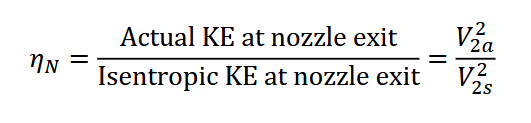Thermodynamics final chap 7
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What is entropy definition in Statisical Thermo
Degree of diorder of substance
Available microstaes and unterainty of molecular positions
What is the defintion of entropy in classical thermodynamics
mesure of irreversibilites (entropy is nort consever but is a thermdynamic property)
What happens to entropy when Isothermal increase in volume
Temp remain constant but added possible positions (increasing entropy)
What happens to entropy when isometric increase in temprature
volocity distibution range increases giving more possible kinetic energyies(entropy increses)
WHat happens to entropy there is a Isometric/Isothermal increase in amass
More possible positions and distributions of energy (entropy increses)
What happensto the entropy if there is a mixing of multiple substances isothermally
Temp remains constant, but added possible postions and particle types(increase in entropy)
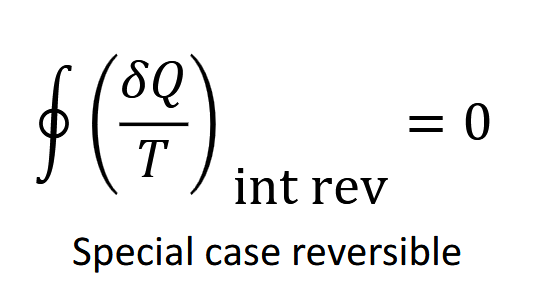
When is the clausis inequality equal ot zero
When the system is totally reversible
When is the clauis inequallity not equal to zero. And what is it equal to
Irreversible cycles;less than zero
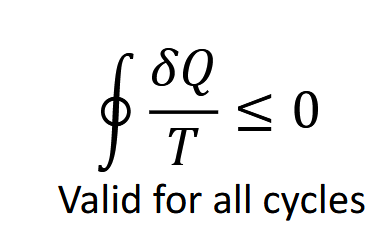
When can changes in entropy be calculated?
when the system is internally reversible
What is the special isothermal reversible case

What is the carnot cycle relationship to entropy change. what ist he derivatio
The carnot cycle returns to its origianl state in reversible process; therfore entropy is a state function that returns to the original state.
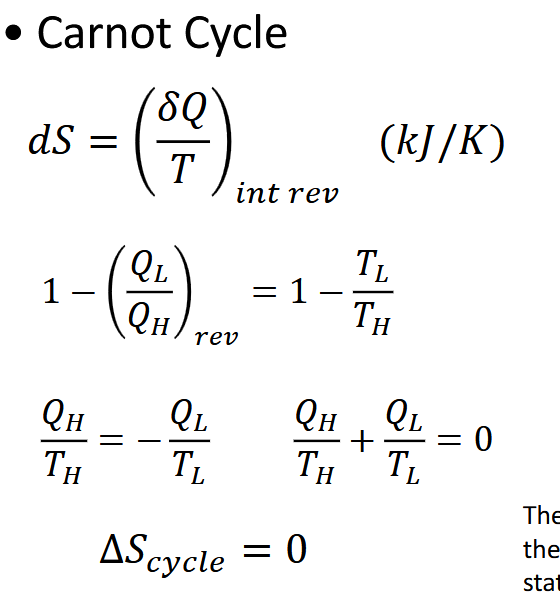
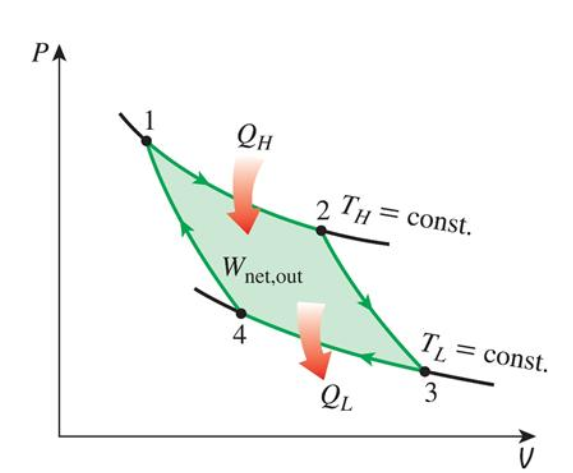
What is the carnot cycle look like on Pv diagram
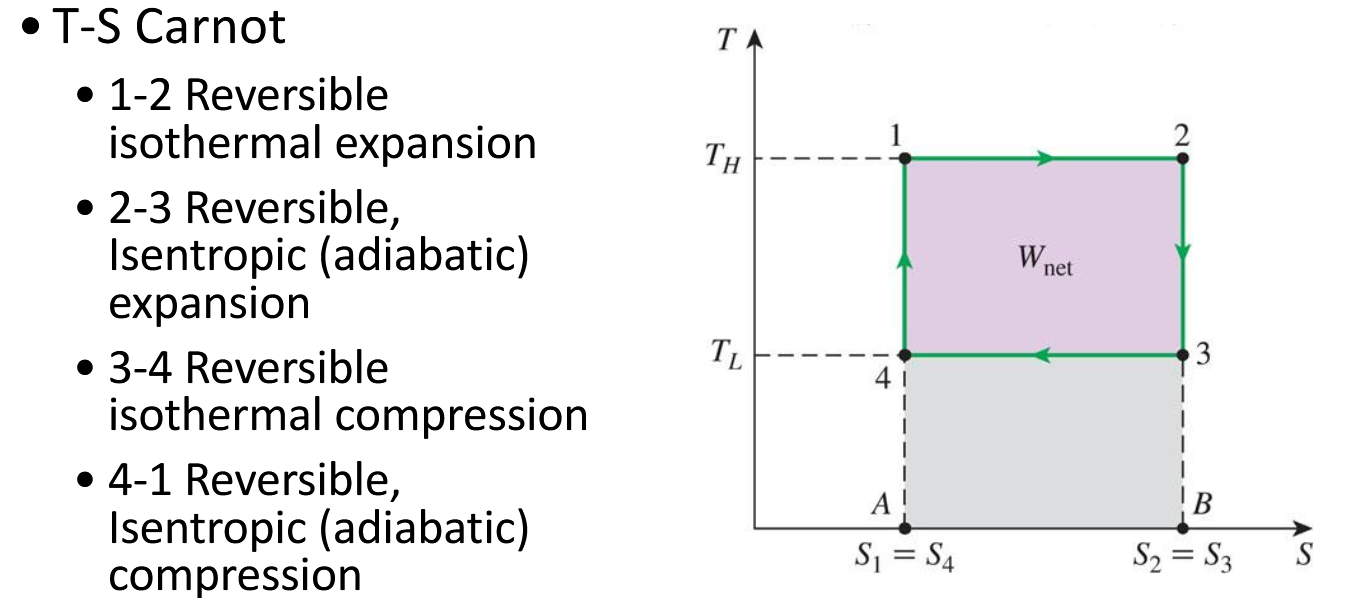
What does the carnot cycle look like on the Ts diagram
What is the increase of entropy principle
Some entropy is created during an irreverible process,due to irribvesibliles
What are the rule s of the increase of entropy principle
Process must occur in direction of Sgen > 0
Entropy is not conserved it is allways increases
S gen is a quantiy mesure of the irriversibilty of the system _thus it can be used to msure the prefornce of devices.
What are some caustions about increase in entropy principle
Change in entropy of a closed system can be negative,but s gen of the whole sustem can never be negative.
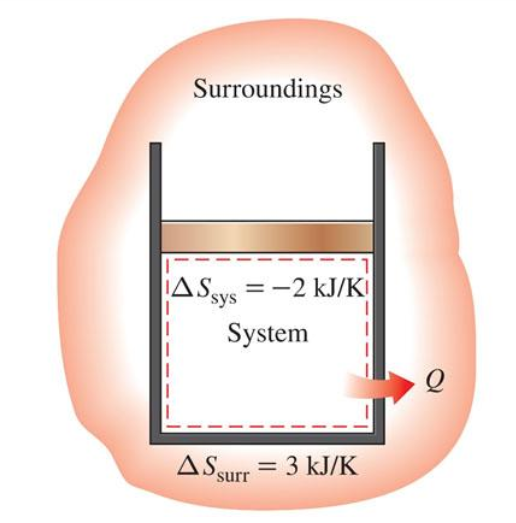
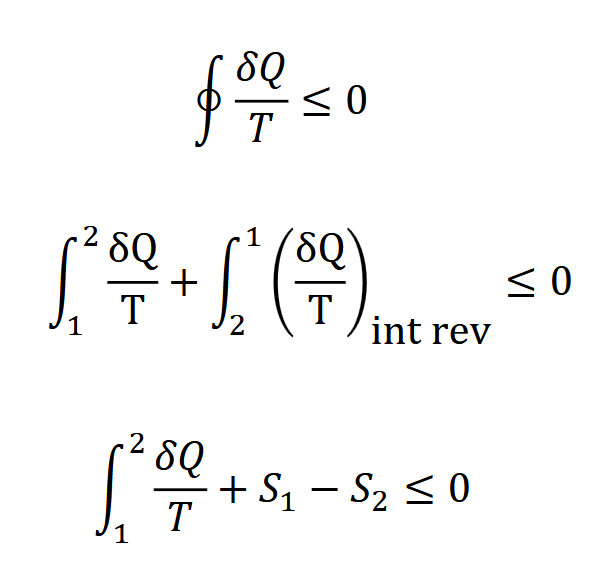
what is the rest of this derivation for Sgen
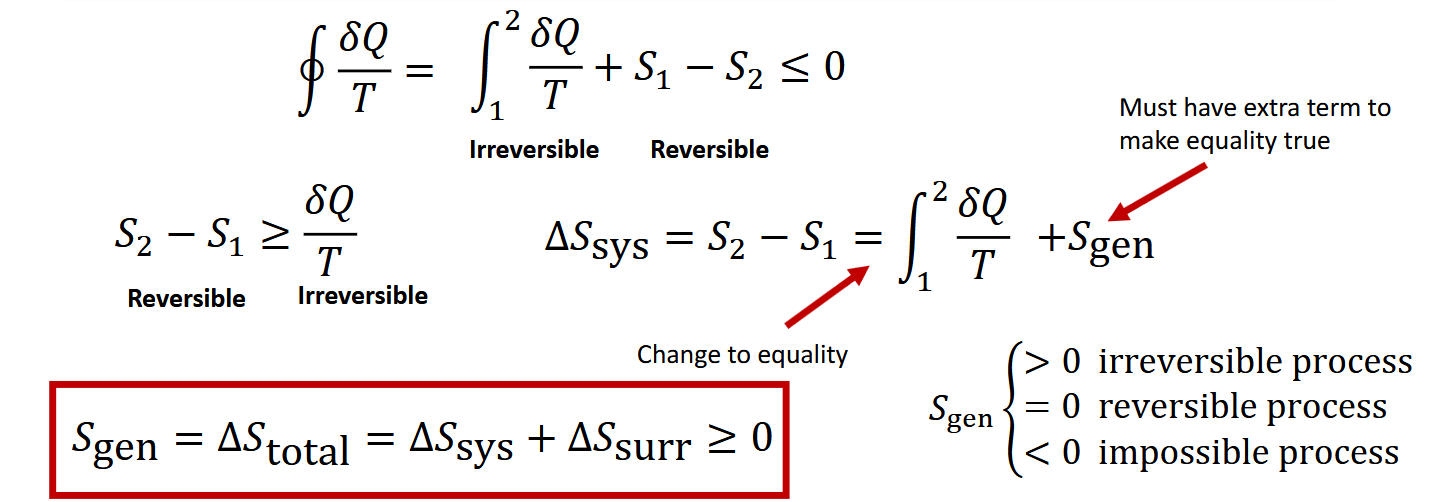
What are the assumptions of isentropic processes
It is internally reversible and adiabatic
Which composnets can be modeeled as near adiabatic(isentropic)?
Pumps Nozzles turbines diffusers compressor
When is the area inder t-s diagram the total heat transfer
Only when it is an internally reversible process.
When are the T-ds reations valid
it is valid for all process in closed and open system. Reversible or irreversible Valid only if realtionships are known
What assumptions are made for entropy change of liquids and solids
Negligible change in volume/specific volume
What assumptions are made for entropy changes of ideal gases(constant volume). Which equation will you use?(CONSTANT VOLUME)

What assumptions are made for entropy changes of ideal gases(constant Pressure)? Which equation do you use CONSTANT PRESUURE

What is exact analysis and how is it used to solve changes in entropy of ideal gas
You use the charts to reqfrence the the entropy (A-17 through A-18) and then useing the s dot for the places in the equations
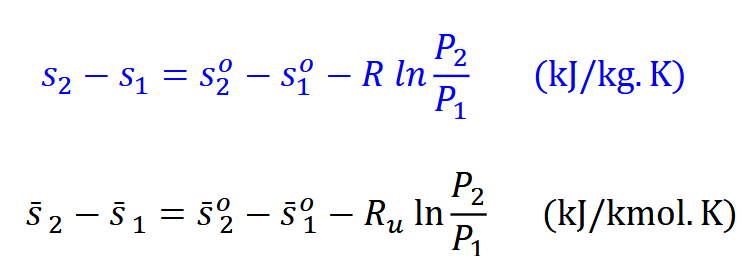
What is average analysis?
You use the average temprature value to determine the cp or cv value
What do the y and x axis repersent on a mollier diagram? what do mollier diagram represent in terms of work and irreversibility?
The veritical distance repersent change in enthalpy. for adiabatic steady flow devices thits is a mesure of work.
The horizontal diance is the change in entropy. For adiabatic flow devices this is a mesure of irreversibilty
What is the third law of thermodynamics nad how does it realate to entropy
Entropy of a pure crystaline subatance at absolute zero temp is zero. THe absolute entropy is deffines from reffecne point zero
this is why all the tables have s dot on them
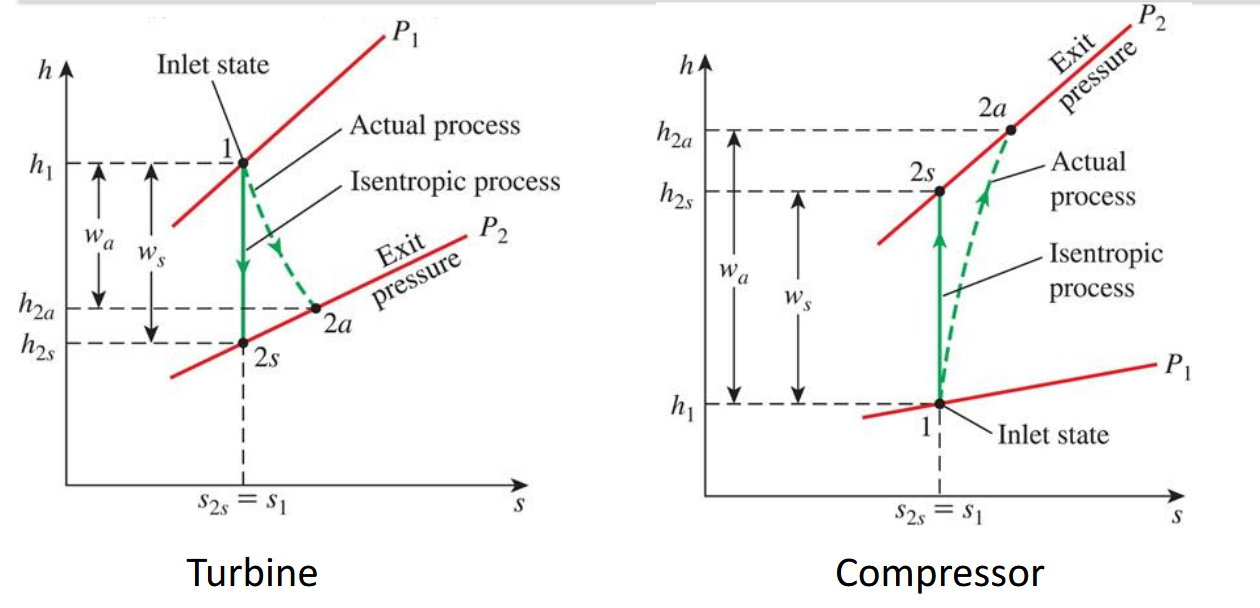
What does isentropic efficency and what does it repersent in Turbine, compressor, and pump?
Isentropic process the highest performing for steaady flow componets value is less than 1
Turbines outputs the most work
compressors and pumps it puts in the least abount of work

What does hte isentropic ves a real process look like on ts diagram
What do isentropic efficy look like for nozzle and diffuser
It is teh ratio of actual kinstic energy vs isentropic kinetic energy
isentropic nozzle will maximizr kietic energy at the exit state