3.10: Aromatic Chemistry
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
What is the structure of benzene?
Planar molecule
Each carbon has 3 bonds
One unhybridized 2p orbital with 1 electron
Overlap of 6 parallel 2p orbitals form continuous pi bond
Electron density lies in 1 cloud above and below the plane
Equal bond lengths
Compare the structures of benzene and cyclohexa-1,3,5-triene
Benzene is more stable than cyclohexa-1,3,5-triene
The enthalpy of hydrogenation of benzene is less exothermic
Due to the delocalization of e- in benzene
Both are planar molecules
Benzene has equal C-C bond lengths, whereas cyclohexa-1,3,5-triene as varied bond lengths as the hexagon is distorted
Why is benzene more stable than cyclohexa-1,3,5-triene
Delocalisation of p e- in benzene means they form continuous pi bond
Why does electrophilic substitution of benzene take place instead of addition?
The continuous pi bond of delocalised e- would have to be broken
So stability would be significantly reduced
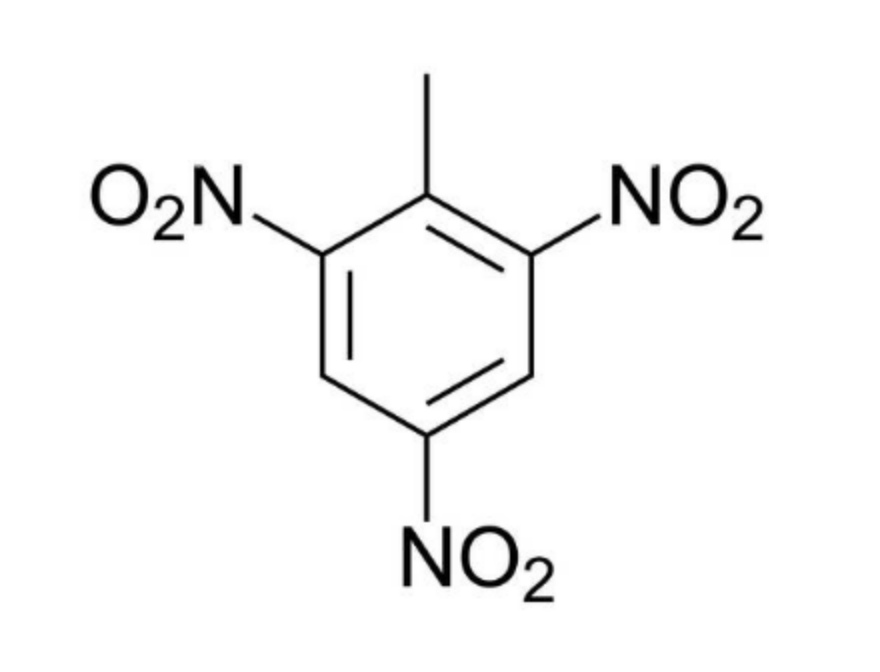
What is the structure of TNT?
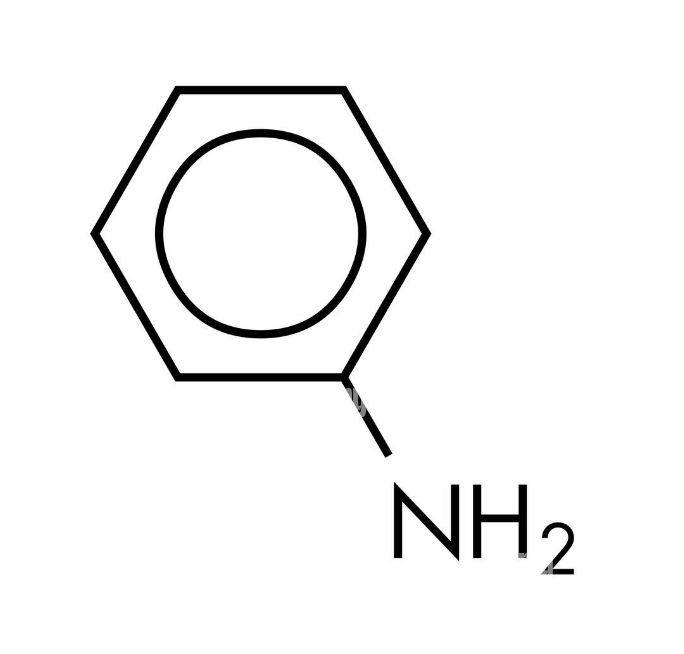
What is the structure of aniline?
What is the name of the mechanism for the nitration of benzene/friedel-crafts acylation?
Electrophilic substitution
What are the conditions required for electrophilic substitution?
500C
Conc HNO3+ conc H2SO4
What is the equation for the formation of reactive catalyst in Friedel-Crafs Acylation?
R-COCl + AlCl3 → R-CO+ + AlCl4-