physical organic chemistry
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
show plot of distance C-OH vs distance C-I
distance between nucleophile, central carbon and leaving group
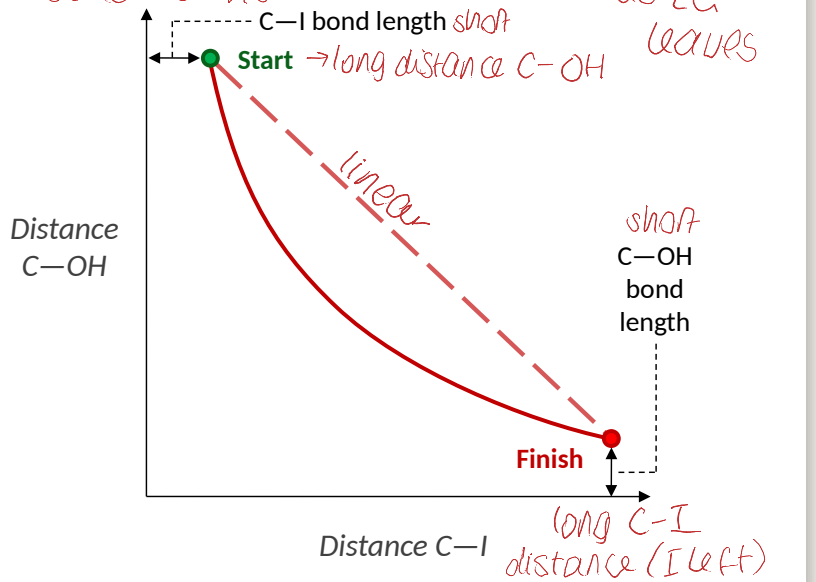
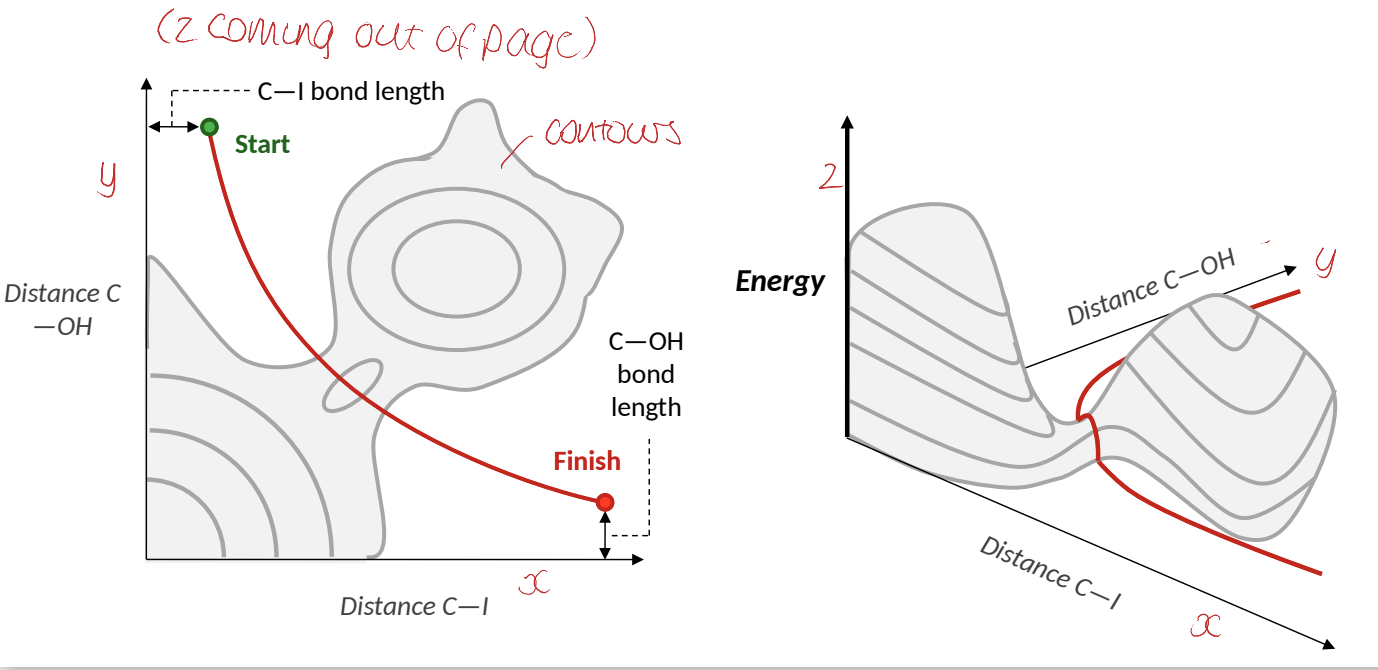
what does this show about the Sn2 reaction?
it doesn’t go straight over high energy hill (higher energy barrier)
that is why distance of C-OH vs C-I graph is not linear
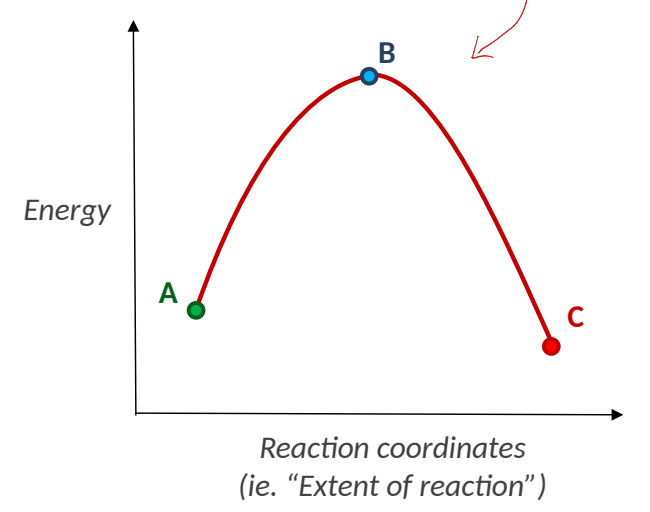
by labelling as A, B, C show energy profile diagram of SN2 reaction
show SN1 reaction of tert-butyl iodide and OH- (show transition states)
label as ABC etc and show energy profile diagram
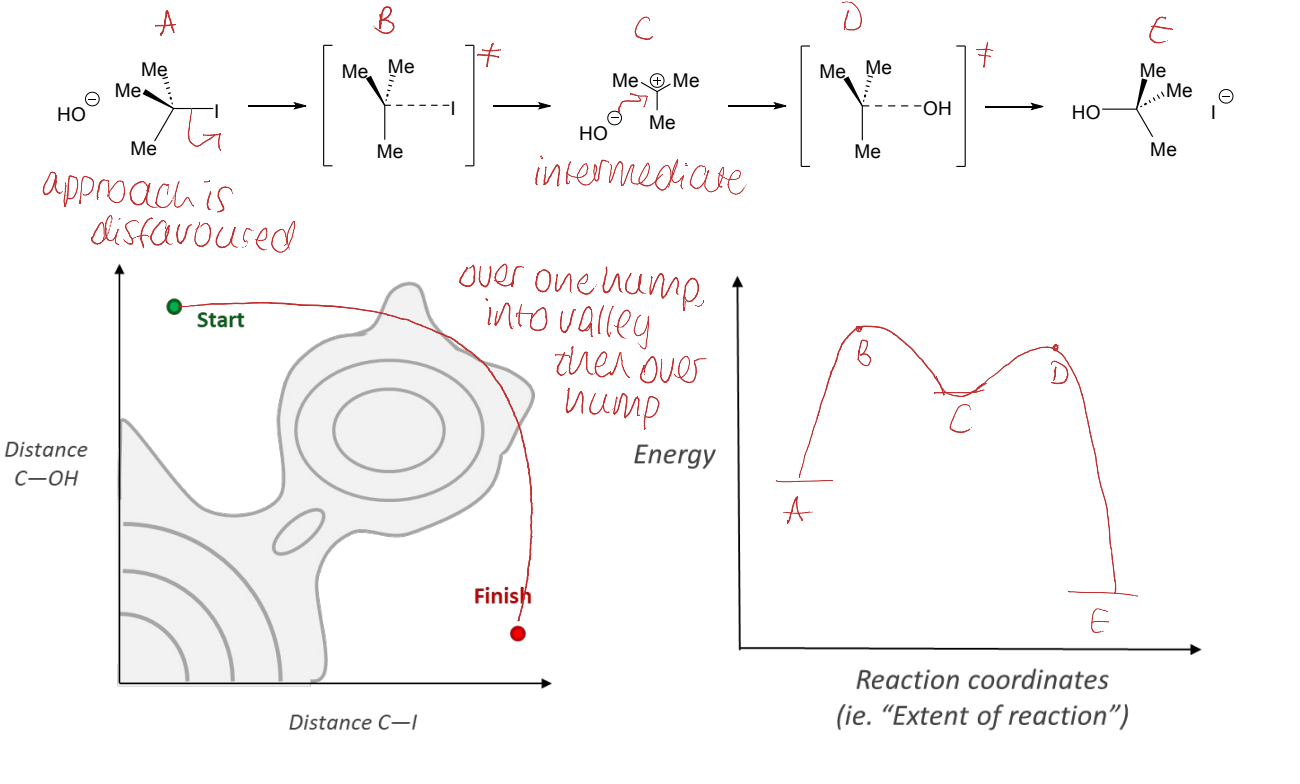
where is an intermediate shown on an energy profile diagram?
are they stable? can they be detected?
energy minimum (valley) that is not the starting material or product
metastable
can be detected by IR or NMR
where is an transition state shown on an energy profile diagram?
can they be detected?
energy maximum
not directly measurable
how does an energy profile diagram show the rate determining step?
TS with highest energy max is the RDS
how do charged intermediates differ from neutral on energy profile diagrams?
charged intermediates are higher in energy than neutral
how does entropy penalty change for transition states involving multiple molecules?
higher entropic penalty than those involve just one molecule
greater increase in order/decrease in disorder
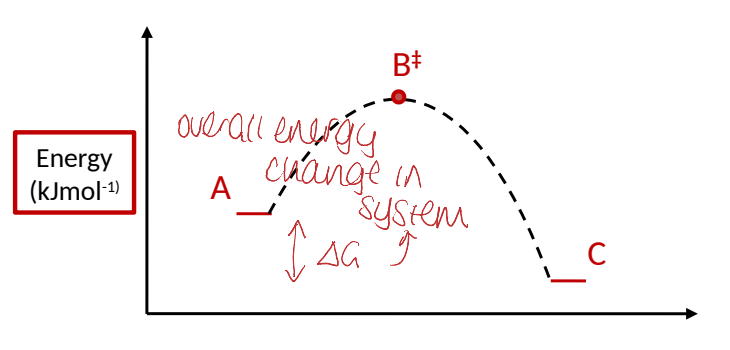
on an energy profile diagram, where is ΔG? what is it?
what does enthalpy generally mean? what is entropy?
what is equation linking these two and Gibbs?
when does a reversible reaction spontaneously proceed forwards?
H = heat a reaction absorbs or releases
S = overall disorder of a system
when ΔG<0

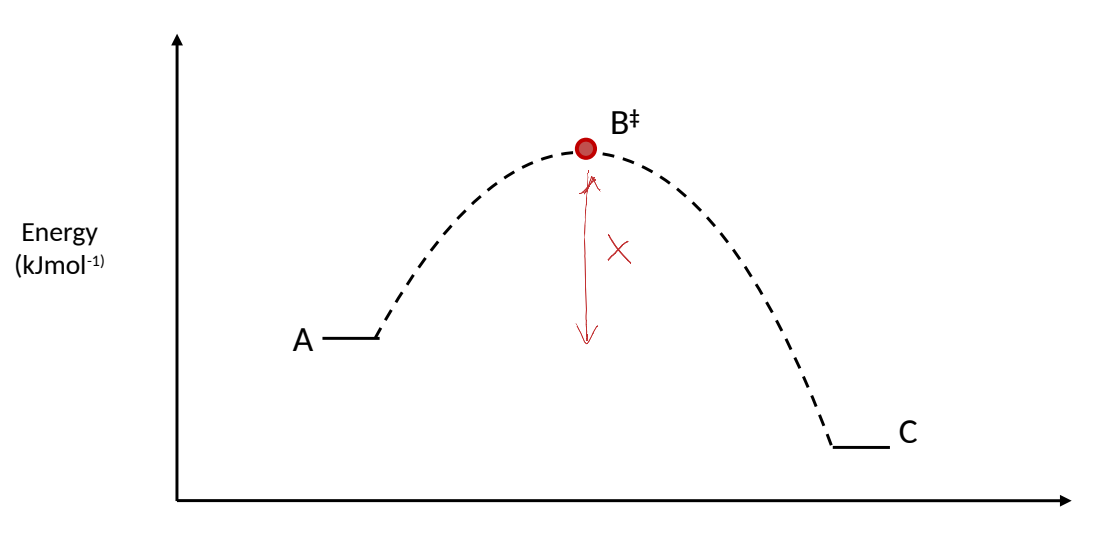
what is ΔG‡? 2
change in free energy between starting material and TS in RDS
the amount of energy available to perform work on the system
where is ΔG‡ on an energy profile diagram?
what does ΔS‡ <0 mean?
what does ΔS‡ >0 mean?
<0 means TS involves the association of multiple molecules = more order
>0 means TS involves dissociation of single molecule = less order
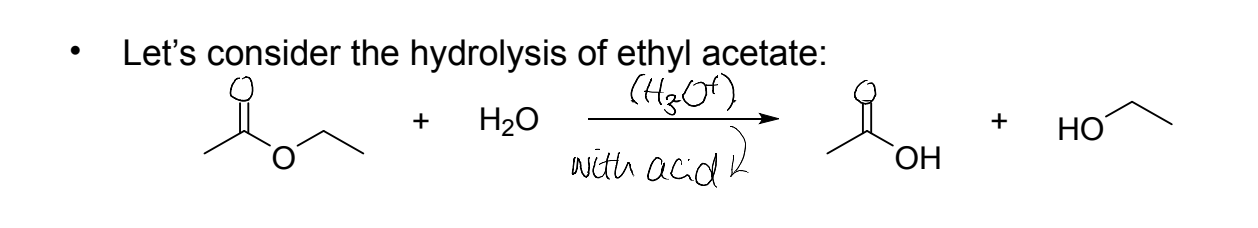
draw mechanism with and without catalyst until the hemiacetal product (note this is higher in energy than starting material)
show on energy profile diagram
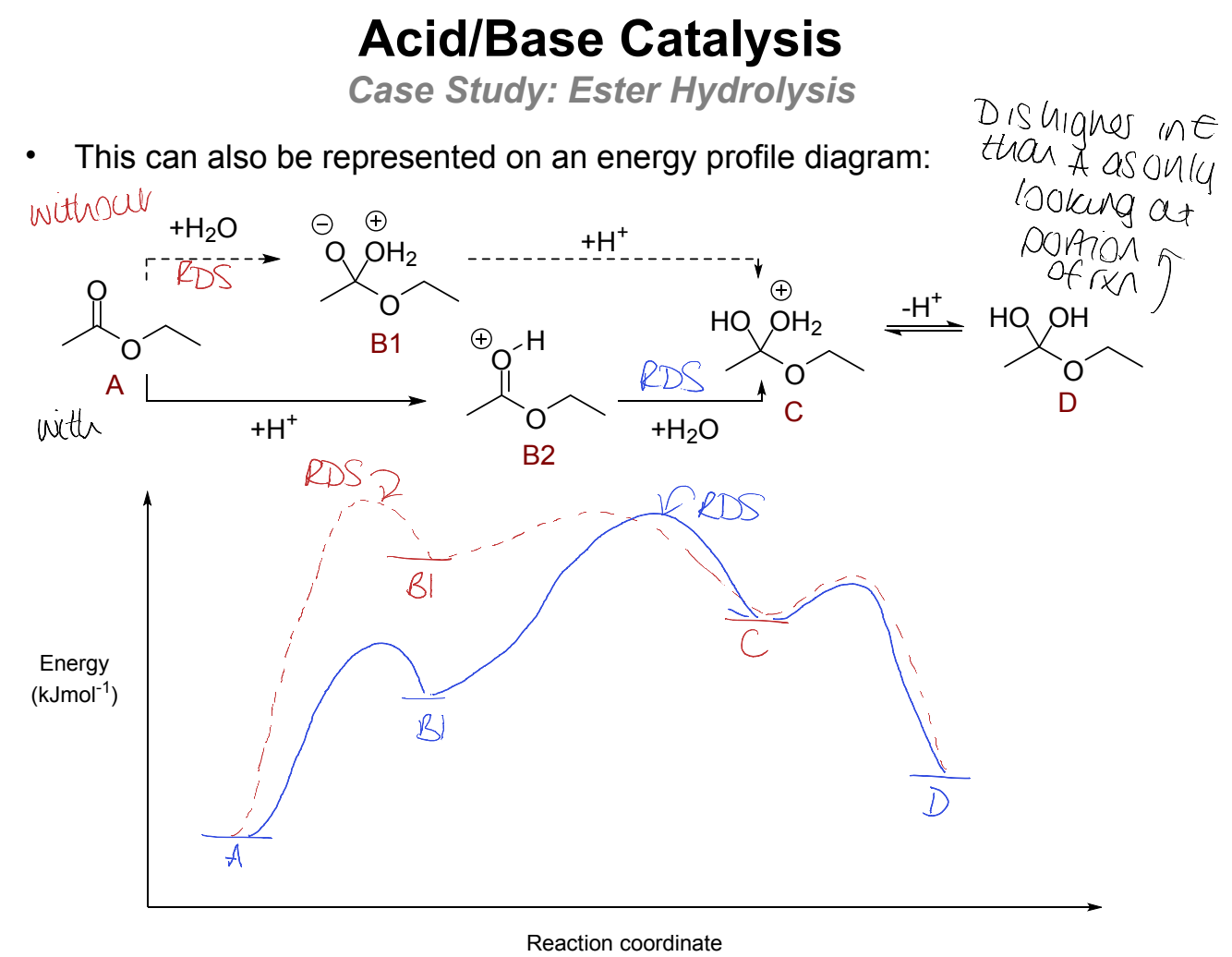
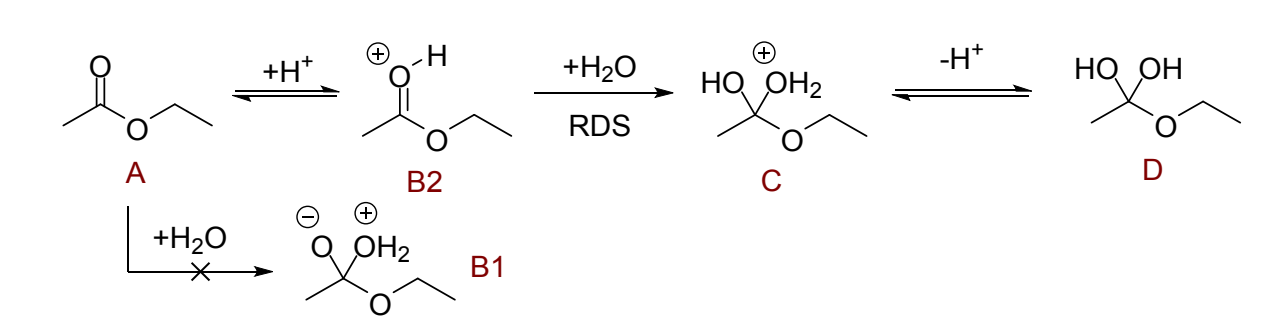
why is first uncatalysed step unfavourable?
why is it better with catalysis?
rate limiting, formation of doubly charged intermediate B1 is not favourable
with catalysis there is a fast proton transfer from solvent - followed by slower, rate determining track on carbonyl by water
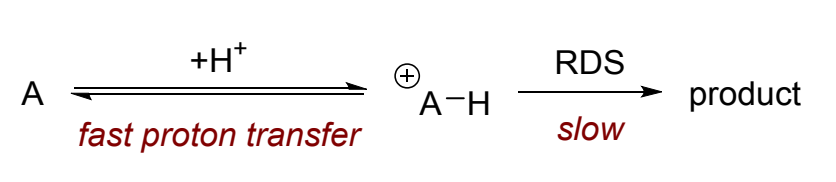
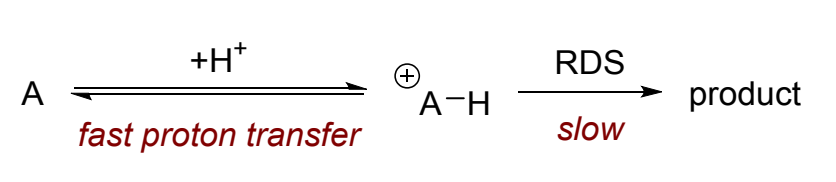
what is this an example of?
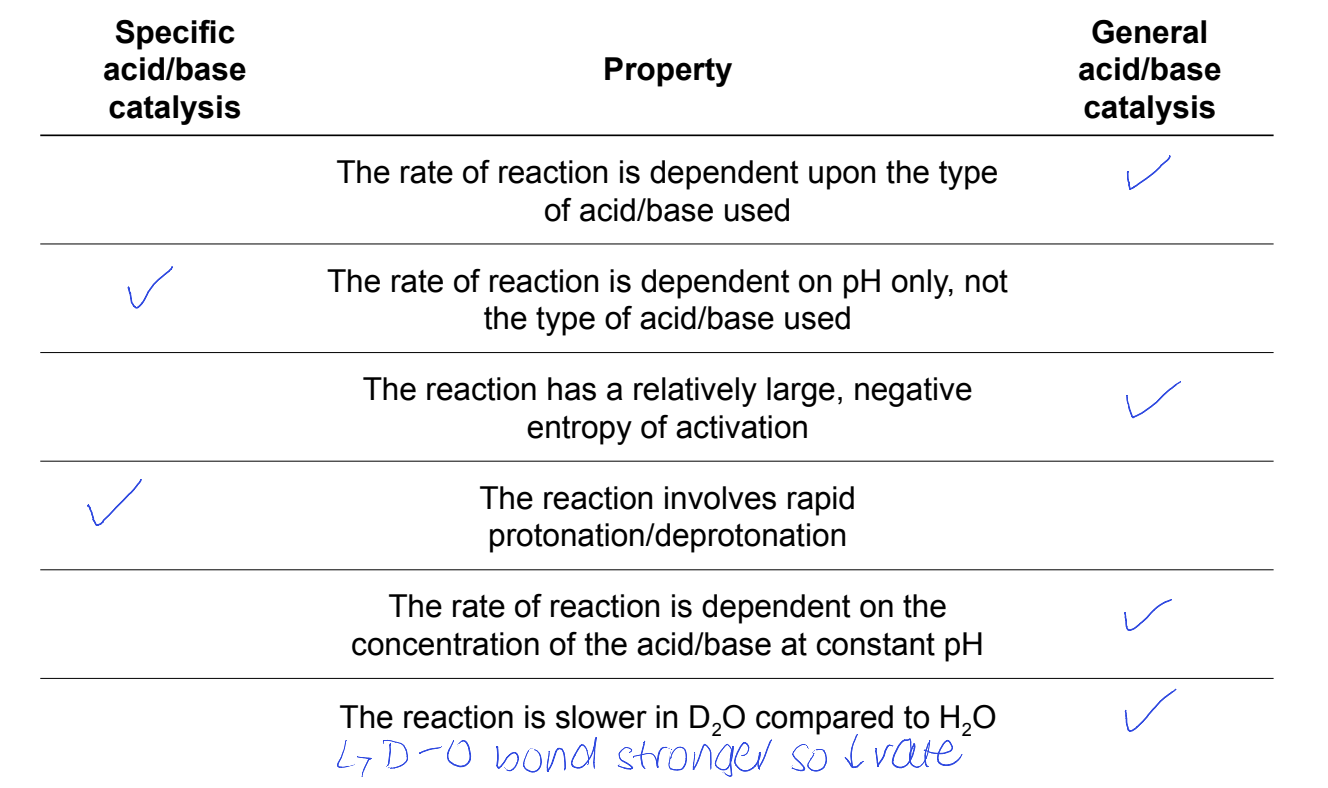
specific acid/base catalysis
= rapid protonation or deprotonation followed by slower RDS
![<p>write a rate equation and write in terms of [H<sup>+</sup>]</p>](https://assets.knowt.com/user-attachments/2c053953-a292-46c5-81d0-dda71d0534c4.png)
write a rate equation and write in terms of [H+]
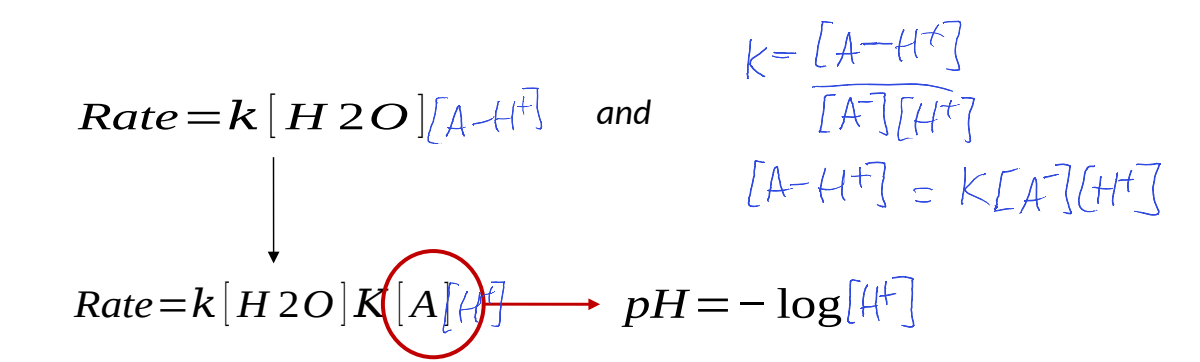
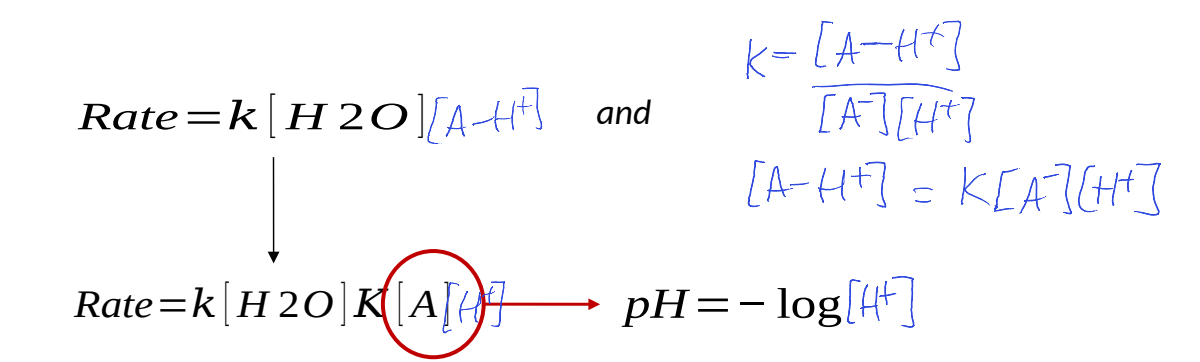
what does this show?
rate is correlated to pH of reaction mixture
actual acid or base used doesn’t affect reaction rate
what is general acid/base catalysis?
when protonation/deprotonation is slow and occurs during RDS
RDS involves the acid or base
nature of acid/base and its concentration impact the rate
why do general acid/base catalysed reaction have ΔS‡ and ΔG‡ values?
rate determining transition states generally involve multiple molecules (high entropic penalty)
high energy barrier
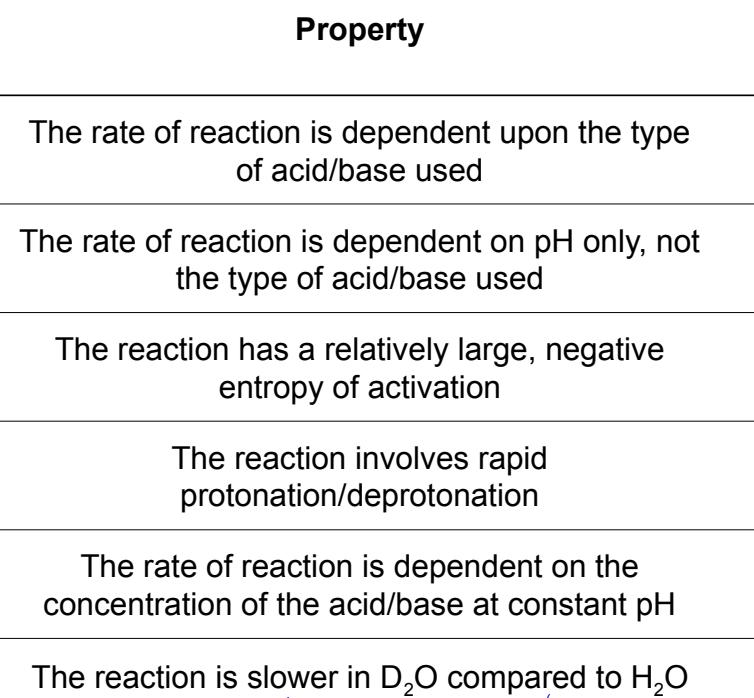
which is for specific and which is for general?
what are nucleophile catalysts?
catalysts that act as a nucleophile then as a LG
the intermediate they form is more reactive than the starting material