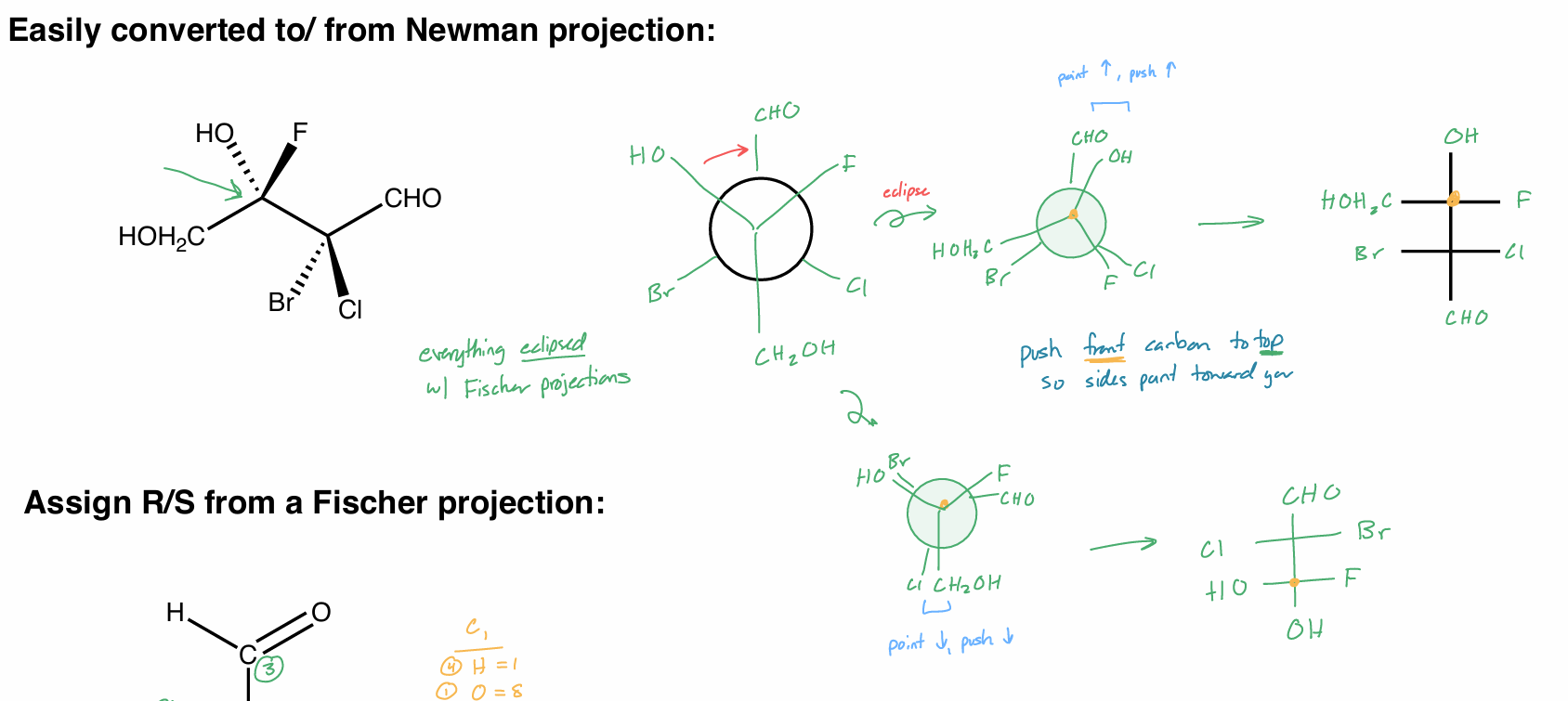
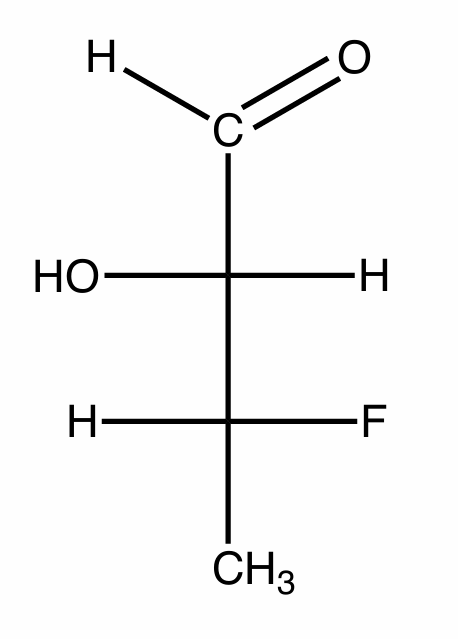
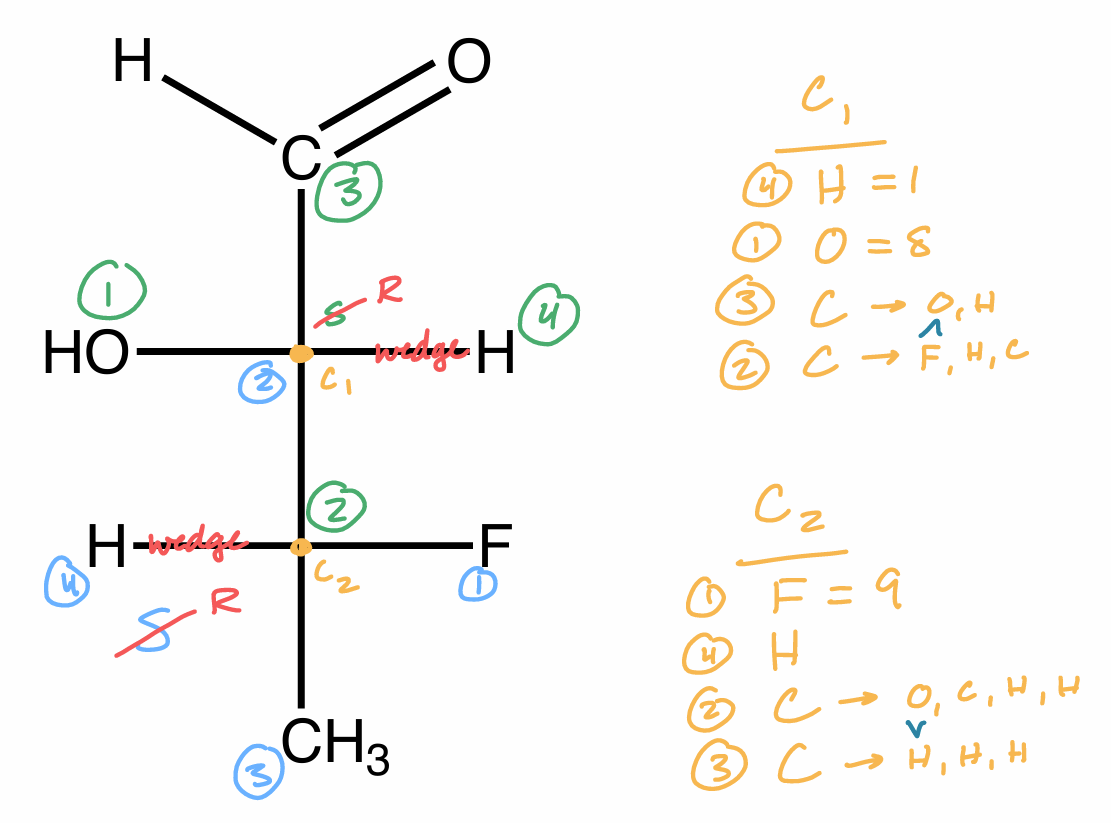
Assign R/S from Fischer Projection
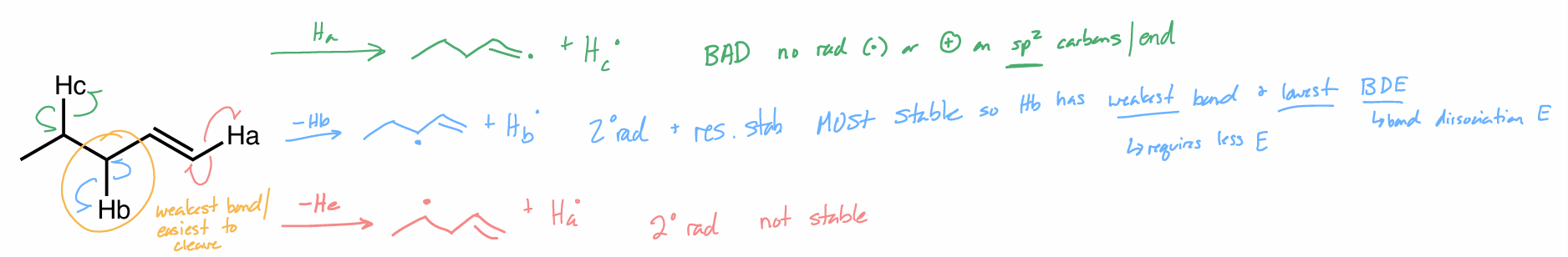
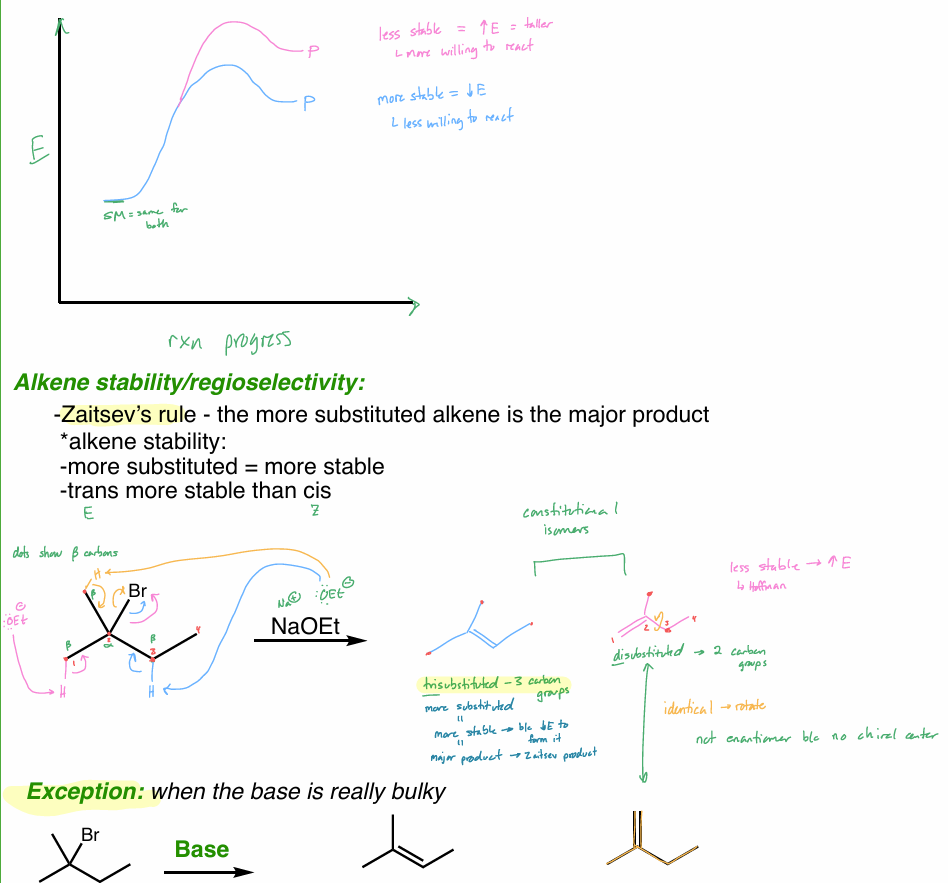
Which substituent forms the most stable product?
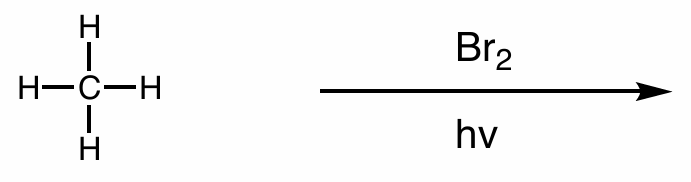
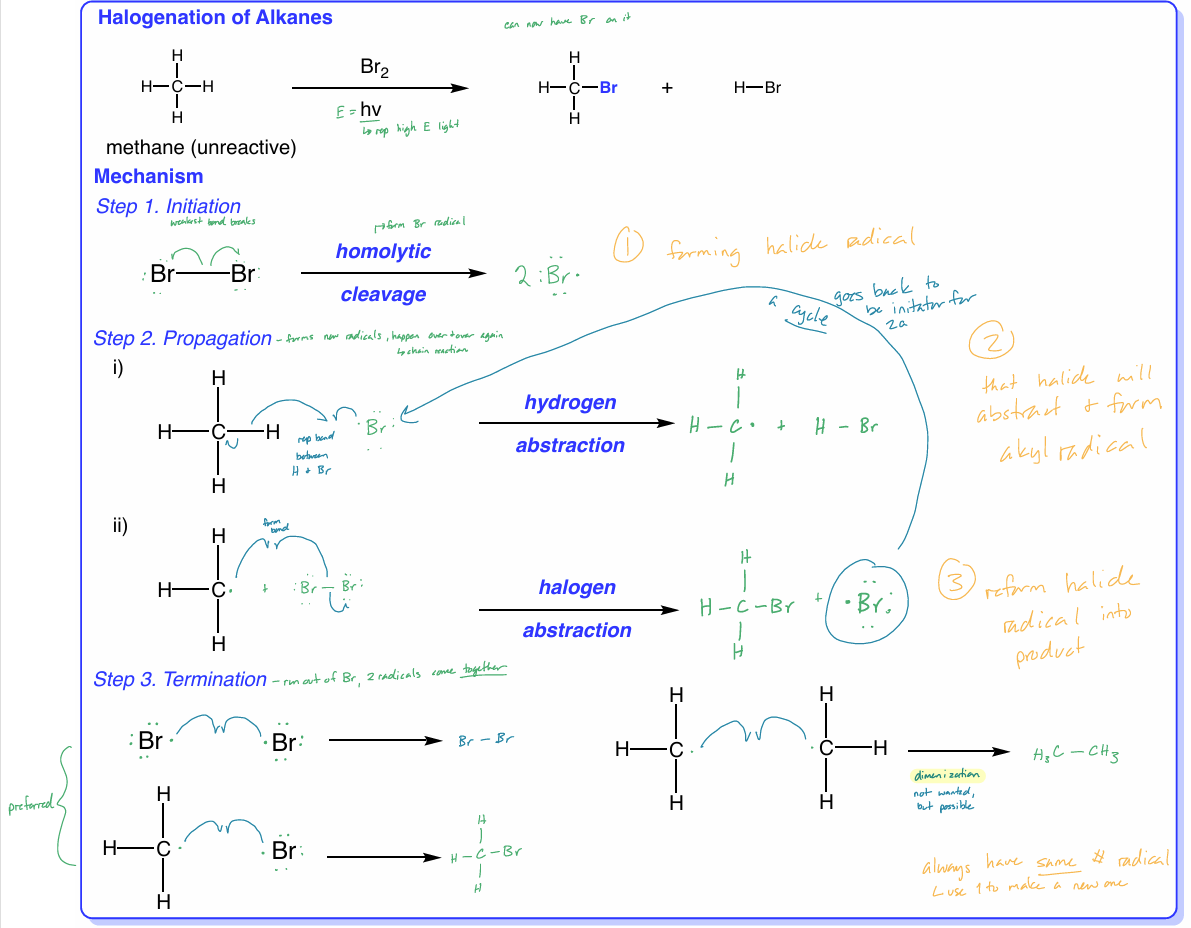
perform the 3-step mechanism of the halogenation of alkanes
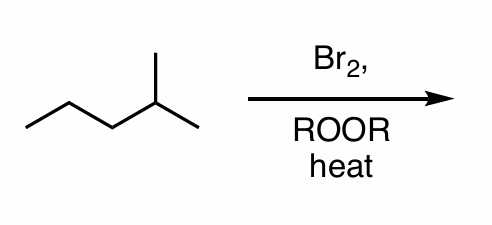
What is the product of this reaction?
Show all steps (hint: halogenation)
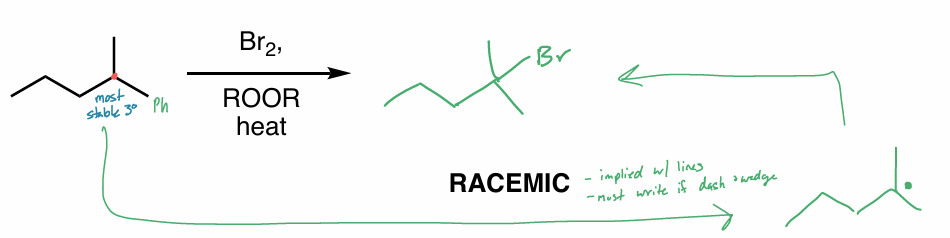
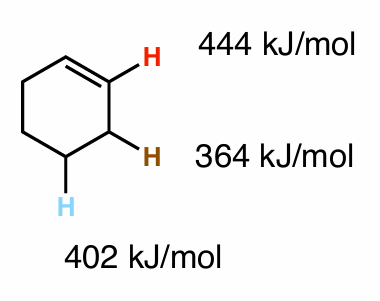
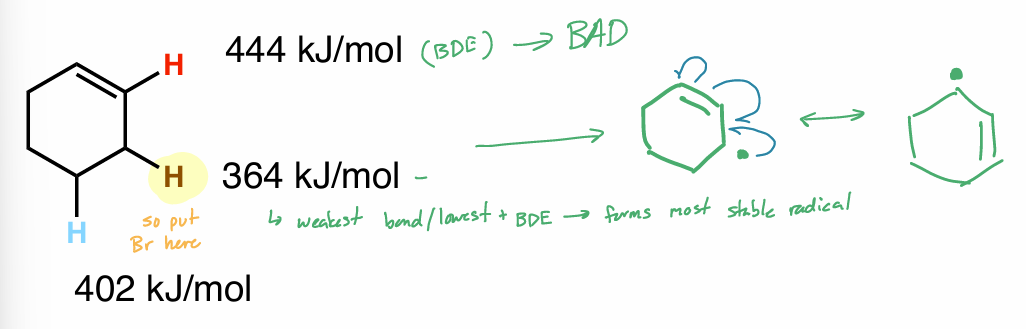
Which hydrogen would for the most stable radical?
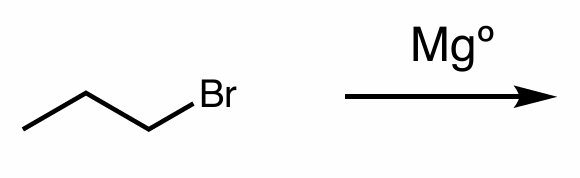
Find the product of this reaction
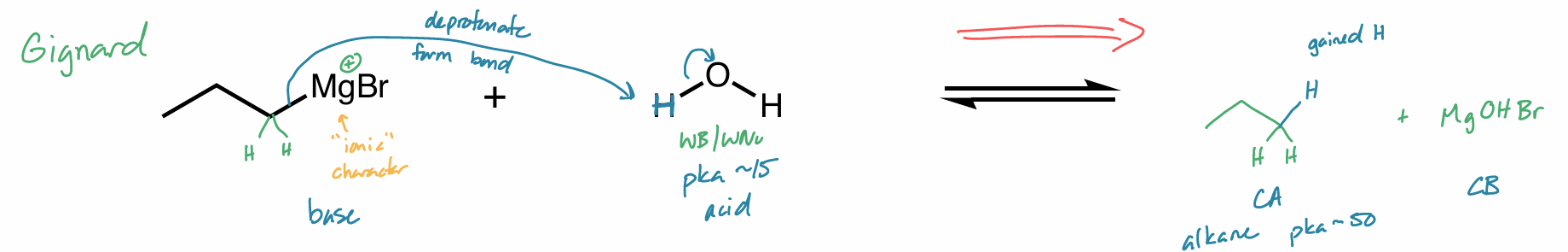
hint: organometallic compounds
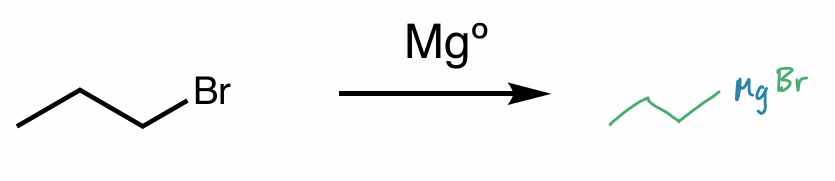
Find the product of this reaction
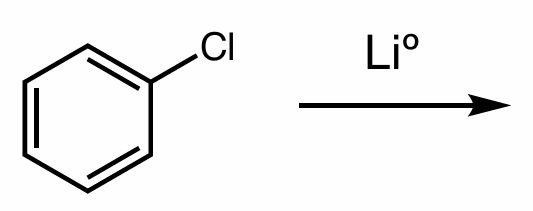
hint: organometallic compounds
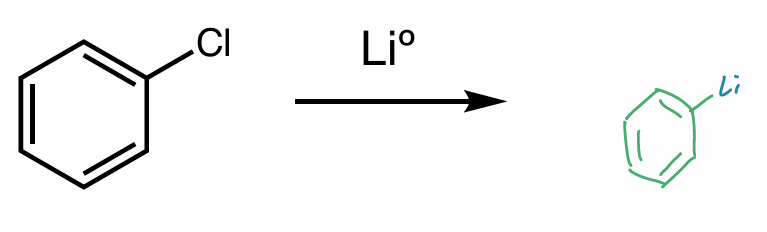
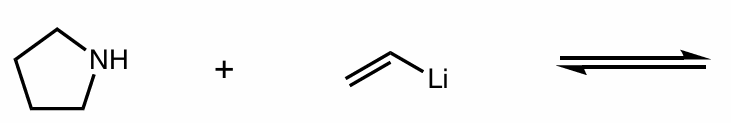
Find the product of this reaction
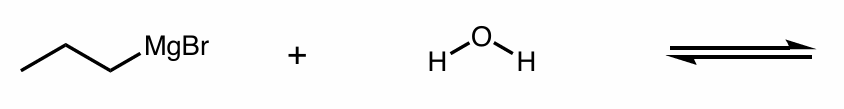
Which side does equilibrium go to?
hint: organometallic compounds
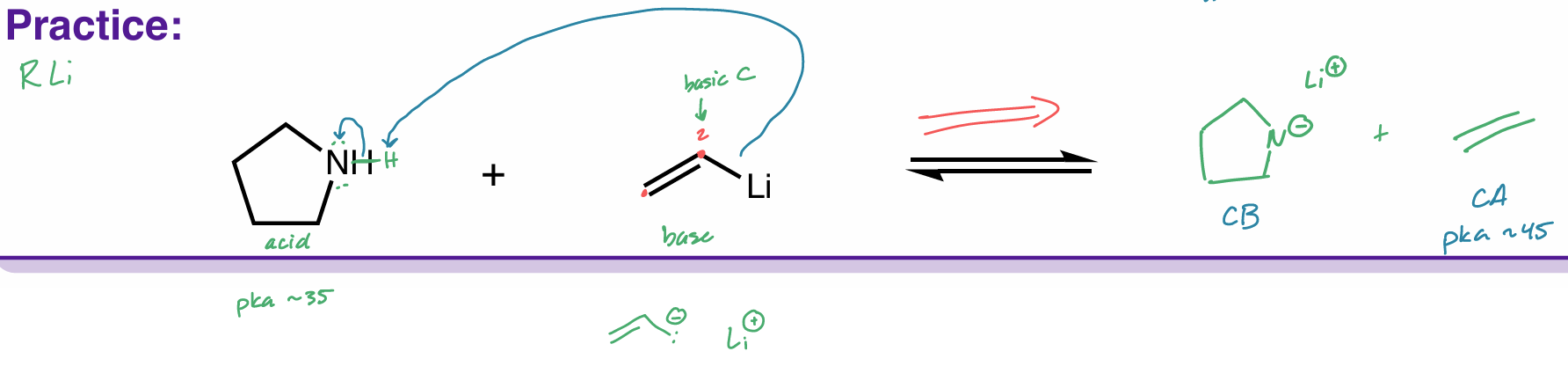
Find the product of this reaction
Which side does equilibrium go to?
hint: organometallic compounds
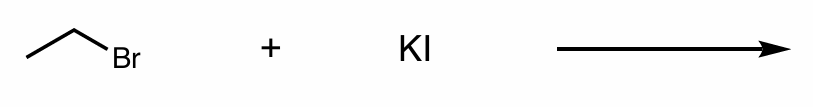
fill in the products
will the following reaction proceed as written?
No

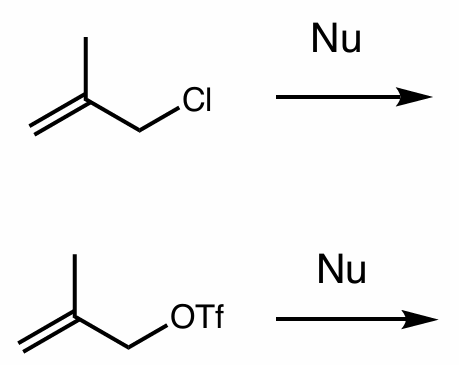
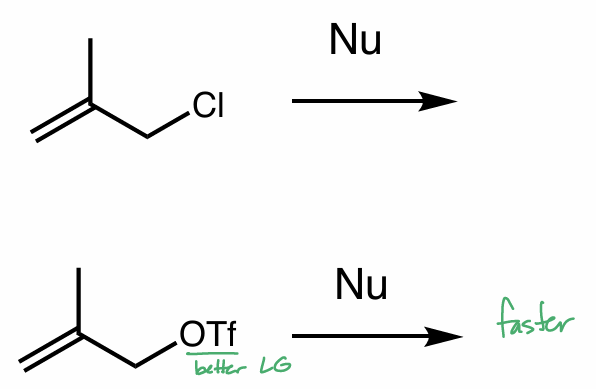
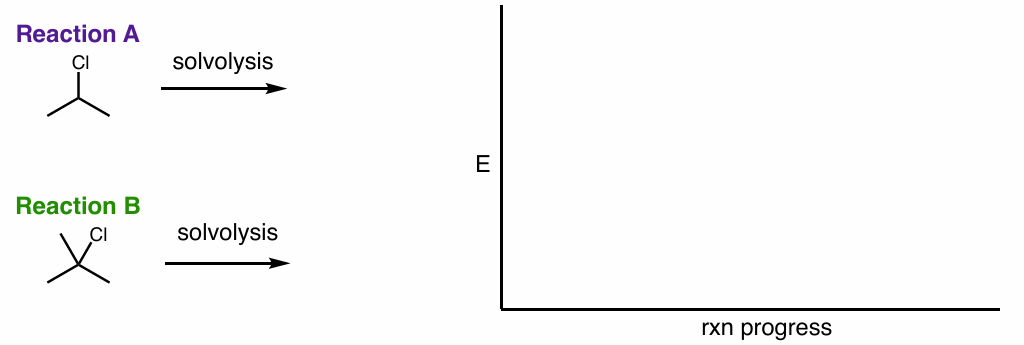
which of these reactions is better/would happen faster and why?
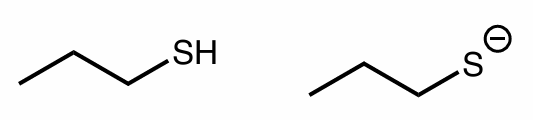
which is a strong/better nucleophile?

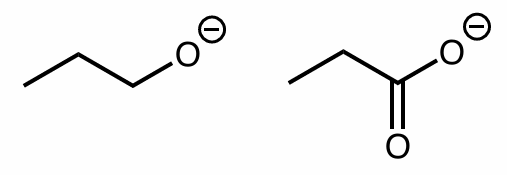
which is a strong/better nucleophile?
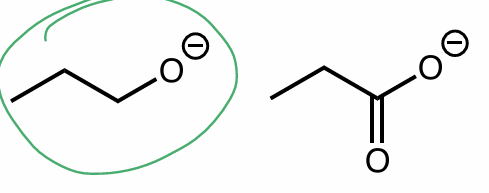
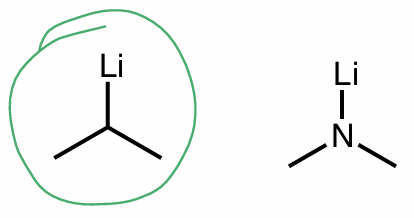
which is a strong/better nucleophile?
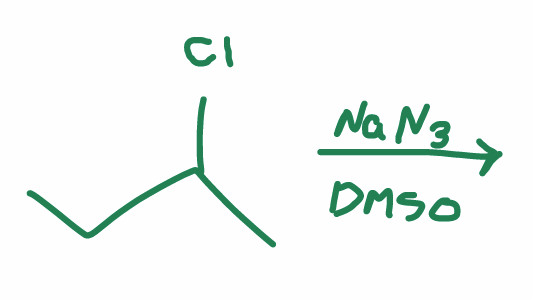
what is the reaction scheme?
find the products
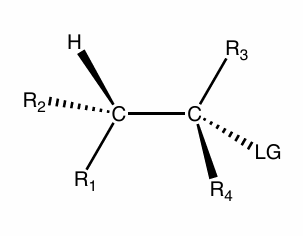
SN2
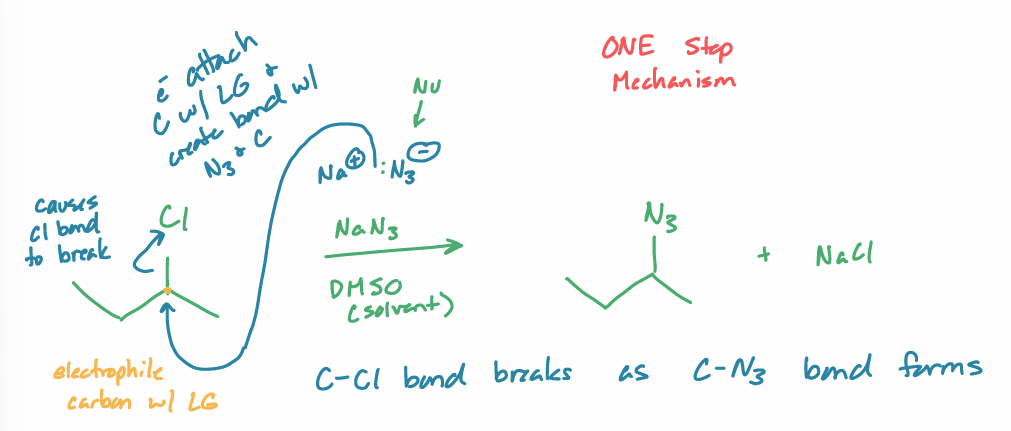
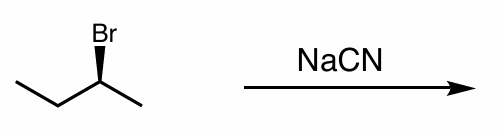
what is the reaction scheme?
find the products
SN2
backside attack
can only tell if have chiral center
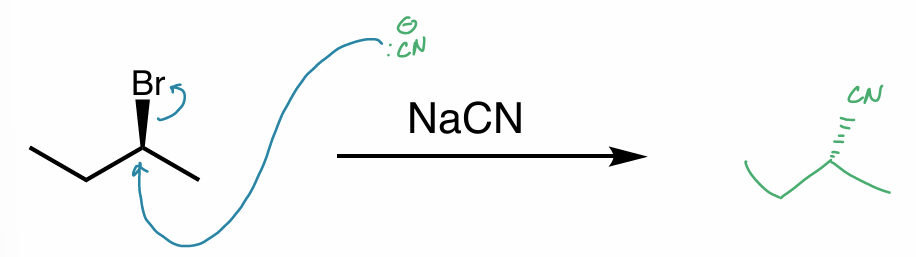
what is the reaction scheme?
find the products
is the reverse reaction possible?
SN2
picture
No b/c OMe is NOT a good LG
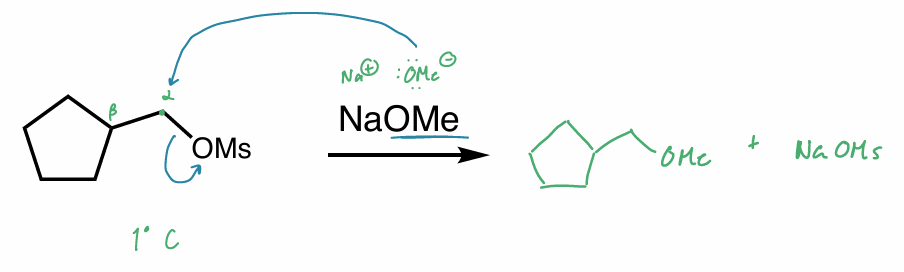
what is the reaction scheme?
find the major product
draw the rxn coordinate diagram
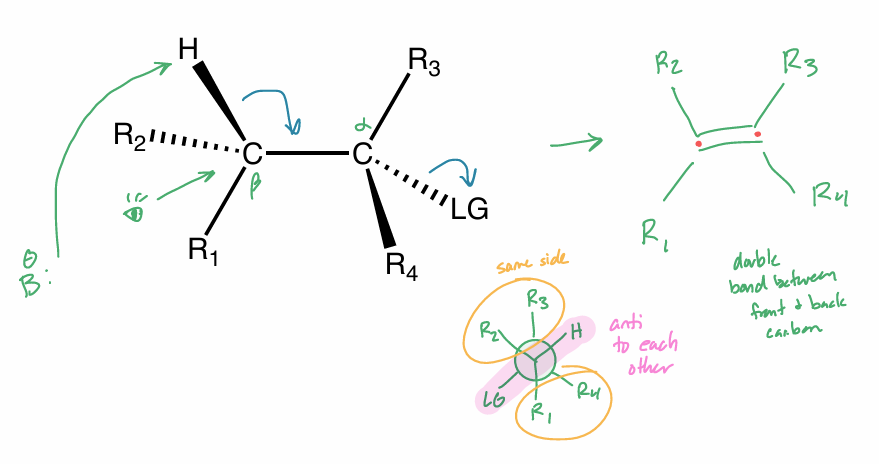
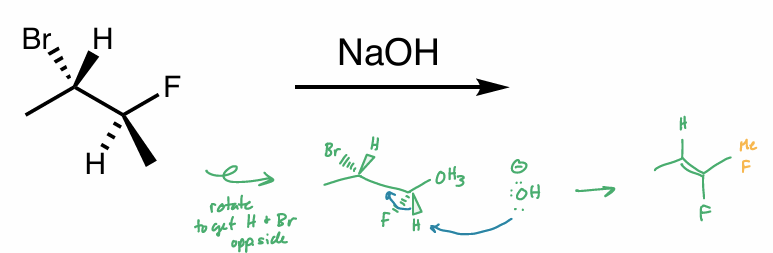
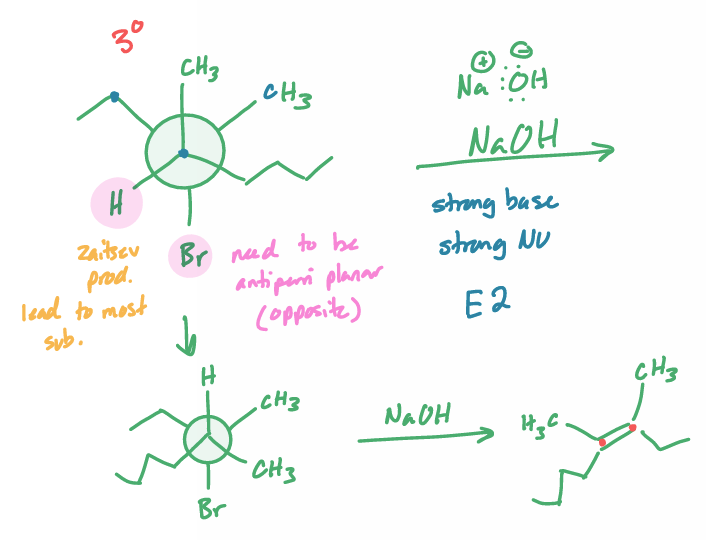
E2
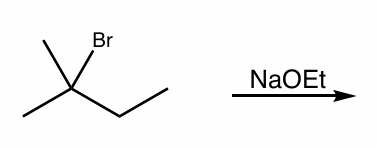
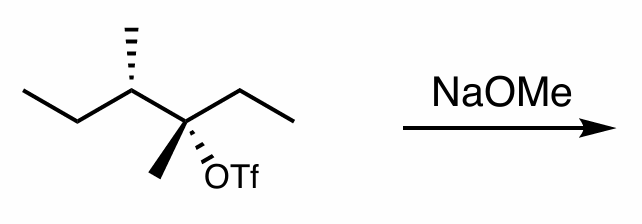
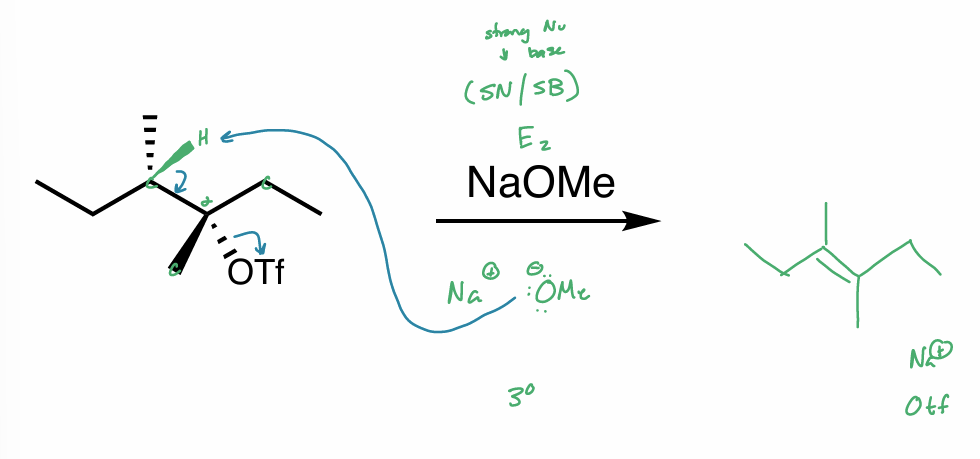
what is the reaction scheme?
find the products (hint: convert to NP)
E2
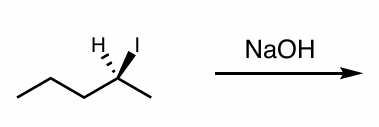
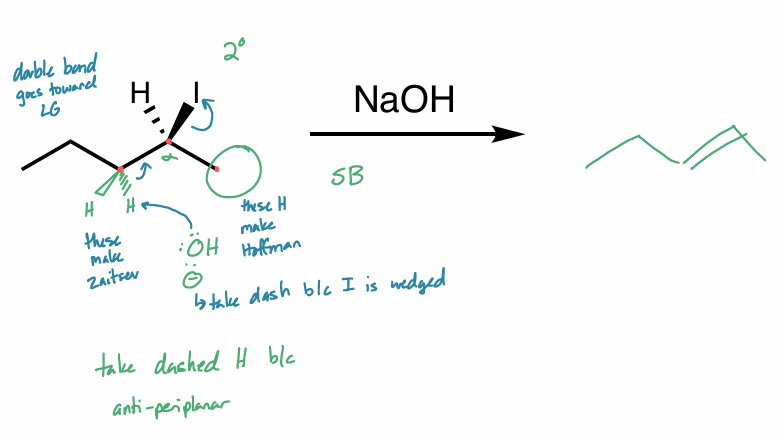
what is the reaction scheme?
find the products
E2
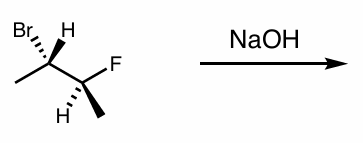
what is the reaction scheme?
find the products
E2
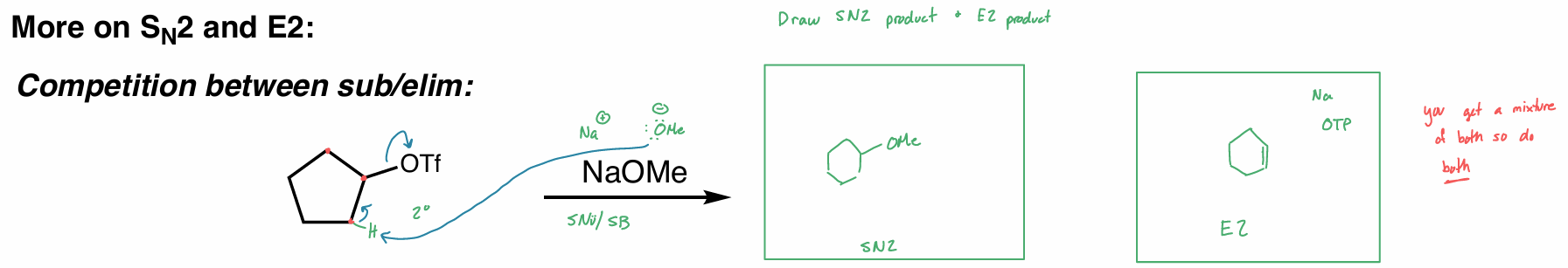
find all of the products (hint: SN2 + E2)
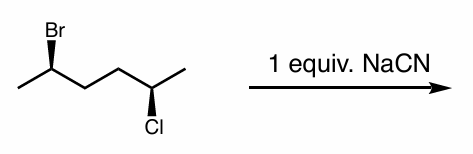
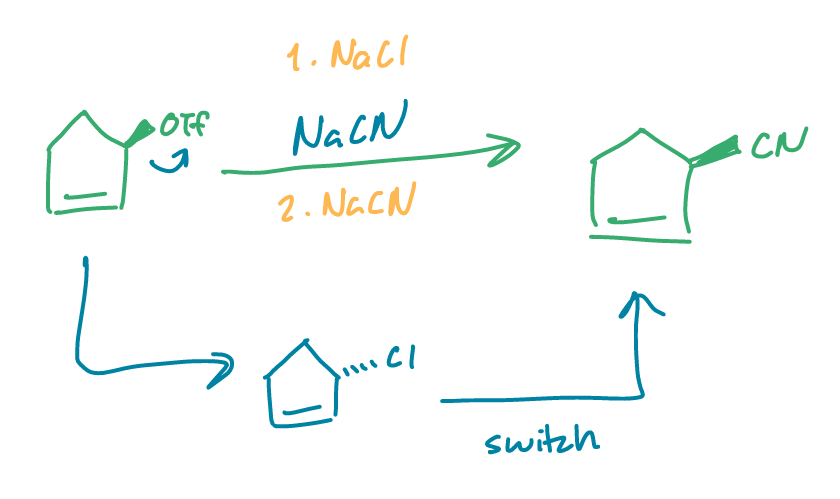
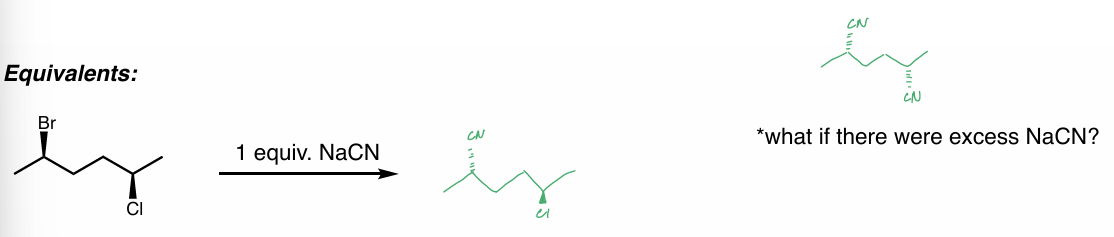
find the product
what if there were excess NaCN?
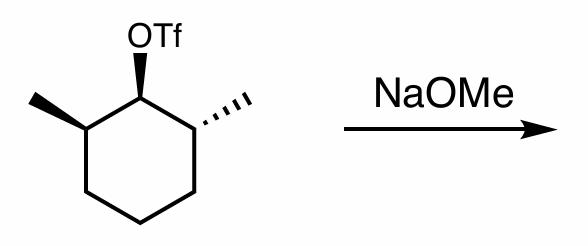
what is the reaction scheme?
find the major product
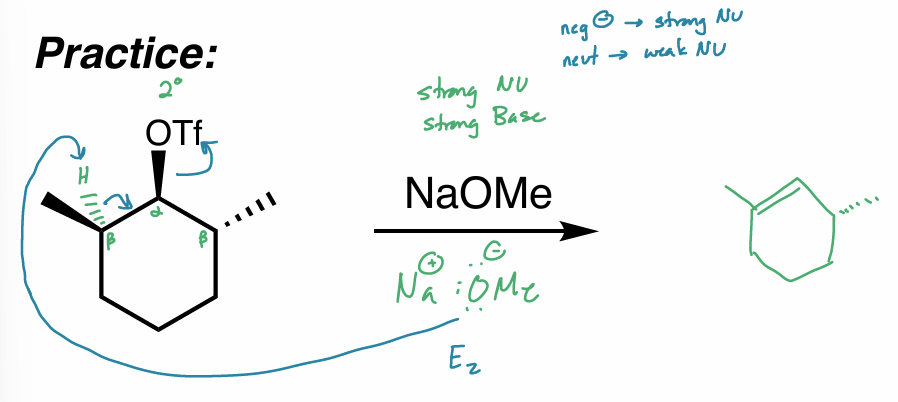
what is the reaction scheme?
find the major product
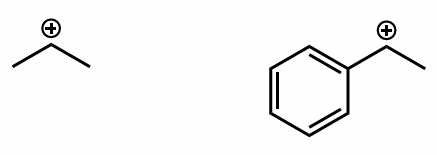
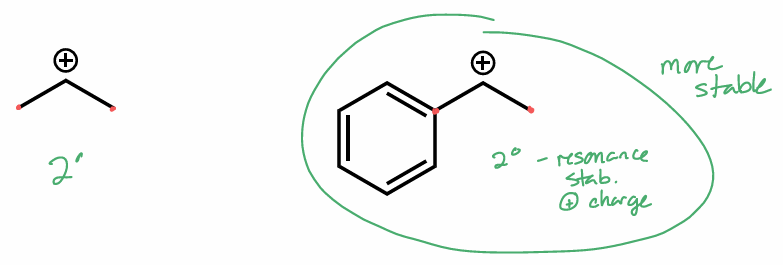
which molecule is the more stable and why?
find the product of each reaction
draw the rxn coordinate diagram
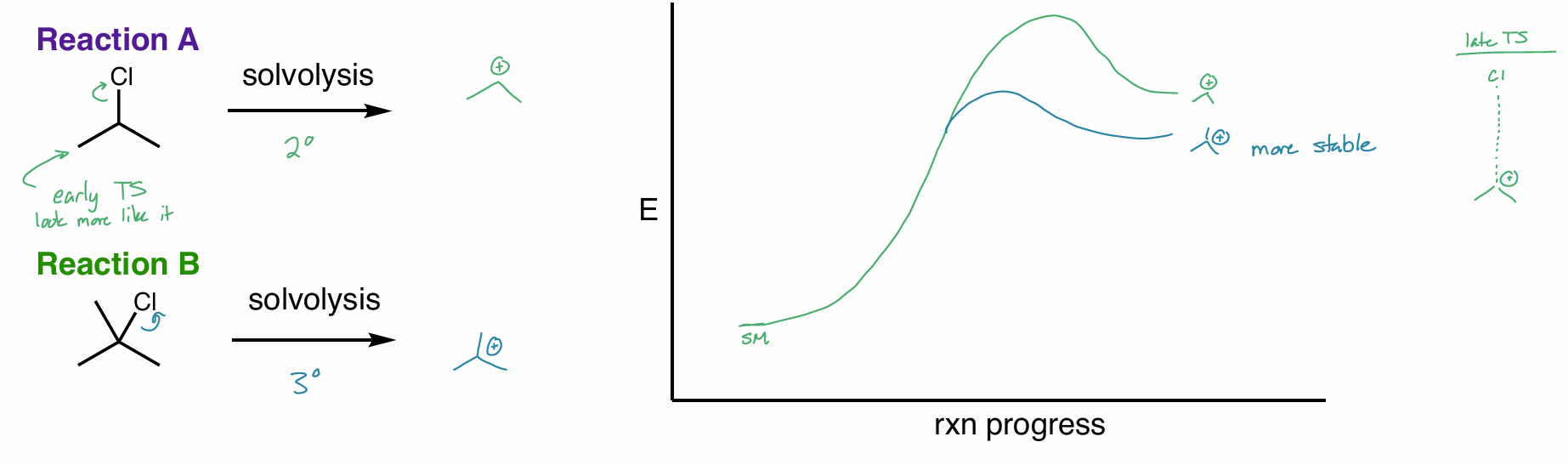
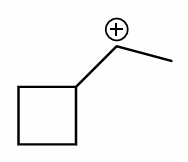
perform a ring expansion on this molecule
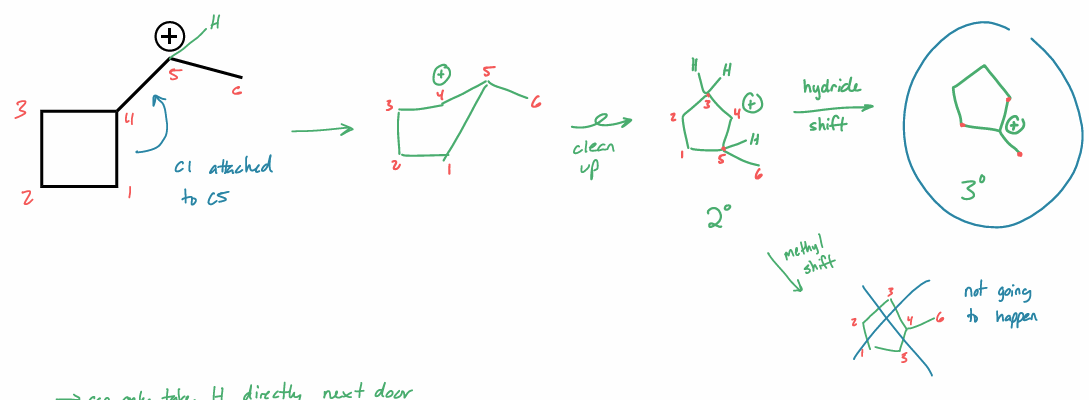
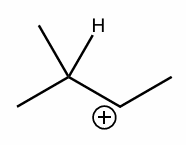
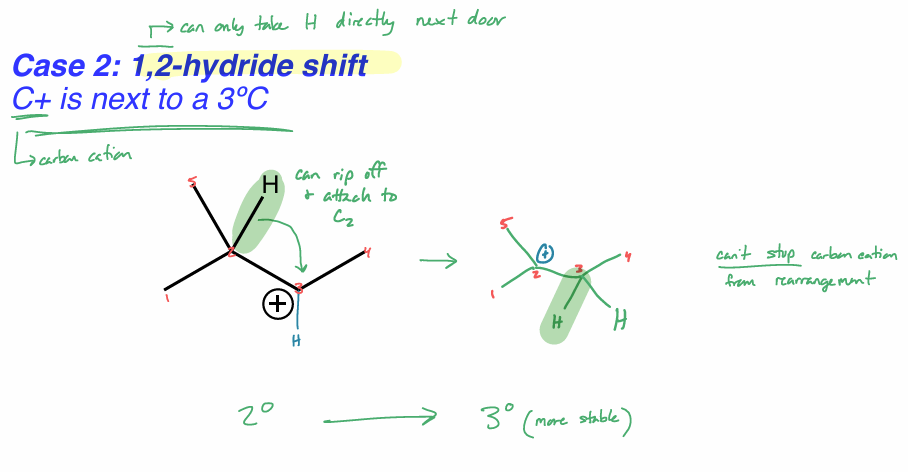
perform a 1,2-hydride shift
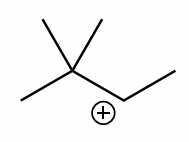
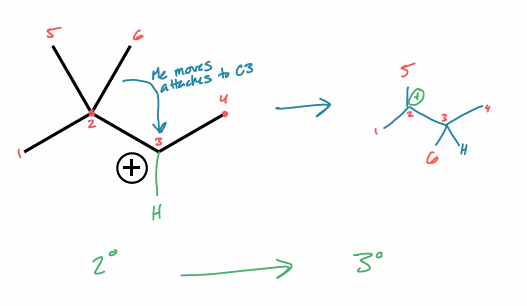
perform a 1,2-methyl shift on this molecule
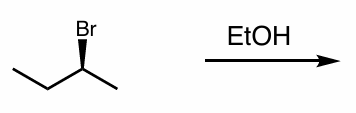
what is the reaction scheme?
find the product (show your work)
is this reaction always racemic?
SN1
pic
No
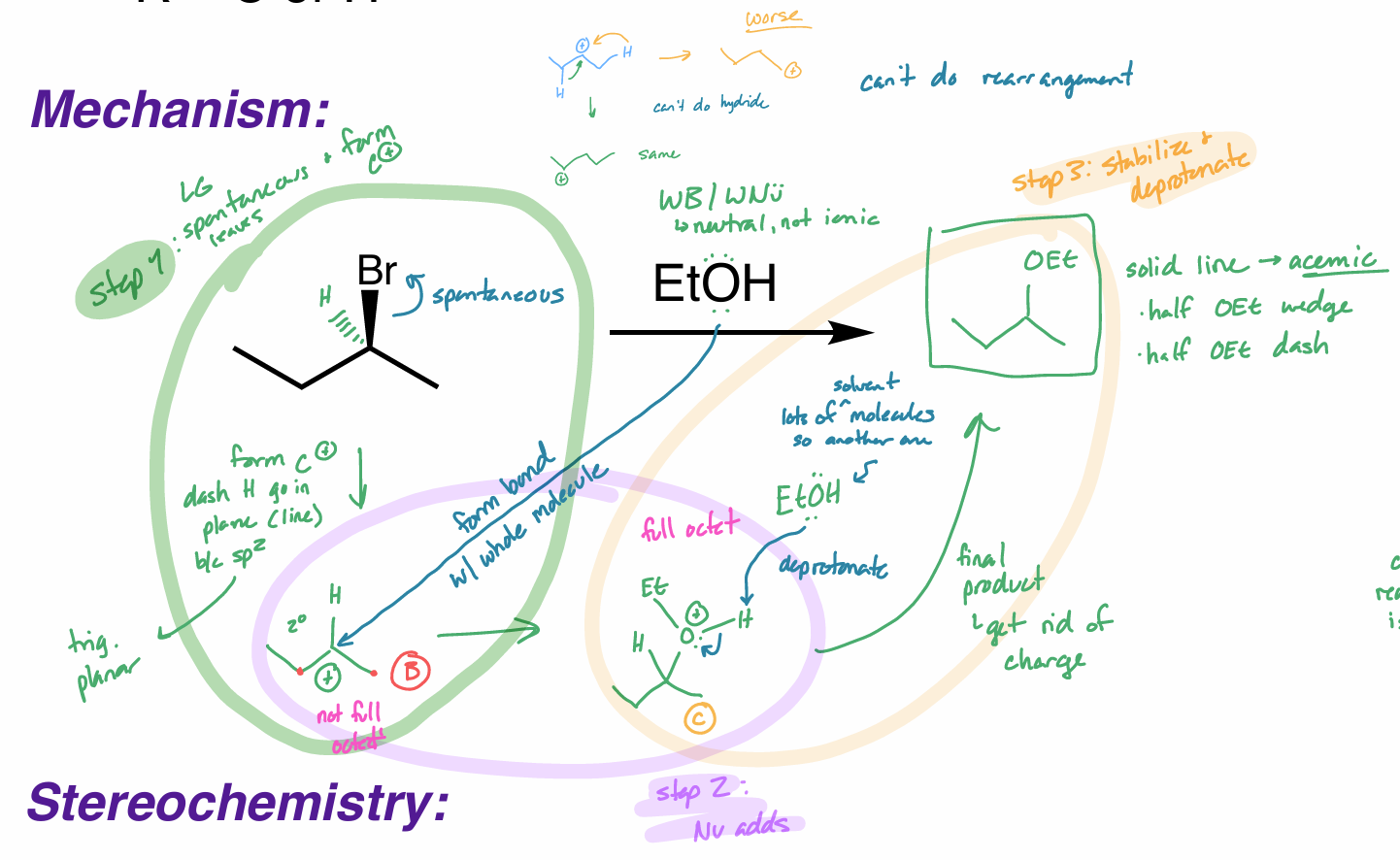
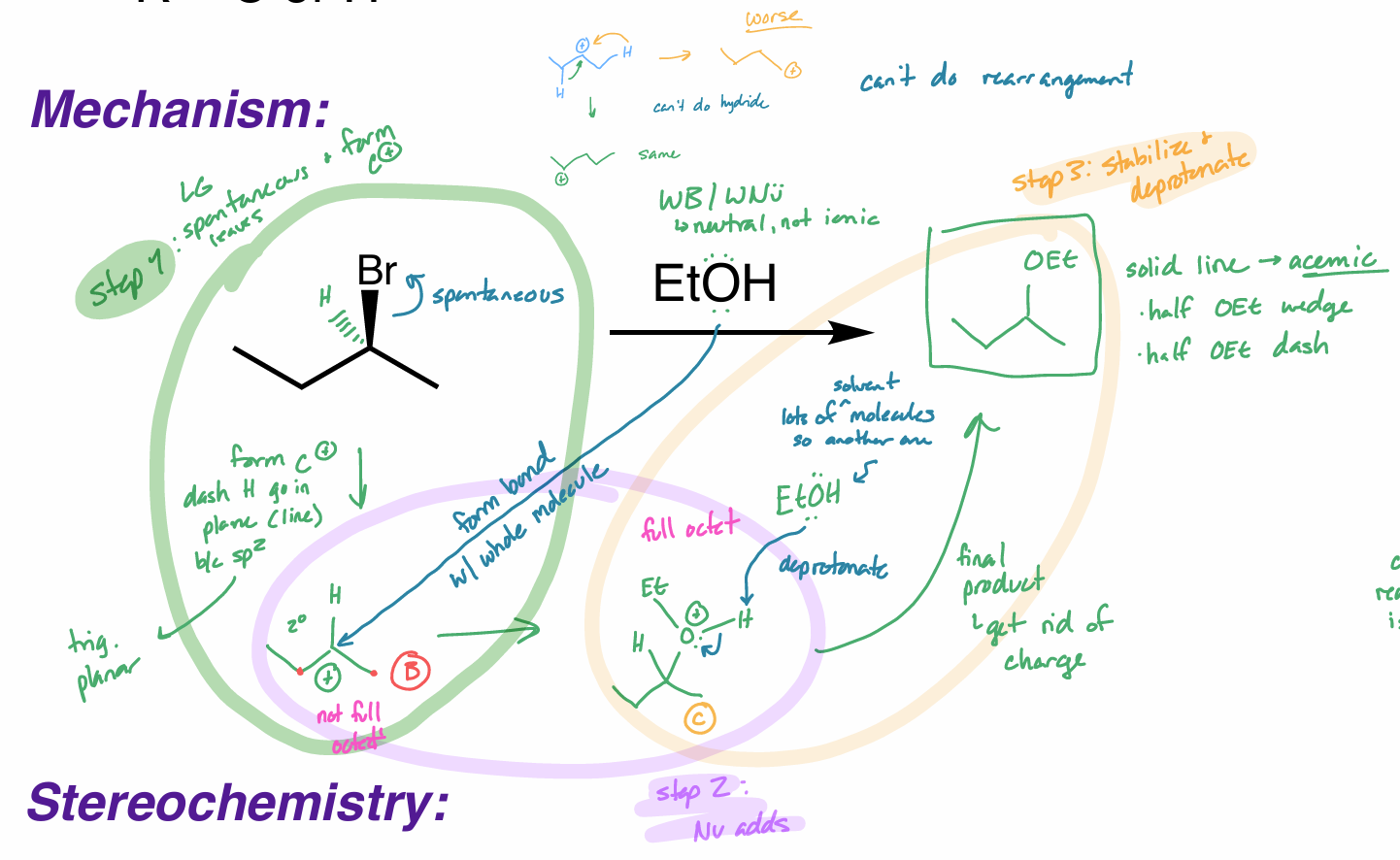
what is the reaction scheme?
find the product (show your work)
E1
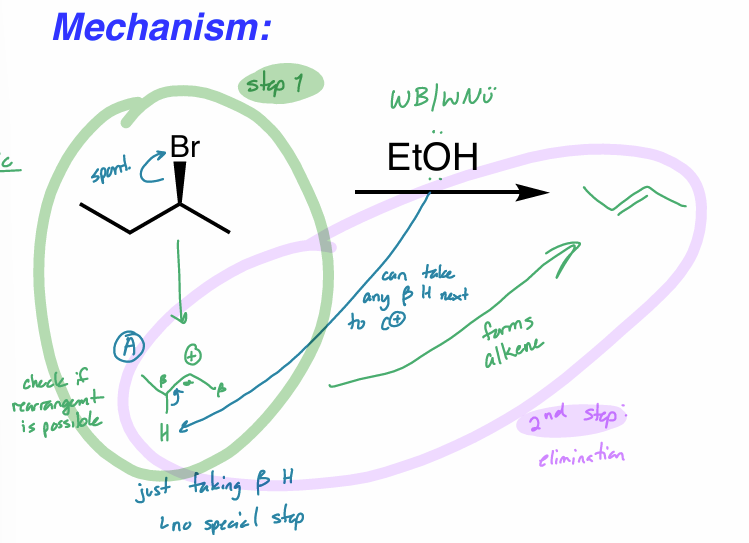
draw the rxn coordinate diagram for this reaction
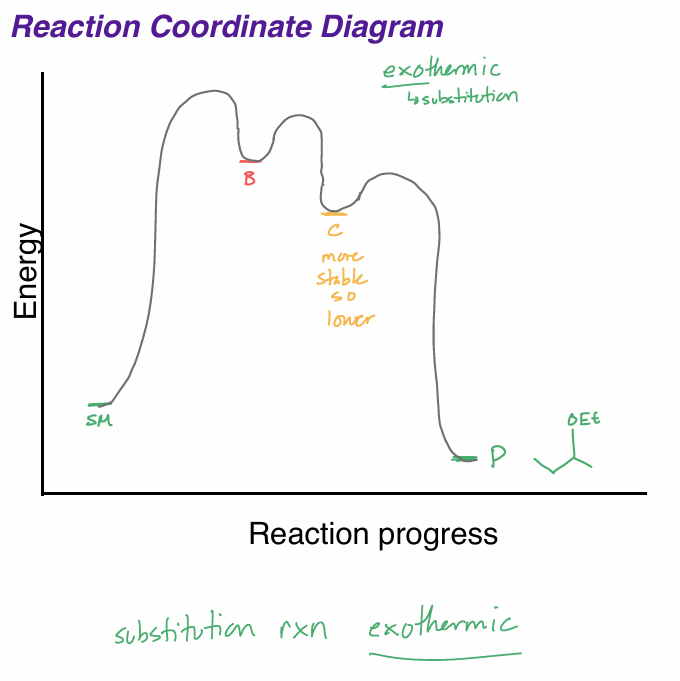
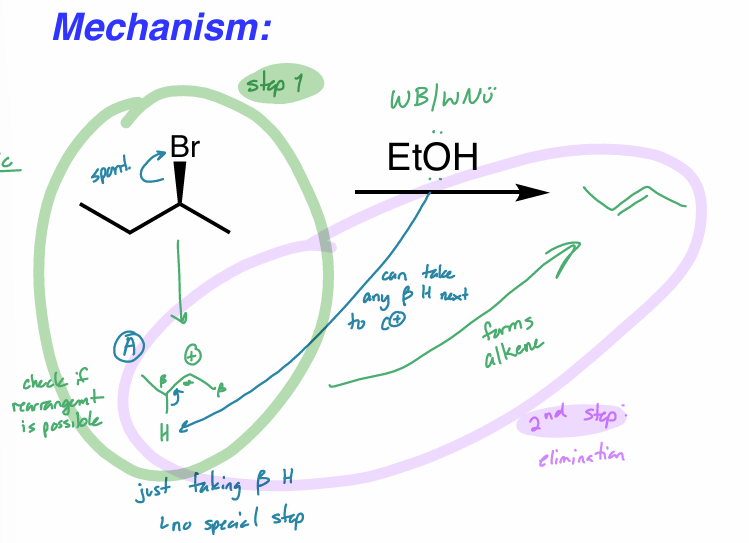
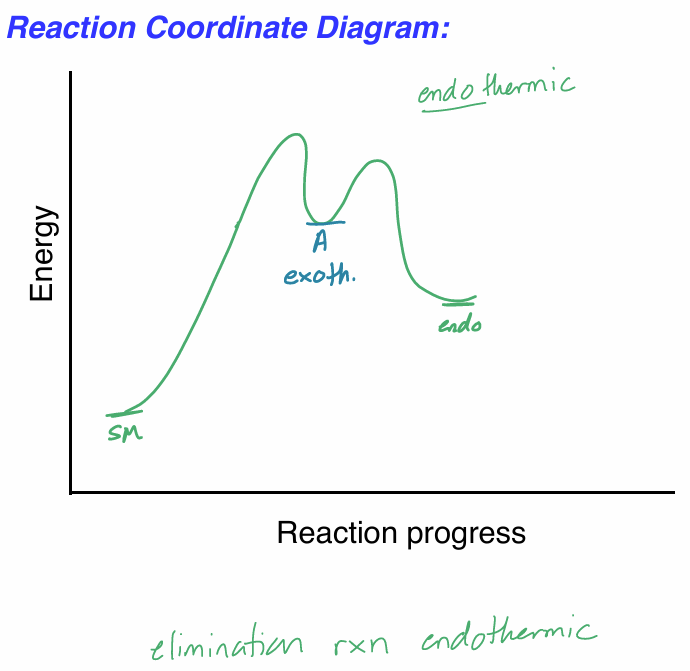
draw the rxn coordinate diagram for this reaction
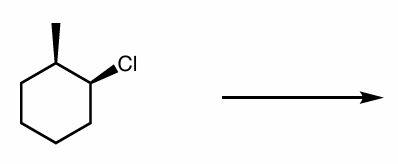
predict the major organic product(s)
specify stereochemistry if applicable
draw the rxn coordinate diagram
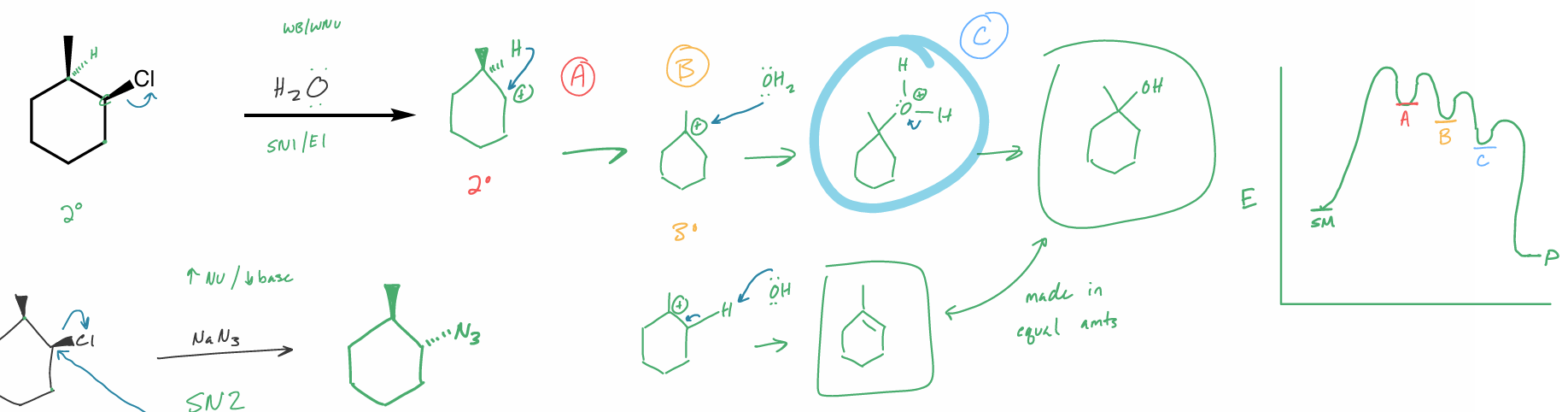
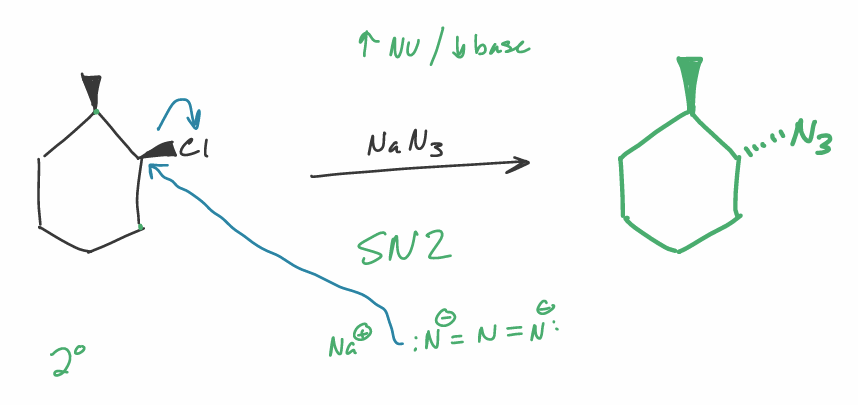
predict the major organic product(s)
specify stereochemistry if applicable
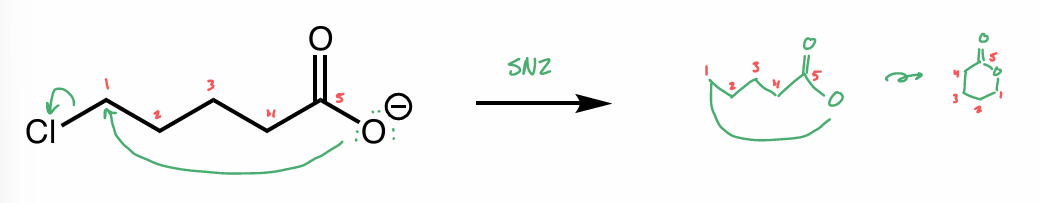
perform this intramolecular reaction
is this the same molecule?
pic
yes
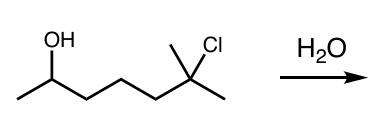
perform this intramolecular reaction
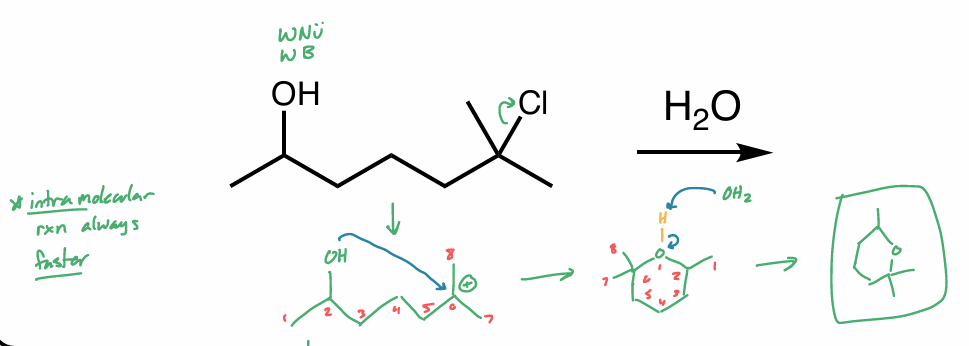
is this the same molecule?
pic
yes
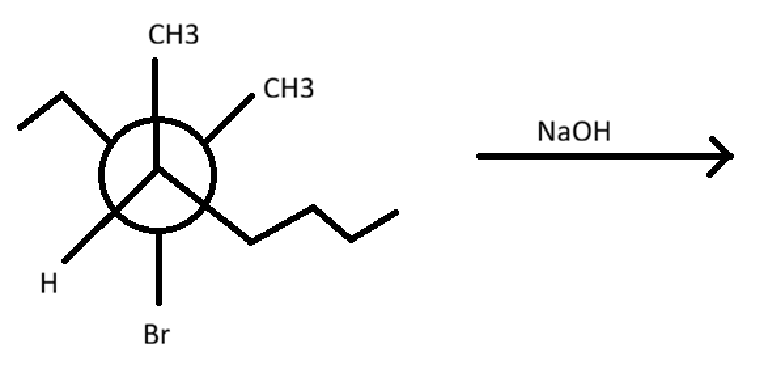
what is the reaction scheme?
find the major product
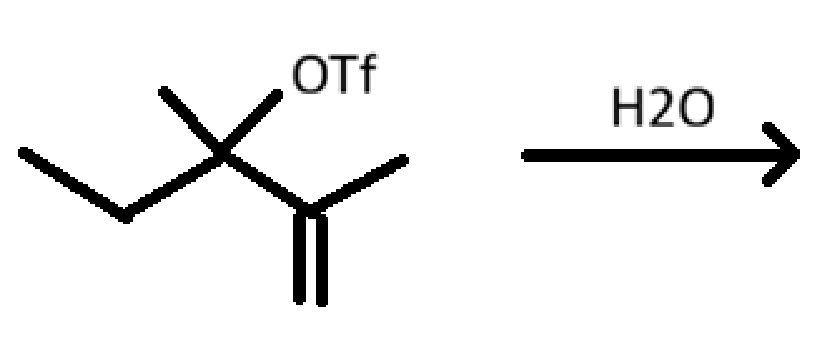
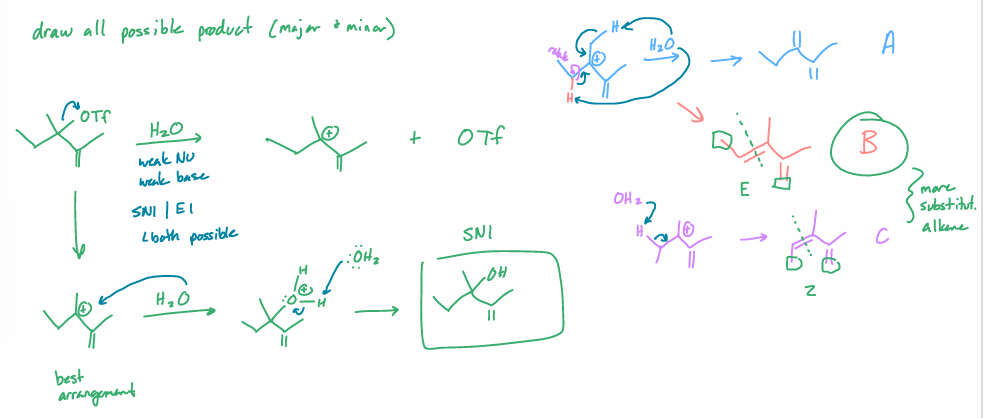
draw all possible products (major & minor)
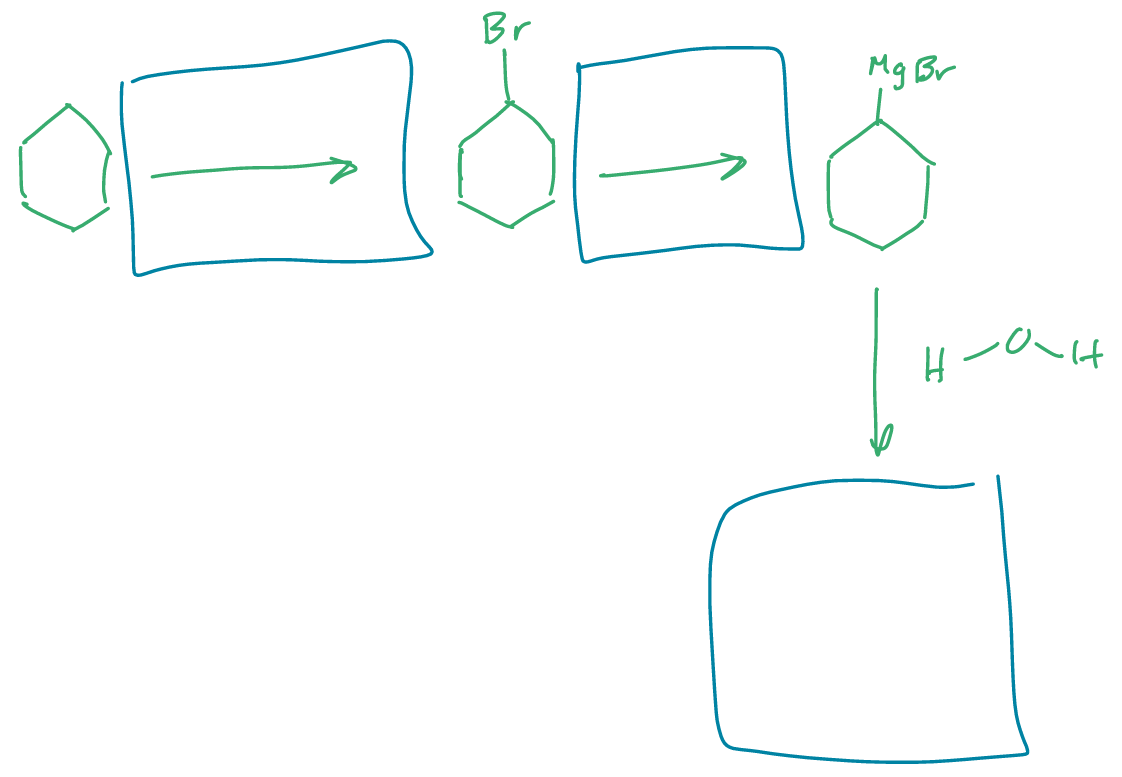
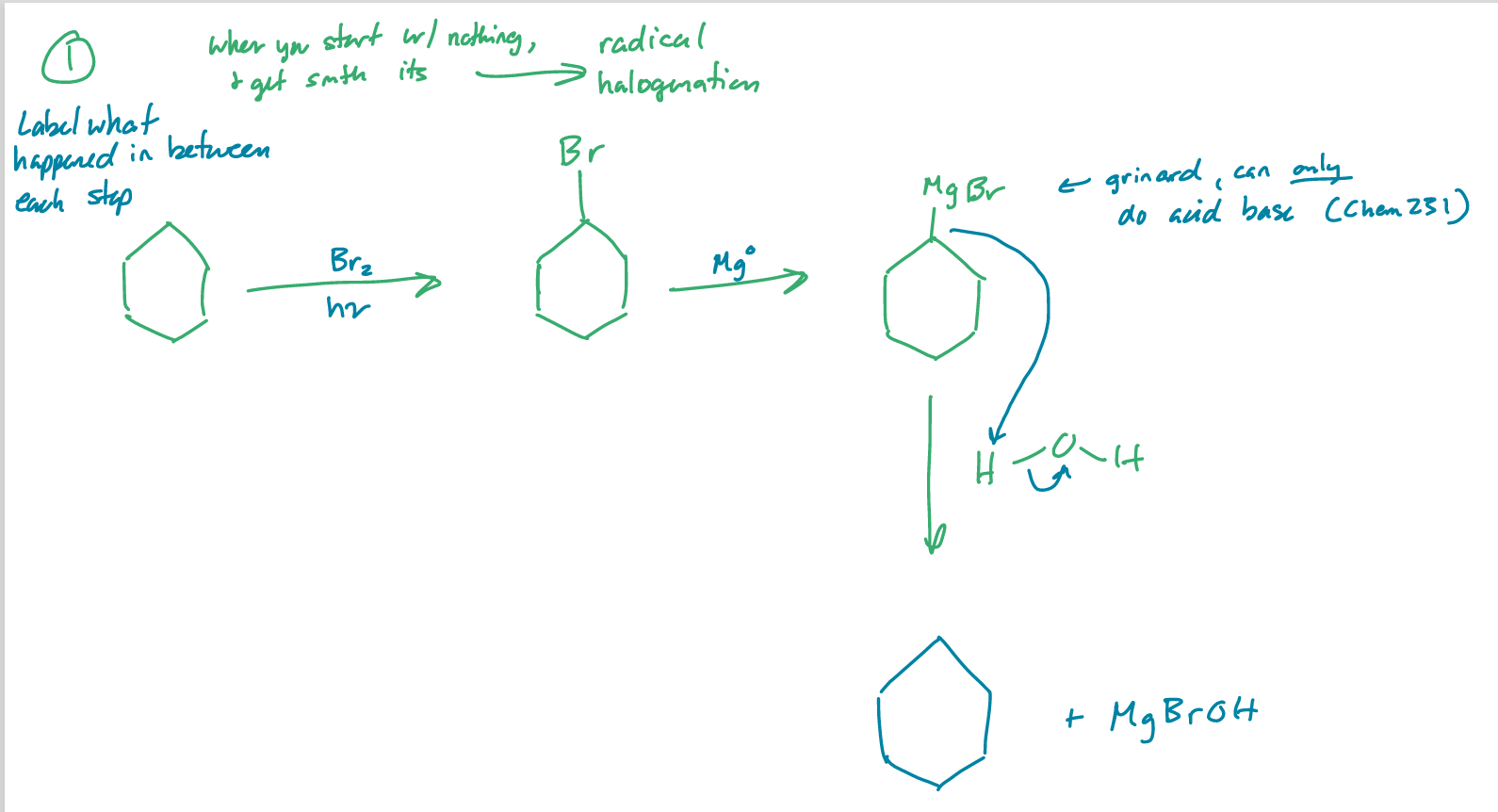
label what happened in between each step
draw the product
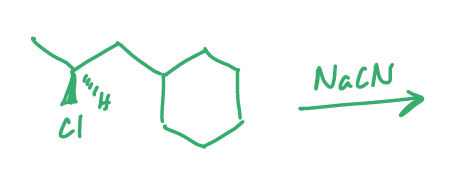
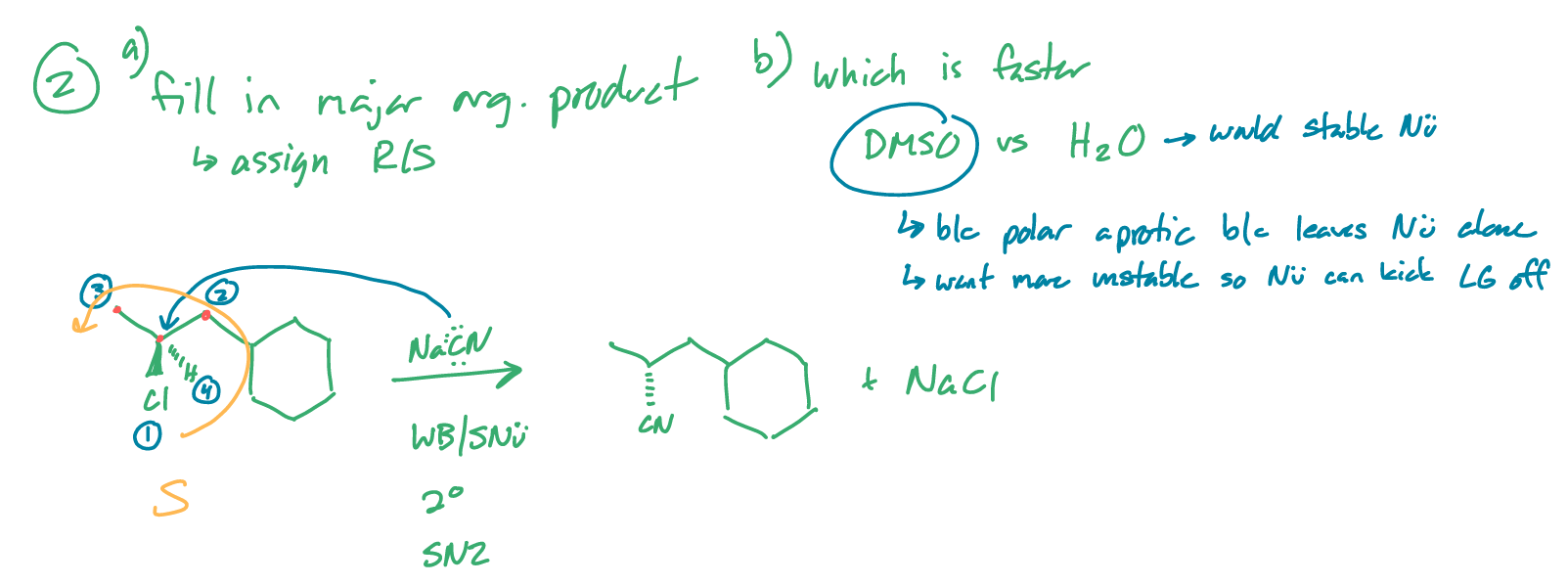
fill in the major organic product
assign R/S
which is a faster solvent and why?: DMSO vs H2O
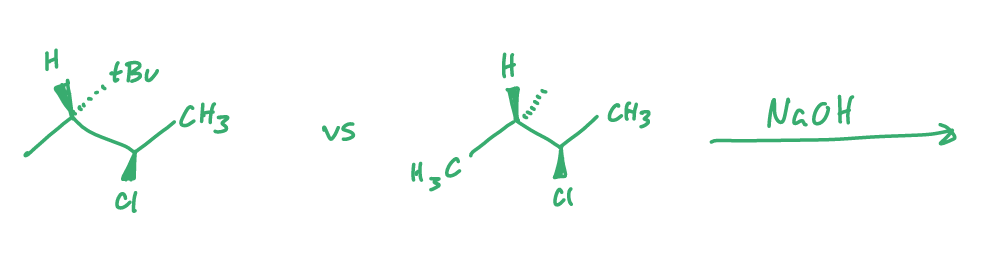
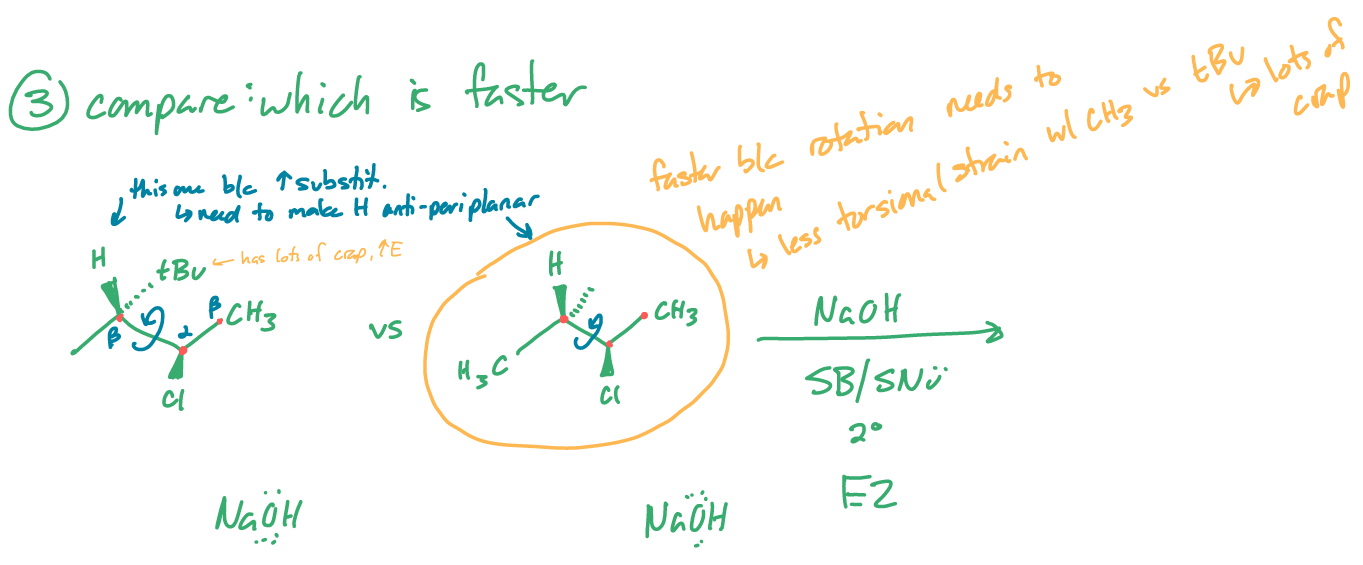
compare which is faster and why?
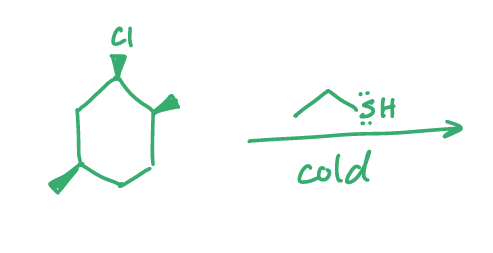
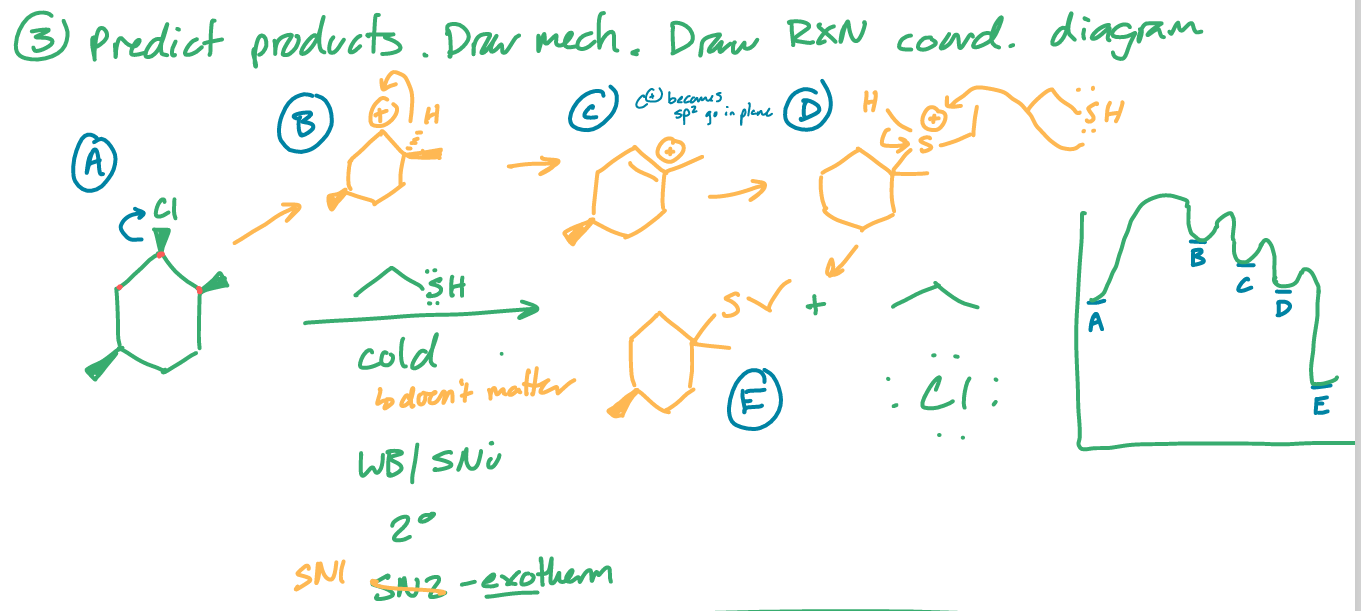
predict the product(s)
draw the mechanism
draw the rxn coordinate diagram
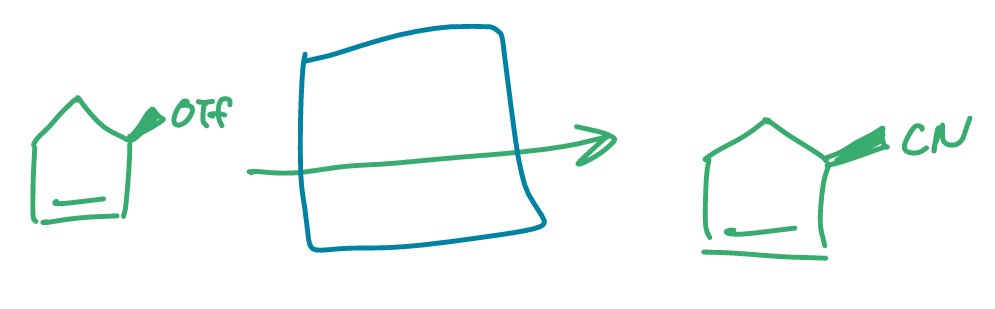
how did this reaction take place?
fill in what reacted with the molecule (hint: more than 1 step)