Module 3 Energy and Biological Building Blocks
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
photoautotrophs
use light as an energy source to produce organic compounds
chemoautotrophs
use environmental inorganic compounds as an energy source to produce organic compounds
heterotrophs
must obtain energy and carbon by consuming other organism or organic matter
photosynthesis
the process used by plants, algae, and certain bacteria to turn sunlight, water, and carbon dioxide into sugar (food) and oxygen
sunlight + 6H2O + 6CO2 → C6H12O6 (glucose) + 6O2
chemosynthesis
uses energy from chemical reactions to build sugars out of smaller molecules
properties of light
the amount of energy in light is inversely proportional to its wavelength
how photosynthesis harvests light energy
photosynthetic pigments with different absorption peaks collect solar energy
reflect unabsorbed wavelengths → we see different colors
chlorophyll a
the primary photosynthetic pigment in plants, algae, cyanobacteria
chlorophyll b
an accessory pigment that broadens light harvesting in plants, green algae and a few cyanobacteria
carotenoids
masked by chlorophyll
anthocyanin
pigment made in the fall
where photosynthesis takes place
most photosynthesis takes place in the ground tissue cells of leaves called the mesophyll cells
mesophyll cells
contain many chloroplasts where photosynthesis occurs
chloroplast morphology
chloroplast: double membraned organelles descended from cyanobacteria that are found in plants
stroma: the space inside the inner membrane of the chloroplast, site of chemical reactions that produce biomass in photosynthesis
thylakoid membranes: stacked into grana and are a key component for energy production of photosynthesis
photosystems
complexes of proteins and pigments (chlorophylls and carotenoids) that harvest light energy to generate high energy electrons
2 types
photosystem 1
photosystem 2
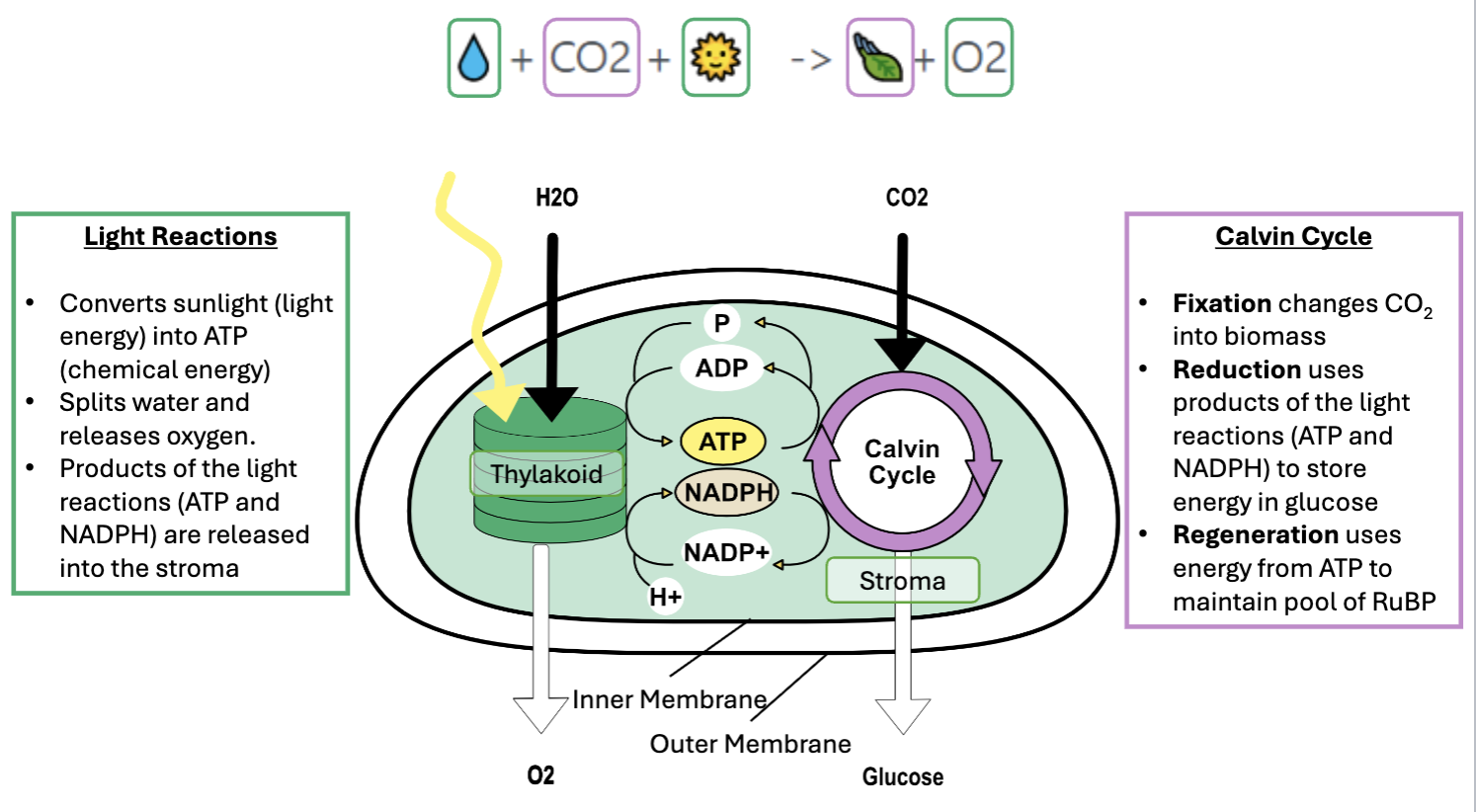
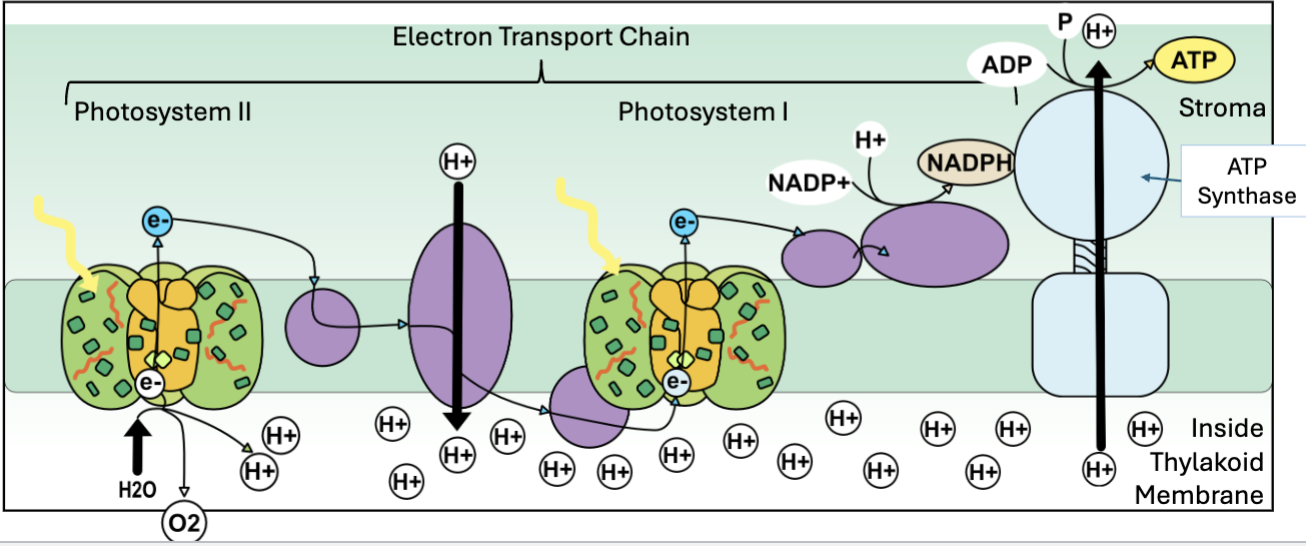
Light reactions (photosynthesis part 1)
uses light to produce ATP
light excited electrons in photosystem 2, water is split releasing O2, protons and electrons
electrons travel down an electron transport chain to photosystem 1, this releases energy to pump H+ into the thylakoid lumen, building a gradient and generates NADPH
H+ flow down the gradient back into the stroma, passing through ATP synthase which drives ATP production
summary
Converts sunlight (lightnenergy) into ATP (chemical energy)
Splits water and releases oxygen
Products of the light reactions (ATP and NADPH) are released into the stroma
calvin cycle (photosynthesis part 2)
Converts CO2 to biomass (glucose)
summary
Fixation changes CO2 into biomass
Reduction uses products of the light reactions (ATP and NADPH) to store energy in glucose
Regeneration uses energy from ATP to maintain pool of RuBP
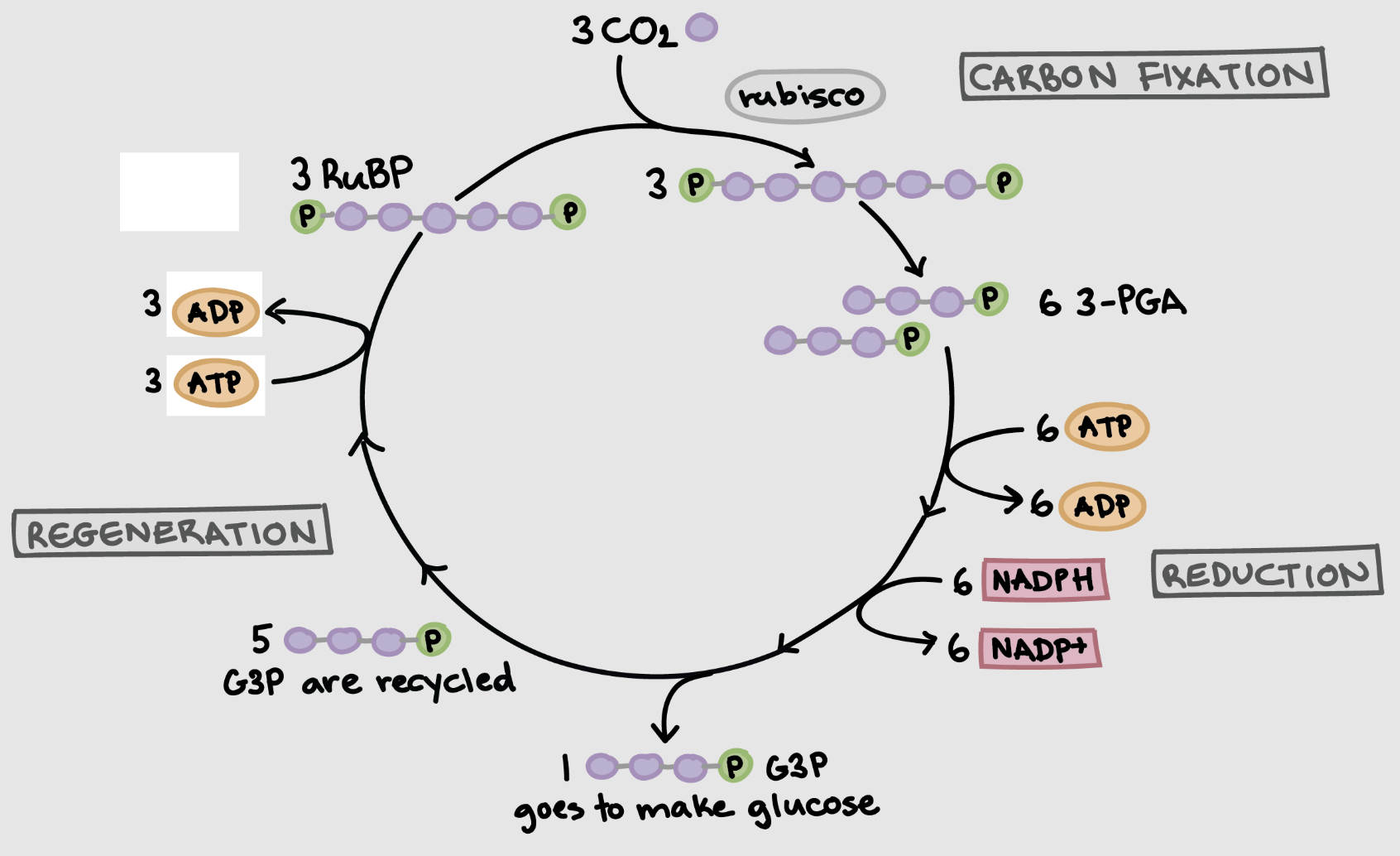
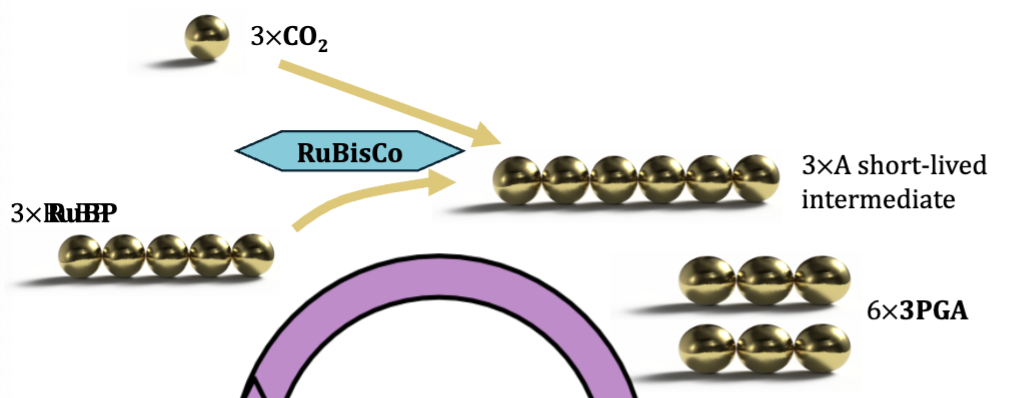
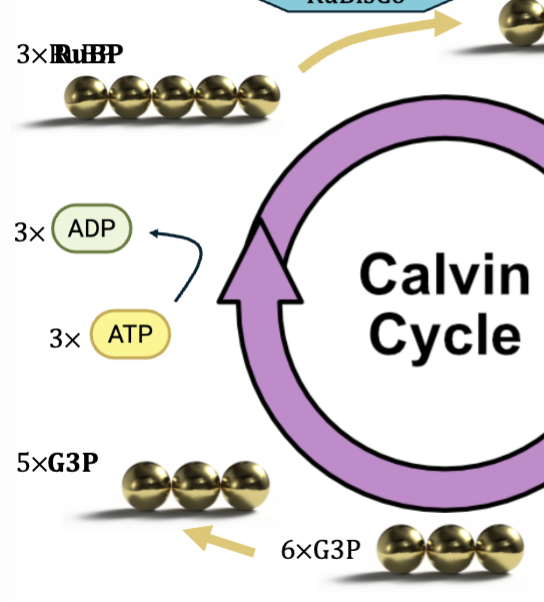
Carbon Fixation
enzyme called RuBisCo exists in stroma
RuBP is a five carbon sugar produced by the calvin cycle
RuBisCo combines CO2 gas with RuBP to “fix” it into a solid but unstable six carbon sugar
six carbon sugar splits into two three carbon molecules - 3PGA
Reduction
ATP and NADPH turn low energy 3PGA into high energy G3P
takes 2 G3P to generate 1 glucose molecule
calvin cycle must turn 2x to generate 1 glucose molecule and regenerate RuBP
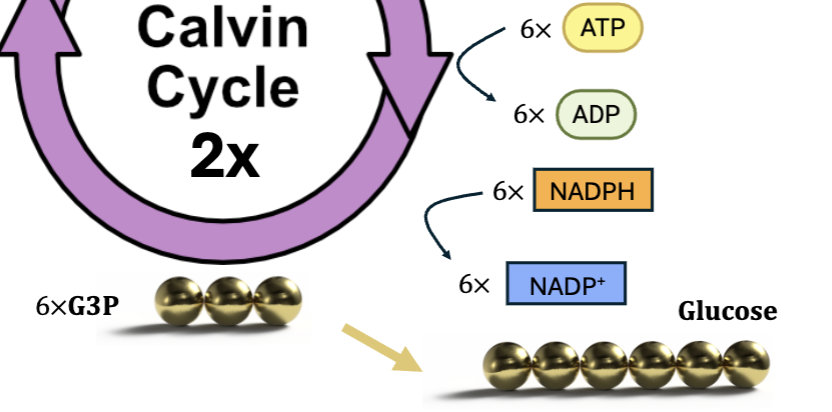
Regeneration
of the 6 G3P molecules 1 is used to form glucose
ATP used to rearrange 5 remaining G3P into 3 RuBP molecules
maintains pool of RuBP to continue cycle of CO2 fixation
C3 photosynthesis
CO2 fixation produces 3 carbon molecule
this is the ancestral form of photosynthesis and most common
the C3 pathway is driven by RuBisCo enzyme
only works well in cool, humid environments
RuBisCo
drives C3 photosynthesis
RuBisCo is slow but plentiful
RuBisCo makes mistakes (will us O2 instead of CO2)
photorespiration (C2 cycle)
process in plants where the enzyme RuBisCo mistakenly uses O2 instead of CO2 during photosynthesis
leads to 20-50% reduction in efficently
if O2 is used it makes 3PGA and 2-phosphoglycolate which is useless and toxic to plants
2-phosphoglycolate can then be made back into 3PGA
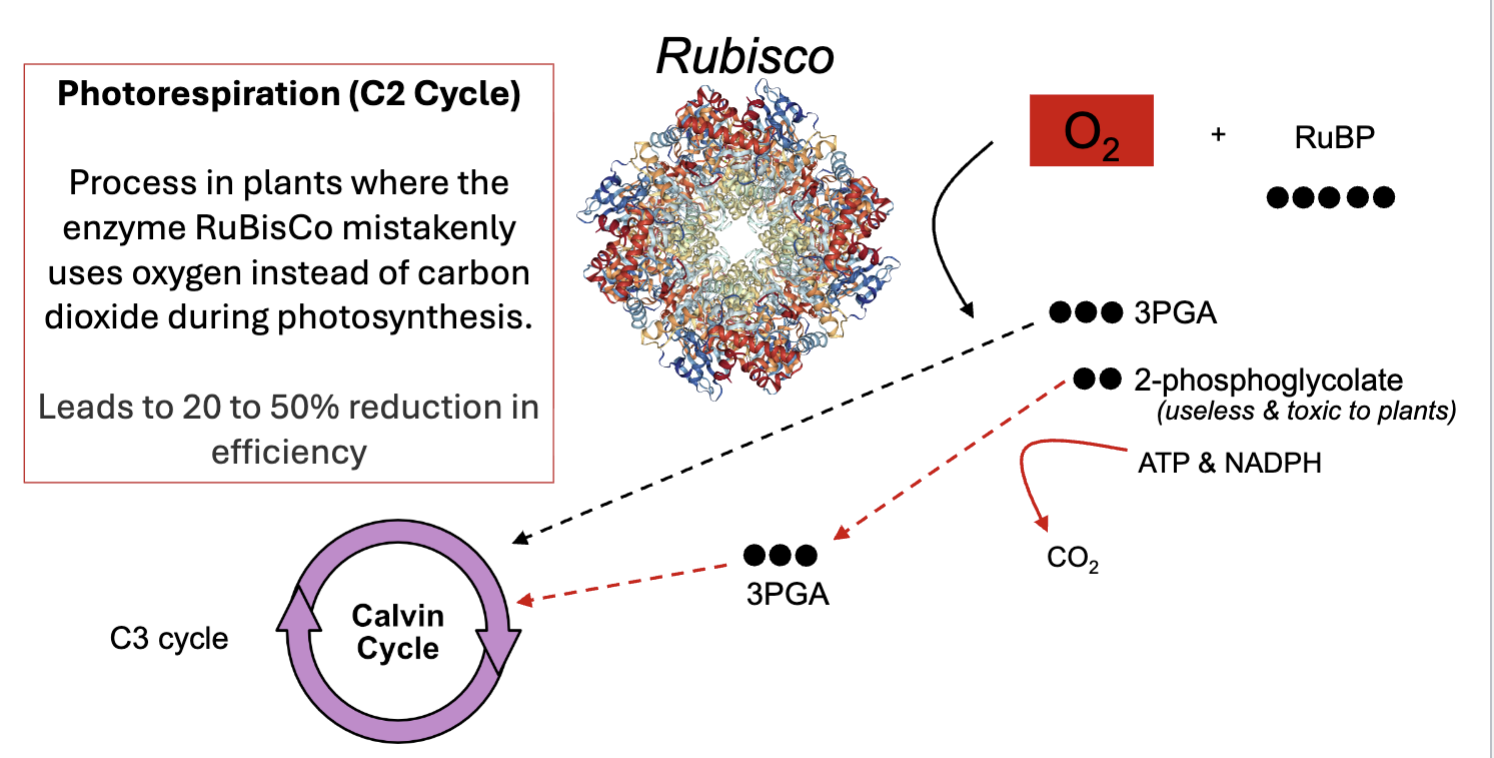
waste product of photosynthesis
O2 and builds up inside the leaf
stomata
pores of the dermal tissue of plant leaves
CO2 in, water and O2 out
different → open pores allow water to evaporate out
guard cells
open and close the stomata to allow gas exchange
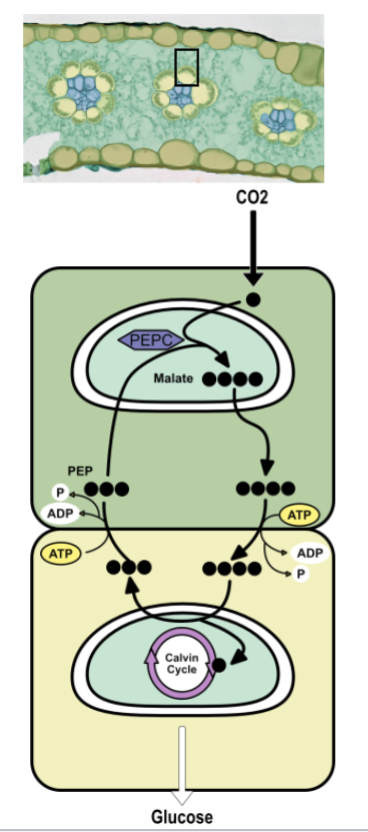
C4 photosynthesis
physically separate carbon fixation from calvin cycle in different cells
carbon fixation → mesophyll cells
calvin cycle → bundle sheath cells
C4 mesophyll cells → carbon fixation
enzyme called PEPC exists in stroma, had higher attraction to CO2 than O2
PEP is a three carbon sugar
PEPC combines CO2 gas with PEP to “fix” it into malate a four carbon sugar
malate actively pumped into bundle sheath cells
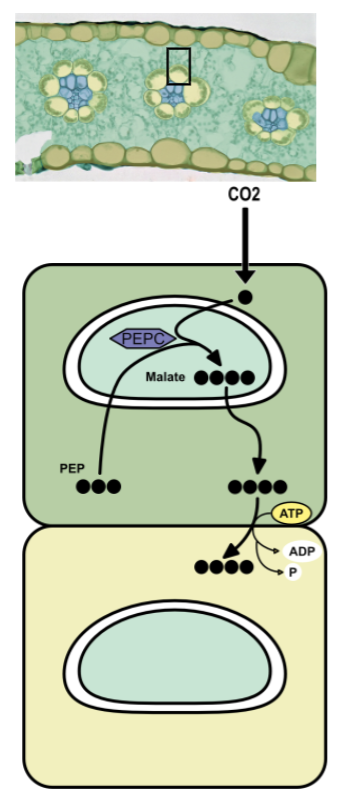
C4 bundle sheath cells → calvin cycle
malate split apart
1 carbon enters regular calvin cycle (with RuBisCo) → glucose
3 carbons → pyruvate
pyruvate diffuses into the mesophyll cell and the ATP is used to regenerate PEP
physically separating steps isolates RuBisCo from O2
common in grasses like corn and sugarcane adapted to hot, sunny environments
CAM photosynthesis
carbon fixation from calvin cycle in mesophyll cells is separated in time
carbon fixation occurs at night
4 carbon molecule stored in vacuoles
calvin cycle occurs during the day
typical of dessert plants like cacti and succulents
metabolism
the chemical reactions that occur in a living organism
obtain energy from food by converting glucose to ATP for use in cellular processes
convert biomass of food into biological building block (macromolecules)
excrete metabolic wastes
anabolic processes
build larger molecules from smaller ones
require energy
catabolic processes
break down larger molecules into smaller ones
release energy
how do you measure metabolic activity
the amount of oxygen use dup (or carbon dioxide produced)
gas exchange use is more easily measured
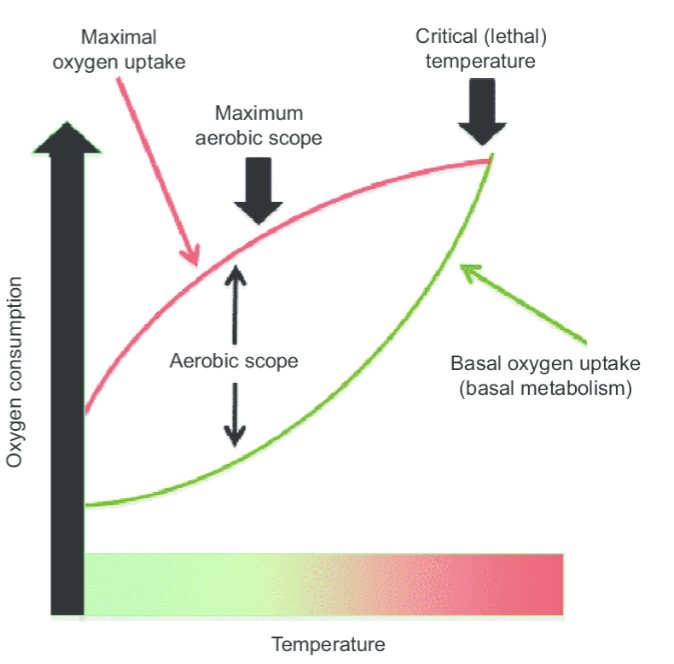
basal metabolic rate (BMR)
the rate at which an animal consumes oxygen while at rest, no digestion of food, no physical, thermal or psychological stress, normal temperature conditions
minimal resting lifestyle
maximum metabolic rate (MMR)
the maximal rate at which oxygen can be transported from the environment to the tissue mitochondria
induced by activity/stress
metabolic rate and enviroment
BMR and max MMR are fundamental physiological parameters providing the floor and ceiling in aerobic energy metabolism
it takes more energy to maintain metabolic function at environmental extremes
as environmental conditions change BMR and MMR will change to different extents
aerobic scope
capacity of an organism to increase its aerobic rate above maintenance level
aerobic scope = MMR - BMR
it determines how much excess energy is available for growth and reproduction
glucose
six carbon sugar that stores energy in chemical bonds
all multicelluar eukaryotes store glucose and oxidize it to provide chemical energy in the form of ATP
once glucose is in a cell it may be used for cellular respiration or fermentation
the site of these processes is in the mitochondria
mitochondrial morphology
double membraned organelles that are descended from bacteria that underwent endosymbiosis found in all eukaryotes
mitochondrial matrix: the space inside the inner membrane of the mitochondrion, the site of aerobic respiration
cristae: inner membrane folds, increase surface area for energy reactions to occur
cellular respiration
breaks sugar down to generate ATP
C6H12O6 + 6O2 + ADP + P → 6H2O + 6CO2 + ATP + heat
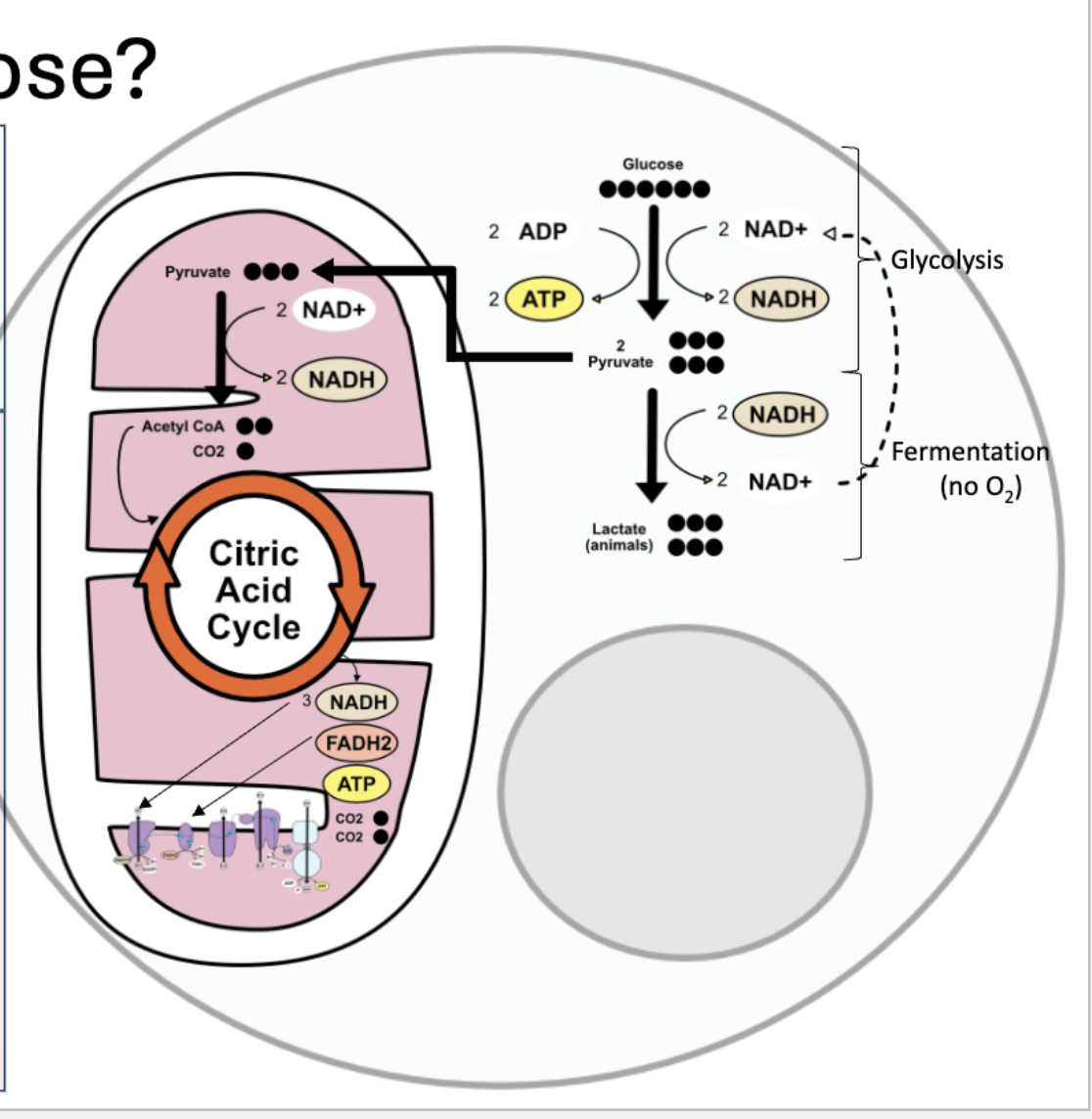
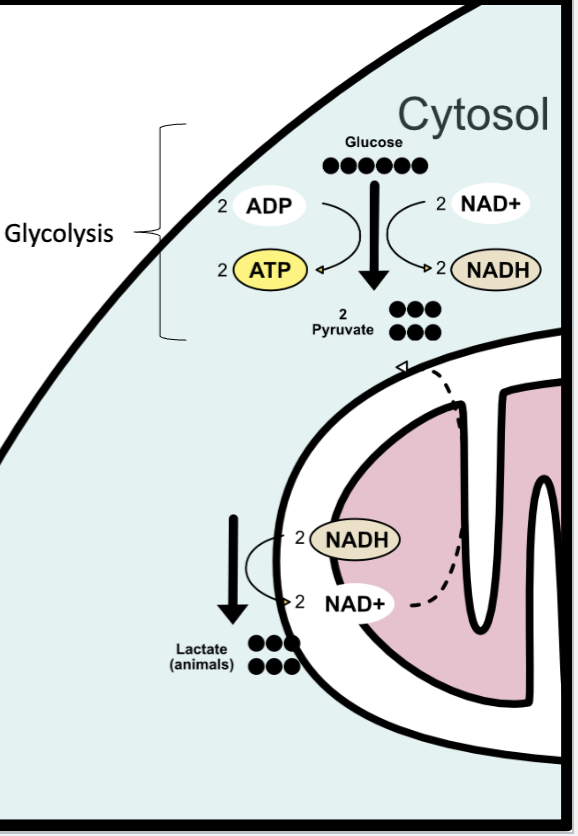
glycolosis
“breaking sugar” in cytosol
glucose breaks down into
2 pyrivate (3 carbon molecules)
2 ATP
2 NADH
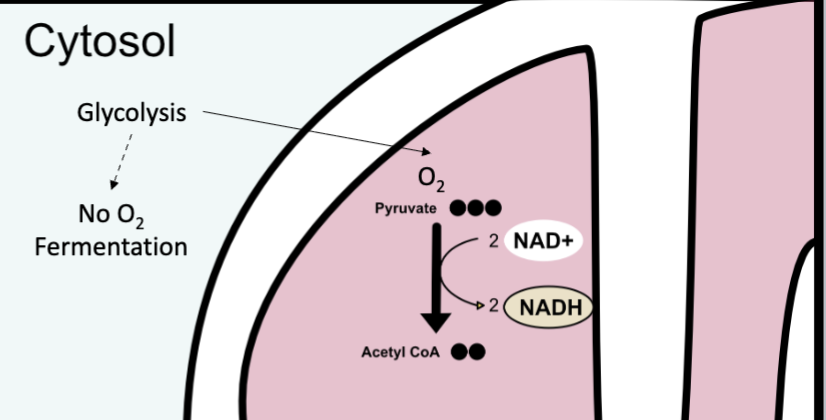
if there is oxygen
pyruvate enters mitochondrion → aerobic respiration
goes to link reaction
if there is NO oxygen
pyruvate remains in cytosol → fermentation (anaerobic)
regenerates NAD+ from NADH
allows glycolysis to continue
end products: lactate (animals), ethanol and CO2 (yeast)
Link reaction (only if oxygen is present)
in mitochondrial matrix
end products: acetyl CoA (2 carbons), NADH, CO2
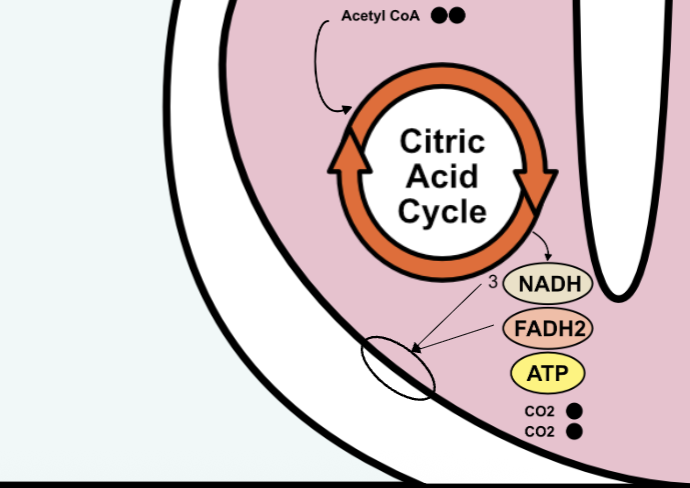
citric acid cycle
multi-step cycle
stores energy in reduced molecules
3 NADH, FADH2, ATP (all going to the electron transport chain)
waste product: 2 CO2
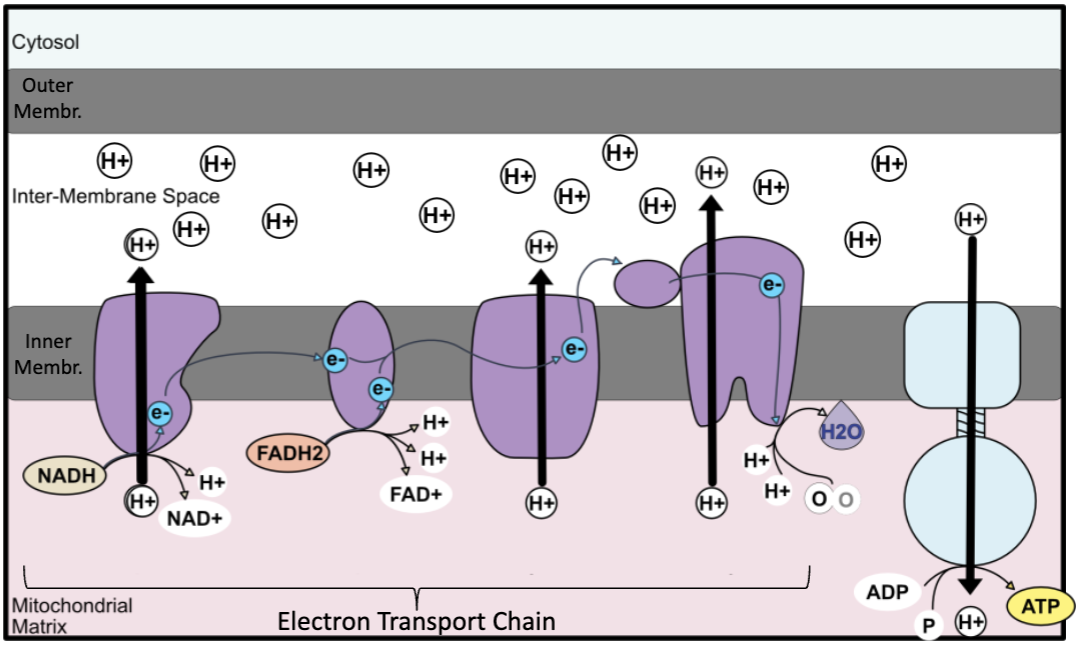
electron transport chain
NADH and FADH2 donate electrons to the ETC on the inner mitochondrial membrane
electrons travel down an electron transport chain releasing energy to pump H+ into the inner membrane space, building a proton gradient
oxygen (1/2 of an O2 molecule) is the final electron acceptor → forms water
protons flow down gradient through ATP synthase rotor, from high to low concentration, proton flow drives rotation in ATP synthase and converts ADP + Pi to ATP
cellular respiration simple summary
glycolysis (cytosol)
1 glucose → 2 pyruvate + 2 ATP + 2 NADH
if no O2: fermentation (cytosol)
2 pyruvate + 2 NADH → lactate + 2 NAD+
if O2: link reaction (mitochondrial matrix)
2 pyruvate → 2 acetyl CoA + 2 NADH + 2 CO2
citric acid cycle (mitochondrial matrix)
2 acetyl CoA → 6 NaDH + 2 FADH2 + 2 ATP + 4 CO2
electron transport chain and ATP synthesis (inner mitochondrial membrane)
NADH and FADH2 donate electrons to establish proton gradient
O2 final electron acceptor
ATP synthase → 28 ATP
why is breakdown required
it is difficult for macromolecules (large and complex molecules) to enter a cell → break them down into micro molecules that can
primary producers
make biomass from environmental CO2
ex. photoautotrophs (photosynthesis) and chemoautotrophs (chemosynthesis)
food web
a graphical representation of the flow of energy through an ecosystem
trophic level
an organisms position in a food web
animal body plans arranged around gut used for internal digestion & absorption
ingestion: acquire and mechanically process food, taken in through the mouth to the gut
many specialized structures exist to allow animals to acquire and mechanically process food
mechanical breakdown increases surface area from chemical digestion to occur
digestion: chemically (and mechanically) process food in the gut
tissues secrete digestive enzymes to breakdown macromolecules and the gut may house microbes to help break down of complex molecules
gut has high surface area to maximize absorption
allows breakdown of macromolecules to micromolecules → required for growth, development, and biological functions.
herbivores (primary consumer)
consume primary producers
suspension/filter feeders (primary consumer)
consume food particles taken out of the water column
passive filter feeders
strain suspended matter from environment
active filter feeders
create water current to strain matter
carnivores (secondary consumer)
consume other consumers
omnivores (secondary consumer)
consume both primary producers and other consumers
defense mechanisms
chemical
produce chemicals to make tissues toxic or unpalatable
consume and repurpose chemical defenses from food
behavioral
move away from predator
physical
avoid predation
crypsis allows animals to blind in with surroundings and hide
aposematism: bright colors act as warning of chemical defenses
deter predation → spines
parasites (secondary consumer)
organisms that live on or inside another organism at the expense of the host
detritivores
consume organic waste and decaying matter, helping to return nutrients to the biosphere
deposit feeder (detritivore)
consume organic matter found on and within the substrate (the material that forms the floor of the enviroment)
fungi body plans maximize surface area for absorption via hyphae
fungi can secrete enzymes to externally break down macromolecules for absorption
many are decomposers: break down dead matter and release nutrients back to the biosphere, especially on land
fungi are the only organisms able to break down lignin, a plant material found in woody plants
many fungi form symbiotic relationship with other organisms to obtain energy and nutrients
ex. lichens are a symbiosis of fungi and algae/cyanobacteria
nutrients
the raw materials organisms use to build structures and supply chemical reactions
macronutrients
elements required in large amounts for growth and development
ex. C, N, P, K, O, Mg
micronutrients
elements required in trace amounts for vital biochemical functions
ex. Fe, Cu, Ni, Zn, Mn, Cl
decomposers
key to breaking down complex molecules and returning nutrients to environment in forms that other organisms can use
ex. detritivores, fungi, microbes
microbiology
the study of microscopic organisms such as bacteria, viruses, fungi, protozoa, and algae
microbiome
the collective genomes of all microbes in an environment
microbiota
the community of microorganisms themselves
microbes
tiny living organisms, they are found everywhere, many are crucial for human health, digestion, and the environment
gut biomes → microbes help animal digestion
fewer than 1% are pathogenic
the rest form the invisible infrastructure of life: breaking down organic matter, recycling nutrients, and sustaining both our own physiology and global ecosystems.
limiting nutrient
nutrients that limit the growth, abundance, or distribution of a population of organisms in an ecosystem due to their scarcity
phosphorus (P) and nitrogen(N) are common limiting nutrients in both soil and aquatic systems, and sometimes iron (Fe)
law of the minimum
productivity dictated not by total resources available, but by the scarcest resource (limiting factor)
nitrogen cycle
although N2 is abundant it cannot be used for growth by living organisms
N2 gas is “fixed” by microorganisms to form nitrogen compounds that are used by other organisms to sustain life
summary
nitrogen Fixation: specialized microbes convert nitrogen gas into inorganic forms organisms can use
ammonification: decomposers convert organic N compounds to Ammonia
nitrification: 2-step process where nitrifying bacteria convert Ammonia to Nitrate
nitrogen cycle: nitrogen fixation
Specialized microbes convert nitrogen gas (N2) to Ammonia
(NH3) or Ammonium (NH4+) in soil
Energy intensive process – requires ATP
Some plants have a symbiotic relationship with nitrogen-fixing bacteria in root nodules
Plants can take-up ammonium directly, but too much can damage cells and movement in soil is limited
nitrogen cycle: ammonification
Dead organisms contain organic N (proteins, etc.)
Decomposers use enzymes to break-down organic N in dead matter → inorganic Ammonia (NH3) and Ammonium (NH4+)
Plants can take-up ammonium directly, but too much can damage cells and movement in soil is limited
nitrogen cycle: nitrification
Nitrifying bacteria convert Ammonia (NH3) → Nitrite (NO2-) → Nitrate (NO3-)
2-step process:
Nitrosomonas converts Ammonia (NH3) → Nitrite (NO2-)
Nitrobacter converts Nitrite (NO2-) → Nitrate (NO3-)
Nitrate preferred by most plants, can move more readily in the soil, but may be returned to atmosphere by de-nitrifying bacteria
spatial subsidy
a resource (nutrients, prey, detritus) gets transported from one habitat to another, increasing the productivity of the organisms living there