3.2.2 - group 2
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
why does atomic radius increase down the group
atoms have more shells of electrons
order of elements down grp 2
Be, Mg, Ca, Sr, Ba
trend of melting points down the group and why
decreases
as atomic size increases, metallic bonding weakens
because distance btw positive ions and delocalized e-s increases
so force of attraction btw them weakens
trend in 1st ionization energy down the group and why
Decreases down the group
Bc distance between nucleus and outer electrons successively increases
Also increased shielding of nucelus by inner shells of e-s
So there is a weaker force of attraction between the nucleus and outer e-
So outermost e- = requires less energy to remove
Trend of reactivity down group 2
Increases
REACTIONS OF GROUP 2 METALS WITH OXYGEN - what do they all do in the presence of oxygen
Burn in it
What does Mg burn with
Give an equation for this
What is the product of this rxn (colour and state)
A bright white flame
2Mg + O2 —→ 2MgO
White solid, has a high m.p.
IDK IF YOU NEED THIS BUT
Can Mg react slowly with oxygen without a flame
Equation for this
what will the magnesium ribbon have a thin layer of on it
Yes
2Mg + O2 —→ 2MgO (same as before)
MgO - formed by the rxn w/ oxygen in the air
SAME IDK IF YOU NEED THIS
WHEN TESTING FOR RXN RATES WITH Mg AND ACID
what will give a false resukt
why
give 2 equations to show this
an uncleaned Mg
both the the Mg and MgO would react but at diff rates
1. Mg + 2HCl —→ MgCl2 + H2
2. MgO + 2HCl —→ MgCl2 + H2O
what does Mg react in steam to produce
what does the Mg burn with
equation for this
magnesium oxide and hydrogen
a bright white flame
Mg (s) + H2O (g) —→ MgO (s) + H2 (g) REMEMBER THE RIGHT STATE SYMBOL !!!
what temp of WATER does Mg react with
warm water
what does it react in water to porduce
give an equation for this
is this rxn faster or slower than the steam rxn
is there a flame
magneisum hydroxide
Mg + 2 H2O —→ Mg(OH)2 + H2
slower
no
what temp of water will the other grp 2 metals react w/
they react w/ increasing vigour down the group to form… (type of product)
give all equations for this
cold water
hydroxides
Ca + 2 H2O (l) —→ Ca(OH)2 (aq) + H2 (g)
Sr + 2 H2O (l) —→ Sr(OH)2 (aq) + H2 (g)
Ba + 2 H2O (l) —→ Ba(OH)2 (aq) + H2 (g)
format for equation of grp 2 metals (except Mg) reacting with cold water
metal + 2H2O (l) —→ metal hydroxide (aq) + hydrogen (g)
the hydroxides produced make the water a___________ (if they are soluble in water)
alkaline
observations for this rxn
fizzing, (more vigorous down group)
the metal dissolving, (faster down group)
the solution heating up (more down group)
with Ca, a white ppt appearing (less precipitate forms down group with other metals)
why is titanium a useful metal
it can be used for making strong, light alloys in…
abundant
has a low density
corrosion resistant
aircraft
what is titanium extracted from?
why cant it be exttracted with carbon
why can’t it be extracted by electrolysis
titanium oxide
titanium carbide (TiC) it is formed rather than titanium
it has to be very pure
instead, its extracted by a rxn with ____________
why
magnesium
because Ti it is a highly reactive metal that forms brittle impurities (titanium carbide) if reduced with carbon. Magnesium, being more reactive than carbon, serves as a stronger, more efficient reducing agent to remove chlorine from titanium(IV) chloride
steps in extracting titanium:
1.t___________ o_________ (TiO2) is converted to t___________ c_________ (TiCl4) (liquid) at 900 degrees. c________ m___________ is also produced.
2. The TiCl4 is purified by f___________ d_____________ in an a______ atmosphere.
3. The Ti is extracted by m___________ in an argon atmosphere at 500 degrees. m____________ c__________ is also produced.
1. TiO2 (solid) is converted to TiCl4 (liquid) at 900 degrees. carbon monoxide is also produced
2. The TiCl4 is purified by fractional distillation in an argon atmosphere
3. The Ti is extracted by Mg in an argon atmosphere at 500 degrees C. magnesium chloride is also produced.
equation for step 1
equation for step 3
TiO2 + 2Cl2 + 2C —→ TiCl4 + 2CO
TiCl4 + 2Mg —→ Ti + 2 MgCl2
why is TiO2 converted to TiCl4
it can be purified by fractional distillation as TiCl4 is molecular (which means its liquid at room temp)
rather than ionic like TiO2 (solid at room temp)
why is titanium expensive (even though it is a relatively abundant metal)
1. The expensive cost of the Mg
2. High temps required in both steps
3. This = batch process. makes it expensive bc the process = slower (having to fill up and empty reactors takes time). It also requires more labour + energy is lost when the reactor is cooled down after stopping
4. The process = expensive due to the argon, and the need to remove moisture (because TiCl4 is susceptible to hydrolysis)
trend in solubility of grp 2 hydroxides down the grp
become more soluble
when grp 2 hydroxides are not soluble, what do they appear as
white ppts
magnesium hydroxide is classed as s__________ s___________ in water
what does this mean
Simplest Ionic Equation for formation of Mg(OH)2 (s)?
sparingly soluble
a (solute) that dissolves only slightly in a solvent (e.g. water), often leaving most of the solid undissolved
Mg2+ (aq) + 2OH-(aq) —→ Mg(OH)2 (s)
how do we know the magneisum hydroxide slightly dissolved
A suspension of magnesium hydroxide in water will appear slightly alkaline (pH 9)
so some hydroxide ions must therefore have been produced by a very slight dissolving
NOTE: as Group 2 hydroxides become more soluble (e.g., from to ), they release more OH- ions
use of Mg(OH)2 in medicine
equation for how this works
used in milk of magnesia / indigestion tablets to reduce stomach acid (HCl)
Mg(OH)2 + 2HCl —→ MgCl2 + 2H2O
solubility of calcium hydroxide
what will it appear as
will a suspension of calcium hydroxide appear more or less alkaline than magnesium hydroxide? why?
slightly soluble
a white ppt
more alkaline (pH 11) cause its more soluble
so more OH- ions present in solution
what is an aqueous solution of Ca(OH)2 called
what can it test for
why does limewater turn cloudy
equation for this
lime water
CO2
white calcium carbonate is produced
Ca(OH)2 (aq) + CO2 (g) —→ CaCO3 (s) + H2O(l)
use of Ca(OH)2 in agriculture
found in lime to neutralise acidic soils
solubility of Ba(OH)2
its solubility makes it strongly alkaline. why?
easily dissolves in water
lots of OH- ions present
Ba(OH)2 (S) —→ Ba2+ (aq) + 2OH-(aq)
solubility of grp 2 sulfates down the grp
less soluble
grp 2 hydroxides = more soluble down the grp
grp 2 sulfates = less soluble down the grp
what sulfate is insoluble
BaSO4
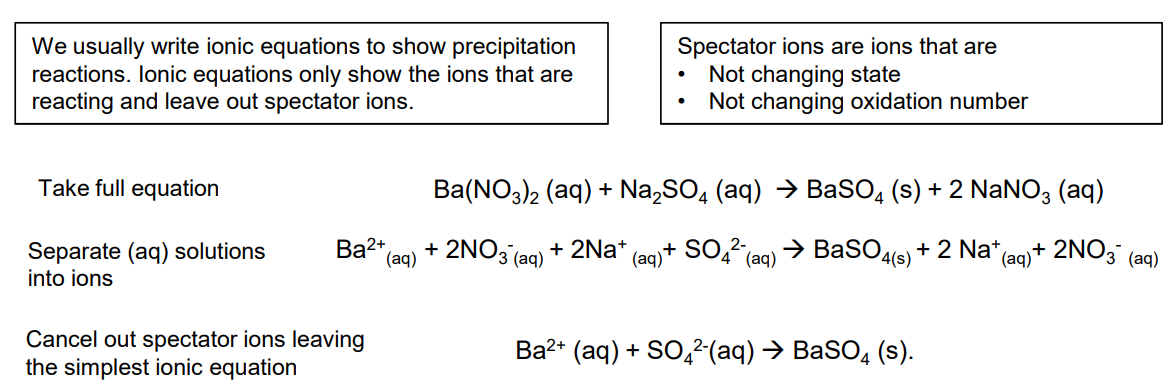
how to write equations for formation of the ppts
Full equation : SrCl2(aq) + Na2SO4 (aq) —→ 2NaCl (aq) + SrSO4 (s)
Ionic equation: Sr2+ (aq) + SO4 2-(aq) —→ SrSO4 (s).
the use of toxic BaSO4 in medicine?
why can it be used despite its toxicity?
used in barium meals to show up the intestines during x-rays. the barium absorbs the x-rays so the gut shows up during the x-ray
its insoluble, so it passes through the gut + not absorbed into the blood
rxn of barium metal with sulfuric acid: fast or slow? why?
equation for this
slow
bc the insoluble barium sulfate produced will cover the surface of the metal and act as a barrier to any further attack
Ba + H2SO4 —→ BaSO4 + H2
as you go UP grp 2 and solubility of sulfates increases, this same effect will happen to a l________ extent
why does this same effect not happen with other acids like hydrochloric or nitric acid
lesser extent
they form soluble grp 2 salts
reagent used to test for presence of sulfate ions
positive test result (contains sulfate ions)
simplest ionic eq. for this
BaCl2 solution acidified with HCl
white ppt forms (this is barium sulfate)
Ba2+ (aq) + SO4 2-(aq) —→ BaSO4 (s)
what is the HCl needed for
why cant we use H2SO4
to react with carbonate impurities that are often found in salts. these would form a white barium carbonate ppt and so give a false positive result
contains sulfate ions and so would give a false positive result.
what two things (?) can be used to remove sulphur dioxide from flue gases in power station?
why is this done
calcium oxide
calcium carbonate
to prevent it escaping into the atmosphere
environmental problem caused by sulphur dioxide
acid rain
what does sulphur dioxide react with to form sulfuric (VI) acid
give an equation for this
water and oxygen
SO2 + H2O + 0.5O2 —→ H2SO4
what does calcium oxide react with sulphur dioxide to form
give an equation for this
what type of reaction is this
calcium sulfate (CaSO3)
CaO + SO2 —→ CaSO3
neutralisation rxn
what does calcium carbonate react with sulphur dioxide to form
give an equation for this
what type of reaction is this
calcium sulfate (but now CaSO4)
CaCO3 + SO2 + 0.5O2 —→ CaSO4 + CO2
neutralisation
REMEMBER: ALL METAL SULFATES (E.G. BARIUM SULFATE) ARE SALTS!!!
common rules for solubility of salts? no syllabus requires these to be learnt but a good chemist does know them.
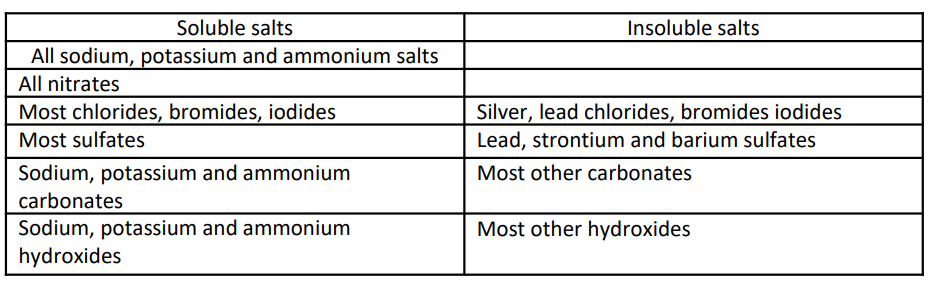
NOT EXACTLY REQ. BUT GOOD TO KNOW
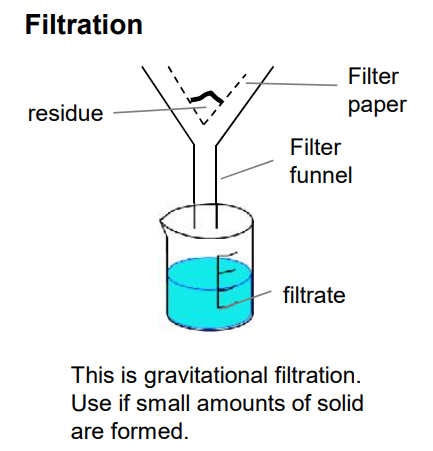
general method to make an insoluble salt
he salt would be removed by filtration, washed with distilled water to remove soluble impurities and then dried on filter paper
how to write ionic eqs. for precipitation rxns