Cell Biology and Genetics, Test 2 Study Flashcards - Yui Hisanaga
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What are some similarities and differences between Prokaryotic and Eukaryotic cells?
Similarities: Both have DNA, cell membrane, and ribosomes
Differences: Prokaryotic cells lack all membrane bound organelles, including their nucleus. Instead, they store their genetic information in their nucleoid.
What are the structures and functions of prokaryotic cells?
Cell Wall: made of peptidoglycan (type of carbohydrate)
Plasma Membrane
Nucleoid
Ribosome
Flagellum: (not all bacteria has this)
Lack of all membrane bound organelles
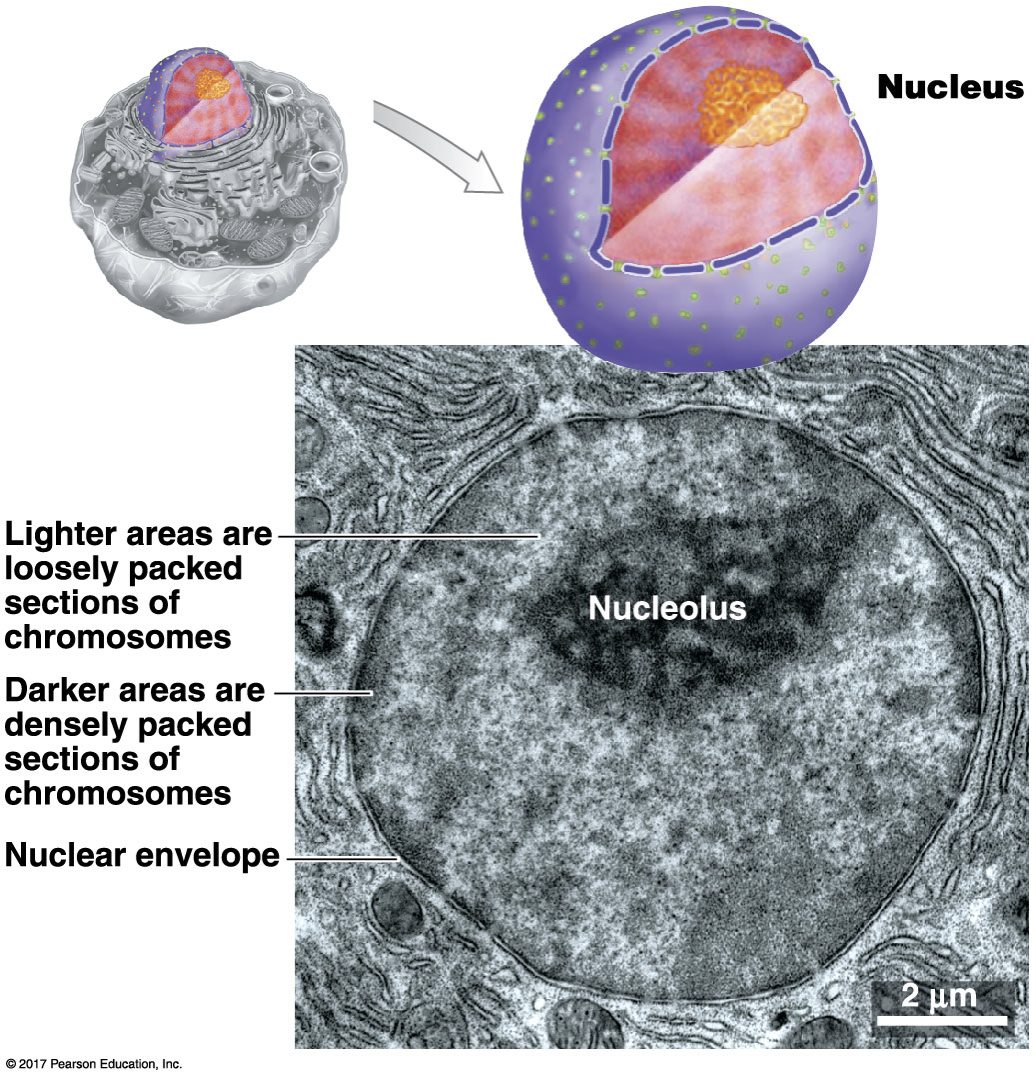
What is the structure and function of a nucleus?
The nucleus stores genetic information in the form of chromosomes. (DNA is protected by proteins called histones.)
Site of transcription (RNA synthesis)
Site of ribosome manufacturing: nucleolus
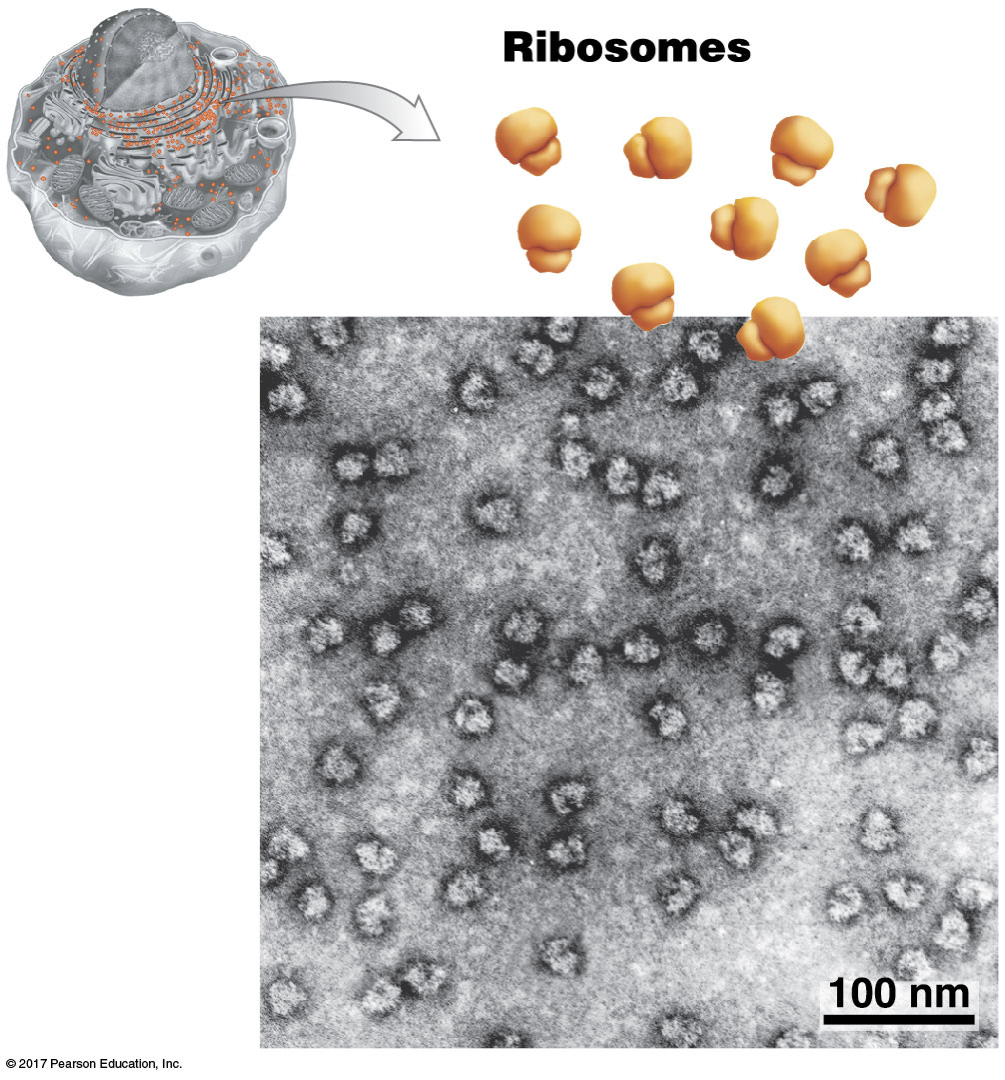
What is the structure and function of ribosomes?
Site of translation (protein synthesis)
Inside of cytoplasm but sometimes attached to rER.
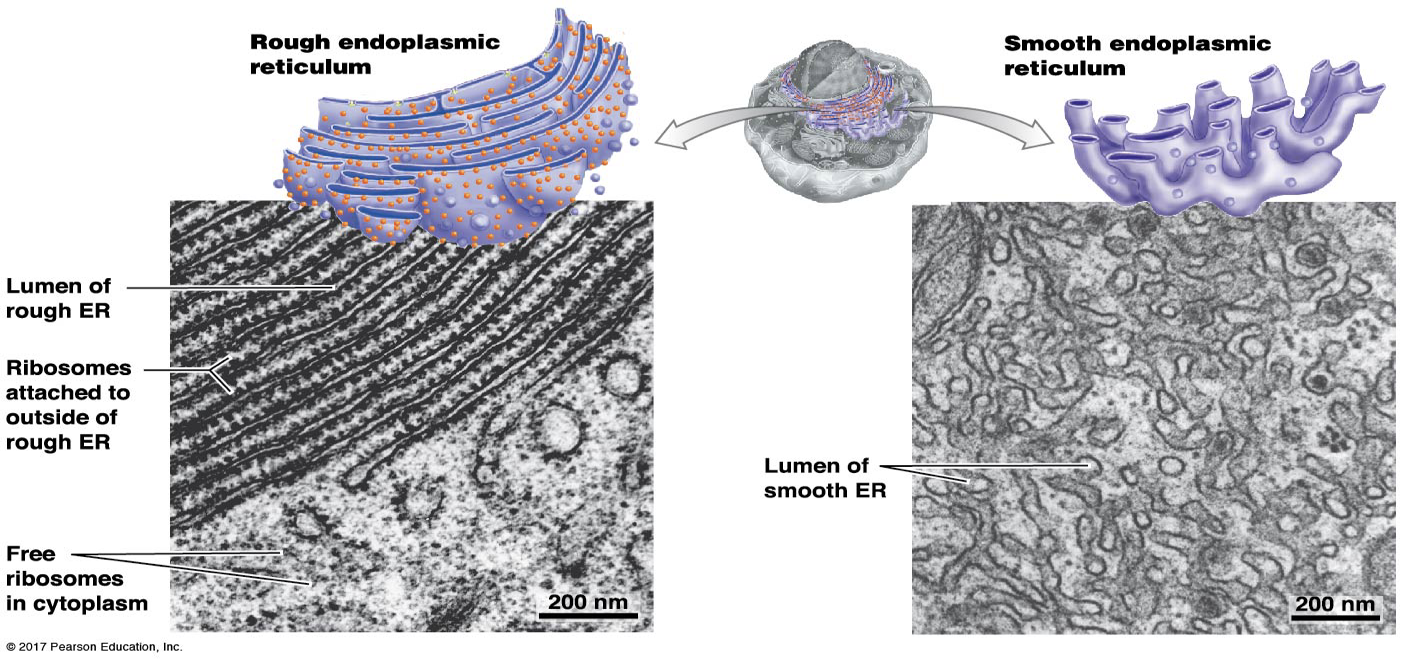
What is the structure and function of the Rough ER?
Site of the synthesis of plasma membrane proteins, lysosomal proteins, and secretory proteins. Packages proteins to go to the Golgi next.
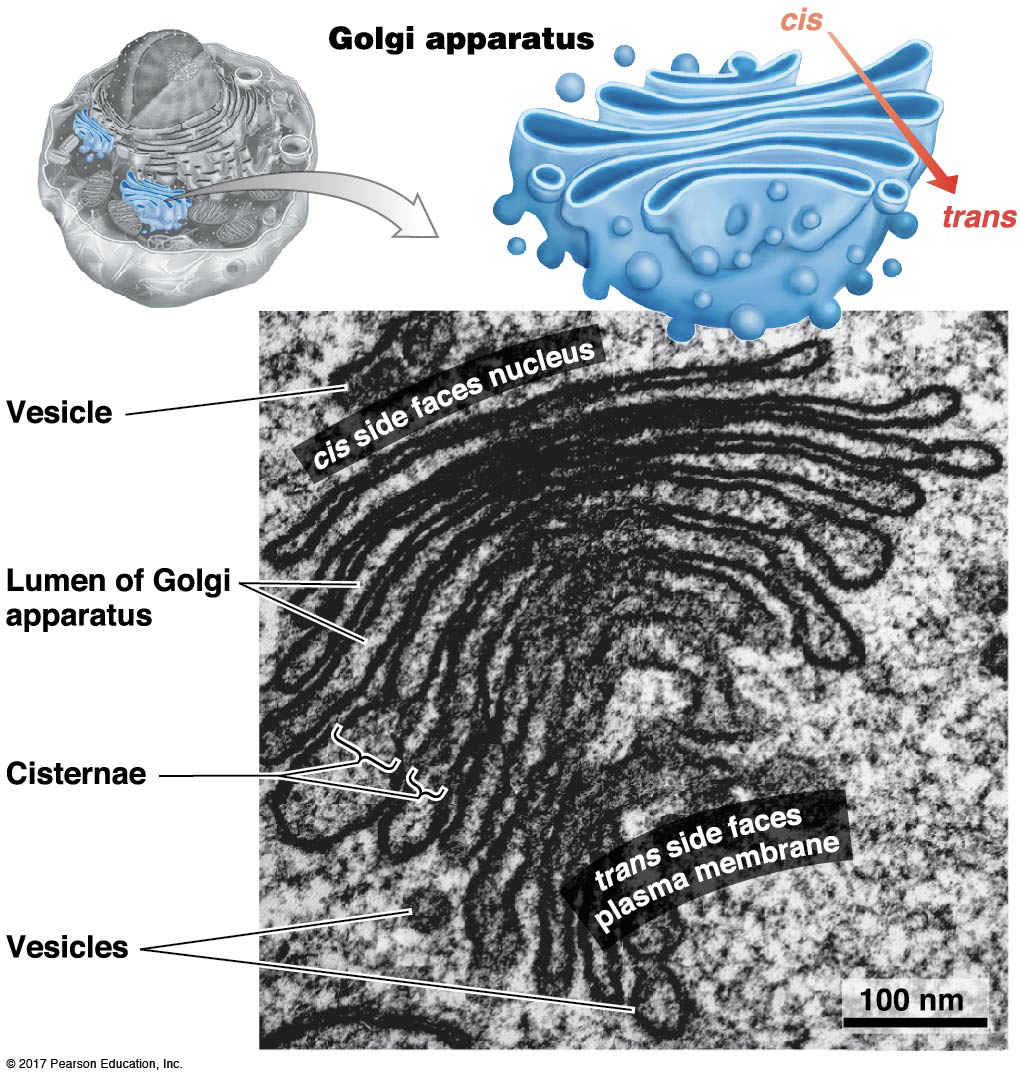
What is the structure and function of the Golgi?
Site of further protein processing (glycosylation: glycoproteins are added)
Site of protein sorting
Ship proteins in vesicles to go to lysosome, vacuole, or plasma membrane.
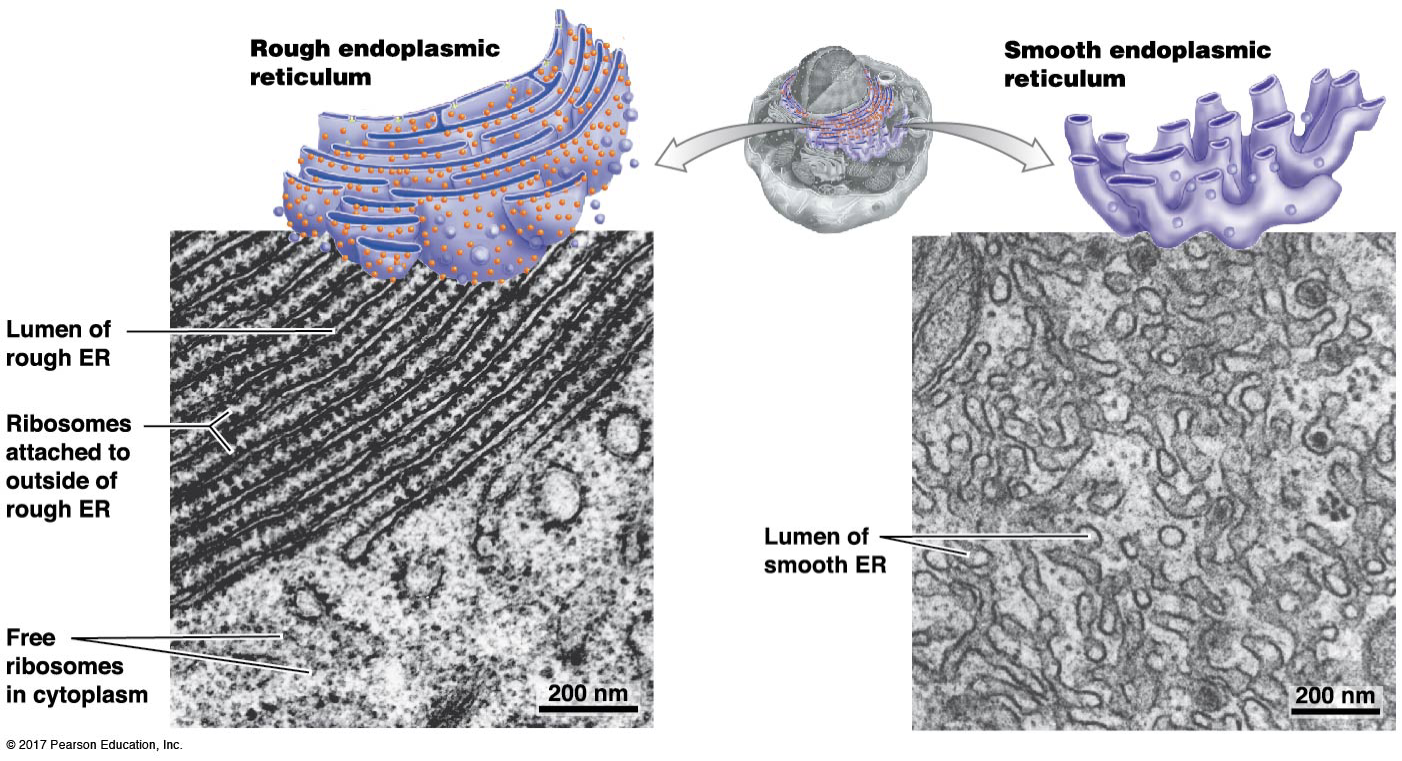
What is the structure and function of the Smooth ER?
Site of synthesis and breaking down lipids (lipid metabolism: making, breaking_
Signal transduction events: Storage and release of Ca2+
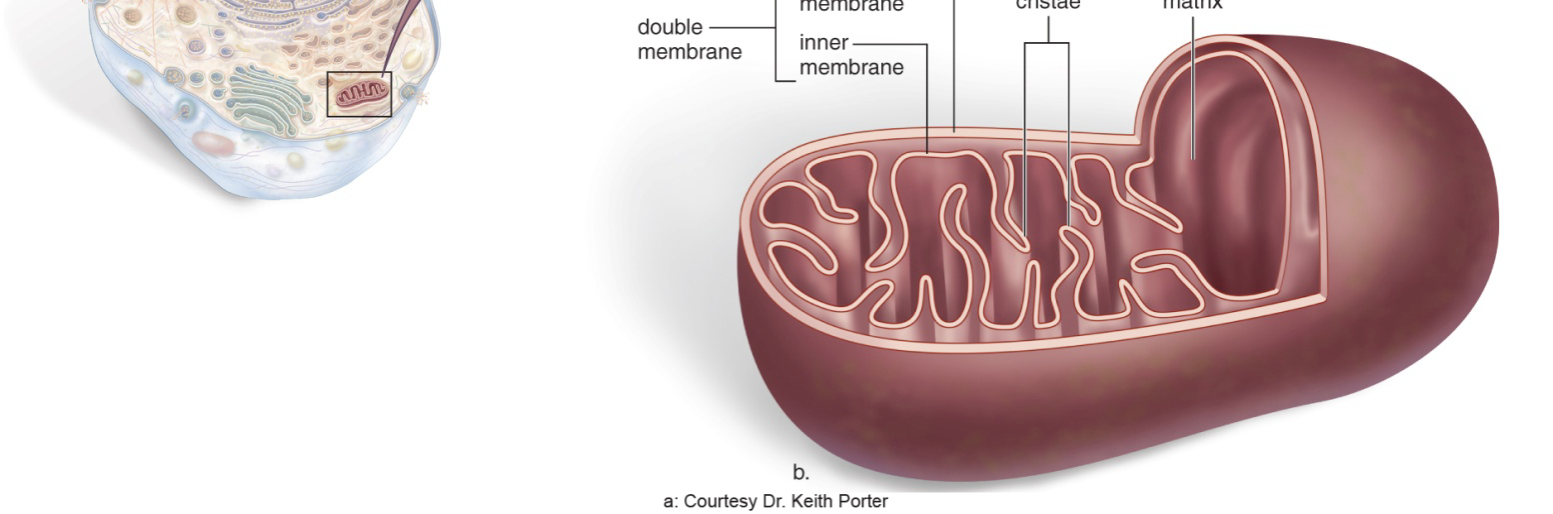
What is the structure and function of the Mitochondria?
Has it’s own DNA
“Powerhouse” generates energy from sugar (Cellular Respiration: Carbohydrate + O2 = CO2 + H2O + ATP)
Double membrane, makes it’s own proteins
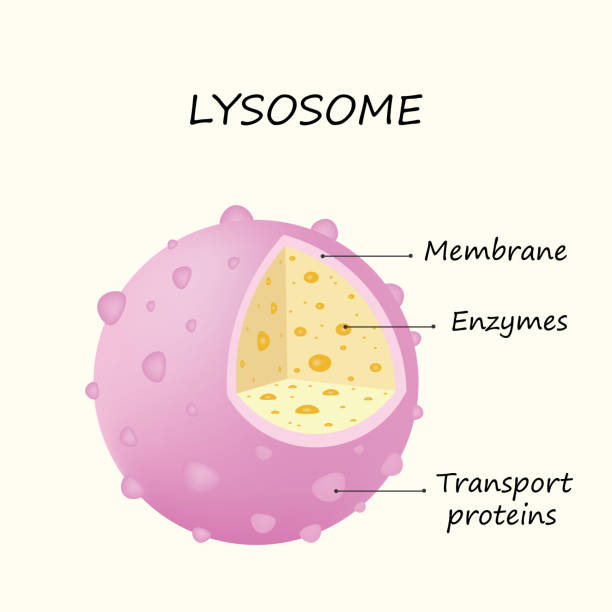
What is the structure and function of Lysosomes? Additionally, what happens when lysosomes malfunction?
Only in animal cells.
Recycling center and digestive system
Has it’s own lysosomal enzymes to aid in that
Breaks down old organelles and molecules
Lysosomal malfunction: Tay Sachs
Accumulation of glycolipids in the brain which leads to premature death
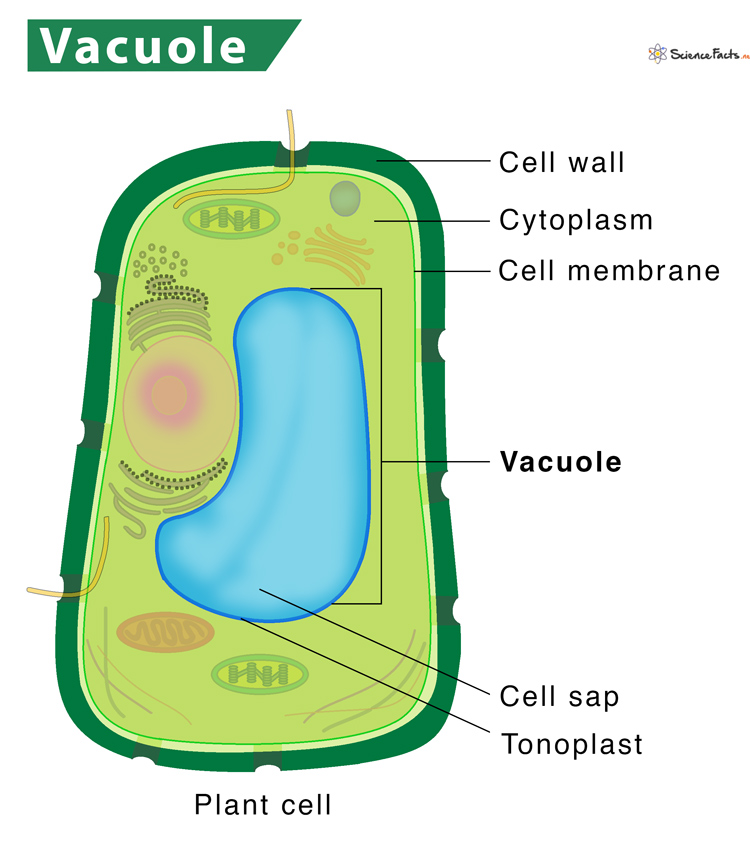
What is the structure and function of Vacuoles?
Only in plant cells.
Like lysosomes but in plants
Storage unit that stores proteins, ions, and water.
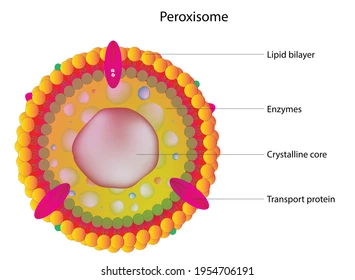
What is the structure and function of Peroxisomes? Additionally, what happens when there is a malfunction in Peroxisomes?
Detox center: contains many enzymes
Oxidize various organic compounds: antioxidants
Catalase converts H2O2 to H2O + O2, hydrogen peroxide is a byproduct of cellular respiration. (idk if we need to know that but that was just in my notes.)
Adrenoleukodystrophy (ALD)
Defect in Peroxisome makes it unable to break down long chain fatty acids. Destroys myelin sheath and causes neurological damage.
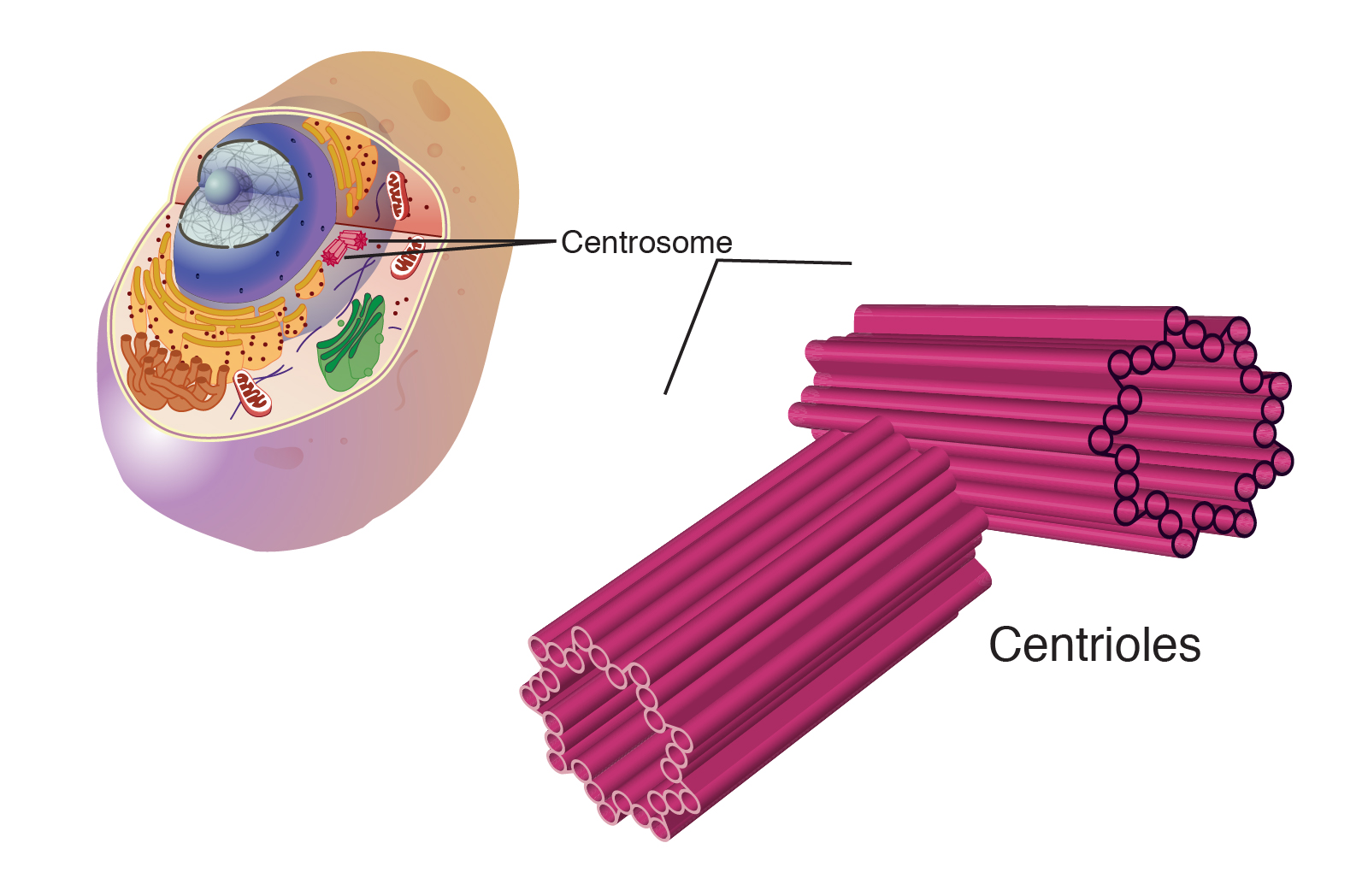
What is the structure and function of Centrioles (Centrosomes)?
Only in animal cells
Only organelle other than ribosomes that are entirely made of proteins and no membrane
Organize microtubules: involved in cell division like mitosis, meiosis
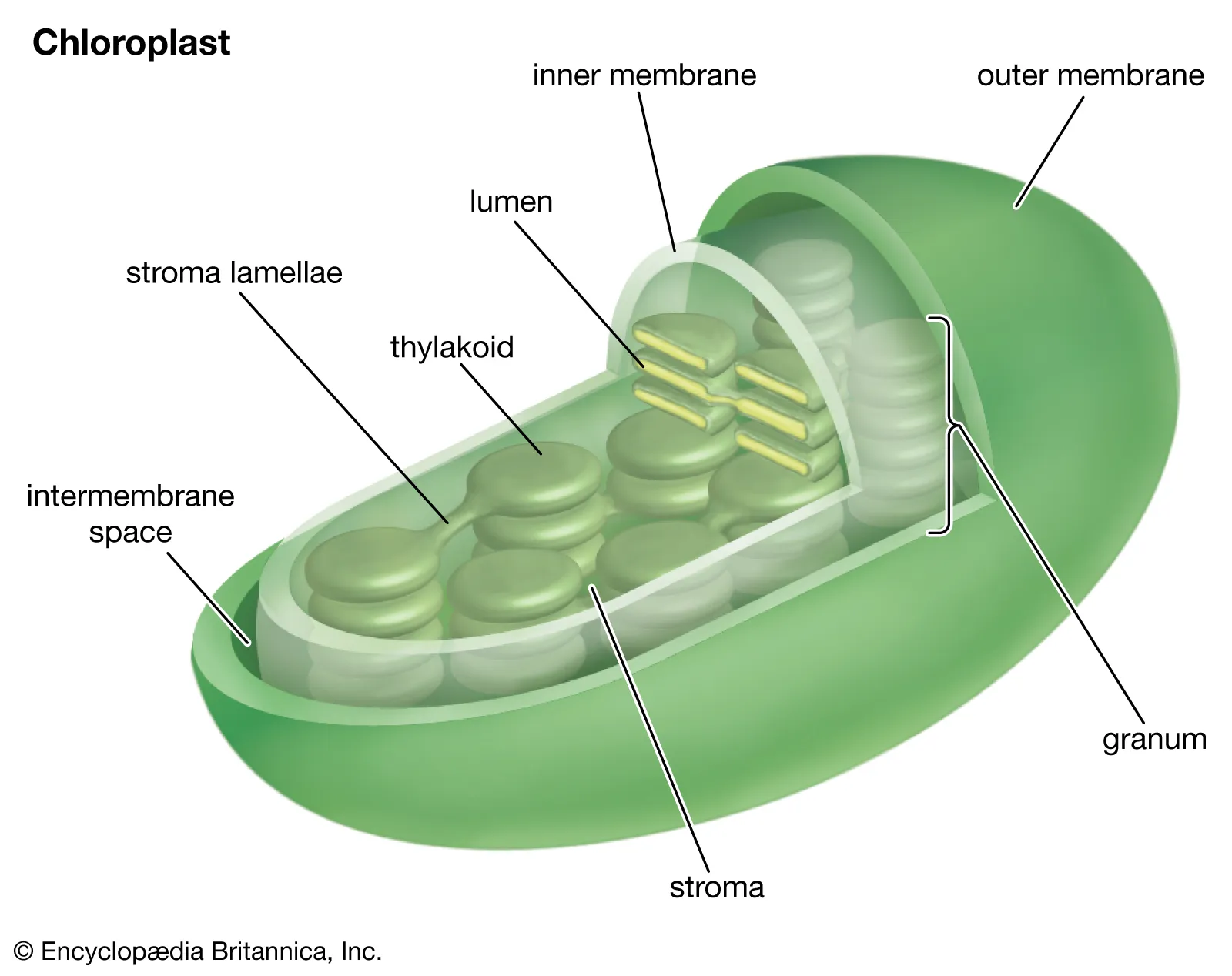
What is the structure and function of Chloroplast?
Only in plant cells
Have their own DNA
Double membrane
Site of photosynthesis
Uses sunlight to convert CO2 into glucose. (6 molecules of CO2 from atmosphere yields 1 glucose)
Which organelles are unique to animal cells and plant cells?
Animals: Lysosomes and Centrioles
Plants: Vacuoles, Chloroplast and Cell Wall
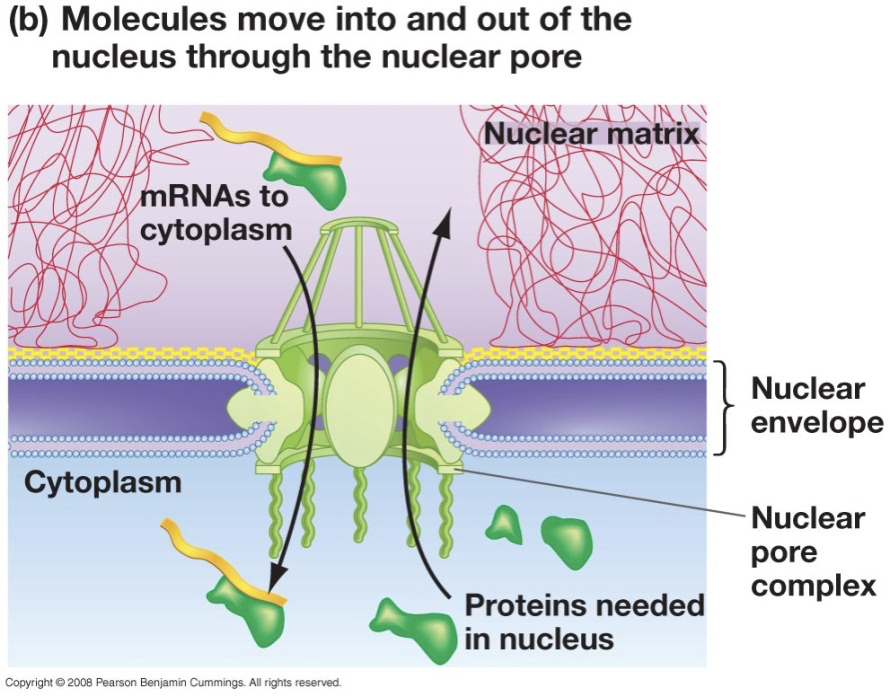
Describe nuclear transport through the pore (Nucleocytoplasmic Transport) What can go in and what can go out?
Import/export of substances between the nucleus and the cytoplasm through nuclear pores.
DNA is not transported. mRNA are exported.
Certain proteins are imported: Ribosomal proteins and transcription factors.
The protein’s sequence determines whether a translated in the cytoplasm or in the rER.
Describe and be able to understand ribosome assembly.
rRNA is made in the nucleolus.
Ribosomal protein is made in the cytoplasm
Ribosomal RNA goes back into nucleus
rRNA + ribosomal proteins are assembled in the nucleolus forms a large subunit a small subunit
Subunits exit nucleus through pores which then enters the cytoplasm
Go to “job site” with signal: goes to rough E.R. without signal: floats
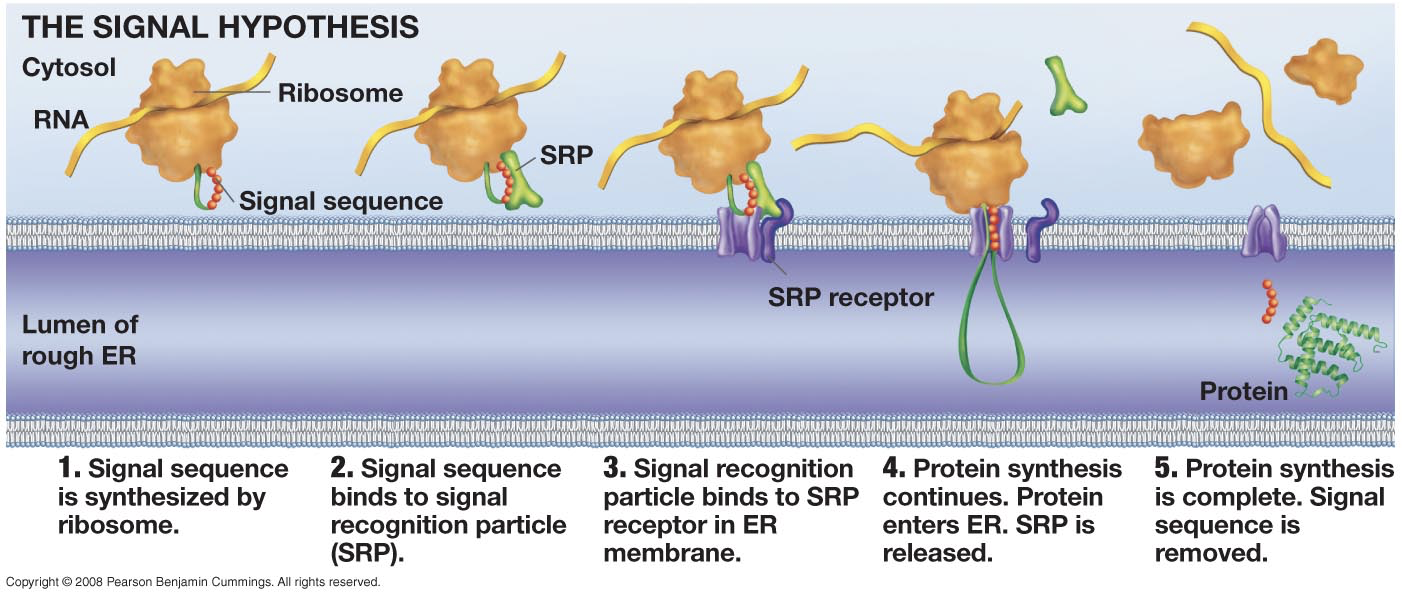
ENDOMEMBRANE SYSTEM
What are the sequence of events in making membrane bound (secreted) proteins?
What determines whether a protein is synthesized at rER?
Sequence of making membrane bound proteins:
mRNA comes into the cytoplasm.
Ribosome binds to mRNA.
Signal peptide is translated
Signal recognition particle (SRP) binds to signal peptide
Ribosome moves to rough ER
Translation of protein completes
Protein ships to Golgi from rER
Protein ships from Golgi to outside of the cell.
The SRP determines whether a protein is synthesized at rER.
What are the different cytoskeleton components and functions?
Microfilaments: Actin, cell contraction, crawling, other movements (muscle contraction)
Intermediate Filaments: Involved in stabilizing and shaping cells
Microtubules:
Mitotic spindles (pulling chromosomes)
“Highway or tracks” in cells for vesicle transport that kinesin can walk on.
Flagella: made of microtubules and dyenein proteins that help with cell movement
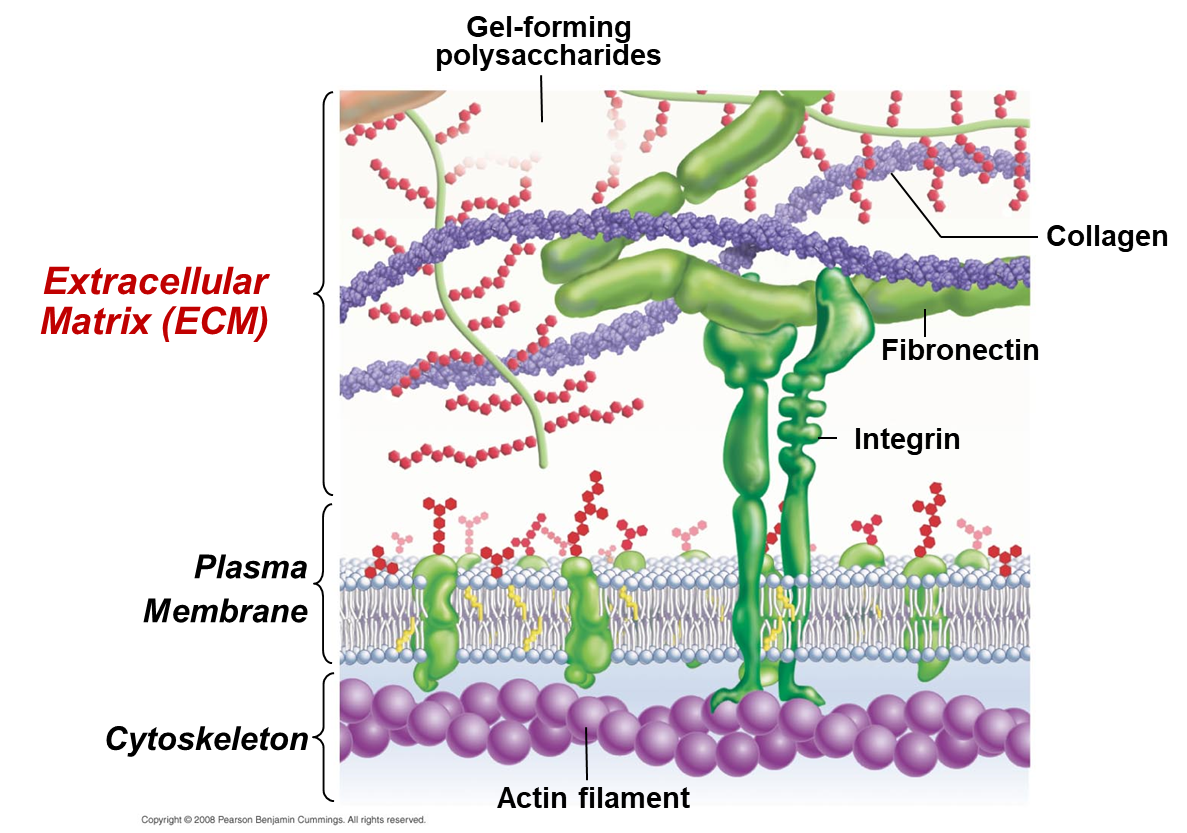
What is the structure and functions and the extracellular matrix?
Only in animal cells.
Functions: Helps to define cell shape, aids in adhesion with other cells and connects to cytoskeleton
Structure: Collagen + Polysaccharides
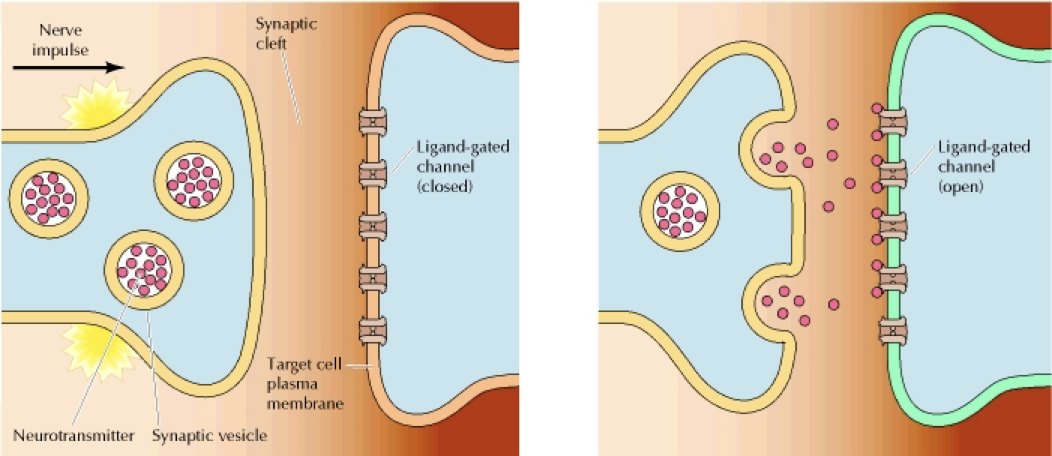
Describe Exocytosis and Endocytosis. How do they work and what are some of their examples?
Exocytosis: Secretion of substances from cell, content of vesicle gets secreted out
Example: Neurotransmitters across a synapse
Build transport mechanism used by immune cells to secret antibody out of the cell.
Endocytosis: Cells bring materials inside the cell
Phagocytosis (large particles, bacteria)
Example: Cell Eating, white blood cell eating bacteria
Receptor-mediated endocytosis (select, small molecules. receptors required)
Example: Familial Hypoercholestrolemia, not having enough LDL receptors.
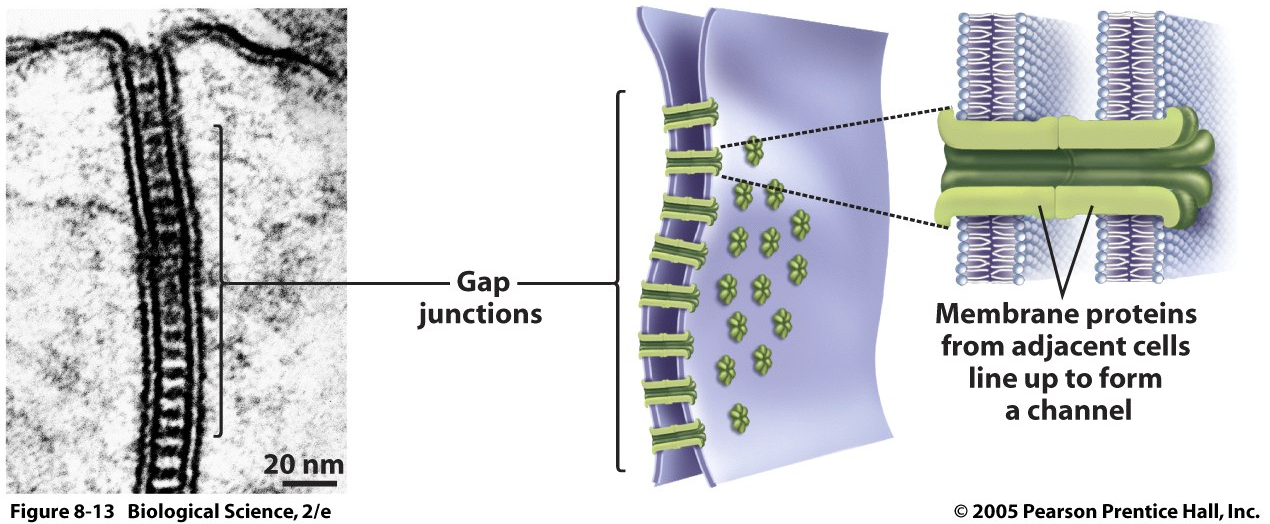
What is the structure and functions of the 3 cell-cell junctions? Provide some examples for what situation and purpose these junctions would serve.
Tight Junctions:
Specialized proteins in plasma membranes of adjacent cells that line up and form a watertight seal. Example: epithelial cells in skin
Adhesion Junctions (eg. Desmosomes)
Join cells from the same tissue. Cell-cell attachments that bind cytoskeletons and composed of membrane proteins that link cells and anchoring proteins. Example: epithelial tissue in muscle
Gap Junctions (eg. Plasmodesmata)
Meant to let things flow between cells, exchange material.
Example: Cell-Cell “tunnels”: direct connections between the cytoplasm of adjacent cells. Enables communication, enables tissue to act in concert
Animal cells: heart tissue, neurons, present in almost all animal tissues.
Plant cells: Plasmodesmata, structures going through cell walls.
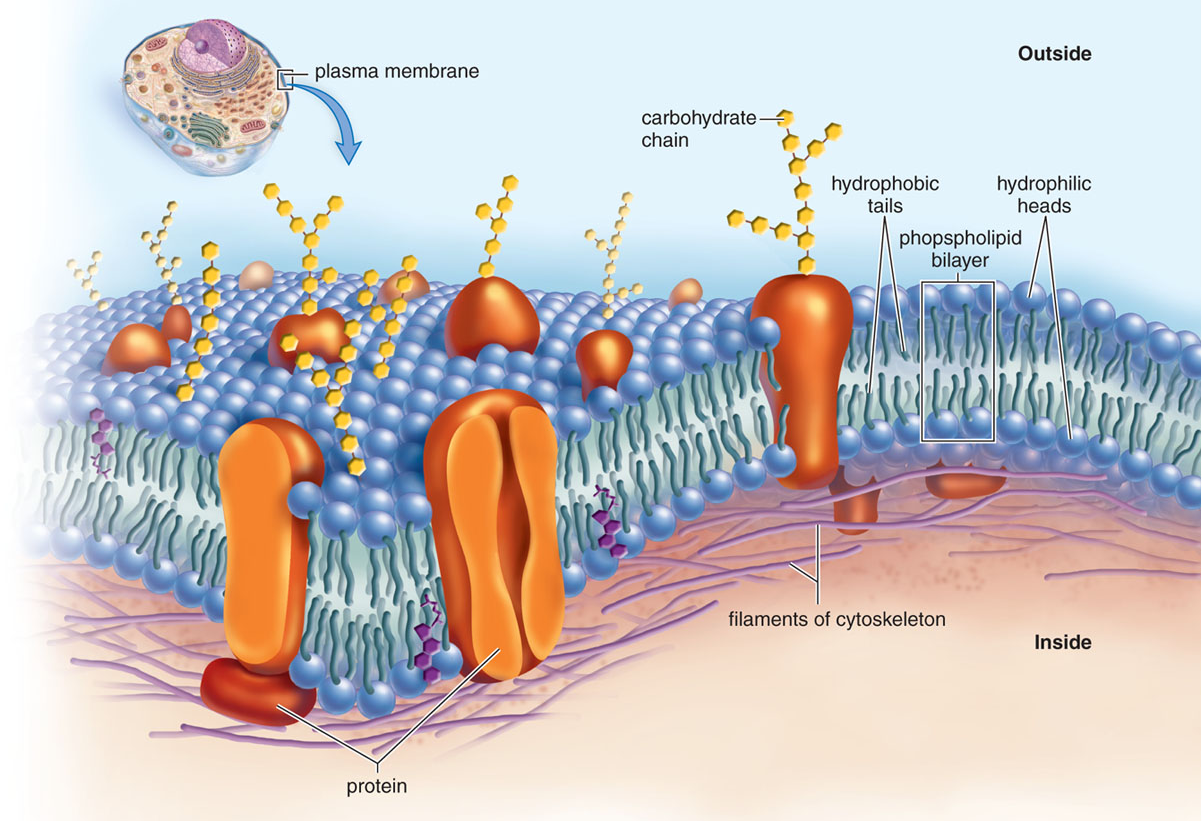
What are the characteristic features of membranes, and what functions do they serve?
Membranes are made of phospholipid bilayers that are selectively permeable.
Phospholipids: Amphipathic: The “head” is polar while the fatty acid chains are nonpolar and hydrophobic.
Cell Membranes also have embedded proteins that help with diffusion.
Example: Integral membrane proteins and peripheral protein.
How are substances transported across a membrane?
Passive Diffusion: High to low concentration
Faciliated Diffusion: Needs channels and carriers, still goes from high to low concentration
Active Transport: Uses pumps through ATP energy which goes against concentration gradient.
What affects high or low permeability? What kinds of molecules would have a high or low permeability?
Many things like temperature, size, polar/nonpolar, cholestrol and saturation affects permeability.
(From high to low): Hydrophobic molecules and small, uncharged polar molecules, large, uncharged polar molecules, and finally ions.
Unsaturated Fatty Acids will have higher permeability because of their “kinked” nature so it makes the membrane more looser.
Higher temperatures will make membranes more permeable.
Higher cholesterol levels make membranes less permeable, will make it harder for things to pass through.
What is passive diffusion and their examples?
Passive diffusion does not require energy and substances will move down with their concentration gradient.
Examples: Gas exchange in lung, oxygen diffusing through cells
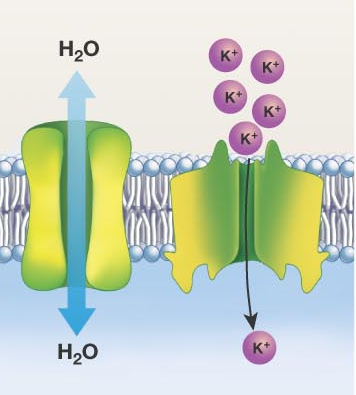
What is Facilitated Diffusion and some examples?
Requires a channel or carrier. It also follows a concentration gradient
Channel Proteins: Allows things to move in/out
Example: Ion Channel (chloride ion channel. mutation is cause of Cystic Fibrosis) and Aquaporins (in kidneys/red blood cells, only allows water in and out)
Carrier Proteins: Only moves out. Highly selective and regulated. It undergoes a conformational (shape) change
Example: Glucose Diffusion (Glucose binds to a carrier protein GLUT, carrier changes shape, then glucose is released, then carrier returns to original shape. Mutation in GLUT-1 deficiency causes developmental delays)
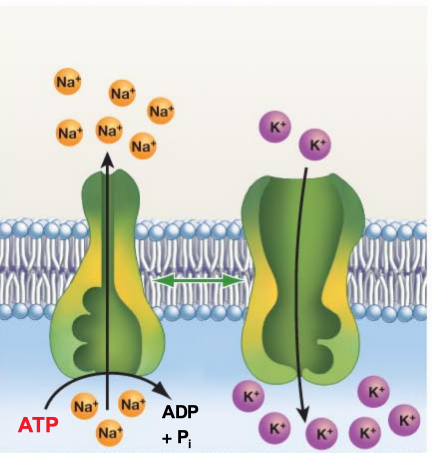
What is Active Transport and some examples?
Solute moves against concentration gradient
Requires ATP and a pump
Example: Sodium Potassium Pump: Important for nerve function, generation of nerve impulse
3 Na+ out and 2 K+ in
What kind of molecules would flow through what membrane? (With channels and pumps)
Able to pass through freely:
Small molecules (H2O, nonpolar molecules)
Unable to pass freely (needs a pump):
Large molecules, polar molecules, charged molecules/ions
What are the cell walls made of in Eukaryotic Cells?
Animals: no cell wall
Plants: cellulose
Fungus: chitin
Protists: Protozoa: none, Algae: cellulose, other polysaccharides
Rough ER protein destinations: (have a signal attached to them)
Cell Surface
Cell Exterior
Lysosome
Vacuole
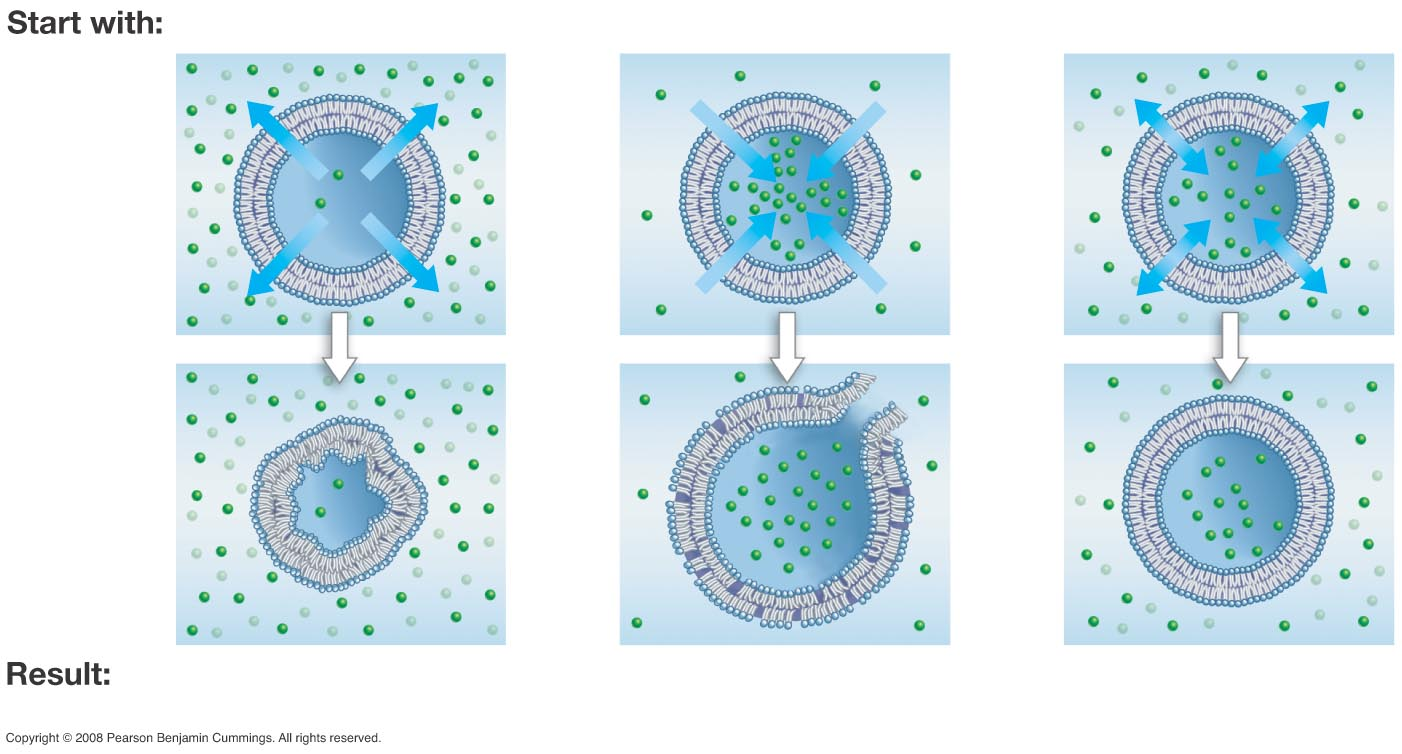
What is Osmosis? What is hypo- hyper- and isotonic solutions?
Omosis: Diffusion of water molecules and water will move against a solute concentration gradient to achieve a more balanced concentration.
This only occurs when solutions are separated by a membrane that is selectively permeable
Hypo: Lower concentration of solute
Hyper: Higher concentration of solutes
Isotonic: Equal concentration of solutes
What is Potential and Kinetic Energy? What are some examples?
Potential: Stored energy
Examples: Chemical energy (bonds. breaking bonds releases energy, forming large molecules takes energy). Lower potential energy = more stable
Kinetic Energy: Things moving
Mechanical motion, light energy (motion of photons), thermal energy (heat) molecular movements
What are the different forms of Energy Conversions? From what form to form?
Concentration Gradient (energy stored in its difference, wants to flow through, flowing through releases energy and takes energy to build up concentration gradient)
ATP breaking down into ADP and Phosphate
What is Entropy, and in what situations does it increase or decrease?
Entropy: Measure of disorder (randomness or evenness) of a system. It naturally wants to increase and spread out.
Higher Entropy: More spread out, breaking things apart
Smaller Entropy: Less evenness, clustered, putting things together
Increases in Spontaneous reactions
-Heat
-pH
What are Endergonic (Non-spontaneous) and Exergonic (Spontaneous) reactions?
Endergonic (Non-Spontaneous) Requires energy, active transport.
Example: Dehydration, Glucose and Glucose to become Maltose
Exergonic (Spontaneous) Does not require energy. Less energy (products) in the products than reactants.
Example: Hydrolysis, ATP to ADP)
What are coupled reactions in energonic and exergonic reactions and oxidation and reduction reactions?
Endergonic and highly exergonic reactions can be coupled to make the overall process exergonic.
Coupling of oxidation and reduction transfers energy (energy conservation)
What are Redox reactions?
OILRIG:
Oxidation is Loss of electrons
Reduction is Gain of electrons
What is the first law of Thermodynamics and some examples?
Energy cannot be created nor destroyed.
Examples: Active transport, photosynthesis
Describe Gibbs Free Energy. What do the different symbols mean, which reactions are they going to be positive or negative in?
Lower potential energy
More entropy
ΔG = ΔH (enthalpy) - T(temp in Kelvin) ΔS (entropy)
Spontaneous Reactions: ΔG will be negative, ΔH will be negative, ΔS will be positive
Non Spontaneous Reactions: ΔG will be positive, ΔH will be positive, ΔS will be negative
On a graph: Spontaneous Reactions will be going down, and non spontaneous reactions will be going up
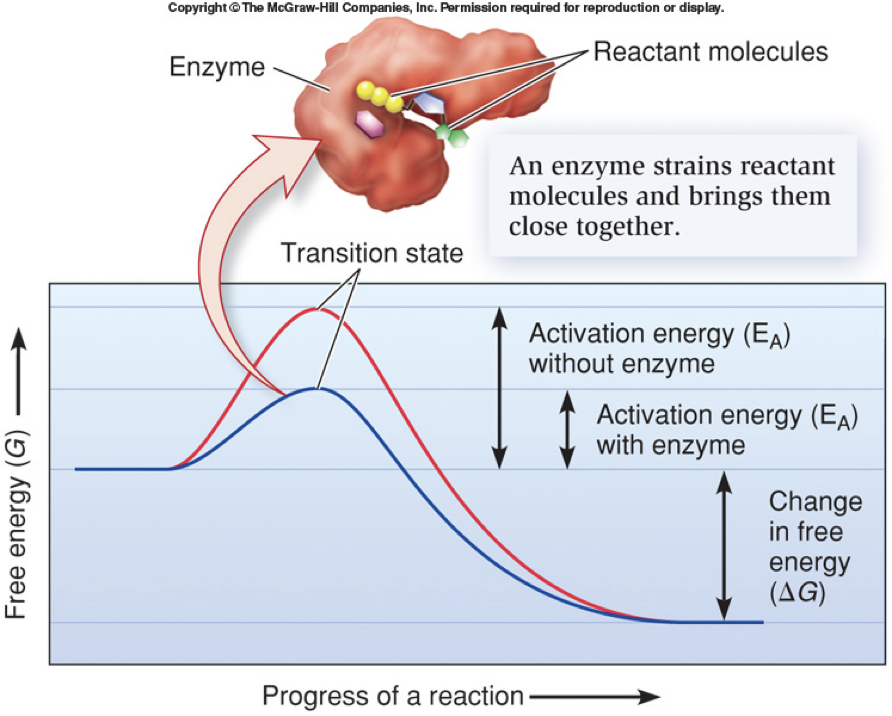
How do Enzymes catalyze reactions?
They bring reactants together in a precise orientation so that “effective collisions” are more likely
Stabilizes the transition state (high energy state halfway between reactants and products) and reduces activation energy
What are some features about Enzymes?
Does not change the nature of a reaction (ΔG)
The lower activation energy makes reactions occur easier and faster.
Are not used up in the reaction (recycled)
Specific for their substrates (reactants)
What affects reaction rate? Why do these things matter?
Substrate concentration
Enzyme concentration
Temperature
pH
Cofactors
Inhibitors/Activators
Temperature and pH matter because some enzymes are adapted to work in those specific conditions like enzymes in our stomach living in an acidic environment. pH only affects ionic bonds. Additionally, temperature will crash due to denaturation of proteins.
What are cofactors and coenzymes?
Cofactors are non-protein molecules that help an enzyme function properly. They provide additional functional groups that amino acids lack.
Some examples are inorganic ions like minerals (aid in the transfer of electrons and oxygen) and then organic molecules which are called Coenzymes. (eg. coenzyme A, NAD+ FAD, vitamins)
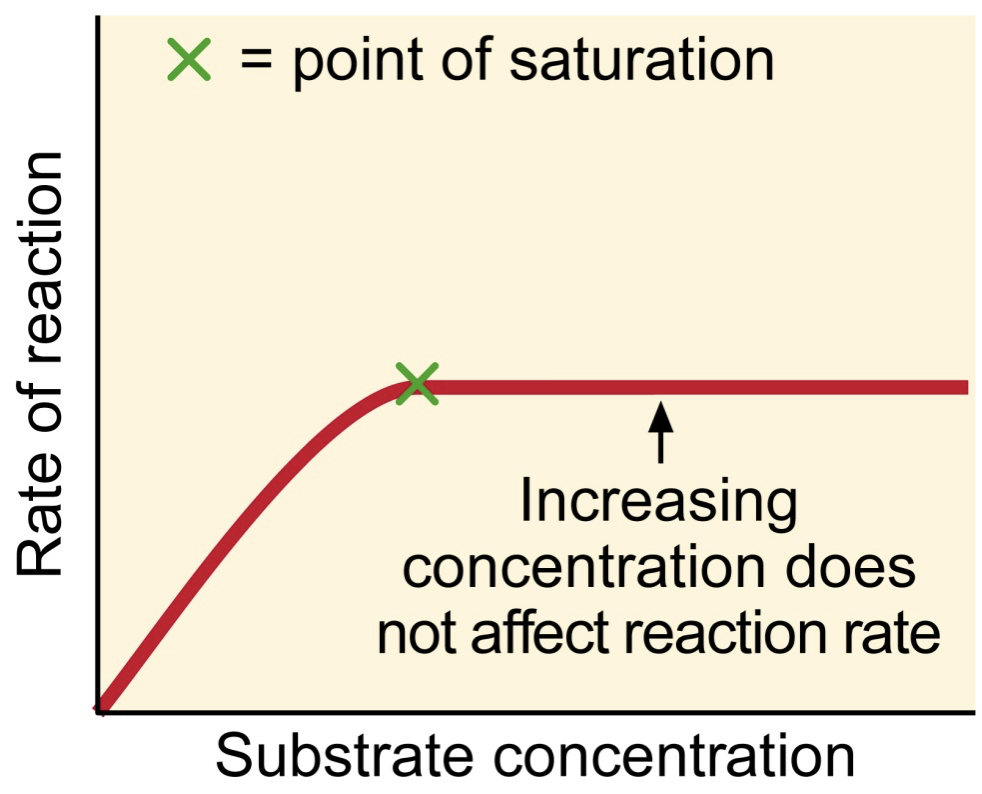
Why does reaction rate plateau with increasing enzyme or substrate concentrations?
Reaction rate is going to plateau because it is going to reach its saturation point. Both enzyme and substrate concentration are going to reach its limit point.
What are competitive and noncompetitive inhibitors? Which requires higher concentration to work?
Competitive: Has an active site and competes with the enzyme so that the substrate cannot bind. It requires a higher concentration of substrate. Looks like substrate (has same shape.)
Noncompetitive: The molecule binds to an allosteric site, which then changes the shape and make the active site unavailable. Because it requires a lower concentration in medications it has fewer side affects.
Example of Noncompetitive Inhibition: Feedback Inhibition
Because the end product of feedback inhibition binds to the allosteric site which changes the shape and therefore the substrate cannot bind to it.
Which ones are reduced and which ones are oxidized, NAD+, FAD, NADH, FADH2?
Reduced: NADH, FADH2
Oxidized: NAD+, FAD
What is the general equation of cellular respiration, and what is being oxidized and reduced?
C6H12O6 + 6O2 —- 6CO2 + 6 H2O + ATP
Oxidized: Glucose
Reduced: Oxygen
*Think OILRIG!
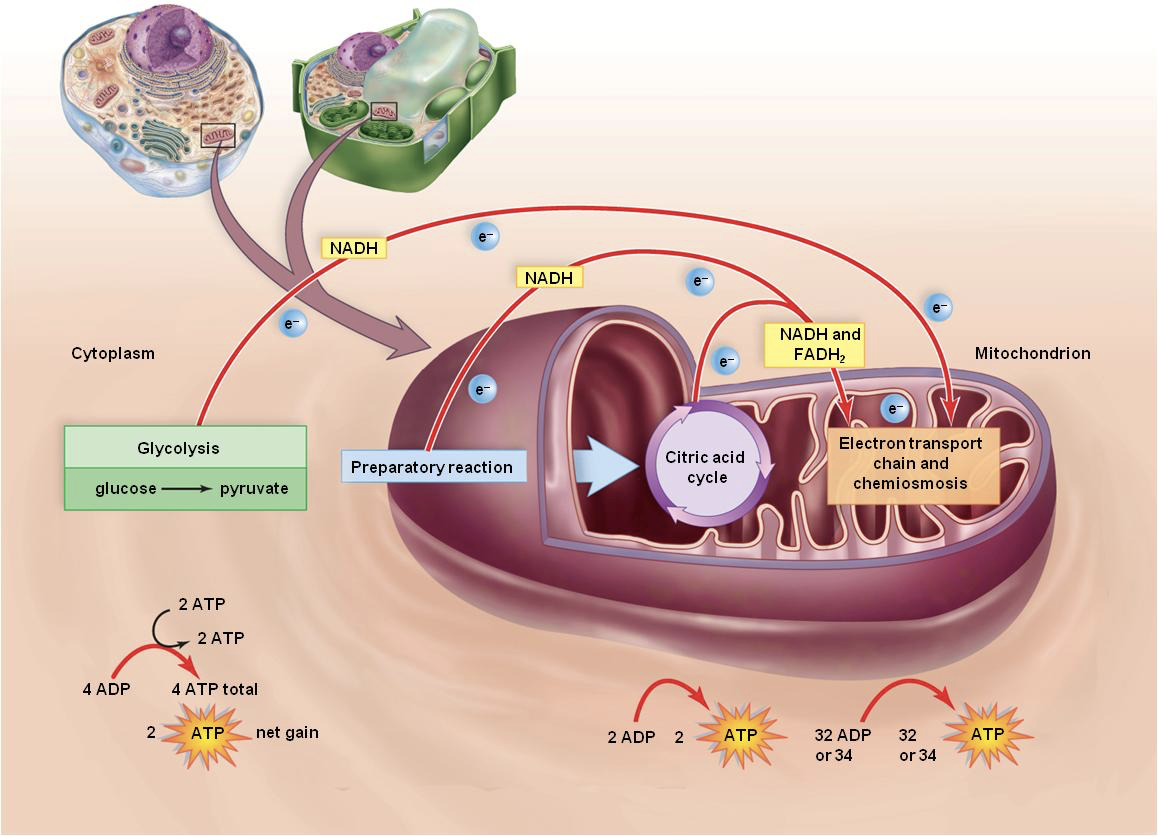
What are the 4 steps in Cellular Respiration?
Glycolysis
Pyruvate Processing
Citric Acid (Krebs) Cycle
Electron Transport Chain + Oxidative Phosphorylation
What comes out of each step in Cellular Respiration? What happens with the carbon containing molecules, electronic carriers, and ATP production?
Glycolysis (6C) —- Pyruvate (3C) — ATP, NADH (CYTOPLASM)
Pyruvate Processing — Acetyl-CoA (2C) —- NADH —- CO2 (2-3 MITOCHONDRIA)
Citric Acid Cycle —- CO2—- NADH, FADH2, ATP
ETC and Oxidative Phosphorylation (ATP synthesis) ATP (MEMBRANE OF MITOCHONDRIA)
In ETC: transfer out e- and pumps out H+
What are the differences of Fermentation and Anaerobic respiration? What is the difference between Lactic Acid Fermentation and Alcohol Fermentation?
Fermentation: Energonic. In Fermentation only glycolysis happens.
Anaerobic Respiration: Exergonic. In Anaerobic Respiration all of the steps happen, except Oxygen is no longer the final electron acceptor.
Fermentation regenerates NAD+ so that glycolysis can continue
Lactic Acid Fermentation: Pyruvate + NADH — lactic acid + NAD
Alchohol Fermentation: Pyruvate —- CO2 + acetaldehyde + NADH — ethanol + NAD+
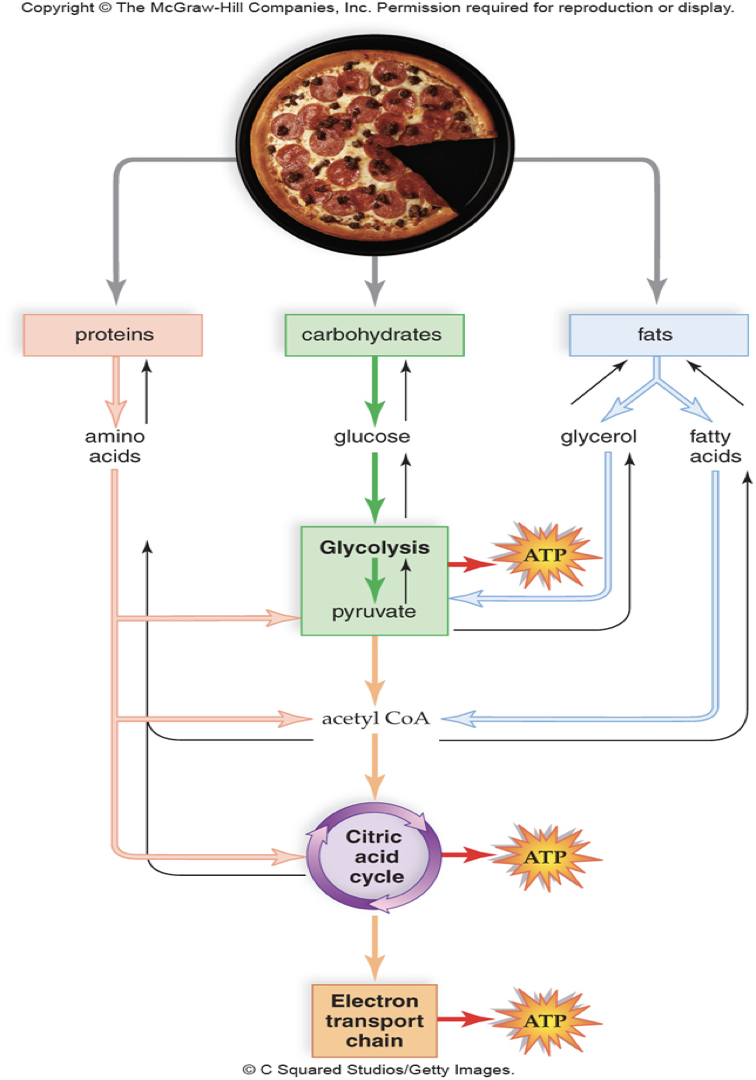
What are Anabolic and Catabolic pathways? How are they coupled, and what are some examples?
*think Ana and Cat
Anabolic: Building things
Catabolic: Breaking things
Metabolism: Catabolism and Anabolism together
Example: In metabolism and how we digest proteins, carbs, and fats to through the pathway to create ATP, but also we take smaller macromolecules to build great molecules.