pharmacology of antimicrobial and antifungal agents pt I & II
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
what are the 2 ways to treat an infection?
empiric- based on most likely microbes and their probable susceptibility
documented/definitive- culture of microbes and determination of susceptibility to drugs
prophylactic therapy
to prevent an infection
what are the principles of antimicrobial therapy?
use the most narrow, least toxic, most cost- effective agent
antimicrobials aid the body’s defenses in clearing infection (eliminate the organism by physical means)
what are the 3 factors of antimicrobial selection?
pathogen factors- likely organism, susceptibility
antimicrobial factors- spectrum of activity, PK/PD, adverse effects, drug interactions, cost
host factors- age, immune status, renal and hepatic function, recent antibiotic exposure, allergy or intolerance, disease severity, pregnancy and lactation
pharmacokinetics
describes what the body does to the drug
absorption, distribution, metabolism, elimination
pharmacodynamics
describes what the drug does to the body/micro-organism
describes antimicrobial effect (bactericidal, bacteriostatic), post antibiotic effect (PAE), PD parameters (time dependent activity, concentration dependent activity)
MBC:MIC above 4, requires immune assistance to clear infection
bacteriostatic
MBC:MIC below 4, cell wall synthesis inhibitors
bacteriocidal
what are the pharmacodynamics of antimicrobials?
aminoglycosides- Cmax:MIC aka concentration dependent
vancomycin and fluoroquinolones- AUC>MIC time and concentration dependent
beta lactams- T>MIC aka time dependent
when a drug is concentration dependent, should it be given in smaller quantities more frequently or larger quantities less frequently?
larger quantities less frequently
when a drug is time dependent, should it be given in smaller quantities more frequently or larger quantities less frequently?
smaller quantities more frequently
cell wall synthesis inhibitors
penicillins
beta lactamase inhibitor combinations
cephalosporins
carbapenems
vancomycin
what is the mechanism of action of beta lactams?
inhibits pencillin binding proteins (PBPs)- involved in the cross linkage of peptide chains, prevents the development of normal peptidoglycan structure
cell lysis- osmostic pressure or activation of endogenous autolysins
what is the basic structure of beta lactams
manipulations of side chains alter spectrum, susceptibility, and pharmacokinetic properties
rapidly bactericidal, nontoxic, bioavailable, resistant to degradation, high affinity for PBPs
time dependent activity
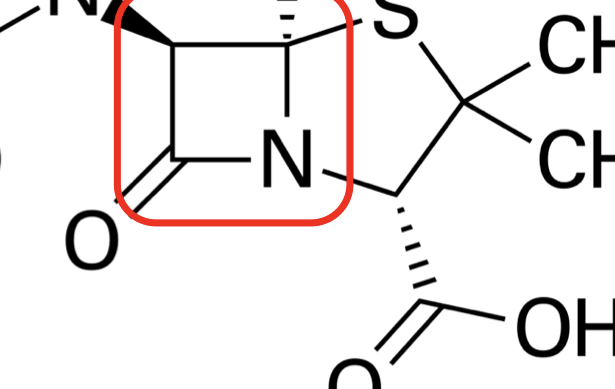
from most narrow to broad, what is the range of penicillins?
penicillin VK, penicillinase resistant PCNs, amoxicillin, amoxicillin/clavulanic acid, carboxyPCNs, ureidoPCNs
what bacteria are susceptible to penicillin VK?
streptoccoci, h. influenzae, G+ anaerobes
what bacteria are susceptible to penicillinase resistant PCNs?
MSSA, streptococci, G+ anaerobes
what bacteria are susceptible to amoxicillin?
streptococci, h. influenzae, enteric G-, G+ anaerobes
what bacteria are susceptible to amoxicillin/clavulanic acid?
MSSA, streptococci, h. influenzae, enteric G-, anaerobes
what bacteria are susceptible to carboxyPCNs / ureidoPCNs?
IV only
MSSA, streptococci, h. influenzae, enteric G-, G+ anaerobes
what are the pharmacokinetics of penicillin and amoxicillin?
absorption- difficult to achieve high serum concentrations with oral formulatons
decent penetration into most tissues except prostate, eye, and uninflamed CNS
primarily excreted renally- adjust dosing with moderate-severe renal impairment
in general, shorter half life than most antimicrobials (amox > peni)
probenecid blocks renal excretion and causes increased serum penicillin levels
what are the adverse effects of penicillins?
hypersensitivity (~1%)- anaphylaxis (0.004-0.015%), cross reactivity with cephalosporins ~2%
phlebitis (1-10% when given IV)
GI disturbances (1-10%)
hematologic (<1%)
neurologic (<1%)
electrolyte disturbances (<1%)
renal (interstitial nephritis) (<1%)
prophylaxis is recommended for dental procedures that involve… ONLY for patients with…
manipulation of gingival tissues or periapical region of teeth or perforation of oral mucosa
underlying cardiac conditions associated with the highest risk of adverse outcome from infective endocarditis
what underlying cardiac conditions are associated with the highest risk of infective endocarditis from dental procedures?
prosthetic cardiac valves, including transcatheter implanted prostheses and homographs
prosthetic material used for cardiac valve repair (annuloplasty rins and chords)
prior history of infective endocarditis
congenital heart disease (CHD)- unrepaired cyanotic CHD, completely repaired but within 6 months of the procedure, or repaired CHD with residual defects
cardiac transplantation recipients who develop cardiac valvulopathy
according to the ADA… current evidence suggests that _____ and ______, NOT _____ are likely to be responsible for the vast majority of cases of IE that originate in the mouth
poor oral hygiene, periodontal disease
dental office procedures
when prophy-ing for IE, what bacteria are we most worried about?
viridans group streptococci
what is the recommended regimen for IE prophy?
oral is 1 hr prior to procedure, IV or IM is 30 min prior to procedure
2g amoxicillin PO
is prophy-ing recommended for patients with joint implants?
according to the ADA, no
according to AAOS, in select circumstances
intrinsic antimicrobial resistance
characteristics of the drug prevent an antimicrobial from reaching the target site, or the antimicrobial is inactivated
acquired antimicrobial resistance
resistance which develops horizontally or vertically
what are mechanisms of antimicrobial resistance?
drug cannot reach the target- efflux pumps, porin channel mutations
drug is inactivated- enzymatic inactivation
target site is altered- modification/protection of target sites
t/f drugs are inactivated by beta lactamases
true-ish? not all drugs, penicillins
clavulanic acid
compounded with amoxicillin
binds to beta lactamase, preventing it from breaking down the beta lactam
enhances the spectrum of the parent drug if decreased activity is a result of beta lactamases
t/f the lower MIC number, the better
true!
from most narrow to broad, what is the range of cephalosporins?
1st gen cephalexin, 2nd gen, 2nd gen cephamycins, 3rd gen cefpodoxime, 4th gen
what bacteria are susceptible to 1st gen cephalexin?
MSSA, streptococci, enteric G-
what bacteria are susceptible to 2nd gen cephalosporins?
MSSA, streptococci, h. influenzae, enteric G-
what bacteria are susceptible to 2nd gen cephamycins?
MSSA, streptococci, h. influenzae, enteric G-, anaerobes
what bacteria are susceptible to 3rd gen cefpodoxime?
MSSA, streptococci, h. influenzae, enteric G-
what bacteria are susceptible to 4th gen cephalosporins?
MSSA, streptococci, h. influenzae, enteric G-
has best G- coverage
what are the pharmacokinetics of cephalosporins?
absorption- IV for severe infections, PO for mild/moderate infections
distribution- similar to penicillins
excretion- primarily renal
what are the adverse effects of cephalosporins?
generally well tolerated
GI disturbances (1-10%), phlebitis (1-10% when given IV), hypersensitivity (<1%), hematologic (<1%), renal (<1%), neurotoxicity (<1%)
how is resistance to beta lactam antibiotics conferred?
drug is inactivated- beta lactamases, penicillinases, cephalosporinases, extended spectrum beta lactamases (ESBLs)
drugs cannot reach the target site- alterations in membrane permeability or efflux
target site is altered- alterations in PBP (PRSP and MRSA)
carbapenems
meropenem, ertapenem, imipenem-cilastatin
broad spectrum agents- reserved for last line treatment of resistant (primarily G-) organisms but have activity against G+, G-, and anaerobic bacteria
what are the pharmacokinetics of carbapenems?
IV only, decent distribution, renally excreted
what are the adverse effects of carbapenems?
hypersensitivity, seizures (caution in patients with seizure history or renal disease)
which of the following is true of beta-lactam antimicrobials?
a) common mechanisms of resistance include enzymatic and activation, and altered target sites
b) intravenous and oral formulas are equally bioavailable
c) responsible for many drug-drug interactions due to inhibition of CYP 450, isoenzyme 3A4
d) primarily bacteriostatic
e) anti-bacterial activity is concentration dependent (higher peaks have greater antibacterial activity)
a) common mechanisms of resistance include enzymatic and activation, and altered target sites
what is the spectrum of vancomycin?
G+ anaerobes and aerobes, including MRSA
NO G- activity because the molecule is too large to cross the outer membrane in G- bacteria
what is the mechanism of action of vancomycin?
inhibits glycopeptide cell wall synthesis, acts at a different (earlier) site than penicillin
bactericidal for s. aureus
AUC dependent activity
what are the uses of vancomycin?
G+ organisms resistant to beta lactams, G+ organisms in patients unable to tolerate beta lactams, C. difficile colitis
what are the pharmacokinetics of vancomycin?
poor oral absorption, oral only for C. difficile colitis
widely distributed (outside of uninflamed CNS)
minimal metabolism; renal excretion
monitor trough/AUC concentrations if course is planned for >3 days
what are the adverse effects of vancomycin?
infusion related effects (>10%)- slow infusion rate, nephrotoxicity (>10%), hypersensitivity (1-10%), ototoxicity (<1%), hematologic (<1%)
when did the first vancomycin resistant enterococci (VRE) appear?
1980s
we got over 30 years of clinical use without resistance
when was VISA/VRSA first reported?
1996
what is the mechanism of action of macrolides (azithromycin)?
reversibly binds 50S ribosomal subunit, suppressing RNA dependent protein synthesis
bacteriostatic
what is the spectrum of activity of macrolides (azithromycin)?
G+ organisms, a few G- organisms (h. influenzae, m. catarrhalis), atypicals (l. pneumophila, m. pneumoniae, c. pneumoniae)
what are the pharmacokinetics of azithromycin?
decent oral absorption
extensive distribution (except CNS), intracellular, concentrates in tissues, low serum levels
hepatically metabolized
elimination in the bile, minimal renal excretion, long half life
what are the drug interactions with azithromycin?
CYP3A4 substrate and inhibitor (erythro and clarithro »»» azithro)
increases levels of: amioderone, digoxin, warfarin, statins, calcium channel blockers, carbamazepine, and others
QTc prolonging agents
what are the adverse effects of azithromycin?
primarily GI disturbances (up to 30% of pts), QTc prolongation
what types of resistance to azithromycin can develop?
drug cannot reach target site- decreased penetration into the cell or pumped out of the cell through efflux pumps
drug is inactivated through hydrolysis
target site is altered- increased enzyme production, modifies ribosomal target
what is the mechanism of action of tetracyclines (doxycycline)?
reversibly binds 30S ribosomal subunit
bacteriostatic, time-dependent killing
what is the spectrum of activity of tetracyclines (doxycycline)?
G+ organisms, G- organisms, anaerobes, atypicals (l. pneumophila, m. pneumoniae, c. pneumoniae), lyme disease (b. burgdorferi)
what are the pharmacokinetics of tetracyclines (doxycycline)?
decent oral absorption- binds Ca, Mg, Al, Fe (dairy products, antacids) causing decreased absorption when administered together
widely distributed (high GI concentrations)
partial liver metabolism
elimination in bile and urine (1/2 life prolonged in renal dysfunction, but no need for dose adjustment)
what are the adverse effects of tetracyclines (doxycycline)?
GI disturbances, photosensitivity, tooth/bone discolortation, hepatotoxicity (rare), renal toxicity (rare)
how can resistance be developed to tetracyclines (doxycycline)?
drug cannot reach the target site- active efflus is most common
drug is inactivated- enzymatic inactivation
target site is altered- ribosomal protection proteins
what is the mechanism of action of clindamycin?
binds to 50S ribosomal subunit (similar to macrolides); time dependent killing
what is the spectrum of activity of clindamycin?
G+ aerobes and anaerobes but never first line
what are the uses of clindamycin?
respiratory, pelvic/abdominal, dental infections, third line alternative for pts truly allergic to penicillin
what are the pharmacokinetics of clindamycin?
great oral absorption, widely distributed (except CNS), primarily metabolized by the liver, excreted in bile and urine
what are the adverse effects of clindamycin?
GI disturbances, C. difficile colitis (which can be worse than the initial infection), metallic taste
what is the mechanism of action of aminoglycosides?
irreversibly binds to 30S ribosomal subunit, inhibiting protein synthesis
bactericidal
concentration-dependent killing
what is the spectrum of activity of aminoglycosides?
G-, synergy / limited G+
post antibiotic effect (PAE)
persistent suppression of bacterial growth following exposure to an antimicrobial OR the time it takes an organism to recover from the effects of antimicrobial exposure
what are the pharmacokinetics of aminoglycosides?
poor oral absorption (IV only)
distribution into extracellular space- not used as monotherapy for infections outside of the urine
renally eliminated
how is resistance to aminoglycosides conferred?
impaired drug uptake (usually intrinsic)
altered target site: ribosomal mutations (usually acquired)
enzymatic modification of aminoglycoside (usually acquired)
what are the adverse effects of aminoglycosides?
nephrotoxicity (variable but generally >10% reported)
ototoxicity (variable, but generally >1% reported)
neuromuscular blockade (<1%)
requires routine monitoring of serum concentrations for efficacy and toxicity
t/f cell wall synthesis inhibitor in combination with aminoglycosides…
increases bacterial killing
what are the uses of aminoglycoside combination therapy?
definitive for select endocarditis/endovascular G+ infections
empiric to improve activity against G- organisms
what are the protein synthesis inhibitor antibiotics?
macrolides (azithromycin), tetracyclines (doxycycline), clindamycin, aminoglycosides
which of the following combinations are most likely to be synergistic if used together?
a) cefazolin and ceftriaxone
b) doxycycline and clindamycin
c) penicillin and gentamicin
d) clindamycin and azithromycin
c) penicillin and gentamicin
what is the mechanism of action of quniolones (ciprofloxacin)?
inhibit topoisomerases II (DNA gyrase) and IV (both enzymes responsible for maintaining DNA supercoiling, replication, and separation)
inhibition of these enzymes subsequently arrests DNA replication
bactericidal, concentration dependent
what is the spectrum of activity of quniolones (ciprofloxacin)?
limited G+ coverage, primarily G- including pseudomonas, atypicals
what is the difference in spectrum of activity of ciprofloxacin and levofloxacin?
ciprofloxacin- nosocomial G- are susceptible, but MSSA and streptococcus pneumoniae are not
levofloxacin- nosocomial G-, MSSA, and streptococcus pneumoniae are susceptible
what are the pharmacokinetics of quniolones (ciprofloxacin)?
excellent oral absorption (decreased by Al, Mg, Fe, Ca / food delays peak concentration)
widely distributed
minimal metabolism
renal elimination
what are the drug interactions of quniolones (ciprofloxacin)?
divalent and trivalent cations
ciprofloxacin inhibits CYP1A2 (strong), CYP3A1 (weak)
increases levels of corticosteroids and warfarin
what are the adverse effects of quniolones (ciprofloxacin)?
GI disturbances (<5%), photosensitivity, QTc prolongation, C. difficile colitis, tendon rupture, dysglycemias
how can resistance to quniolones (ciprofloxacin) develop?
drug cannot reach target site- efflux pumps
target site is altered
increasing resistance with widespread use
what warnings did the FDA communicate about quinolones?
tendinitis and tendon rupture, worsening myasthenia gravis, peripheral neuropathy, joint pain and CNS issues, hypoglycemic risk, increased risk of ruptures or tears in aorta
what is the mechanism of action of metronidazole?
enters cell and reduced to cytotoxic produce, damages DNA, RNA, and proteins
bactericidal, concentration dependent killing
what is the spectrum of activity of metronidazole?
anaerobes
what are the uses of metronidazole?
first line for anaerobic infections
C. difficile infection- guidelines no longer recommend first line
what are the pharmacokinetics of metronidazole?
excellent oral absorption
widely distributed, including CNS
liver metabolism
excreted in bile and urine
what are the adverse effects of metronidazole?
generally well tolerated
metallic taste
GI disturbances
CNS- cumulative peripheral neuropathy
disulfiram reaction with alcohol
which antibiotics are nucleic acid structure inhibitors?
quniolones (ciprofloxacin), metronidazole
what is the mechanism of action of trimethoprim/sulfamethoxazole?
interferes wtih bacterial folic acid synthesis
sulfonamides competitively inhibit to prevent dihydrofolic acid synthesis
trimethoprim stops production of tetrahydrofolic acid
what are the uses of trimethoprim/sulfamethoxazole?
urinary tract infections, skin and soft tissue infections
drug of choice for nocardia, p. jirovecii, and s. maltophilia
what is the spectrum of activity of trimethoprim/sulfamethoxazole?
G+ aerobes including MRSA, G- aerobes, PJP, listeria, nocardia
what are the pharmacokinetics of trimethoprim/sulfamethoxazole?
good oral absorption (similar levels to IV or PO)
distributes into most tissues, including CNS
minimal metabolism
renal excretion
what are the adverse effects of trimethoprim/sulfamethoxazole?
hypersensitivity, skin disorders (25-50% of HIV patients, <10% of non-HIV patients)
GI disturbances, bone marrow suppression, photosensitivity, hyperkalemia, renal toxicity (pseudo-creatinine elevation, but also may cause true acute kidney injury)