Digestion, Absorption, and Metabolism
1/85
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
86 Terms
digestion
macromolecules into absorbable units; breaking down
absorption
mostly takes place in the small intestine; moving across cell membrane
water and ions in the large intestine
motility and secretion
digestion/absorption are influenced primarily by ___ in the GI
monosaccharides (glucose/galactose/fructose)
carbs can only be absorbed as ___
amylase
breaks down larger carbs into smaller glucose chains and disaccharides
brush border enzymes
on the microvilli on gut enterocytes apical side, target already digested carbs (disaccharidases) and breaks them further into monosaccharides
Na/K ATPase on basolateral side: provides Na/K gradient
GLUT 2: found on basolateral side
SGLT1: found on apical side
GLUT5: found on apical side
where are the 4 main transporters (Na/K ATPase, SGLT1, GLUT5, GLUT2) on gut enterocytes (in the mucosa layer) involved in carb absorption found within the cell?
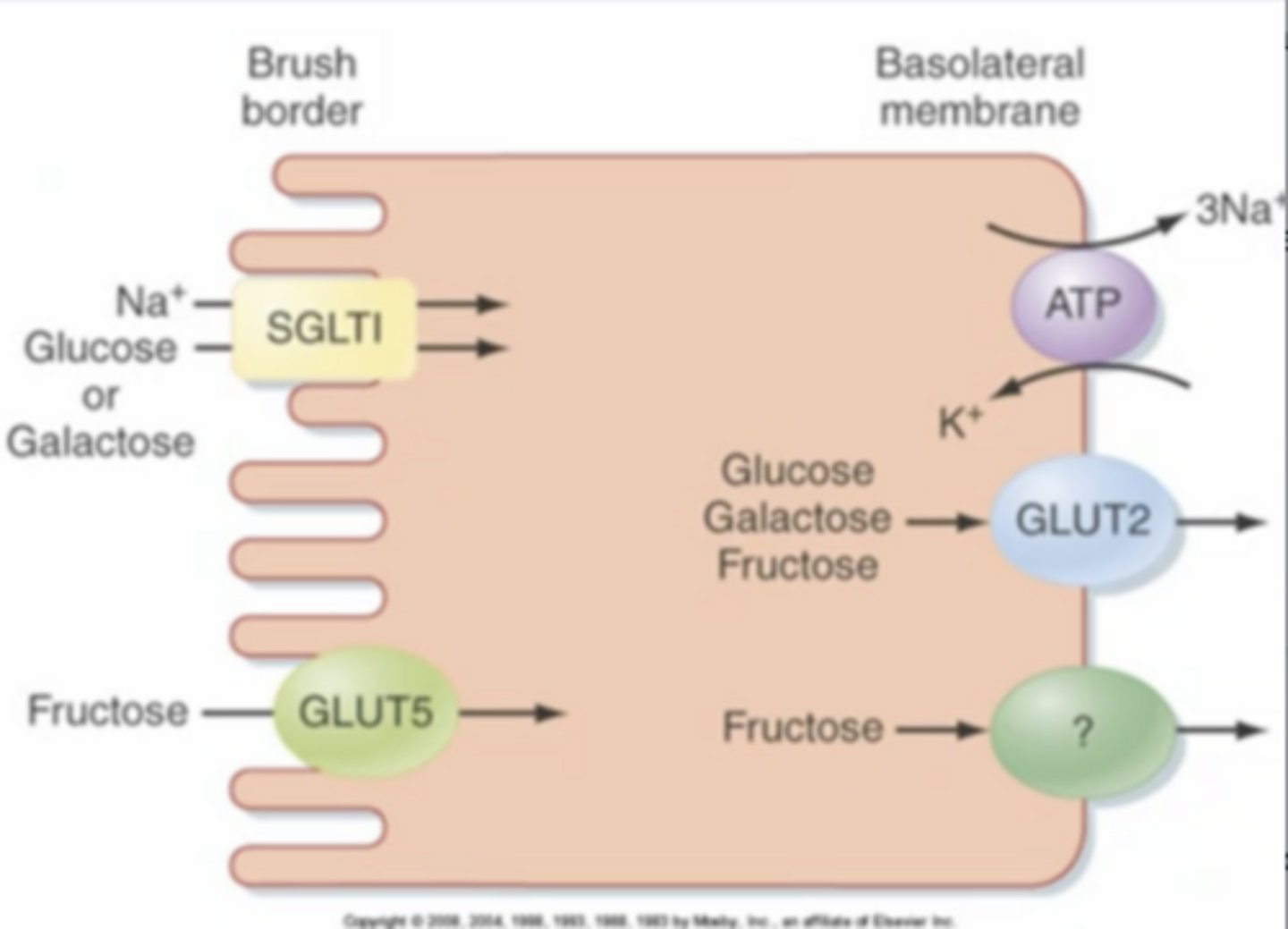
SGLT1
secondary activate transporter symporter that uses existing Na gradient to bring glucose/galactose into cell (with higher affinity for glucose); on apical side of enterocytes, main driver of glucose entering cells
regulated by glucose in the intestine; increased glucose in lumen = increased expression of this
even if you have little glucose in the lumen or an equal amount as inside the cell, you can still transport it through the cell into the blood bc the Na gradient it follows is still present (and transport relies on that)
if you have low Na in the lumen, the driving force for glucose movement decreases so less is brought into the cell
if you have high glucose in the lumen, more will move into the cell bc it will follow its gradient
how will the activity of SGLT1/the movement of what it transports be altered in low glucose, low Na, and high glucose concentrations in the lumen?
GLUT5
facilitated diffusion (passive) of fructose; relies on concentration gradient
found on apical side
GLUT2
transport of all monosaccharides; found on basolateral side
glucose
most favorable monosaccharide for energy conversion (primary energy source to initiate glycolysis); taken into body from diet and levels in blood are monitored closely
stored in liver, and other carb compounds are converted to it
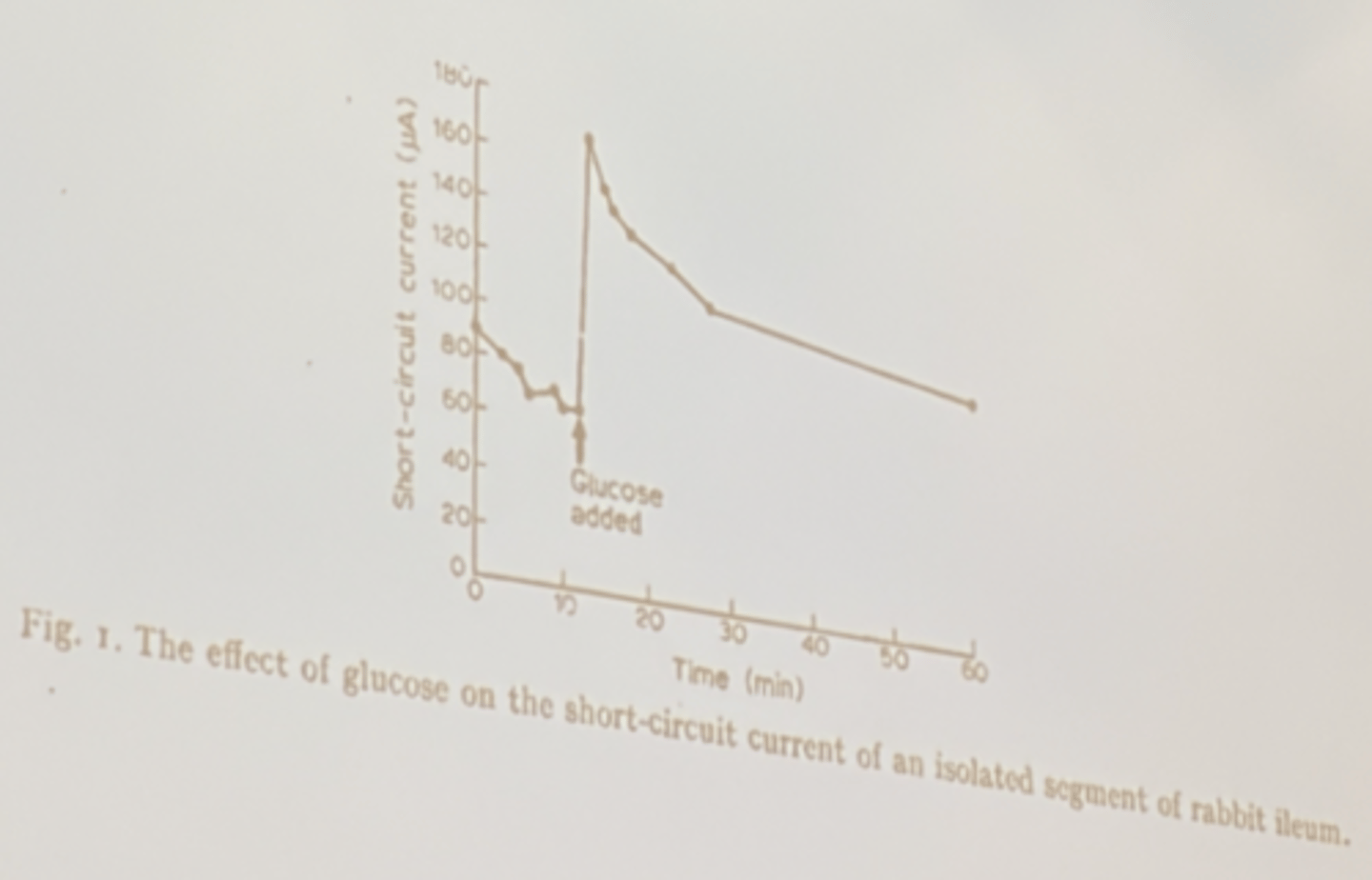
if you add glucose, it causes more Na to be able to be moved into cells bc it increases activity of SGLT1 (co-transporter of glucose and Na) → as glucose increases extracellular the co-transport mechanism continuously moves Na+ inside the cell as well, so short-circuit current charge increases
if you increased glucose concentration in the lumen, how would you expect short-circuit current to change?
you get hydrated faster if the water you drink has glucose/electrolyte ions in it; water follows their concentration, so increasing their concentration in cells draws water into cells to hydrate you faster
however if you have too high of a concentration of glucose outside your cells, you will get dehydrated (ex. diarrhea) very quickly bc there won't be enough transported to cells and it will draw water/ISF out into the GIT lumen
what happens to hydration levels/water movement in the body at small and large increases of extracellular glucose/ions?
endopeptidases and exopeptidases
what are the two methods/agents of protein digestion?
endopeptidases (proteases)
attack peptide bonds to break up large peptides into smaller ones
secreted by stomach (pepsin), intestine, and mainly pancreases as inactive proenzymes
ex. pepsin, trypsin
exopeptidases
release single amino acids into GI lumen by breaking terminal peptide bonds; secreted by pancreas
ex. carboxypeptidase
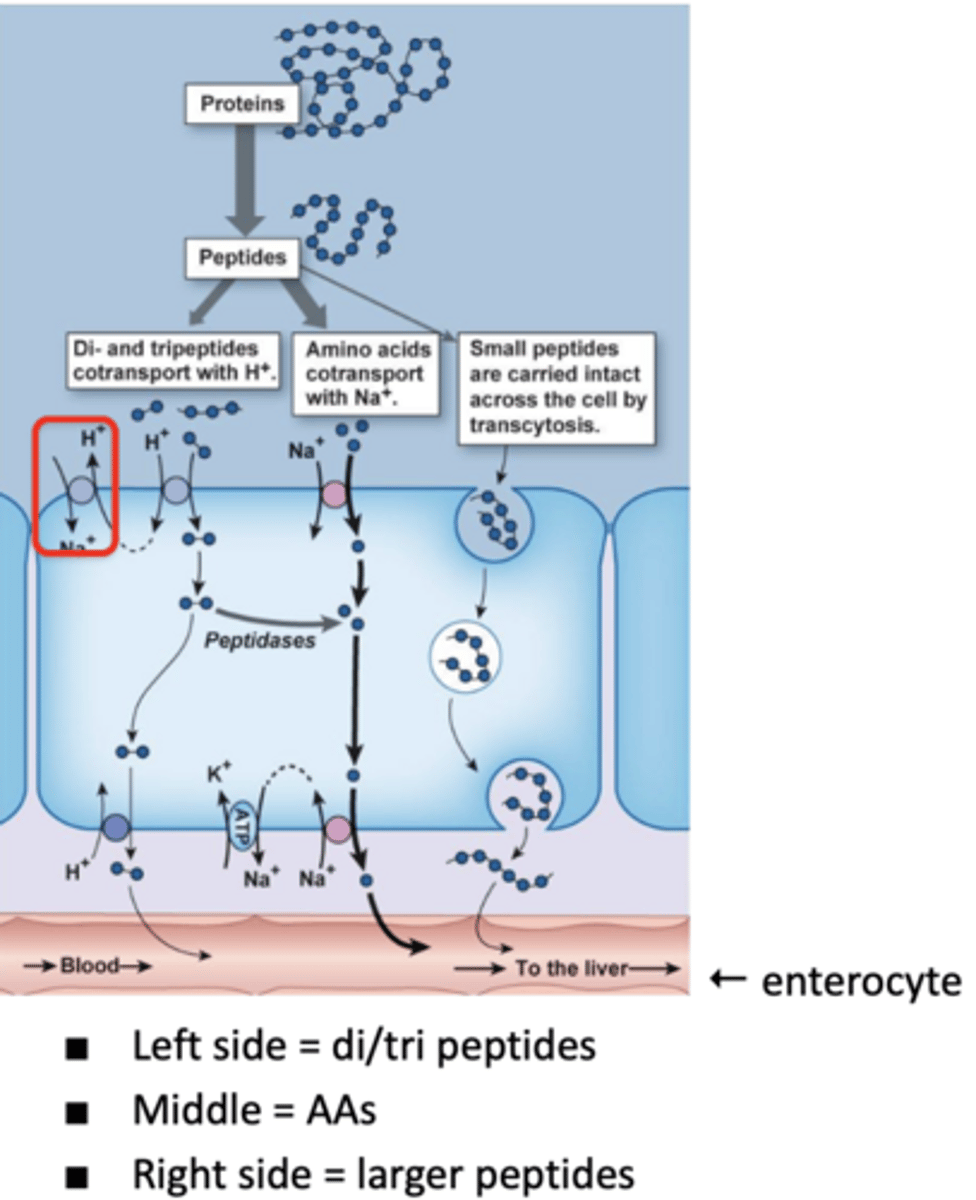
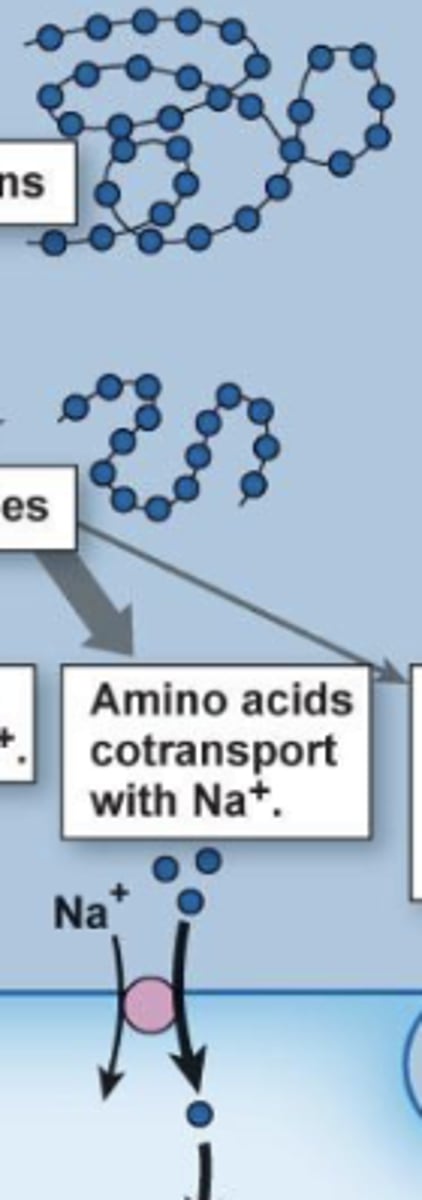
amino acids: secondary active transport
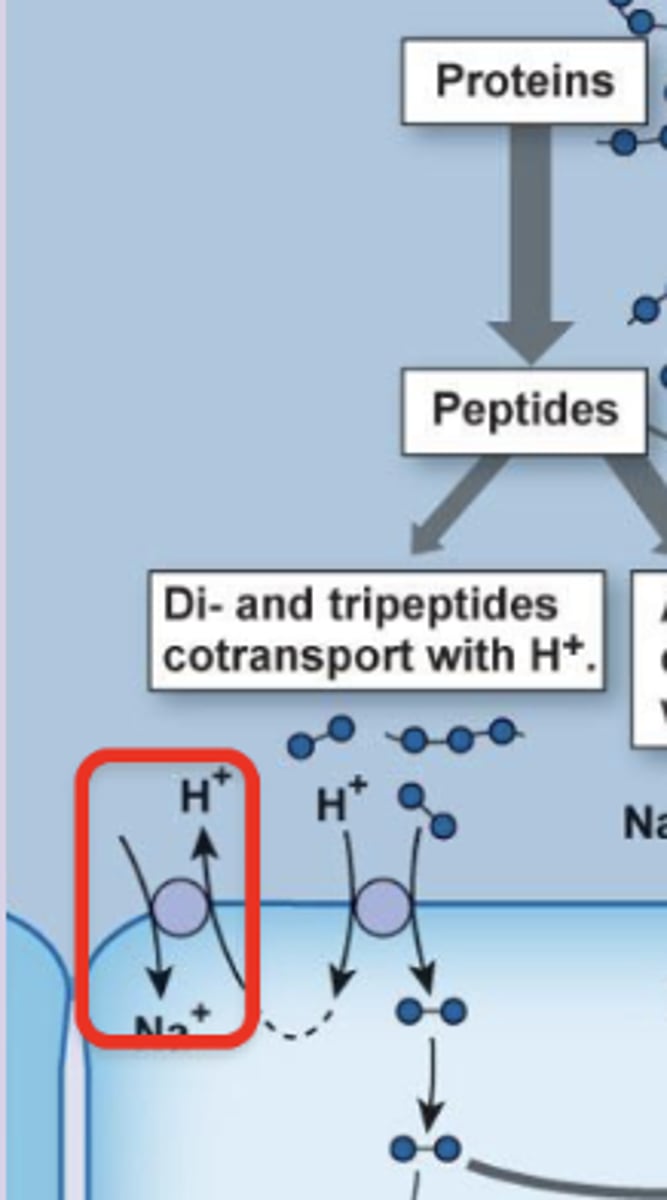
di/tri peptides: tertiary active transport (PepT1)
large peptides: transcytosis
what are the ways/structures proteins can be absorbed, and what is the mechanism for each?
primary active transport = Na/K ATPase → establishes Na gradient (bottom middle)
secondary active transport = Na/H+ exchanger and Na/AA cotransporter → use Na gradient from ATPase to move H+ out of cell or AAs into cell
tertiary active transport = PepT1 → uses H+ gradient from secondary Na/H exchanger to move dipeptides and tripeptides into cell
what is the primary, secondary, and tertiary active transport involved in protein absorption?
amino acid/Na co-transporter
follows existing Na gradient (from Na/K ATPase) to move AAs into cells (along with Na) into gut enterocytes
secondary active transport
PepT1
tertiary active transport following H+ gradient (established by H+/Na+ antiporter) that moves dipeptides and tripeptides into gut enterocytes
sometimes (less frequently) large peptides can come in via transcytosis
may elicit allergic response (if a foreign protein is absorbed)
more in infants with less developed digestive system; proteins in mother's breastmilk can be absorbed by child (passive immunity)
how are larger peptides brought into cells?
in this disease, the Na/AA secondary transporter is not functioning
having PepT1 still working means that you can still have transport of di/tri peptides so they are not AA deficient
having Na/K ATPase is also important for this, but not the main reason (to answer the question)
answer the question and provide reasoning for your answer choice

emulsification (bile salts)
process of breaking down large lipid droplets (fats) into smaller ones; physical/mechanical break down
increases SA for lipase action
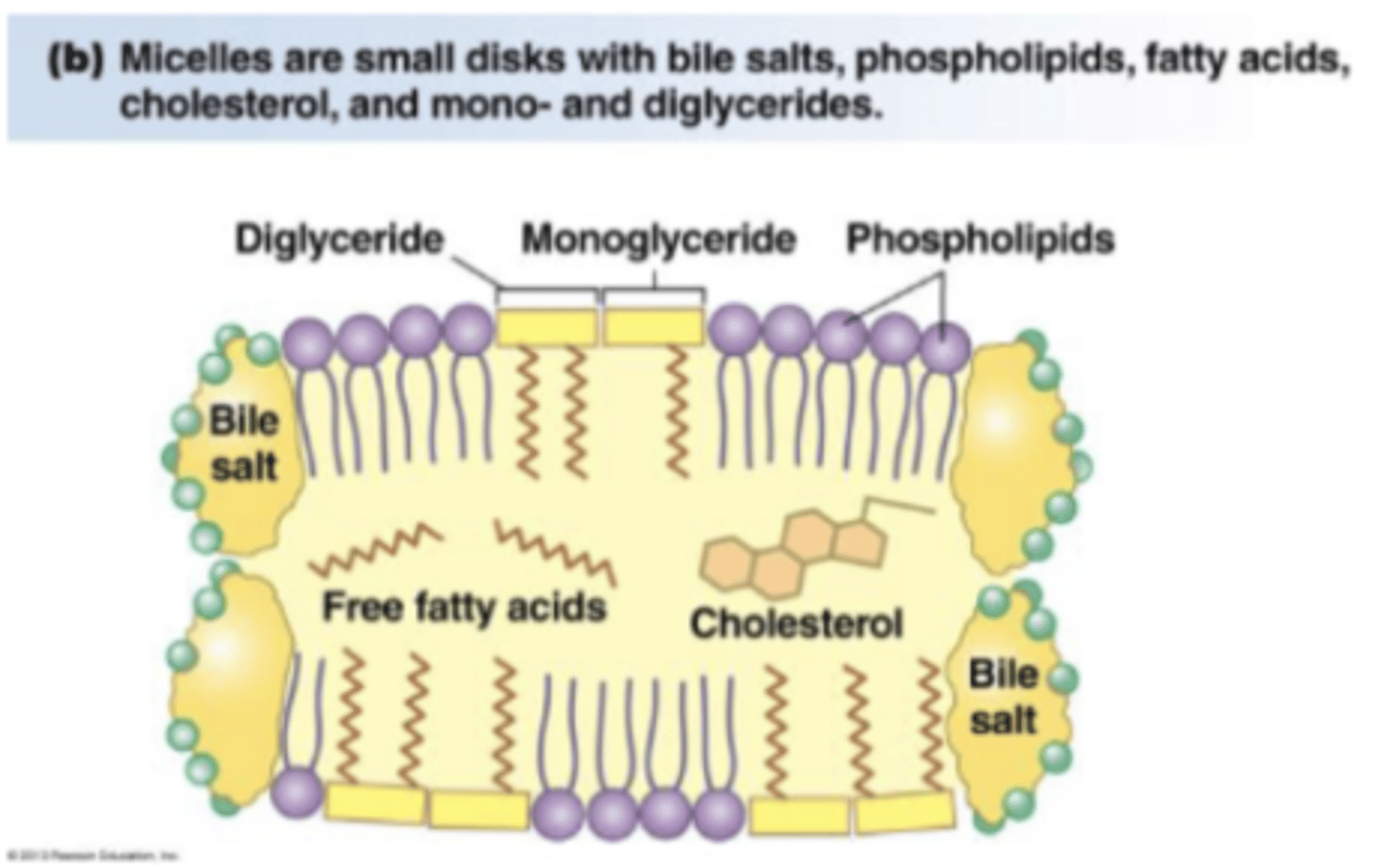
bile salts
amphipathic agents; have one hydrophobic (interact with lipids) and one hydrophilic (interact with water) side
enzymatic fat digestion
lipase and colipase (helps anchor lipase so it can do its job, not involved in chem rxn) digest triglycerides into monoglycerides and free fatty acids; chemical breakdown
bile is from the liver, but these enzymes come from the pancreas
phospholipase digests phospholipids
micelle
form that digested fat from lumen exist in, which is a structure where digested fat (monoglycerides and fatty acids) is enclosed by bile salts; prevents digested fat from clumping together (which would be unable to enter cells), so they must be dissolved by bile salt
break down and reform constantly, and break down allows them to be absorbed; when they dissociate fatty acids/monoglycerides can cross the cell (into interior enterocyte)
emulsification, enzymatic fat digestion, and micelle formation
what are the 3 steps in fat digestion?
enter apical membrane: monoglycerides and fatty acids (when moved out of micelle) can diffuse into cell through membrane bc they are also hydrophobic and small (compatible with phospholipid bilayer)
when they enter cells they become chylomicrons for transport
how do digested fats enter cells?
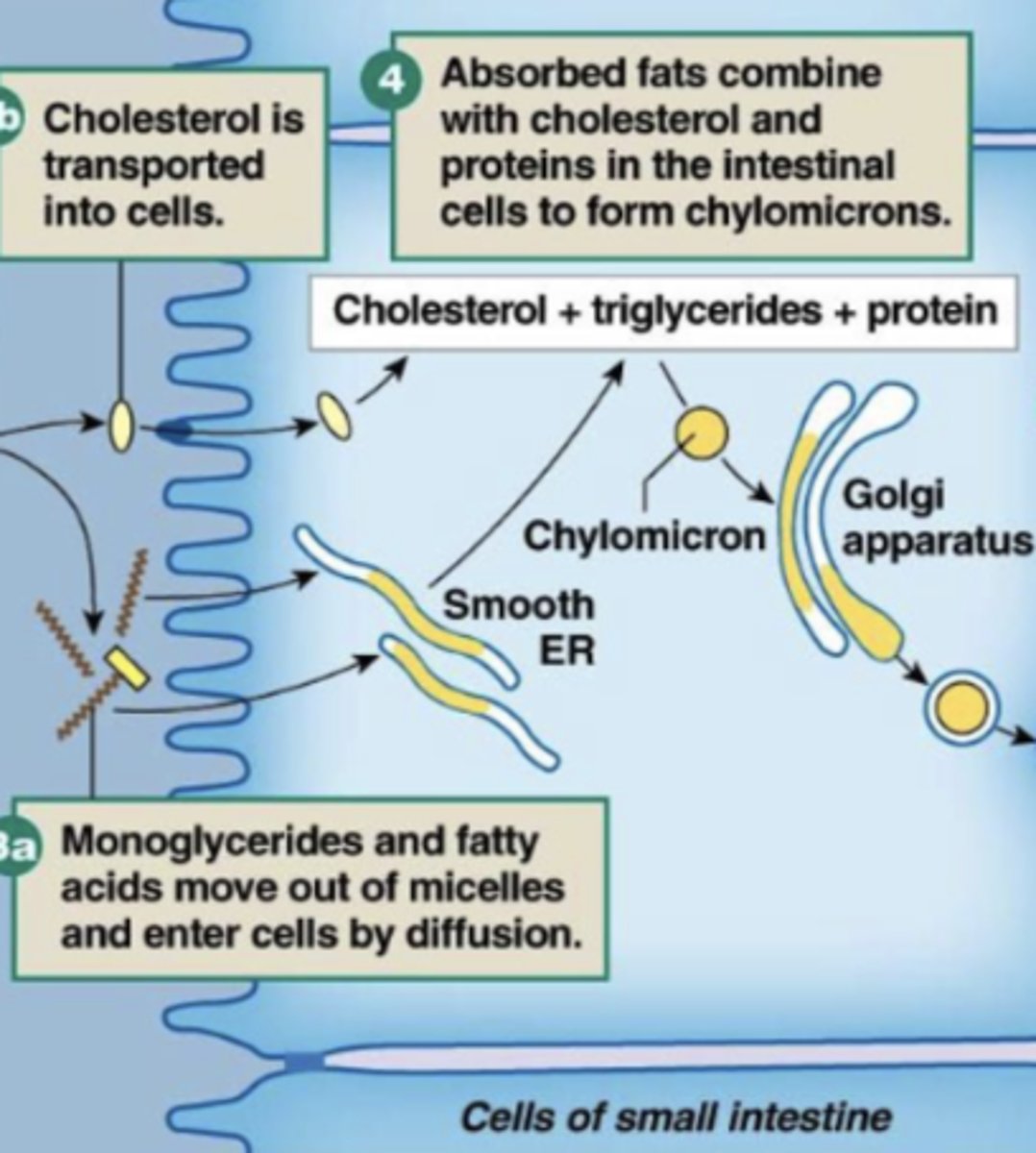
chylomicrons
largest lipoprotein synthesized inside SI enterocytes; monoglycerides and fatty acids recombine into triglycerides and bind with cholesterol to form these
when digested fat enters cell it's packaged and sent throughout the body in these via LYMPHATIC system (not blood bc they are too big for it!)
lacteal
end of lymphatic system that allows chylomicrons to enter and travel within lymph vessels
leave basolateral membrane via exocytosis to enter lacteal → eventually the lymph vessels become leaky (larger gaps between cells/pores) and chylomicrons can diffuse into vena cava and travel to liver
indirectly travels to liver since digested fat goes all around body
describe the process of chylomicrons leaving a cell and traveling through the body (including start and end point)
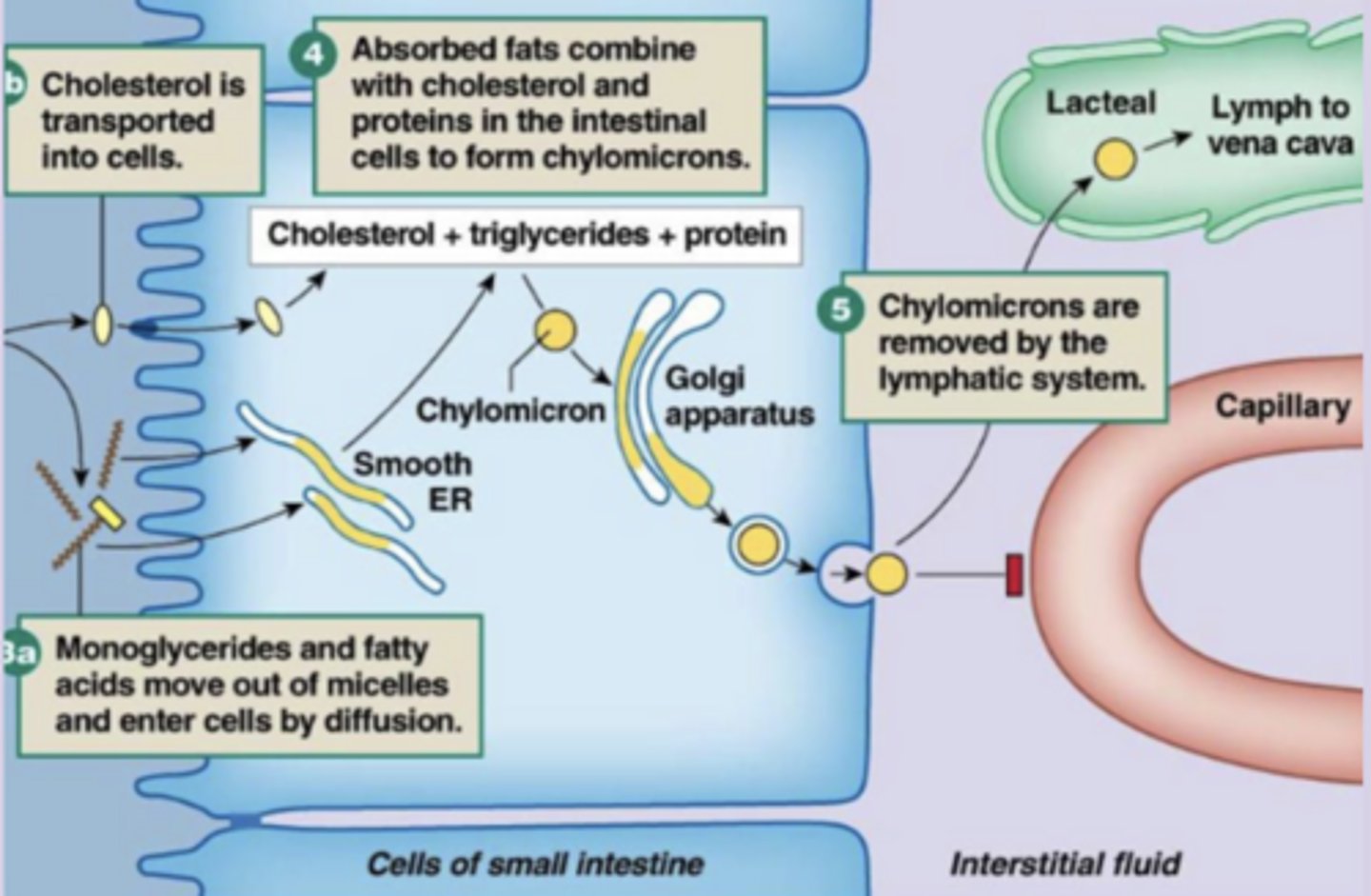
bc you need a concentration gradient for passive diffusion of fats into cell; converting free fatty acids/monoglycerides into chylomicrons lowers intracellular concentration of them so more can move into cells
why is forming and transporting chylomicrons important?
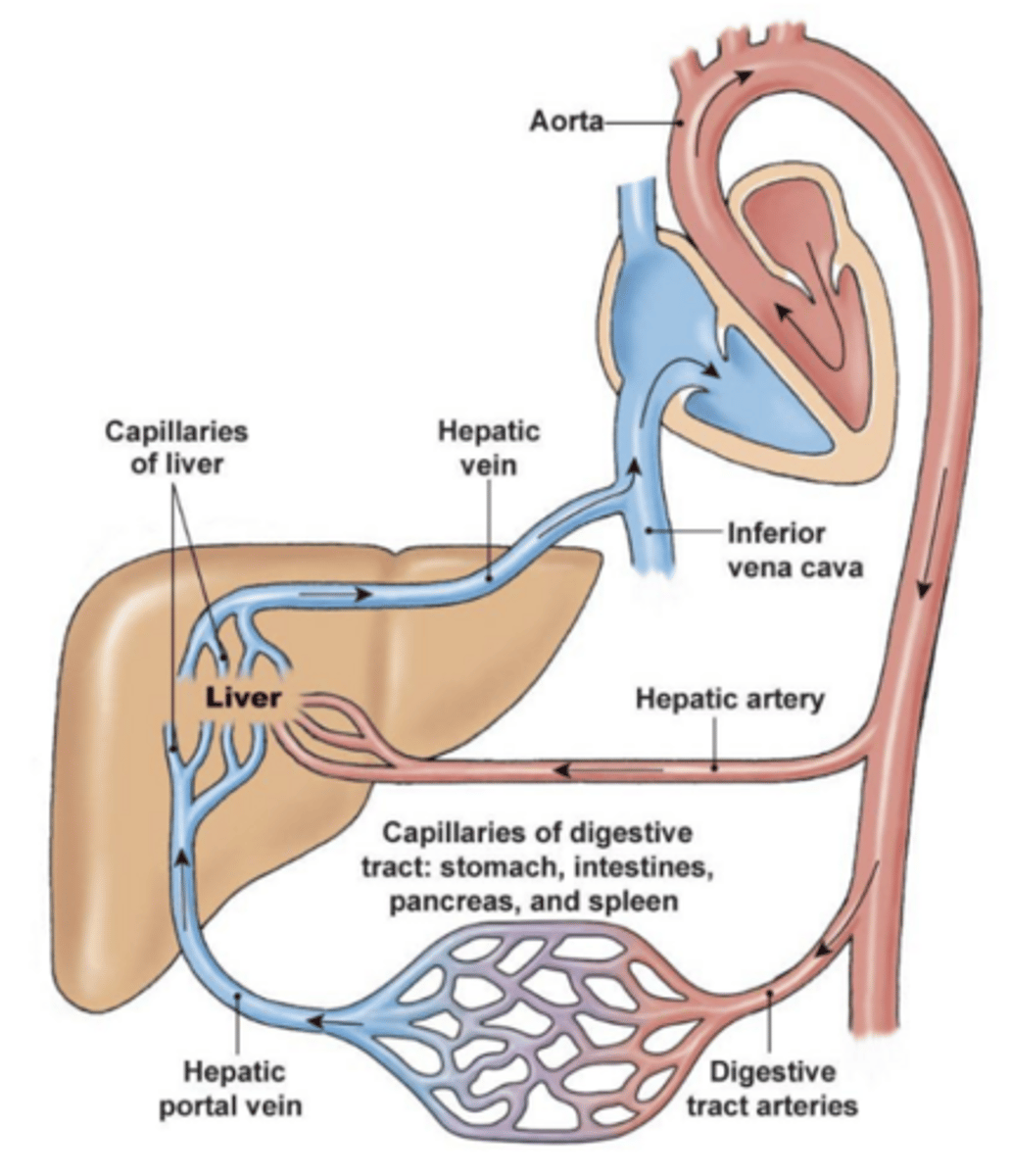
the hepatic portal system (blood), liver (which utilizes those products; most absorbed nutrients go to liver first)
***second exchange site at liver sinusoids
(chylomicrons) enter lacteal (lymphatic system), which goes all over the body bc they enter the vena cava eventually
products of carb/protein digestion are absorbed into ___, which connects directly to the ___
products of fat digestion enter ___
hepatic arteries come from aorta and provide oxygen/nutrients to liver
blood vessels that supply GIT bring it oxygen/nutrients, but also receive digested/absorbed nutrients (except fat) and bring them to the liver (capillaries surrounding GIT go right to the liver via hepatic portal veins)
hepatic veins come out of liver to deliver blood it has processed elsewhere
how does blood enter and leave the liver?
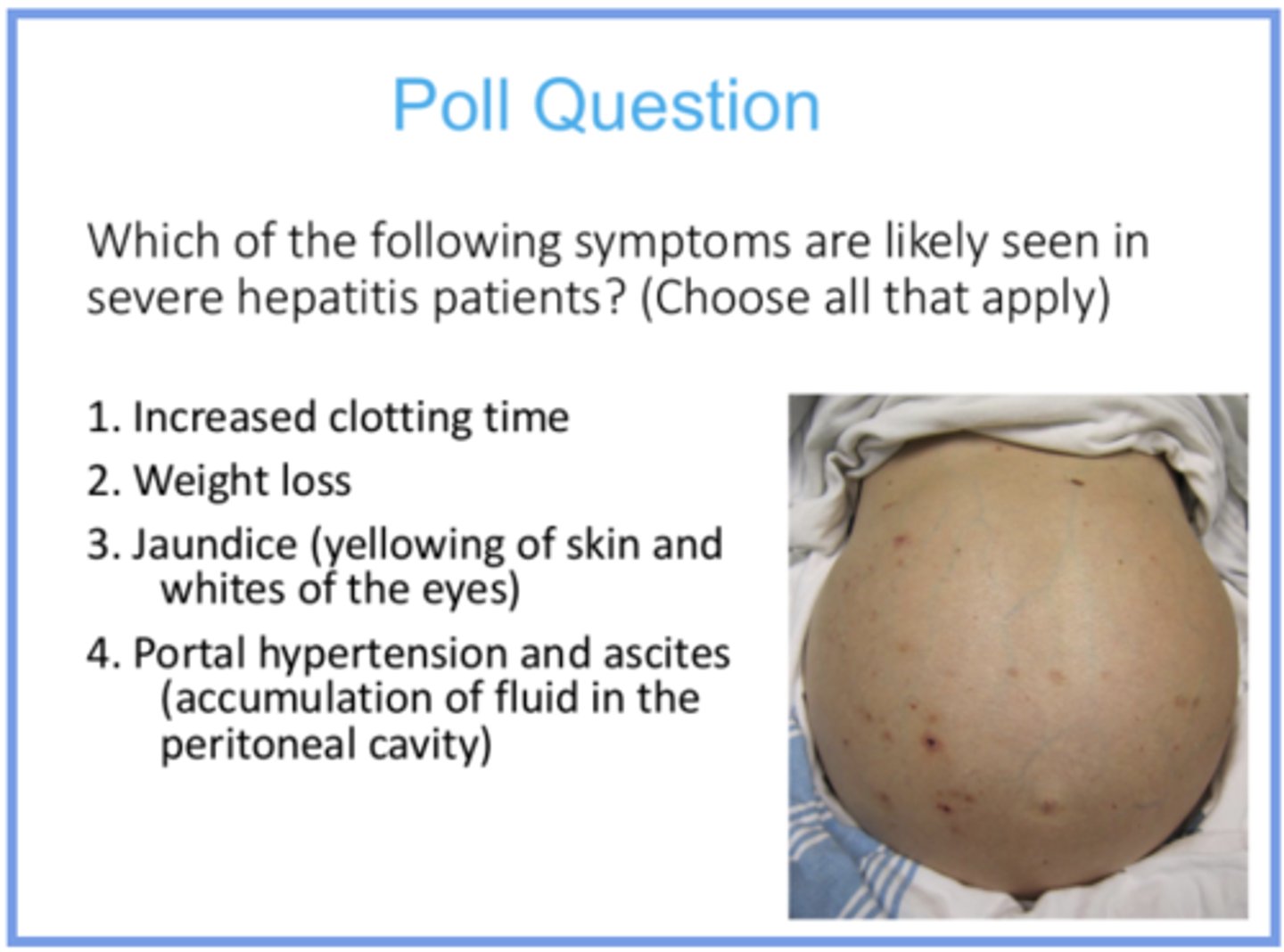
the liver produces clotting factors, so if it is damaged you will not have those proteins being created and thus will have longer clotting time
bc bile salts will be compromised, you won’t be able to digest/absorb fat as much so weight loss is possible
you will have jaundice because bilirubin will not be incorporated into bile (which is made by the liver), so it will accumulate elsewhere
drainage issues in liver can cause fluid to accumulate in portal veins and the capillary beds that support them, leading to accumulation of fluid in peritoneal cavity
answer the question and provide reasoning for all answer options
cecum (where the large intestine begins/where the ileum leads into) → ascending/transverse/descending colon → sigmoid colon → rectum
describe the structures that make up the large intestine in order (from closest to small intestine to end)
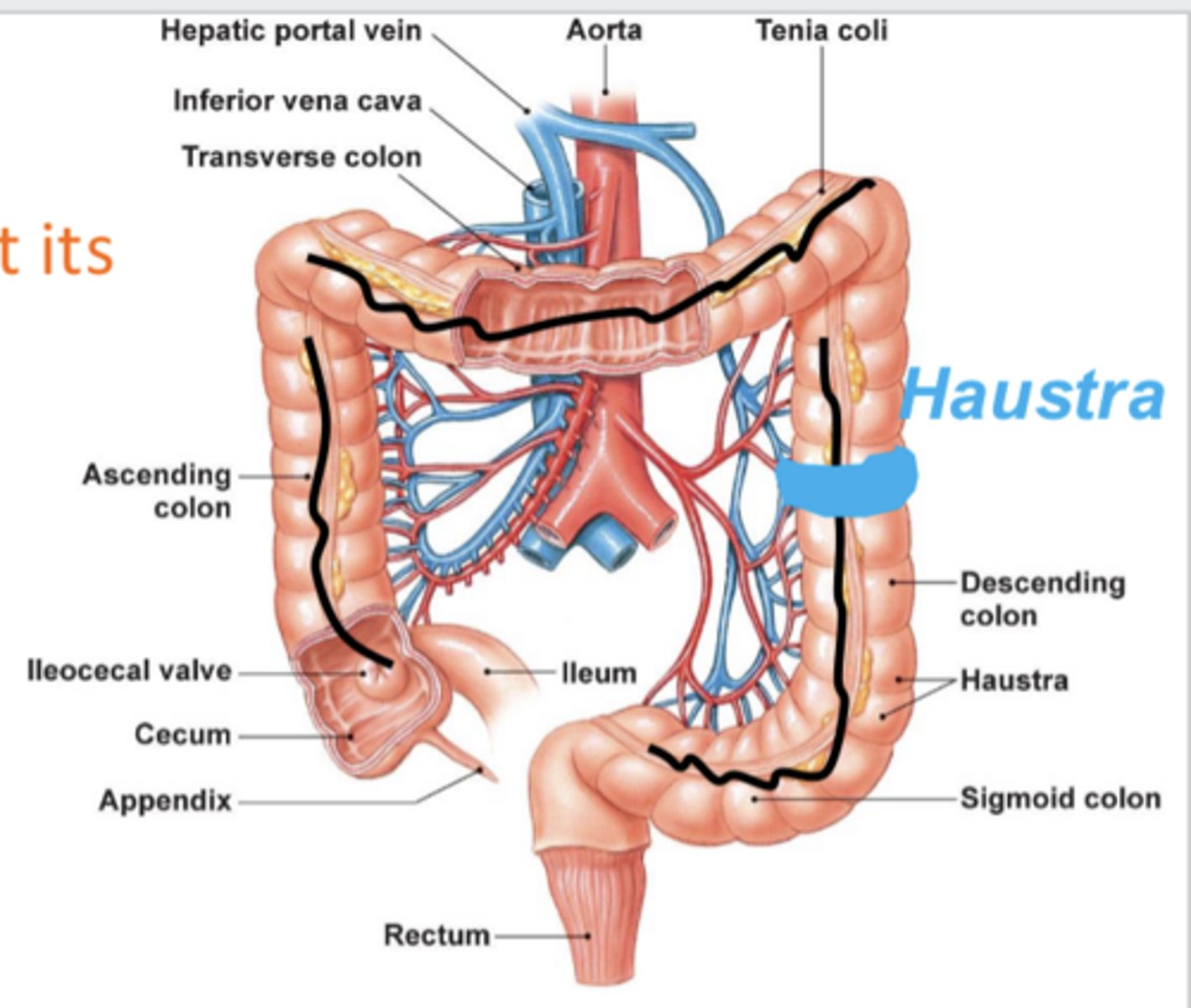
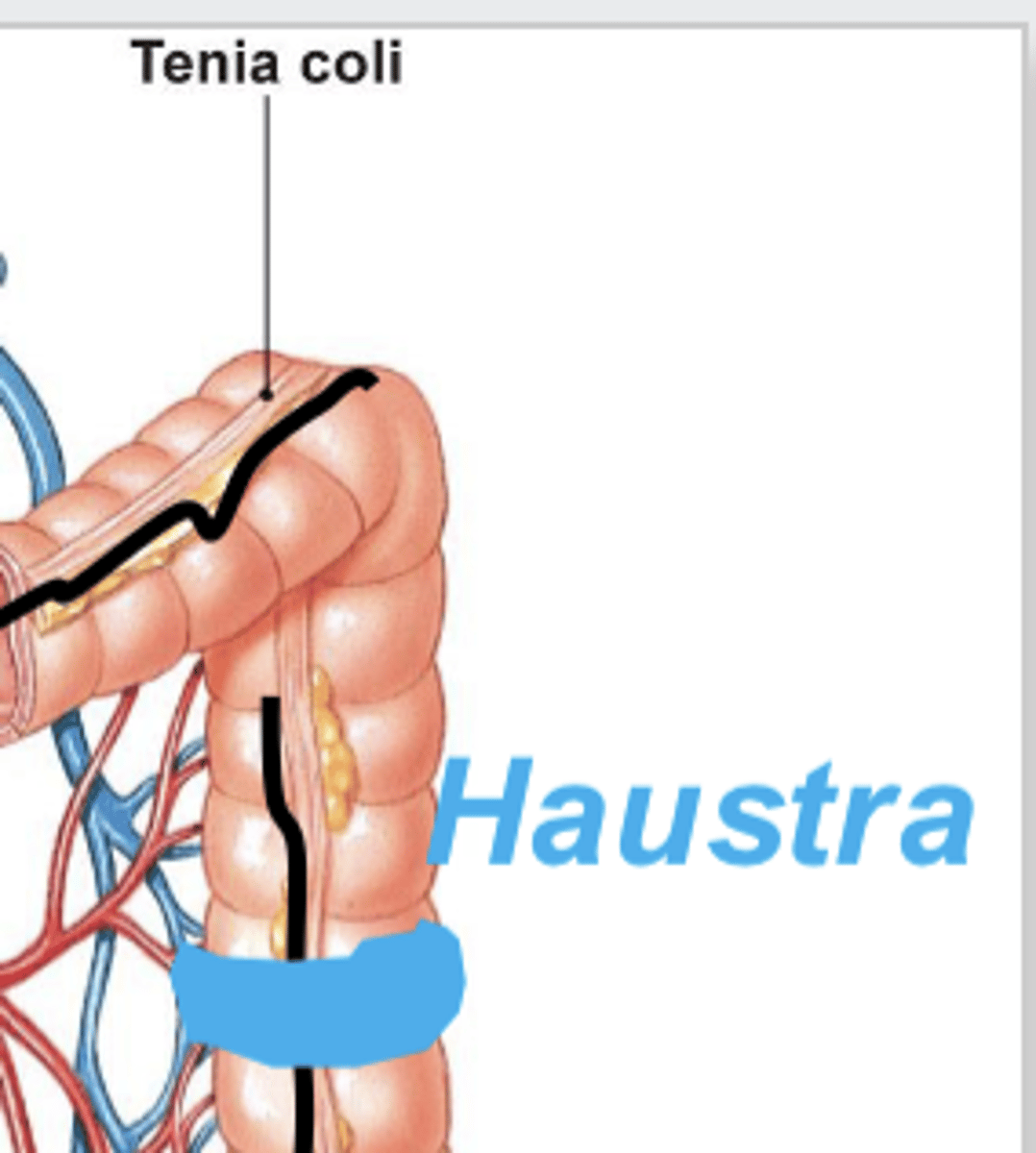
tenia coli
3 bands of smooth muscle that the large intenstine shrinks into; contractions of these "puckers" it into pouch-like segments called haustra
unique feature of large intestine because it has this and not a longitudinal layer (for smooth muscle)
smooth muscle transitions to skeletal muscle (external anal sphincter) in the large intestine
relatively slow movement within it so bacteria has a lot of time to grow and process digested food
movement is mostly segmentation, but has some unique strong peristalsis (mass movement)
describe the movement that occurs in the large intestine
mass movement (large intestine)
unique strong peristalsis of the large intestine; rare (only happens 3-5 times a day, normally after you eat) which increases urge to use bathroom
glucose-6-phosphate
glucose comes into cells via transporters and based on a diffusion gradient (high out/low in = move into cell) → cells add a phosphate onto glucose to "trick" transporters into thinking intracellular glucose is constantly low so it keeps flowing in
to be used in glycolysis, glucose must be converted to ___? Explain why
(1) enter glycolysis to generate ATP and pyruvate or (2) be stored as glycogen
these processes occur in the cytoplasm
what are the two pathways glucose-6-phosphate may enter after it is converted to that form, and where do they occur?
no oxygen = gets converted to lactate (lactic acid) → causes fatigue and lowering of tissue pH (more acidic)
oxygen present = enter mitochondria → interact with pyruvate dehydrogenase complex (contains pyruvate dehydrogenase) → become acetyl CoA and enter citric acid cycle → generate ATP and electron donors for ETC (also to generate ATP)
what happens to pyruvate in an environment with or without oxygen?
pyruvate dehydrogenase
enzyme that converts pyruvate into Acetyl CoA
part of pyruvate dehydrogenase complex
glycogen
branched polymer of multiple glucose units; stored in skeletal muscle or the liver when blood glucose is high
glycogenesis
process that forms glycogen; glucose must have been in the form of glucose-6-phosphate to be stored as glycogen or enter this process
primary enzyme of this process is glycogen synthase
glycogenolysis
when blood glucose is no longer high and a carbohydrate meal is not consumed to replenish it, glycogen may be broken down into glucose through this process
glycogen phosphorylase
primary enzyme in breaking down glycogen to glucose-6-phosphate
glucose-6-phosphatase
enzyme that will remove the phosphate off of glucose and allow it to exit the cell
the liver does have it since its primary role is to regulate blood glucose; it must have it so glucose can be released into the blood and supply tissues with an energy source
***also present in kidneys
muscles do NOT have it since their primary role is to generate force through contraction, which requires energy; they need to use all the glucose-6-phosphate they release when they break down glycogen for their own glycolysis (to make their own ATP)
what parts of the body have and lack glucose-6-phosphatase?
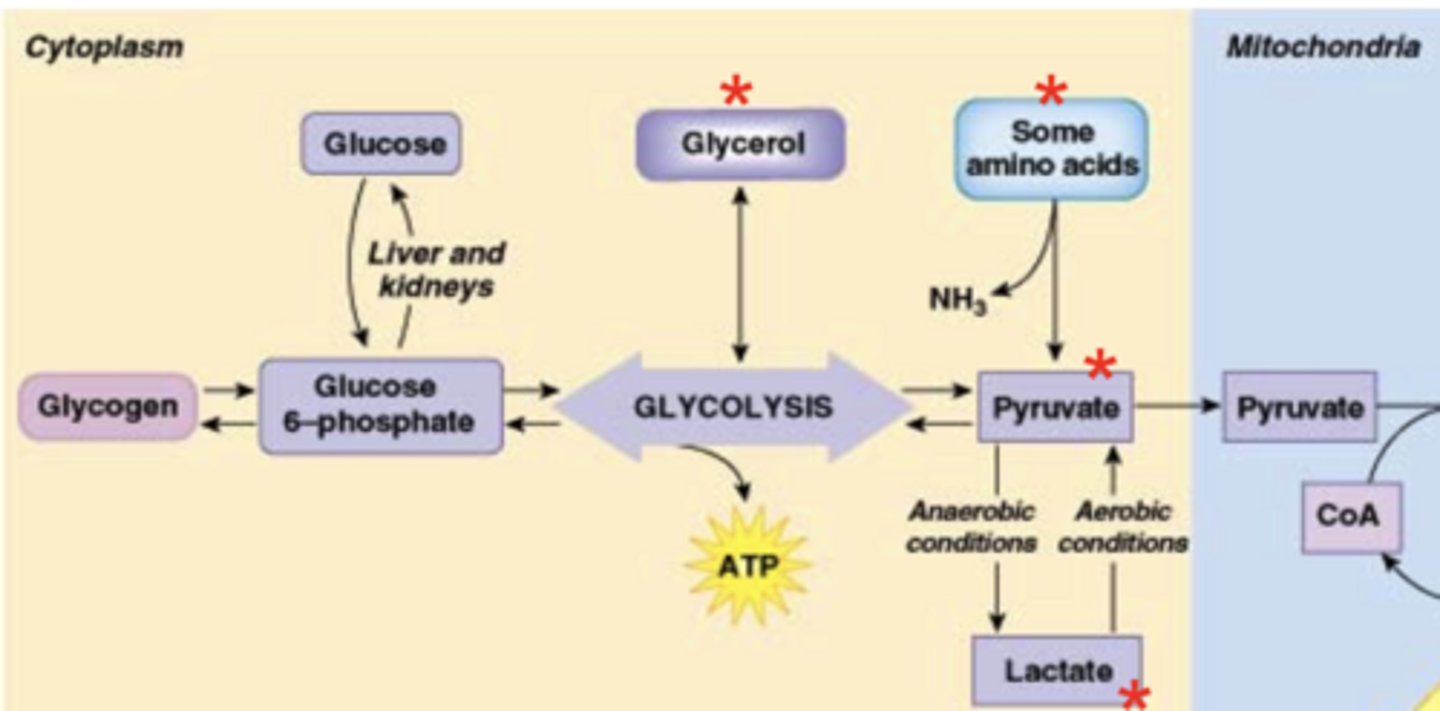
gluconeogenesis
occurs during fasting state, liver using products of digestion to make glucose; process of generating glucose from a non-carbohydrate precursor (including amino acids, pyruvate, lactate, or glycerol)
1) amino acids: via deamination (removal of -NH3) and the cori cycle
***excess protein consumption may lead to fat deposition as amino acids are converted to glucose (may eventually be produced in excess and converted to fat stores)
2) pyruvate: before exiting the cytoplasm
3) lactate
4) glycerol: generated in glycolysis, important for formation of triglycerides
describe the four substrates that may be converted into glucose via gluconeogenesis
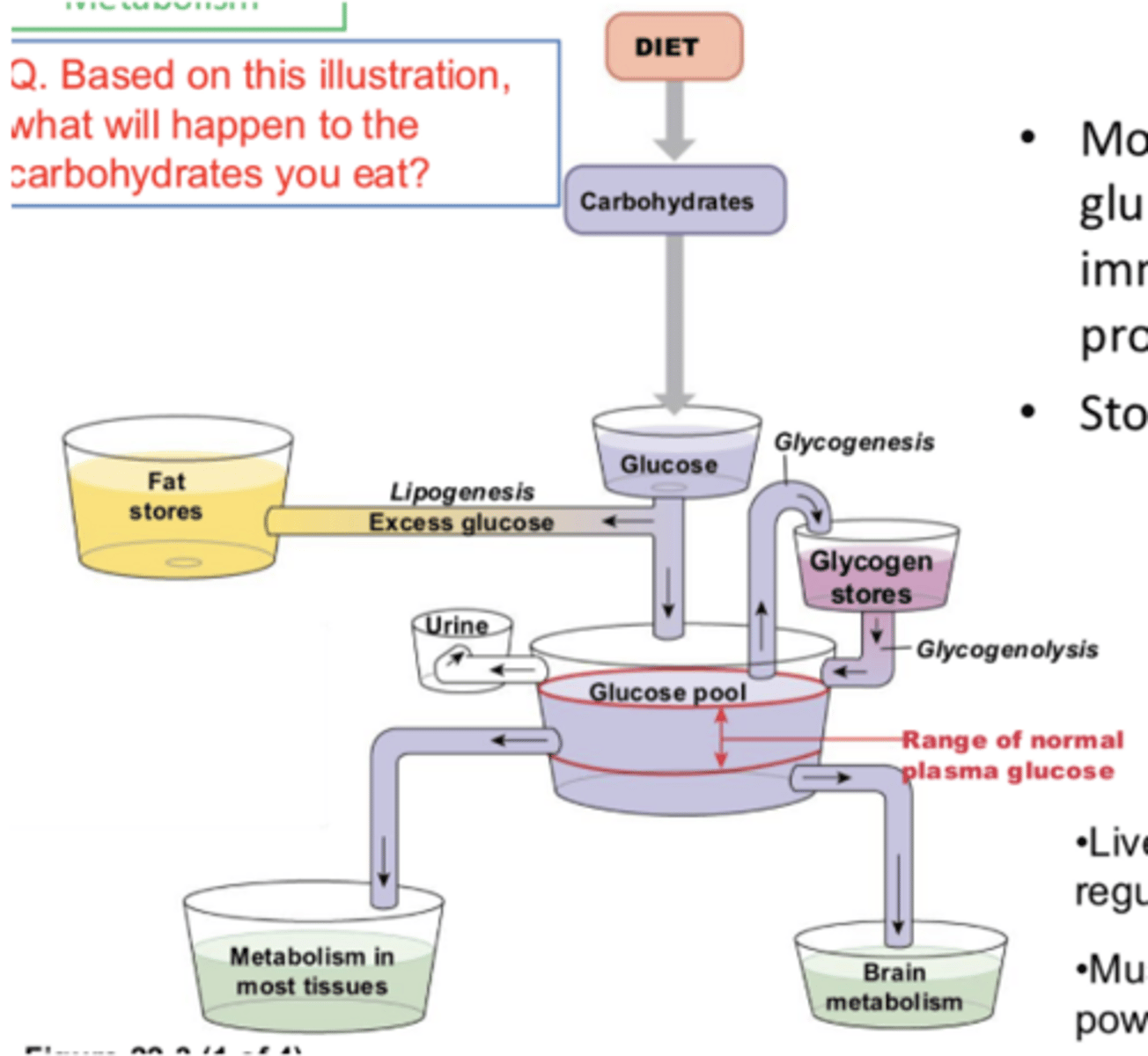
fed state (absorptive state)
following a eating meal when the products of digestion are being absorbed, used, and stored
NET anabolic pathway; small molecules building into larger molecules (ex. glucose/AA monomer → glycogen/protein polymer
***in food we eat, carbs (glucose) is very important → plasma glucose level is critical for body functions
priority/majority usage = metabolism in tissues and brain; immediate energy production
***brain supply is prioritized the most (has high affinity glucose transporters)
next priority usage = storage in glycogen (glycogenesis)
least priority usage = excess glucose excretion in urine and storage as fat (lipogenesis)
how is glucose used in the body (prioritized)
used to regulate blood glucose (liver) or power muscle contraction (glycogenolysis)
stored glucose in the liver (glycogen) lasts 3-4 hours and can be used elsewhere in the body, but stored glycogen in muscles is only used by the muscles themselves
a small bit can be stored in the kidney as well
how is glycogen used by the body?
broken down and absorbed as AAs, which are used in synthesis of new proteins, and with higher amounts can be used in gluconeogenesis
after we eat proteins, they are ___
fats are broken down and absorbed as chylomicrons → enter lacteal and eventually blood → on blood facing side of endothelial cells, lipoprotein lipase breaks down chylomicrons → free fatty acids are used for immediate energy production (in liver in beta oxidation or gluconeogenesis)
describe the path of fat absorption and usage of them after
lipoprotein lipase
on blood facing side of endothelial cells, dietary fats are absorbed as chylomicrons which are broken down by this enzyme into glycerol and free fatty acids for storage (fed state) or energy production (fasted state)
after breakdown, they can be transported into adipose tissue for storage as fat molecules
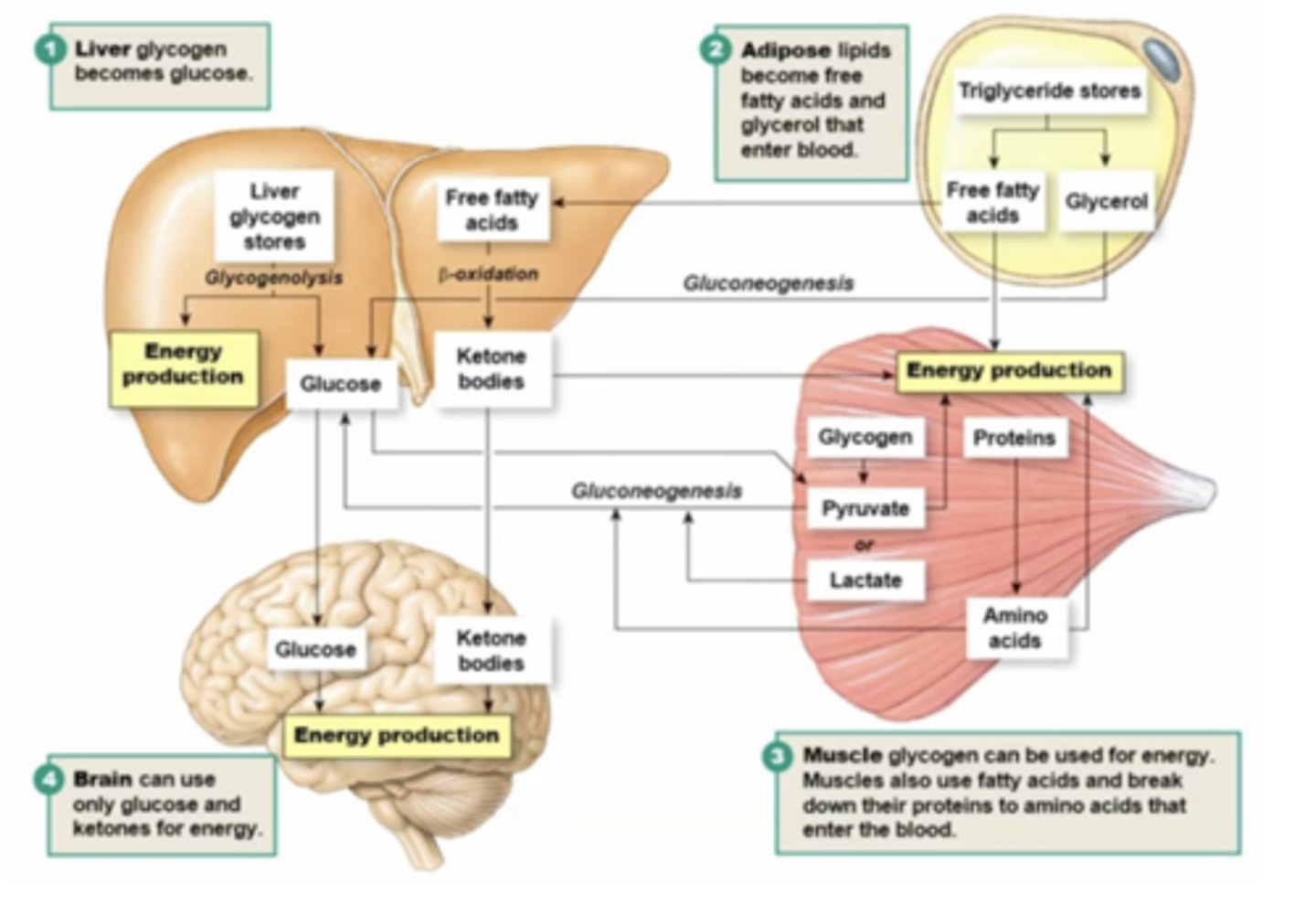
fasted state (posabsorptive)
once all nutrients from a meal have been digested, absorbed, and distributed to various cells
NET catabolic state
blood glucose drops bc storage is dipped into for energy
liver: glycogen stores broken down, beta oxidation, gluconeogenesis
adipose tissue: breaking down triglycerides into free fatty acids (beta oxidation)/glycerol
muscle: cori cycle, glucose sparing, muscle breakdown (of own proteins to produce AAs that can be used in gluconeogenesis (least preferred method))
brain: consumer that uses glucose and ketones for energy
what are the key organs in homeostasis of metabolism in fasted state, and what methods do they use to provide energy?
glycogen stores are broken down to glucose and provide energy to other places in the body (including brain)
glucose can enter systemic circulation bc the liver has glucose-6-phosphatase, which removes phosphate group so it can leave liver cells and enter blood (muscles do not have this so they can't break glycogen directly into glucose)
describe how the liver breaks down glycogen stores to provide energy
beta oxidation
free fatty acids may be converted back to acetyl CoA as a source of energy for the citric acid cycle through this process
only really occurs in prolonged fasting state (in the liver)
acetyl CoA
precursor for cholesterol (lipid steroid with ring structure) and source of energy for citric acid cycle
in fasted state, starts to break down triglycerides into FFAs and glycerol → glycerol can be used in gluconeogenesis, FFAs can go into beta oxidation/generate ketone bodies
normally adipose tissue has cell membrane enclosure (perilipin) so lipase can't get in, but the break down of that barrier (so lipids can be broken down) and activation of hormone sensitive lipase facilitates this
describe how adipose tissues can break down to provide energy
hormone sensitive lipase
ONLY in fasted state, breaks down (phosphorylates) fats when activated by hormones; breaks down stored fats (triglycerides) into free fatty acids and glycerol in the fasted state to provide energy when blood glucose is low/ATP is needed
inhibited in the presence of insulin
perilipin
protects the fat stores from hormone sensitive lipase; when broken down (phosphorylated), becomes leaky and allows active hormone sensitive lipase to get in and access stored fat (mobilize it)
lipase/colipase: in SI/from pancreas
lipoprotein lipase: to break down chylomicrons
hormone sensitive lipase in adipose tissue
what are the 3 main lipases used in the body?
cori cycle
glycogen in muscles can be broken down into glucose-6-phosphate which enters glycolysis to make pyruvate/lactate → brought to liver for gluconeogenesis (which makes new glucose that can be brought back to the muscles/other places in body)
happens bc there's no glucose-6-phosphatase in muscle (can't leave cell)
indirectly, cori cycle
directly, glycogenolysis
muscle can provide glucose ___ via the ___, but not ___ generate glucose via ___
glucose sparing
in muscles, using fatty acids from broken down adipose tissue as an energy source over glucose, but it takes a while
insulin
endocrine signal (hormone) from pancreatic beta cells that promotes energy usage and storage; dominant in fed state and without this glucose can NOT enter cells
stimulates glucose transport into cells/glucose oxidation (using glucose) and glycogen/fat/protein synthesis (glucose storage)
glucagon
endocrine signal (hormone) from pancreatic alpha cells that promotes energy release; dominant in fasted state
stimulates glycogenolysis (breaking down glycogen), gluconeogenesis, and ketogenesis
the ratio of insulin to glucagon
metabolism is primarily controlled by ___, and signals sent to cells in fed or fasted state
insulin: high glucose in blood and high AA level in blood (can afford to store nutrients bc there is excess)
***nutrients being absorbed have receptors on beta cells to stimulate insulin release, but there are also receptors for GIP, GLP1, and NTs from the parasympathetic NS (rest and digest in fed state)
glucagon: low plasma glucose but high AA in blood (gluconeogenesis triggered in fasted state, which is helped by higher level of AAs)
what signals stimulate insulin and glucagon secretion?
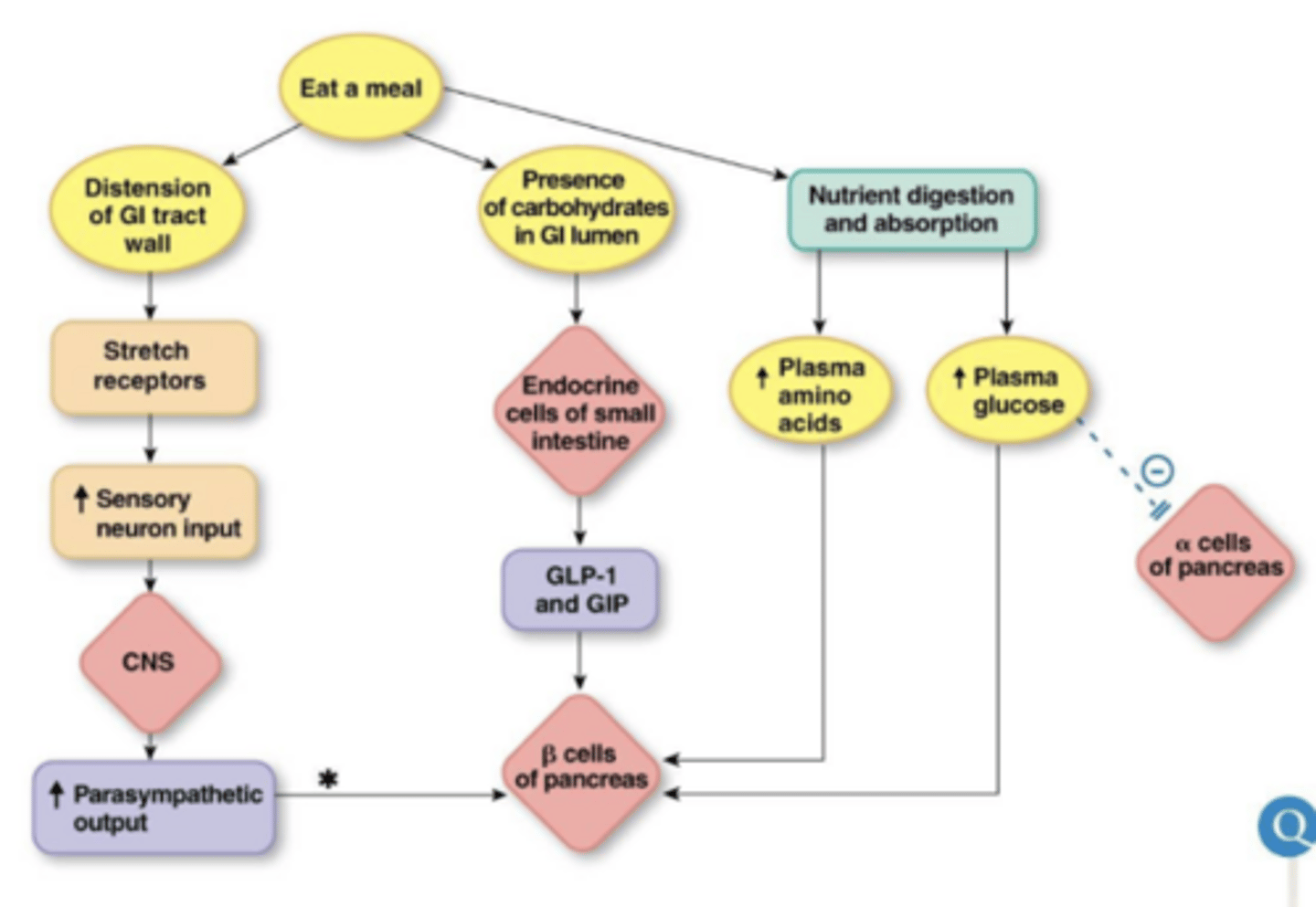
GIP and GLP-1
GLP-1 stimulates insulin secretion → when you have an agonist of this it causes an increased response to insulin
____ are secreted by SI in high glucose to stimulate pancreatic beta cells to secrete insulin
because you want insulin production in anticipation of glucose arrival; even before glucose increases in blood, the body is ready to receive this
prevents glucose level from spiking/fluctuating too much
why do we have lots of parallel pathways to secrete insulin?
parasympathetic, not sympathetic, activates insulin secretion
all other choices are correct
answer the question and explain your answer

GLUT4
glucose transporter in adipose and resting muscle cells; at rest contained in vesicles, but in insulin presence there is increased expression of this via exocytosis of them to insert it into membrane and allow glucose to move into the cell via facilitated diffusion
when we exercise, it also increases expression of this on membrane
lipogenesis
stimulated in adipose tissue in the presence of insulin by G3P; if ATP is abundant it signals we don't need to make more since it's not actively being used → in this fed state (high blood glucose, excess ATP) acetyl CoA is used to build fatty acids
this is why extra carbohydrates often lead to fat deposition
in liver glucose must be transported in/out constantly (both fed and fasted state) → expression of GLUT2 is constant (and indirectly controls this)
in fed state hexokinase is activated to maintain glucose gradient (trap it in cells in a diff form) and help keep it moving into the cell
in fasted state glucose gradient favors glucose leaving the cell (bc if you haven't eaten extracellular glucose is low)
describe how glucose transport is balanced in the liver (concentration gradients in the fed/fasted states)
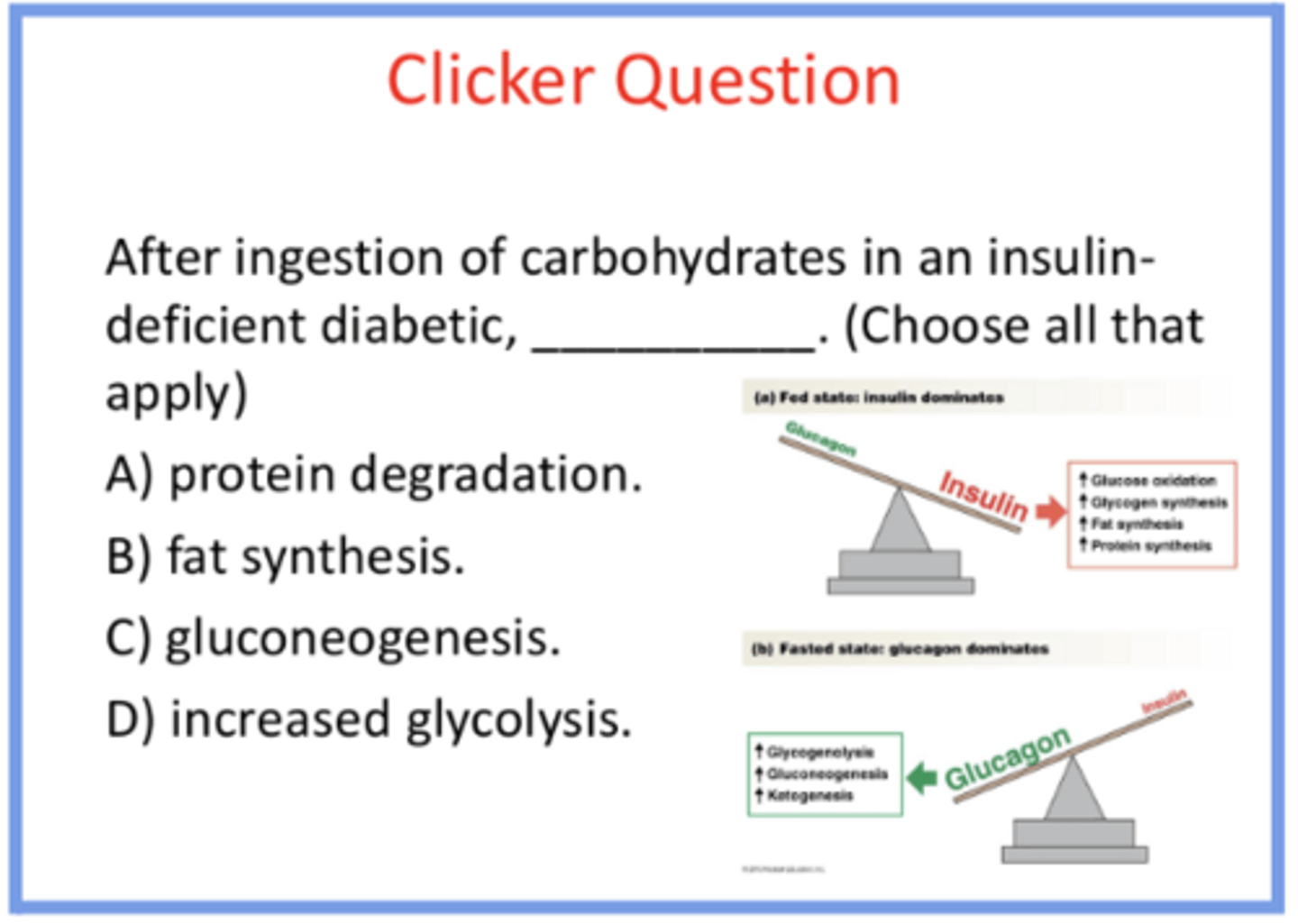
type 1 diabetes
insulin deficiency; people with this have lower body mass because their bodies always think they are in a fasted state
symptoms include hyperglycemia, increased gluconeogenesis, lipolysis and ketogenesis, and protein breakdown
destruction of pancreatic beta cells, genetic defect in MHC on chromosome 6
lipolysis
fatty acids may be released from fat stores through this process
ketogenesis
conversion of fatty acids to ketone bodies as an alternate energy source for the brain (which relies only on glucose and ketones for its high energy demands
occurs in times of starvation when we have very low available glucose
type 2 diabetes
insulin resistance; cells cannot respond to the insulin in the body
genetic defects in glucokinase, insulin molecule, insulin receptor, GLUT 4
***your body interprets this as fasted state even though you just ate because you can’t move glucose into cells without insulin
protein degradation occurs bc its a catabolic state
NOT fat synthesis bc that’s an anabolic process
gluconeogenesis bc glucose is not entering cells (no insulin), and low insulin to glucagon ratio = like fasted state → causes more gluconeogenesis bc that’s what the hormone signal is saying
NOT increase glycolysis because you’re not able to use the glucose you have
***everything in the green box will happen, everything in the red will NOT
answer the question and provide reasoning for all answer options
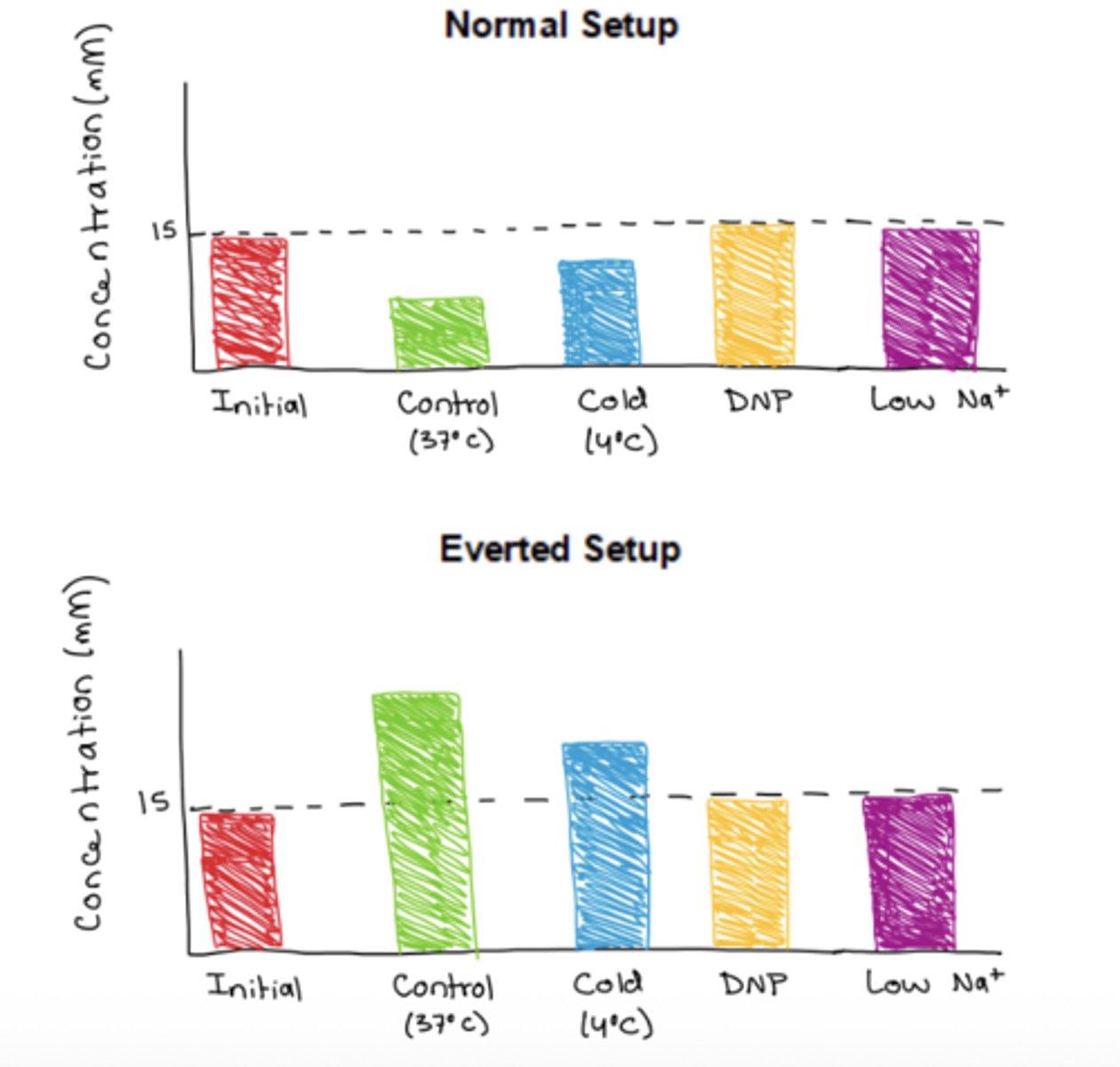
normal: in control conditions (Na and glucose present), glucose will be transported out of the intestinal sac into the exterior solution via SGLT1 (apical), so concentration in the sac would decrease. this would also happen exactly the same way in the cold condition, but just at a slower rate so concentration would not drop as much. however, without an Na gradient (in the low Na+ condition AND DNP condition since you need ATP for transport that creates the Na gradient) glucose will not be able to move out of the intestinal sac since SGLT1 relies on an Na gradient for secondary active transport, so the DNP and low Na+ concentrations would be the same as the initial concentration.
everted: for the same exact reasons, the same processes as described above would occur. however, the control and cold would increase instead (with cold rising less) because the lumen is facing the external solution (so glucose would go into
you have two sections of the small intestine tied at the ends suspended in two beakers, and inside both the intestinal sacs and beakers there is a 15mM glucose solution. in one beaker, the SI is inside out and the lumen side faces out ("everted"), and in the other it is oriented normally.
you have 4 experimental conditions (to alter the solution) for incubation: control (conditions as is), cold temperature, DNP (compound that disrupts ATP generation), and low Na+
describe how the concentration of glucose (mM) in the intestinal sac would change in each of these experimental conditions for the normal and everted setups